Abstract
Background
Renal interstitial fibrosis (RIF) is a common pathological change in chronic kidney disease (CKD). Many CKD patients exhibit varying degrees of disruption in iron and lipid metabolism. Ferroptosis, a unique form of cell death, has been observed in numerous kidney diseases. Regulating ferroptosis presents a promising therapeutic strategy for RIF. Traditional Chinese medicine (TCM) has shown potential in treating RIF, attracting growing research interest in clarifying its mechanisms.
Objectives
This article aims to summarize the role of ferroptosis in RIF and explore the relationship between ferroptosis and TCM monomers, with the goal of identifying new candidate drugs for treating RIF by targeting ferroptosis.
Methodology
Through inductive analysis of existing studies, the effects of active components in TCM—such as flavonoids, terpenes, and phenolic acids—on key proteins and signaling pathways were examined. These proteins include GPX4, TFR1, EMT, α-SMA, KEAP1, SLC7A11, Smad3, ATF3, Nrf2, TGF-β1, Smad-2, and FTH. Signaling pathways such as Nrf2/GPX4, TLR/Nox4, and Nrf2/HO-1 were analyzed to elucidate the regulatory mechanisms.
Results
TCM active components were found to modulate the expression of multiple proteins involved in ferroptosis and fibrosis through key signaling pathways. This regulation alleviates oxidative stress and apoptosis, reduces iron deposition, and ultimately inhibits the development of RIF.
Conclusion
This review highlights the significance of ferroptosis in RIF and illustrates how TCM monomers can interfere with this process. The findings provide innovative ideas and a theoretical basis for the treatment of RIF by targeting ferroptosis, offering promising directions for future drug development.
Keywords
Introduction
Chronic kidney disease (CKD) has become a significant global public health issue. Forecasts indicate that by 2040, it will rank among the top five causes of mortality worldwide (Wang et al., 2021). Renal interstitial fibrosis (RIF) is the main pathological change observed in many kidney disorders and plays a crucial role in disease progression (Humphreys, 2018). Damage and inflammation are triggering factors, with any regulatory cell death, including ferroptosis, driving cellular death and inflammation (Galluzzi et al., 2014). Ferroptosis is an emerging type of programmed cell death characterized by iron dependence, typically marked by iron accumulation and lipid peroxidation (LPO). This process is governed by various cellular metabolic pathways and signaling mechanisms, including redox balance, iron metabolism, mitochondrial function, and the metabolism of amino acids, lipids, and glucose (Jiang et al., 2021). Ferroptosis is fundamentally distinct from other types of cell death—including autophagy, necrosis, apoptosis, and pyroptosis—in terms of its structural features, molecular mechanisms, and genetic regulation (Fang et al., 2023). Recent research indicates that ferroptosis contributes to several renal disorders, including acute kidney injury, diabetic nephropathy, and CKD (Müller et al., 2017; Wang et al., 2020, 2022).
Modulating ferroptosis to alleviate RIF offers potential therapeutic targets for clinical treatment. Increasing research focuses on ferroptosis, exploring the intervention effects of traditional Chinese medicine (TCM) on RIF (Wang et al., 2023). This article provides an overview of the latest mechanisms underlying ferroptosis in RIF. It also evaluates recent advancements in research on TCM monomers for treating RIF.
Mechanisms of Ferroptosis
Disruptions in Iron Metabolism
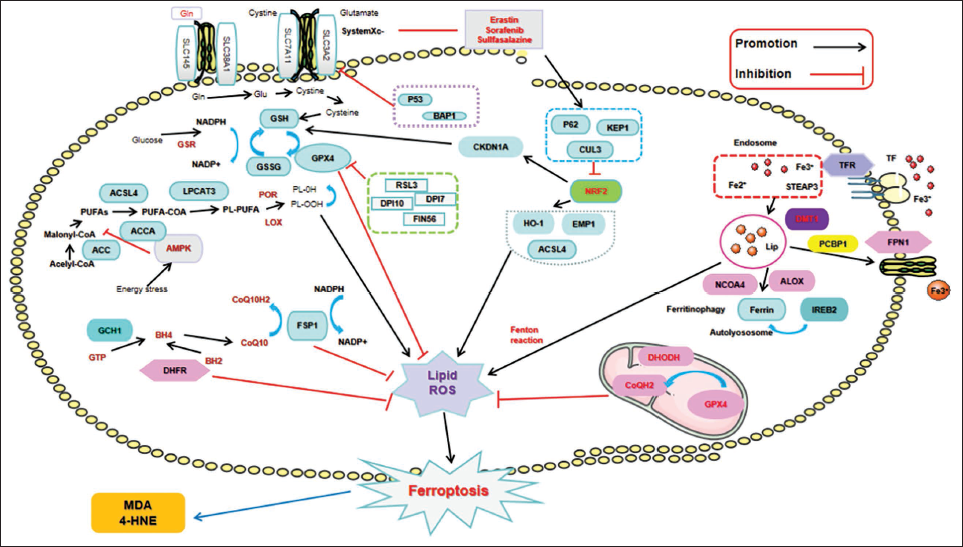
Dysregulations in the processes of iron uptake, absorption, storage, and excretion can trigger ferroptosis through iron-mediated increased generation of reactive oxygen species (ROS) via the Fenton reaction (Dixon et al., 2012). Another pathway involves the activation of iron-containing enzymes, such as arachidonate lipoxygenase (ALOX) and nuclear coactivator 4 (NCOA4)-mediated ferritinophagy, releasing Fe2+ stored in ferritin into the cytoplasm (Hou et al., 2016), leading to the continuous accumulation of iron-dependent lipid ROS. Additionally, Fe2+ generates ROS through participation in the Fenton reaction and catalyzation of iron-dependent oxidases (Ingold et al., 2018; Yang et al., 2016). Moreover, increased concentrations of transferrin (TF) and its receptor, transferrin receptor 1 (TFR1), further facilitate the initiation or progression of ferroptosis (Gao et al., 2015).
LPO
Elevated levels of ROS instigate LPO, which specifically compromises biomembranes enriched with phosphatidylethanolamine (PE) and polyunsaturated fatty acids (PUFAs), ultimately triggering ferroptosis. Following their remodeling by acyl-CoA synthetase long-chain family member 4 (ACSL4) and lysophosphatidylcholine acyltransferase 3 (LPCAT3), PUFAs are transformed into phospholipids. This process results in the generation of lipid peroxides, such as malondialdehyde (MDA) and 4-hydroxynonenal (HNE), via both enzymatic and non-enzymatic pathways. The accumulation of these lipid peroxides causes irreversible damage to membranes, culminating in cellular demise (Dixon et al., 2015; Kagan et al., 2017; Mortensen et al., 2023). Lipoxygenases (LOX) and cytochrome P450 oxidoreductase (POR) often serve as cofactors in enzymatic reactions.
Cystine/Glutamate Anti-porter/GSH/GPX4 System
The glutamate anti-porter, also called System Xc-, is a heterodimer made up of solute carrier family 7 member 11 (SLC7A11) and solute carrier family 3 member 2 (SLC3A2). This anti-porter is essential for regulating the uptake of extracellular cystine (Cys) and for releasing intracellular glutamate (Parker et al., 2021). Cys entering the cell is reduced to Cys, participating in the synthesis of glutathione (GSH). Inhibition of System Xc- reduces cystine influx, decreasing Cys levels and hampering GSH synthesis. GSH, as a cofactor of glutathione peroxidase 4 (GPX4), reduces the toxicity of lipid peroxides, protecting biomembranes from ferroptosis. GPX4 functions as a key regulator essential for maintaining intracellular lipid homeostasis. The loss of function or silencing of this enzyme results in the buildup of lipid ROS, which subsequently initiates the process of ferroptosis (Wang et al., 2023).
Other Pathways
Fibroblast specific protein-1 (FSP1) acts as a new inhibitor, preventing ferroptosis caused by GPX4 deficiency (Doll et al., 2019). FSP1 facilitates the conversion of CoQ10 into its reduced form using NAD(P)H as an electron donor, which boosts its antioxidant properties and consequently diminishes the buildup of lipid peroxides. Concurrently, CoQ10 plays a protective role against ferroptosis by neutralizing free radicals and reducing oxidative stress levels (Liu et al., 2023). The FSP1/CoQ10/NAD(P)H and System Xc-/GSH/GPX4 systems exist as independent parallel systems, collectively exerting anti-oxidative effects, limiting oxidative damage (Bersuker et al., 2019). Overexpression of GCH1 enhances the BH4/BH2 folate biosynthesis pathway, inhibiting LPO to prevent ferroptosis (Kraft et al., 2020). Rapid inactivation of GPX4 significantly upregulates DHODH, leading to increased production of CoQH2, neutralizing LPO to prevent mitochondria-triggered ferroptosis (Porporato et al., 2018). AMPK is activated by stress, which then phosphorylates and inhibits its downstream substrate acetyl-CoA carboxylase 1 (ACC1). This phosphorylation reduces fatty acid synthesis. As a result, the accumulation of lipid peroxides is attenuated, which delays the initiation of ferroptosis (Song et al., 2018). Furthermore, AMPK facilitates the metabolism and utilization of iron within cells through the modulation of autophagy-related gene expression. This regulatory mechanism helps to mitigate excessive iron accumulation and lowers the concentration of lipid peroxides in cellular environments (Wang et al., 2024a) AMPK can additionally suppress ferroptosis by augmenting the activity of the Nrf2 signaling pathway, which in turn stimulates the expression of antioxidant enzymes (Liu et al., 2024). Additionally, P53 and Nuclear factor E2-related factor 2 (Nrf2) are closely related to the occurrence of ferroptosis (Dodson et al., 2019; Lei et al., 2021). For example, P53 can upregulate iron regulatory proteins (IRP) and transferrin receptors (TFR), promoting the uptake and utilization of iron, thereby enhancing the cell’s resistance to oxidative stress (Lei et al., 2021). P53 can inhibit SLC7A11, thereby reducing intracellular glutathione levels and promoting the occurrence of ferroptosis (Shin et al., 2024). Nrf2 can upregulate the expression of iron transport protein ferroportin and iron storage protein ferritin, thereby promoting the expulsion of excess iron from cells, reducing iron accumulation, and lowering the occurrence of oxidative stress (Lu et al., 2021). Nrf2 has the ability to enhance the expression of antioxidant enzymes, including GPX4 and SLC7A11, which are essential for the suppression of ferroptosis (Lee & Roh, 2023). See Figure 1 for details.
The Main Regulatory Mechanism of Ferroptosis.
Role of Ferroptosis in Renal Interstitial Fibrosis
Inflammation
Infiltration of inflammatory cells is a key feature of RIF (Kang et al., 2015). Inflammatory mediators, including tumor necrosis factor-alpha (TNF-α) and IL-6, have the potential to facilitate the development of ferroptosis. This process subsequently intensifies RIF through the secretion of pro-fibrotic agents, prominently transforming growth factor-beta 1 (TGF-β1) (Liang et al., 2024). Moreover, an imbalance in iron metabolism may result in harm and programmed cell death of renal tubular cells, which in turn can exacerbate the advancement of RIF (Chen et al., 2024). Studies on obesity-induced renal ferroptosis found that ferrostatin-1 (Fer-1) mitigates renal tissue fibrosis, inflammatory cell infiltration, and inflammatory factor expression induced by a high-fat diet (Luo et al., 2020). Fer-1 targets the STING/ACSL4 axis to combat inflammatory factor production, macrophage infiltration, and fibrosis in hypertension-associated CKD (Gao et al., 2023). Tocilizumab, an IL-6 receptor-targeted drug, inhibits ferroptosis, reduces macrophage activation and differentiation, alleviates inflammation, and lowers unilateral ureteric obstruction (UUO) surgery mice fibrosis and apoptosis (Yang et al., 2020). Consequently, targeting the interaction mechanisms between inflammation and ferroptosis could offer novel therapeutic approaches for the management of RIF. By regulating the inflammatory response and iron metabolism, it is possible to effectively reduce the occurrence and progression of RIF, thereby improving patient prognosis (Huang et al., 2023).
Oxidative Stress
Oxidative stress plays a crucial role in the generation of ROS and the impairment of the antioxidant defense mechanisms, ultimately resulting in cell death through processes such as apoptosis and ferroptosis (Guerrero-Hue et al., 2019; Linkermann et al., 2014). Research indicates that oxidative stress contributes to the impairment and programmed cell death of renal tubular epithelial cells (TECs) by facilitating the onset of ferroptosis. This process subsequently initiates a fibrotic response within the renal interstitium (Jian et al., 2023). Fer-1 exerts an inhibitory effect on the levels of HIF-1α and HO-1, which leads to a decrease in iron accumulation and the generation of ROS. Consequently, this action mitigates renal fibrosis in mice with Type 2 diabetes (Feng et al., 2021). Nrf2, a major transcription factor defending against oxidative stress (Nezu et al., 2017), transcriptionally activates countermeasures against ferroptosis (Shin et al., 2018). Sulforaphane (SFN) (de Zeeuw et al., 2013) and ferroptosis inhibitor liproxstatin-1 (Lip-1) modulates the Nrf2/GPX4 signaling pathway to suppress ferroptosis, thereby diminishing oxidative stress and mitigating fibrosis in mice subjected to UUO (Yang et al., 2023).
Autophagy
Autophagy plays a critical role in modulating fibrosis by influencing mechanisms such as cell death, interstitial inflammation, and the synthesis of pro-fibrotic secretory proteins, notably fibroblast growth factor 2 (FGF2) (Livingston et al., 2016).
Studies indicate that the prevalence of RIF is significantly associated with the buildup of intracellular iron and the resulting oxidative stress. Furthermore, dysfunctional autophagic processes may result in an excessive accumulation of iron, thereby enhancing the occurrence of ferroptosis (Pan et al., 2024). The renal tubular cells lacking autophagy exhibit more severe cell damage and fibrosis under the influence of ferroptosis. Furthermore, the activation of ferroptosis can further exacerbate renal interstitial damage and fibrosis by affecting the process of autophagy (Zhu et al., 2023). Ferroptosis and autophagy share common regulatory factors, such as SLC7A11, GPX4, Nrf2, and heat shock protein β-1 (HSP90β1) (Li et al., 2019). Mitochondrial autophagy receptor NIX can release Beclin1 to induce mitochondrial autophagy and bind with SLC7A11, blocking the activity of System Xc-, thereby inhibiting ferroptosis (Zhang et al., 2023). Ferritinophagy, mediated by NCOA4, is a selective form of autophagy. In a rat model of CKD induced by 5/6 nephrectomy, the upregulation of NCOA4 expression resulted in ferroptosis, thereby facilitating the progression of RIF (Wang et al., 2022). Melatonin and zileuton mitigate ferroptosis and enhance renal fibrosis resulting from unilateral UUO via the established AKT/mTOR/Nrf2 autophagy signaling pathway (Jung et al., 2023).
Signaling Pathways
TGF-β/Smads signaling pathway: TGF-β, recognized as a primary contributor to fibrosis, plays a pivotal role in the signaling cascades associated with fibrotic processes by stimulating the release of pro-fibrotic agents. The expression levels of ACSL4 exhibit a strong correlation with the incidence of RIF. A research investigation focused on the inhibitors of ACSL4 demonstrated that both the inhibition and knockdown of ACSL4 significantly diminish ferroptosis in TECs. Additionally, it was observed that these interventions lead to a reduction in the expression of pro-fibrotic cellular factors, ultimately mitigating RIF. This phenomenon is attributed to the capacity of ACSL4 inhibition to antagonize TGF-β expression as well as the TGF-β/Smads signaling pathway within TECs (Dai et al., 2023). Hippo signaling: YES-associated protein (YAP) is a transcription coactivator in the Hippo pathway, promoting ferroptosis by regulating ACSL4 expression. YAP facilitates CaOx crystal-induced cell ferroptosis and enhances renal fibrosis by upregulating ACSL4 (Li et al., 2023).
The Role of TCM Active Components in Counteracting Ferroptosis to Improve RIF
TCM is a treasure of Chinese traditional medical science. It is extensively utilized in managing RIF. The effective ingredients of TCM are the fundamental substances that are used to improve the efficacy of Chinese medicine. The active components of Chinese medicine have multi-target, multi-pathway, and multi-level against RIF activities (Yang et al., 2024).
Research indicates that various bioactive constituents found in TCM, including flavonoids, terpenoids, and phenolic acids, possess the capability to mitigate RIF through the suppression of ferroptosis. Specific details are provided in Table 1 and Figure 2.
Monomeric Compounds.
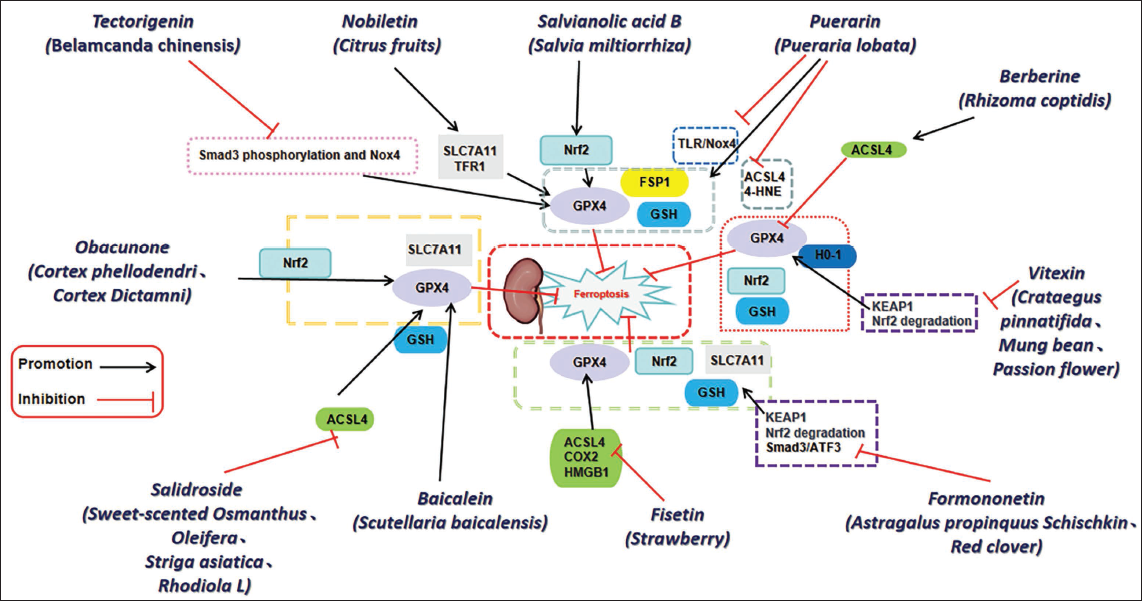
Traditional Chinese Medicine Active Components in Counteracting Ferroptosis to Improve Renal Fibrosis.
Flavonoids
Flavonoids represent a category of polyphenolic substances that are abundantly present in various plant species, predominantly extracted from sources such as fruits, vegetables, tea, grains, and specific herbs. The primary categories of these compounds consist of flavones, flavonols, flavonoid glycosides, and isoflavones. Each of these classes exhibits a range of biological properties, including antioxidant, anti-inflammatory, and anti-fibrotic activities (Song et al., 2023). TCM utilizes numerous medicinal plants, such as Scutellaria baicalensis, Goji berries, and Angelica sinensis, which are abundant in flavonoid compounds. These plants are extensively employed in the treatment of a range of ailments, particularly those affecting the kidneys and liver (Wang et al., 2024b).
Tectorigenin, a bioactive isoflavonoid derived from Belamcanda chinensis, exhibits notable anti-ferroptotic effects, both in vivo and in vitro. Animal model investigations have demonstrated that Tectorigenin markedly suppresses the phosphorylation of SMAD family member 3 (Smad3), as well as the transcriptional and protein levels of its direct downstream target, NADPH oxidase 4 (NOX4). This mechanism appears to lead to an indirect restoration of GPX4 expression. Additionally, in vitro assessments have indicated that Tectorigenin effectively inhibits ferroptosis induced by erastin and RSL3, as well as renal fibrosis stimulated by TGF-β1 (Li et al., 2022). Nobiletin (Nob) is an important flavonoid derived from citrus fruits. Research has demonstrated that Nob significantly decreases the levels of renal fibrosis and EMT markers in mice subjected to UUO, improve oxidative stress and apoptosis, inhibit leukocyte infiltration and inflammatory response, significantly reduce GPX4, SLC7A11/xCT, and TFR1, and reduce ferroptosis against RIF (Lo et al., 2022). Puerarin, an isoflavone derived from Pueraria lobata, exhibits potent antioxidant properties and has the capacity to either directly or indirectly augment energy metabolism while mitigating oxidative stress. It plays a significant role in the pathophysiology of various diseases (Lv et al., 2022). Puerarin mitigates the increased expression levels of TLR/Nox4 and suppresses the expression of factors associated with oxidative stress and ferroptosis. Additionally, it decreases collagen deposition, interstitial fibrosis, and α-SMA expression within the renal tissues of a CKD model induced by ischemia/reperfusion (I/R). Puerarin could reverse the changes of SOD, MDA, GSH, and Fe2+ levels induced by I/R and hypoxia/reoxygenation (H/R). The results show that Puerarin pretreatment can alleviate renal fibrosis in vivo and in vitro models. It provides a new target for the treatment of renal fibrosis (Jian et al., 2023). Berberine (BBR), a flavonoid from Rhizoma coptidis, is an effective treatment for many diseases, including inflammation, diabetes, and cancer (Deng et al., 2019; Zhang et al., 2019). BBR shows considerable potential as a therapeutic agent for addressing fibrotic disorders, and studies in mouse UUO models have confirmed that BBR can reduce kidney pathological injury, kidney interstitial fibrosis, and the number of α-SMA positive cells. In vitro studies have found that BBR can effectively reduce the expression of α-SMA and GPX4 in renal myoblasts, increase ACSL4, then activate ferroptosis in renal myoblasts, inhibit the activation of renal myoblasts, and alleviate mouse RIF (Liu et al., 2023). Vitexin (VIT), a naturally occurring flavonoid derived from various sources including Crataegus pinnatifida, Mung beans, and passion flowers, exerts its effects by inhibiting the degradation of Nrf2 mediated by KEAP1 and ubiquitination. Additionally, it activates the Nrf2/HO-1 signaling pathway, enhances the expression of GPX4, and alleviates LPO and ferroptosis. Furthermore, VIT demonstrates a protective role against tubular injury, interstitial fibrosis, and inflammatory responses (Song et al., 2023). Research has demonstrated that VIT possesses the capability to diminish levels of ROS, Fe2+, and MDA, while simultaneously enhancing GSH concentrations and the expression of GPX4 and SLC7A11 proteins within renal tissue. Additionally, in vitro studies have validated that VIT effectively mitigates iron-induced cell death resulting from elevated glucose levels in HK-2 cells. This compound reduces the concentrations of ROS, Fe2+, and MDA, increases GSH levels, and upregulates the expression of GPX4 and SLC7A11, thereby inhibiting ferroptosis and ameliorating diabetic kidney disease (DKD) (Zhang et al., 2023). Formononetin (FN), a bioflavonoid derived from various plants including Astragalus propinquus Schischkin and red clover, has demonstrated therapeutic potential in models of CKD induced by UUO and folic acid (FA). Additionally, in pTECs models subjected to RSL3 and Erastin, FN treatment significantly ameliorated renal damage and fibrosis linked to ferroptosis. Regarding its mechanistic action, FN was observed to inhibit the interaction between Smad3 and ATF3, thereby preventing their nuclear translocation. Furthermore, FN enhanced the activity of Nrf2 and SLC7A11, exhibiting a notable inhibitory effect on both renal fibrosis and ferroptosis in CKD, as evidenced by both in vivo and in vitro studies (Zhu et al., 2023). Fisetin, a flavonoid present in strawberries and other fruits and vegetables. It has strong antioxidant and anti-inflammatory activities and can combine with iron and copper to play a beneficial role (Maher, 2020). It can inhibit ACSL4, COX2, and HMGB1, increase GPX4 expression, effectively restore the ultrastructural morphology of mitochondria in CKD mice kidneys, and alleviate renal fibrosis (Wang et al., 2024c). Fisetin promotes the translocation of Nrf2 to the nucleus, thereby enhancing its antioxidant properties. Additionally, it influences the expression of markers associated with iron-dependent cell death, including HO-1 and GPX4, while also mitigating oxidative stress-related damage in mice with DKD (Qian et al., 2023).
Baicalein (BAI), a polyphenolic flavonoid isolated from the roots of S. baicalensis, exhibits significant antioxidant properties. This compound has been utilized in the treatment of fibrotic disorders affecting various organs, including the kidneys, heart, lungs, and liver (Wang et al., 2015). Studies have shown that BAI reduces kidney weight, improves renal function, and mitigates renal tubule damage in UUO-induced rat models. In the UUO rat model, the inhibition of nephrohydrosis was achieved through the reduction of ROS and MDA levels, alongside the elevation of SOD and GSH levels. It can effectively reduce the expression of TGF-β1, α-SMA, SamAD-2, and other fibrosis-related proteins in vitro and in vivo, prevent RIF, increase the expression of SLC7A11, GPX4, FTH, and other ferroptosis-related proteins, and inhibit ferroptosis (Liang et al., 2024). In recent years, there has been a notable increase in investigations concerning the influence of flavonoids on ferroptosis-mediated RIF. Empirical studies have revealed that flavonoids exhibit considerable antioxidant and anti-inflammatory properties, underscoring their potential therapeutic value in addressing renal disorders. Furthermore, clinical investigations have suggested that flavonoids positively impact kidney function enhancement and the reduction of urinary protein levels, thereby furnishing clinical evidence that supports their use in the management of RIF (Liu et al., 2021). Although there has been some progress in the study of flavonoids in ferroptosis-mediated RIF, there are still many challenges. First, the low bioavailability of flavonoids limits their widespread clinical application. Therefore, future research needs to explore methods to enhance their bioavailability, such as through nanocarrier technology or chemical modification to improve their efficacy (Wang et al., 2022). Moreover, while previous research has uncovered the mechanisms through which flavonoids exert antioxidant and anti-inflammatory effects, their specific action targets and signaling pathways still need to be explored in depth to better understand their role in RIF. In addition, the design and implementation of clinical trials also need to be more rigorous to ensure the reliability and reproducibility of the research results. Despite these challenges, research on flavonoids is still full of opportunities, especially in the context of personalized medicine and precision therapy, where the potential of flavonoids as natural drugs deserves further exploration and development (Burke et al., 2024). Through interdisciplinary collaboration and research, new breakthroughs in the treatment of RIF are expected in the future.
Terpenoids
Terpenoids represent a significant category of naturally occurring compounds within the plant kingdom, frequently located in essential oils, resins, and the nectar of numerous plant species. These compounds can be categorized into distinct classes based on their structural variations, such as monoterpenes, sesquiterpenes, terpenes, diterpenes, and triterpenes. Furthermore, Terpenoids are known to display a wide range of pharmacological properties, encompassing anti-inflammatory, anti-bacterial, anti-tumor, antioxidant, and analgesic activities. Research has shown that terpenoids can influence the occurrence of ferroptosis through various mechanisms. For example, certain terpenoids can inhibit ferroptosis by activating antioxidant enzymes such as GPX4, thereby enhancing cell survival (Yi et al., 2024). Consequently, a comprehensive investigation into the regulatory function of terpenoids in ferroptosis may uncover their prospective uses in therapeutic interventions for various diseases.
Salidroside (SAL) is a glycoside of phenylpropanoid origin, derived from various plants including sweet-scented Osmanthus, Oleifera, Striga, and Rhodiola L. It is classified as a diterpene glycoside and is recognized for its extensive anti-aging properties. SAL can significantly reduce LPO in kidney, significantly lowers TGF-β and α-SMA levels, regulate TfR1 protein level, minimize the accumulation of iron, regulate SLC7A11 and GPX4 protein expression, inhibit cell ferroptosis to delay renal aging, thereby delaying renal aging and inhibiting aging-related glomerular fibrosis (Yang et al., 2022). Obacunone is a natural compound extracted from plants such as Cortex phellodendri, Cortex Dictamni, and other plants. This compound is classified as a limonoid triterpenoid and exhibits a range of pharmacological properties, such as anti-inflammatory, antioxidant, and anti-cancer activities (Gao et al., 2018; Murthy et al., 2015; Zhou et al., 2019). Obacunone functions as a powerful agonist of Nrf2, leading to a notable decrease in the levels of Fe2+ within the renal tissue of murine subjects, as well as a reduction in MDA concentration in serum. Concurrently, there is a significant elevation in the expression of Nrf2, GPx4, and SLC7A11 within kidney tissue. These findings indicate that Obacunone mitigates ferroptosis through the activation of the Nrf2/GPx4 signaling pathway, consequently enhancing RIF in mice subjected to the UUO model (Qiu et al., 2023). Within the framework of RIF, the activation of the Nrf2/GPX4 signaling pathway is recognized as a pivotal mechanism that safeguards renal TECs against oxidative stress and ferroptosis. Through the inhibition of Keap1, Nrf2 undergoes stabilization and subsequently translocates to the nucleus, where it facilitates the upregulation of GPX4. This process ultimately bolsters the antioxidant defenses of the cells and mitigates the progression of RIF (Li et al., 2022). The importance of terpenoid compounds in ferroptosis and RIF is increasingly gaining attention, and related research provides us with new perspectives to understand these complex biological processes. Terpenoid compounds may influence ferroptosis and fibrosis processes by regulating intracellular iron homeostasis, antioxidant stress, and other mechanisms. However, despite preliminary results, the specific mechanisms of terpenoid compounds in these pathological processes still require in-depth exploration. Subsequent investigations ought to concentrate on examining the biological activities of various terpenoid compounds along with the factors that affect them, including dosage, administration routes, and interactions with other drugs. This will elucidate the possible applications of terpenoid compounds in the treatment of CKD.
Phenolic Acids
Phenolic acids represent a prevalent category of bioactive substances found in plant species, predominantly characterized by the presence of benzene ring structures coupled with carboxyl functional groups (Lu & Han, 2024). These compounds mainly originate from fruits, vegetables, grains, and their processing by-products, particularly in higher concentrations in the peels, seeds, and roots of plants (Rashmi & Negi, 2020). Phenolic acids mitigate oxidative stress through the neutralization of free radicals and the suppression of LPO, thus safeguarding cells from oxidative injury induced by iron (Shang et al., 2024). Salvianolic acid B (Sal B), an important water-soluble component of Salvia miltiorrhiza, it is one of the most powerful natural products known to have antioxidant effects. It can alleviate the epithelial-interstitial transformation process in the process of renal fibrosis, thus alleviating end-stage renal injury (He et al., 2020). In UUO rat models of RIF, Sal B may reduce MDA content and increase SOD activity, inhibit ferroptosis and alleviate oxidative damage by activating Nrf2/GPX4 pathway, and thus improve RIF (Sun et al., 2022). In the model of acute renal injury induced by H/R in renal TECs, it was observed that Sal B effectively mitigated ferroptosis. This protective effect was achieved through the reduction of mitochondrial damage and lipid accumulation, alongside the upregulation of FSP1 and GPX4 expression while downregulating ACSL4 expression (Zhao et al., 2024). With the deepening of research, phenolic acid compounds have been confirmed to have potential roles in regulating intracellular iron homeostasis, alleviating oxidative stress, and inhibiting the process of fibrosis. Nevertheless, existing research findings regarding the correlation among phenolic acids, ferroptosis, and RIF exhibit inconsistencies, posing a significant challenge in reconciling diverse perspectives and outcomes in clinical practice. On one side, certain research findings demonstrate that phenolic acid compounds have the potential to significantly hinder the advancement of RIF. Conversely, other investigations propose that the underlying mechanisms by which these compounds operate require additional scrutiny, particularly given that their effects may differ across various pathological contexts. Therefore, future research should focus more on clarifying the mechanisms of action of phenolic acid compounds, exploring their effects in different models, and integrating clinical data to validate their true therapeutic potential.
Discussion
The underlying mechanisms involved in the pathogenesis of RIF are intricate, and as of now, no targeted and effective pharmacological treatments have been established. However, continuous research and an enhanced comprehension of ferroptosis in renal conditions have facilitated advancements in the formulation of ferroptosis-related agents, both inducers and inhibitors, aimed at the therapeutic management of CKD. However, the majority of these developments are still in the animal experimental stage. This review indicates that many natural medicinal components have shown efficacy in regulating ferroptosis for treating RIF, potentially bringing innovation to CKD treatment. Nonetheless, a number of challenges still require attention: First, it remains to be determined if ferroptosis serves as a fundamental mechanism in the development of CKD. Second, the relationship between ferroptosis and other cell death modalities during the progression of the disease—whether they function synergistically or antagonistically—necessitates additional investigation. Third, many genes regulate ferroptosis, but most cannot serve as sensitive indicators for detecting ferroptosis, making the identification of specific biomarkers a challenge. Fourth, TCM compounds are less applied in cell and animal experiments, gradually losing the characteristics and advantages of Chinese herbal compatibility. It can be seen that the mechanisms of action of monomeric compounds in TCM in the treatment of RIF and related diseases have not been fully understood.
Future Perspectives
Subsequent investigations ought to concentrate on examining the distinct impacts of these compounds on ferroptosis and the modulation of its associated signaling pathways. For example, researchers can utilize modern molecular biology techniques, such as gene editing and transcriptomics, to analyze the effects of monomeric compounds in cell models, revealing their regulatory effects on key genes and proteins. Additionally, by combining network pharmacology methods, it is possible to systematically identify the targets of monomeric compounds in TCM and their interaction networks, thereby providing new perspectives for mechanism research. At the same time, considering the multi-component characteristics of TCM, future studies should also focus on the synergistic effects of various components in TCM formulas, with the aim of finding more effective treatment strategies (Ning et al., 2024). In the process of promoting the clinical application of single compounds from TCM, safety and feasibility assessment are crucial. To begin with, it is essential to conduct thorough clinical trials to assess the safety profile of these compounds. This evaluation should encompass adverse reactions, possible drug interactions, and the risks that may arise from prolonged usage. Second, researchers should focus on the pharmacokinetic characteristics of single compounds from TCM to determine their absorption, distribution, metabolism, and excretion in the body, which is essential for assessing their clinical applicability. Furthermore, considering the individualized treatment characteristics of TCM, future research should explore the differences in responses to single compounds among different patient populations to achieve the goal of personalized medicine. Through these measures, a solid foundation can be established for the clinical application of single compounds from TCM in RIF and other related diseases (Xuan et al., 2023).
Conclusion
Through this review, by summarizing the role of active Chinese medicinal components in counteracting ferroptosis for RIF treatment, it is hoped that more attention will be paid to the more in-depth and comprehensive study of TCM compounds in the mechanism of ferroptosis. This could highlight the unique features and advantages of TCM, providing new insights for its therapeutic application. To this end, researchers should actively engage in interdisciplinary collaboration to promote the translation of TCM compounds into clinical applications, providing new treatment options for patients with RIF.
Footnotes
Abbreviations
ACC1: Acetyl-CoA carboxylase 1; ACSL4: Acyl-CoA synthetase long-chain family member 4; ALOX: Arachidonate lipoxygenase; BAI: Baicalein; BBR: Berberine; CKD: Chronic kidney disease; Cys: Cystine, FA: Folic acid; Fer-1: Ferrostatin-1; FGF2: Fibroblast growth factor 2; FN: Formononetin; FSP1: Fibroblast specific protein-1; GPX4: Glutathione peroxidase 4; GSH: Glutathione; H/R: Hypoxia/reoxygenation; HNE: 4-Hydroxynonenal; HSP90β1: Heat shock protein β-1; I/R: Ischemia/reperfusion; IRP: Iron regulatory proteins; Lip-1: Liproxstatin-1; LOX: Lipoxygenases; LPO: Lipid peroxidation; LPCAT3: Lysophosphatidylcholine acyltransferase 3; MDA: Malondialdehyde; NCOA4: Nuclear coactivator 4; Nob: Nobiletin; NOX4: NADPH oxidase 4; Nrf2: Nuclear factor E2-related factor 2; PE: Phosphatidylethanolamine; PUFAs: Polyunsaturated fatty acids; RIF: Renal interstitial fibrosis; ROS: Reactive oxygen species; SAL: Salidroside; Sal B: Salvianolic acid B; SFN: Sulforaphane; SLC3A2: Solute carrier family 3 member 2; SLC7A11: Solute carrier family 7 member 11; Smad3: SMAD family member 3; System Xc-: Glutamate anti-porter; TCM: Traditional Chinese medicine; TECs: Tubular epithelial cells; TF: Transferrin; TFR: Transferrin receptors; TFR1: Transferrin high-affinity receptor 1; TNF-α: Tumor necrosis factor-alpha; UUO: Unilateral ureteric obstruction; VIT: Vitexin; YAP: YES-associated protein.
Authors Contribution
Xiaoming Yan: Conceptualization, writing—original draft, data curation. Yao Chen: Formal analysis, investigation, methodology. Qiang Zhao: Data curation, investigation, methodology. Shihua Yi: Conceptualization, resource, supervision, writing—review & editing. Ming Chen: Visualization, writing—review & editing. Yingying Fang: Data curation, investigation.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was obtained from the relevant ethics committee or Institutional Review Board (IRB).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by funding from “Supported by Heilongjiang Provincial Natural Science Foundation of China (LH2021H070)” and “TCM research Project of Heilongjiang Province (ZHY2022-104).”
Informed Consent
The participant has provided informed consent for the submission of the article to the journal.
