Abstract
Background
Quercetin possesses anti-oxidative stress effect with potential in the treatment of tumor-related diseases. Whether it plays a role in liver cancer remains unclear.
Objectives
This study aims to explore its role in the ferroptosis of liver cancer cells and the action of the nuclear factor erythroid-related factor 2 (Nrf2) signaling pathway in this process.
Materials and Methods
Liver cancer cells (MHCC97-H) were cultured in vitro and assigned into two groups: the control and the experimental groups. MHCC97-H cells were given different concentrations of intervention and divided into five groups, with concentrations of 0.5, 1.0, 2.0, 4.0, and 8.0 mg/mL, and detected by the CCK8 method to observe cell viability. The culture time was set at three time points (24, 48, and 72 h). The cell proliferation activity, clone cell number, cell morphology under Hoechst staining, and cell apoptosis number of each group were compared, and the optimal dose was selected. MHCC97-H cells were further intervened with Nrf2 inhibitor ML385, Nrf2 agonist dimethyl fumarate (DMF), ferroptosis agonist Erastin, and ferroptosis inhibitor Fer-1 to observe the expression of various proteins, reactive oxygen species (ROS), and malondialdehyde (MDA). Among them, ROS was detected by flow cytometry, Nrf2, NADPH oxidase 4 (NOX4), and glutathione peroxidase 4 (GPX4) levels were detected by Western blot (WB), and MDA and glutathione (GSH) content were detected by MDA and GSH corresponding kits.
Results
Compared with the control group, the cell viability of the experimental group decreased, the number of cell proliferation decreased, the number of apoptosis increased, and the cell nucleus was condensed, which showed a dose-dependent change with the change of drug concentration. The levels of GPX4 and Nrf2 in the Que+8 group decreased, and the contents of NOX4 and ROS increased significantly. Levels of Nrf2, GSH, and MDA in the ML385 group and Que+8 group decreased, and the contents of GPX4, NOX4, and ROS increased significantly. Compared with the Que+8 group, levels of Nrf2, GSH, and MDA in the Que+8+DMF group increased, and GPX4, NOX4, and ROS levels decreased significantly. Compared with the Fer-1 group, levels of MDA and ROS in the Erastin group increased, and Nrf2 levels decreased.
Conclusion
Quercetin can effectively activate Nrf2 signaling to upregulate the oxidative stress response and induce ferroptosis in liver cancer cells, which is helpful for treating liver cancer.
Introduction
Hepatocellular carcinoma (HCC) accounts for up to 75% of liver cancer (Sung et al., 2021). Its etiology includes factors such as genetics and lifestyle, and its pathogenesis is diverse. When the levels of free radicals and lipid peroxides are too high, cells are damaged, and HCC is induced, which is one of the pathogeneses (Suzuki et al., 2022). Ferroptosis, as a non-apoptotic cell death, not only can effectively inhibit the rapid growth of tumor cells but also improve the body’s immunity to a certain extent, thereby playing a role in hindering the progression of tumor diseases (Wang, Chen, et al., 2022; Zhao et al., 2022). The nuclear factor erythroid-related factor 2 (Nrf2) signaling pathway can inhibit ferroptosis by regulating glutathione peroxidase 4 (GPX4) and other pathways (Deng et al., 2023). In recent years, drugs and related experimental studies on oxidative stress have gradually attracted widespread attention in the field of HCC treatment.
One of the flavonoid compounds represented by quercetin not only has a significant anti-oxidative stress effect but also shows certain potential in the treatment of tumor-related diseases (Carrillo-Martinez et al., 2024; Li et al., 2021). Quercetin inhibits the growth of liver cancer cells by scavenging free radicals, maintaining oxidative balance, increasing the level of glutathione (GSH), and enhancing antioxidant activity (Ding et al., 2024). Further animal experimental observations also verified that quercetin promotes apoptosis of liver cancer cells (Luo et al., 2024). However, quercetin-promoting liver cancer cell ferroptosis may involve the combined effects of multiple mechanisms and multiple targets. Currently, there is no specific report on the specific mechanism of quercetin-induced liver cancer cell ferroptosis. Therefore, this study further explores the effects of quercetin on liver cancer cell ferroptosis in order to provide new ideas for inhibiting tumor growth.
Materials and Methods
Experimental Materials
Human hepatoma cell MHCC97-H (Shanghai Lianmai Bioengineering Ltd.); quercetin (Shanghai Tongtian Biotechnology Ltd., catalog number 01250840); GPX4 (Boersen Biotechnology Ltd., catalog number KM040396); Erastin, Fer-1 (Shanghai Maokang Biotechnology, catalog numbers A100700, A4371); GSH (A006-1-2), malondialdehyde (MDA) (A003-1-1) kits were purchased from Shanghai Xinyu Biotechnology Ltd.; Nrf2 (Jianglai Biotechnology Ltd., catalog number JL18277); reactive oxygen species (ROS) detection kit (Shanghai Xinyu Biotechnology Ltd., catalog number XY-47051-1); Nrf2 agonist dimethyl fumarate (DMF) (China National Pharmaceutical Chemical Reagent Ltd., catalog number 81007728); ML385 (Nanjing Feihong Technology Ltd., catalog number FH0541); annexin V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) apoptosis kit (Jiangsu Huizhi Biotechnology Ltd., catalog number KGA106).
Methods
Cell Culture
MHCC97-H cells were cultured in a Dulbecco’s Modified Eagle Medium (DMEM) medium (100 µg/mL streptomycin solution, 10% fetal bovine serum, and 100 U/mL penicillin solution were added) in an incubator (temperature: 37°C, 5% CO2). The old medium was discarded after 2–3 days and replaced with a new medium until the cells were fused to 80%–90%. The cells were digested and passaged by trypsin (containing 0.25% ethylenediaminetetraacetic acid (EDTA)).
Grouping and Intervention
MHCC97-H cells were assigned to a control group and an experimental group. The experimental group was intervened by adding quercetin solution at 0.5, 1.0, 2.0, 4.0, and 8.0 mg/mL, and further divided into Que+0.5, Que+1, Que+2, Que+4, and Que+8 groups.
CCK8 Detection of Cell Viability
The CCK8 cell proliferation experiment was performed using a 96-well plate. First, 5 × 104 (100 µL) of “2.2.2” cells were inoculated into a 96-well plate, and then the cell growth rate was detected by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) method, and the morphological changes of cells were recorded. The cell growth rate was calculated as (drug group − blank control group)/control group × 100%.
Hoechst Staining to Detect Cell Morphology
Prepare six-well plates for cell inoculation, with an inoculation density of 5 × 105 cells per well. After 24 h of culture, remove the original culture medium, and add trichloroisocyanuric acid (TCCA) solutions with concentrations of 0, 97, 194, and 387 µmol/L to each well, respectively, and continue to treat for 24 h; wash with phosphate-buffered saline (PBS) 3 times, and observe the cell morphology under a fluorescence microscope; further add Hoechst 33342 staining solution to each well for staining, set the staining time to 10 min, and after the staining is completed, further observe and record the changes in cell morphology and color reaction under a fluorescent inverted microscope.
Plate Cloning Experiment to Detect Cell Proliferation
Each group of MHCC97-H cells was prepared into a cell suspension, and a cell suspension with a density of 5 × 102/mL was added to each well of a six-well plate. A dimethyl sulfoxide (DMSO) control group and a quercetin treatment group with different concentrations were set up and then incubated. After the cell culture was completed, it was washed with PBS 2–3 times, fixed with paraformaldehyde, and stained with crystal violet for 20 min. The crystal violet dye used was recovered, and after being thoroughly washed to remove the residues, the specific number of clones formed was recorded by taking pictures.
Annexin V-FITC/PI Detection of Cell Apoptosis
Prepare six-well plates for cell inoculation, with an inoculation density of 5 × 105 cells per well, and add trypsin for digestion. After digestion, further centrifuge, set the speed (200 g), time (5 min), and then stain the cells. The kits are FITC and PI, and finally, use a cell analyzer for further detection and analysis.
Flow Cytometry Detection of ROS
MHCC97-H cells were grouped according to the grouping method in “2.2.2,” and the corresponding drug intervention was implemented. Wash the cells twice with a serum-free culture medium, and then add dichloro-dihydro-fluorescein diacetate (DCFH-DA) at a concentration of 50 µmol/L to the cells, and incubate them at a certain temperature for 20 min. During this process, in order to ensure that the fluorescent dye can be evenly distributed in cells and fully penetrate, the culture dish must be gently shaken once every 5 min. After incubation, wash the cells to completely remove the dye that is not bound to the cells. The fluorescence intensity was measured by flow cytometry, and the oxidative stress state of the cells was evaluated accordingly.
Detection of MDA and GSH
The cells of each group were digested with pancreatic enzymes, collected, and placed in PBS solution, and further broken by ultrasonic disruption technology at a low temperature (4°C) until a uniform cell homogenate was formed. According to the instructions provided by the kit, the levels of MDA and GSH in the cell homogenate were measured, respectively.
Western Blot (WB) Detection of Nrf2 Signaling Pathway and Ferroptosis-related Protein Expression
The sample cells of all groups were collected, and phenylmethylsulfonyl fluoride (PMSF) and radioimmunoprecipitation assay (RIPA) lysis buffers were added. After mixing them evenly by blowing, they were lysed with an ice bath shaker for 30 min. After completion, the supernatant was separated by centrifugation (12,000 r/min). Then, protein supernatant buffer was added and heated for 8 min, and then stored at a certain temperature for use. Proteins were separated by polyacrylamide gel electrophoresis (PAGE) gel electrophoresis (12%). After the electrophoresis, the proteins were transferred to the nitrocellulose (NC) membrane, which was blocked for 20 min. Then, primary antibodies of β-actin, NADPH oxidase 4 (NOX4), Nrf2, and GPX4 (dilution ratio 1:1,000) were added and incubated for 4 h. After incubation, the NC membrane was washed with phosphate-buffered saline with Tween 20 (PBST) solution three times, and the secondary antibody (1:10,000 dilution) was added for 1.5 h. Finally, the membrane was exposed to an enhanced chemiluminescence (ECL) chemiluminescent reagent, and the band was analyzed using ImageJ software.
Statistical Methods
The data obtained from the above experiments were analyzed using Statistical Package for the Social Sciences (SPSS) 25.0 and GraphPad Prism software. The calculated data were (mean ± standard deviation (SD)), which conformed to the normal distribution and met the homogeneity of variance; the data were calculated using the F test, and the comparison of data between groups used the least significant difference (LSD) method. p < .05 was the test standard.
Results
Quercetin Reduces the Activity of Liver Cancer Cells, Inhibits Cell Proliferation, and Further Promotes Apoptosis
In this study, it was found that after adding quercetin, the cell viability and proliferation number decreased significantly (vs. the control group, Figure 1A–1D), and the cell viability and proliferation number of the Que+8 group were the lowest (vs. the Que+0.5, Que+1, Que+2, and Que+4 groups, Figure 1A–1D). Further observation of quercetin’s effect on the cell number and morphology of liver cancer cells MHCC97-H by Hoechst staining found that the number of MHCC97-H live cells had decreased. The cell nucleus was condensed under the intervention of quercetin (vs. the control group, Figure 1E), among which the number of live cells in the Que+8 group was the least. The cell death phenomenon was the most obvious (vs. the Que+0.5, Que+1, Que+2, and Que+4 groups, Figure 1E). In addition, apoptotic cell numbers increased significantly in MHCC97-H cells after quercetin intervention (vs. the control group, Figure 1F), and were the largest in the Que+8 group (vs. the Que+0.5, Que+1, Que+2, and Que+4 groups, Figure 1F). This indicates that quercetin reduces the proliferation activity of liver cancer cells and further increases their apoptosis rate, and with the increase in drug concentration, it shows concentration dependence.
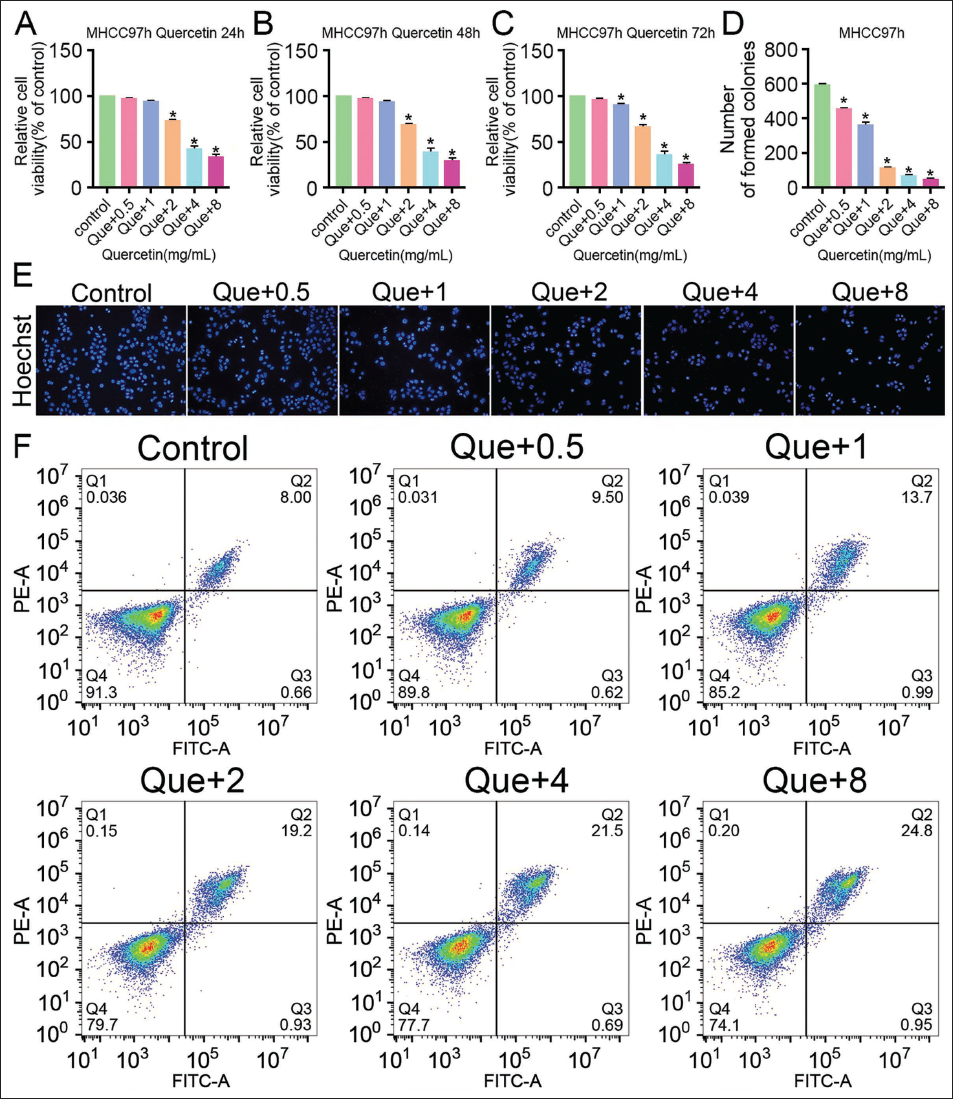
Quercetin Can Inhibit the Nrf2 Pathway to Increase ROS Content and Changes in Cell GPX4 and NOX4
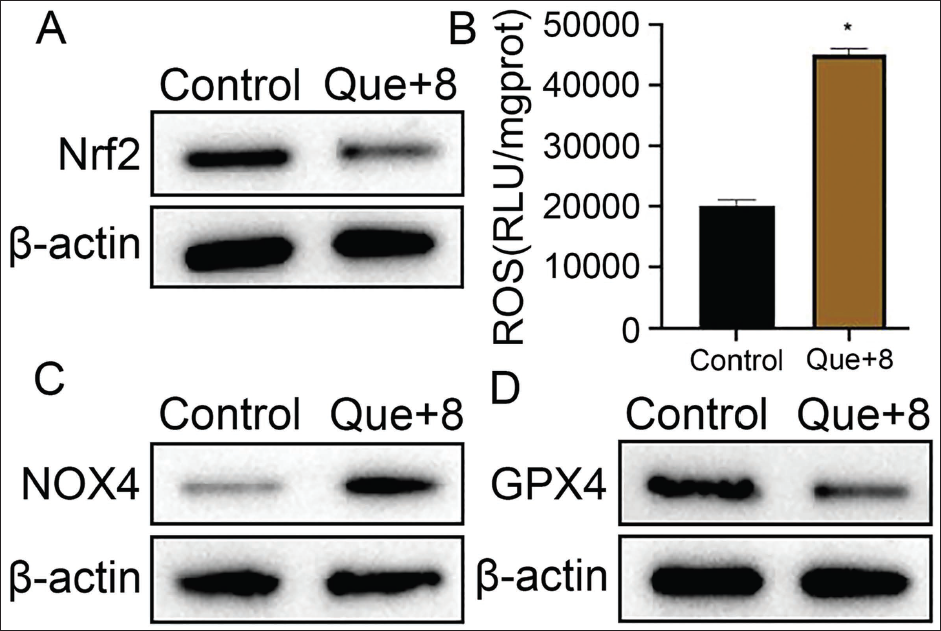
The Nrf2 and ROS expression levels of MHCC97-H cells were observed, and it was found that the Nrf2 level of the Que+8 group was significantly lower than that of the control group (Figure 2A). In contrast, the ROS level was significantly increased (Figure 2B). The study also observed that the NOX4 protein expression increased, while the GPX4 expression decreased (vs. the control group, Figure 2C and 2D). This shows that quercetin can inhibit the Nrf2 pathway to increase ROS release, upregulate the body’s oxidative stress response, and inactivate the GPX4 protein, which may be related to the expression of NOX4, a downstream protein of Nrf2.
Quercetin Inhibits the Nrf2 Pathway and Induces Ferroptosis
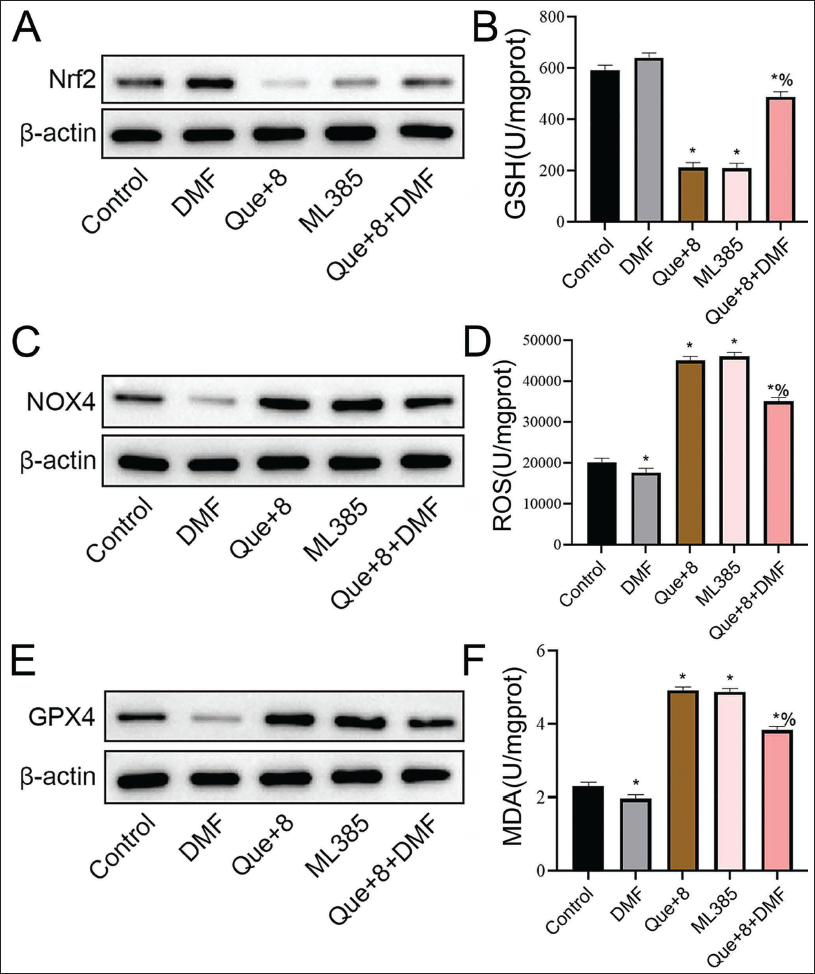
Using the Nrf2 inhibitor ML385 and the Nrf2 agonist DMF to intervene in MHCC97-H cells, it was found that the levels of Nrf2 and GSH in the Que+8 group and ML385 group showed a significant downward trend (Figure 3A and 3B), and the content of NOX4 and ROS increased significantly (vs. the control group, Figure 3C and 3D). The changing trend of the DMF group was opposite to that of the Que+8 group and ML385 group (vs. control group, Figure 3A–3D), while the Que+ expression levels of Nrf2 and GSH in cells in the 8+DMF group increased (vs. the Que+8 group, Figure 3A and 3B). The NOX4 and ROS contents decreased (vs. the Que+8 group, Figure 3C and 3D). Further observation of the contents of GPX4 protein and MDA found that the expression of GPX4 protein in the Que+8 group and the ML385 group decreased (vs. the control group, Figure 3E), while the MDA level increased significantly (vs. the control group, Figure 3F). The DMF group showed an opposite trend (Figure 3E and 3F). Further addition of DMF based on the Que+8 intervention found that the expression level of GPX4 increased (vs. the Que+8 group, Figure 3E), and MDA decreased (vs. the Que+8 group, Figure 3F). It shows that quercetin can downregulate the GSH and ferroptosis-related protein GPX4 levels by inhibiting the Nrf2 pathway, causing the ROS content to increase, increasing MDA expression, and ultimately inducing ferroptosis. In addition, this process is related to the expression of NOX4.
Ferroptosis Further Aggravates Cellular Oxidative Stress Response
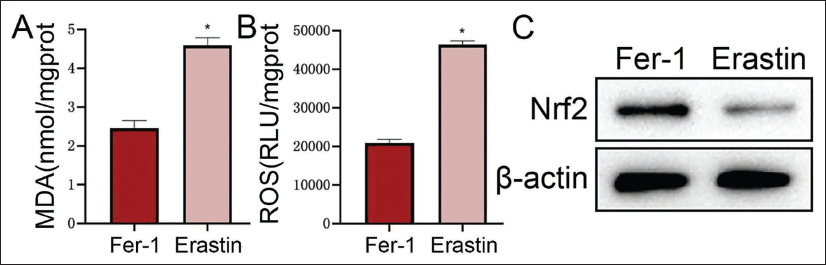
Fer-1 (ferroptosis-specific inhibitor) and Erastin (ferroptosis-specific agonist) were added to each group of MHCC97-H cells, respectively. Compared with the Fer-1 group, the MDA and ROS levels of the Erastin group are lower. The levels showed an increasing trend (Figure 4A and 4B), while the Nrf2 level was significantly reduced (vs. the Fer-1 group, Figure 4C). This shows that ferroptosis can further upregulate the oxidative stress response and affect the expression level of Nrf2.
Discussion
According to literature reports, HCC is a malignant tumor in China, and its incidence rate is increasing year by year (Sidali et al., 2022). This study mainly explored the ferroptosis-inducing effect of quercetin on liver cancer cells. Quercetin is a natural, non-toxic flavonoid compound with extensive functions such as antioxidant, anti-apoptosis, and anti-cancer (Cui et al., 2022; Georgiou et al., 2023), and quercetin has been proven to induce tumor cell apoptosis (Guo et al., 2021). In order to explore quercetin’s effect on ferroptosis in liver cancer cells, this study first set up different concentration gradients of quercetin to intervene in MHCC97-H liver cancer cells and observed their activities and apoptosis levels. The results showed that quercetin can decrease the proliferation activity of liver cancer cells to a certain extent and can cause apoptosis. At the same time, as the drug concentration increases, it shows concentration dependence. Therefore, we speculated that quercetin is beneficial for the treatment of liver cancer. We selected the highest concentration of Que+8 to further explore the mechanism of quercetin-induced apoptosis in tumor cells.
One of the antioxidant transcription factors, represented by Nrf2, can play a key role in inhibiting cellular oxidative stress by coordinating cellular protective factors (Wang, Zhang, et al., 2022). Research by Feng et al. (2019) pointed out that tanshinone IIA inhibits silica-induced oxidative stress by inhibiting NOX4 and activating Nrf2/ARE signaling, suggesting that the role of Nrf2 in regulating oxidative stress is closely related to NOX4. In the HCC model established by Palomino-Antolín et al. (2023), it was also found that NOX4 deletion stimulated Nrf2 to increase its activity and activated the myeloid tumor oncogene (Myc proto-oncogene, MYC) transcription to mediate mitochondrial dynamics and transcriptional programs that increased oxidative metabolism and promoted tumor progression. In tumor cells, quercetin inhibits the Nrf2 pathway (Tang et al., 2023) and can regulate ROS content in cells. In addition, knocking down the expression of Nrf2 inhibits the heme oxygenase-1 (HO-1)/GPX4 pathway protein, which can promote ferroptosis (Wang, Liao, et al., 2022; Yang et al., 2022). This study found that the levels of Nrf2 and GPX4 in liver cancer cells in the Que+8 group decreased significantly, while the NOX4 expression and ROS content increased significantly. This suggests that quercetin inhibits the Nrf2 pathway in liver cancer cells and can upregulate the oxidative stress level of liver cancer cells.
GPX4 is a ferroptosis marker protein, and its reduced expression can promote the occurrence of ferroptosis (Xue et al., 2023). Previous studies have also pointed out that the ferroptosis process induced by GPX4 ubiquitination in tumor cells is accompanied by the accumulation of large amounts of lipid peroxides, ROS, and the production of MDA (Zhang et al., 2022; Zhou et al., 2023). Therefore, the low-level expression of GPX4 under quercetin intervention in this study suggests, to a certain extent, that the apoptosis mechanism induced by quercetin may be related to ferroptosis induced by oxidative stress. GPX4 is GSH-dependent and can reduce lipid hydroperoxides to reduce the accumulation of ROS. The GSH/GPX4 pathway is an important way to inhibit ferroptosis (Li et al., 2022; Wang et al., 2023). In the study of Xu et al. (2022), it was found that GSH depletion, related to Nrf2 inactivation, can induce oxidative stress in liver tissue, leading to ferroptosis, and can be rescued by Fer-1. In order to further clarify whether quercetin induces ferroptosis in liver cancer cells via regulating cellular oxidative stress levels, this study added the Nrf2-specific inhibitor ML385 to liver cancer cells and found that levels of Nrf2, GSH, and GPX4 in the ML385 group were reduced, and NOX4, ROS, and MDA content were increased. The trend in the Que+8 group was the same as that in ML385. After further adding the Nrf2 agonist DMF to Que+8 for intervention, the effects of quercetin on various proteins and ROS in liver cancer cells were weakened. This suggests that quercetin can downregulate GPX4 expression by inhibiting the Nrf2 pathway, thereby promoting oxidative stress-induced ferroptosis. During the ferroptosis process, tumor necrosis factor-alpha (TNF-α) may be released, activating the p38 mitogen-activated protein kinase (MAPK) signaling pathway to generate a large amount of ROS, further exacerbating oxidative stress (Liu et al., 2023; Yang et al., 2023). Research by de Souza et al. (2022) found that silencing NRF2 in T98G human glioblastoma cells reduced cell viability after Temozolomide (TMZ) treatment, upregulated cellular GSH levels, and resisted ferroptosis, suggesting that NRF2/GSH is involved in ferroptosis; it also observed that after intervention of the ferroptosis inducer Erastin, the cystine-glutamate anti-porter system (Xc-system) was blocked and GSH was depleted, suggesting that, to a certain extent, ferroptosis induction can inhibit the Nrf2 pathway. In this study, it was also observed that under the intervention of Erastin, the MDA content of cells increased, inducing ferroptosis, and the ROS content in liver cancer cells increased, while the Nrf2 level decreased. This shows that the activation of ferroptosis can cause the accumulation of ROS to reverse regulate the body’s oxidative stress levels, inhibit the activity of the Nrf2 pathway, form a virtuous cycle, and further promote the apoptosis of liver cancer cells. Although we have explored the mechanism in vivo, whether we can show the same results in vitro needs more verification. Next, we will elaborate on the model mice.
Conclusion
In summary, quercetin can accumulate MDA and ROS in liver cancer cells through the Nrf2 pathway, induce ferroptosis, and thereby increase apoptosis. This process is related to the decrease in GSH and GPX4 protein expression and the increase in NOX4 expression. It can be seen that there is a close relationship between quercetin and ferroptosis in liver cancer cells, and it has great potential for treating HCC. However, the specific mechanism of action of quercetin on Nrf2 is not clear. Moreover, this study is still in the basic research stage and is only discussed at the cellular level. The pathological changes in animal models have not yet been observed. This is also a shortcoming of this study. Future research will further construct animal models and normal liver cells. The model improves the results of this study.
Footnotes
Abbreviations
GPX4: Glutathione peroxidase 4; GSH: Glutathione; NOX4: NADPH oxidase 4; Nrf2: Nuclear factor erythroid-related factor 2; ROS: Reactive oxygen species.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the ethics committee of the Second Affiliated Hospital of Nanchang University.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
