Abstract
The aim of the present study was to investigate the potential oxidative damage of di-(2-ethylhexyl) phthalate (DEHP) in the rat testis and to further elucidate the potential modulatory effect of quercetin. DEHP was diluted in corn oil and given to rats by oral gavage at doses 0, 300, 600, and 900 mg/kg/day (groups I, III, IV, or V, respectively) for 15 consecutive days. Group VI was pretreated with quercetin (90 mg/kg), 24 h before starting the experiment and then treated with DEHP (900 mg/kg/day) for 15 consecutive days. Group II was treated with quercetin (90 mg/kg/day). The relative testes weight and sperm motility were significantly decreased by treatment with 900 mg/kg of DEHP. Both sperm count and daily sperm production were significantly decreased by DEHP treatment at doses of 600 and 900 mg/kg. Serum testosterone level and prostatic acid phosphatase (ACP) activity and testicular lactate dehydrogenase-X (LDH-X) activity were significantly decreased in animals treated with 900 mg/kg. Serum total ACP activity was significantly increased in animals treated with 600 and 900 mg/kg of DEHP. DEHP treatment induced oxidative stress and histopathological abnormality. These abnormalities were effectively normalized by pretreatment with quercetin except for LDH-X near normalcy. In conclusion, the findings of this study demonstrate that DEHP impairs testicular function at least, in part, by inducing oxidative stress and quercetin has a potent protective effect against DEHP-induced testicular toxicity in rats.
Introduction
Phthalic esters are compounds widely used as plasticizers. About 3 million tons of phthalic esters per year are produced worldwide and they can be found in many everyday products, such as PVCs, plastic bags, food packaging, cosmetics, industrial paints as well as blood transfusion packs. 1 Due to the non-covalent nature of their link with plastics, phthalates can easily leach from these products and, consequently, be ingested. 2 Very large proportion of the literature on the endocrine disruptors categorized as anti-androgens deals with phthalates. 3 Di-(2-ethylhexyl) phthalate (DEHP) is currently the most commonly used phthalate plasticizer. 4 After exposure, DEHP is rapidly hydrolyzed by esterases in the gut, liver, and blood into mono-(2-ethylhexyl) phthalate (MEHP), which is believed to be the active molecule. 5
Numerous studies have been performed to assess the effects of phthalates, principally on the male reproductive functions. Exposure to phthalate esters results in inhibition of testicular testosterone synthesis associated with the development of abnormalities of the reproductive tract of fetal and prepubertal rat. In postpubertal rats, phthalates were also found to decrease sperm production. 6 –8 DEHP and its main metabolite, MEHP have been deleterious on the male reproductive system of the neonatal, prepubertal, and adult animals and induced dramatic changes in germ cells. 9,10 Although the induction of oxidative stress may represent a common mechanism in endocrine disruptor-mediated dysfunction, especially on testicular cells, 11 recent studies are also providing supporting evidences for such an effect with DEHP . 12 –14
Therapeutic strategies aimed at preventing or delaying generation of reactive oxygen species (ROS) might be a reasonable choice against DEHP-induced toxicity. 15 Dietary intake of antioxidants is a plausible and effective way to augment and fortify endogenous defense systems, since many antioxidants can act as free radical scavengers resulting in cytoprotection. 16 Quercetin, one of the most widely distributed flavonoids in plants including various vegetables, fruits, and herb medicine, is generally regarded as a prominent ROS scavenger. 17 Quercetin offers protection against oxidative stress and cell death induced by a variety of insults in many types of cells. 18 Quercetin has been reported to protect testicular cells from oxidative damage induced by environmental chemicals. 19
Concern has increased regarding the adverse effects of DEHP on reproduction. 20 However, the mechanism by which phthalates and particularly DEHP exerts toxic effects in reproductive system are not yet fully elucidated. Therefore, the aim of the present study was to investigate the potential oxidative damage of DEHP in the rat testis and to further elucidate the potential modulatory effect of quercetin against this environmental pollutant.
Materials and methods
Chemicals
DEHP, quercetin, and DL-α hydroxycaproic acid were purchased from Sigma-Aldrich Chemical Company (St Louis, MO, USA). All other chemicals are of analytical grade.
Animals and treatment
Sixty adult male Wistar rats (220 ± 10 g) were housed in clean polypropylene cages and maintained on a 12-h light/12-h dark cycle and a temperature of 20–25°C with ad libitum access to food and water.
The animals were divided into six groups. Group I served as control receiving corn oil through the experiment. Group II received quercetin (90 mg/kg) suspended in corn oil and administered by oral gavage through the experiment. DEHP was diluted in corn oil and given to rats by oral gavage at doses 300, 600, or 900 mg/kg/day for 15 consecutive days (groups III, IV, and V, respectively). Group VI was pretreated with quercetin (90 mg/kg), 24 h before starting the experiment and then treated with DEHP (900 mg/kg/day) for 15 consecutive days. The doses and duration of treatment of DEHP and quercetin were selected as per previous publications. 21 –23 This study involving experimental animals was conducted in accordance with national and institutional guidelines for the protection of animal welfare.
Necropsy
Twenty-four hours after the last dose, the animals’ body weights were recorded and blood samples were collected from the retro-orbital sinus, under ether anesthesia, and allowed to clot at room temperature. Samples were centrifuged and supernatant serum was separated from the clot as soon as possible and stored at −80°C until total acid phosphatase (ACP) (TACP) and prostatic ACP (PACP) and testosterone assay. The animals were euthanized under anesthetic ether. The testes were removed, cleaned from adhering fat and connective tissues and weighed in grams. The relative testes weight (weight of testes (g)/body weight (g) × 100) was calculated. The cauda epididymides were used for sperm count and motility. One testis from each rat (the right) was processed for evaluation of daily sperm production (DSP), whereas the left one was homogenized in ice-cold 0.15 M KC1 (10% w/v). The homogenate was centrifuged at 9000g for 10 min at 4°C and the supernatant immediately collected and processed for biochemical studies and enzyme activity assays. One testis from each group was processed for histopathological examination. 24 Protein concentrations were determined using a bicinchoninic acid kit (Pierce, Rockford, Illinois, USA) that employed bovine serum albumin as a standard.
Sperm count and motility
Epididymides were dissected out, immediately minced in 5 ml of physiological saline and then incubated at 37°C for 30 min to allow spermatozoa to leave the epididymal tubules. The percentage of motile sperm was recorded using a phase contrast microscope at a magnification of 400×. Total sperm number was determined using a Neubauer hemocytometer as previously described. 25 To determine sperm motility, 100 sperms each were observed in three different fields, and classified into motile and nonmotile sperms, and the motility was expressed as percentage incidence.
Daily sperm production
DSP was determined as previously described. 26 The right testis was decapsulated and homogenized in 50 ml of ice-cold 0.9% sodium chloride solution containing 0.01% Triton X-100 using a Polytron homogenizer (Kinematica, Inc., USA). 27 The homogenate was allowed to settle for 1 min and then was gently mixed, and a 10 ml aliquot was transferred to a glass vial and stored on ice. After thorough mixing of each sample, the number of sperm heads (step 19 spermatid head) in four chambers of Neubauer type hemocytometer was counted under a light microscope with 40× objective. To calculate DSP, the number of spermatids at stage 19 was divided by 6.1, which is the number of days of the seminiferous cycle in which these spermatids are present in the seminiferous epithelium.
Serum testosterone
Testosterone was measured using the Pathozyme Testosterone ELISA kit (DRG international, Inc., USA). Briefly, standards, specimens, and controls were dispensed into appropriate wells, followed by testosterone horseradish peroxidase reagent and anti-testosterone reagent, before mixing thoroughly and incubating at 37°C for 90 min. Wells were then rinsed with deionized water and substrate solution was dispensed into each well, gently mixed, and incubated for 20 min. The reaction was stopped with the “stop reagent” and the absorbance recorded at 450 nm. 28
Serum TACP and PACP activity
α-Naphthylphosphate is hydrolyzed by ACP to phosphate and α-naphthol, which is converted with fast red TR salt into azodye. The increase in the absorbance of the azodye at 405 nm is proportional to TACP activity in the sample. PACP reaction can be blocked by tartarate and determined indirectly (through the non-prostatic ACP) by calculating the difference in the activities. 29
Testicular LDH-X
Testicular lactate dehydrogenase-X (LDH-X) activity was measured using α-ketovaleric acid as the substrate. 30 The activity of LDH-X is expressed as micromoles of NADH oxidized per minute per milligram protein.
Testicular oxidative stress status
The cellular lipid peroxidation (LPO) products within the testis were determined using thiobarbituric acid reactive substances during an acid-heating reaction. The samples were diluted by 1.5 ml trichloroacetic acid (20% w/v) and centrifuged at 3000g for 10 min. Then, the precipitation was dissolved in sulfuric acid and 1.5 ml of the mixture was added to 1.5 ml of thiobarbituric acid (TBA; 0.2% w/v). The mixture was then incubated for 1 h in a boiling water bath. Following incubation, 2 ml of n-butanol was added. The solution was centrifuged, cooled, and the absorption of the supernatant was recorded at 532 nm. The calibration curve of tetraethoxypropane standard solutions was used to determine the concentrations of TBA–malondialdehyde adducts in samples. 31
Superoxide dismutase (SOD) was assayed by the method of Marklund and Marklund. 32 Briefly, the assay mixture contained 2.4 ml of 50 mM tris(hydroxymethyl)aminomethane–hydrochloric acid buffer containing 1 mM ethylenediaminetetraacetic acid (pH 7.6), 300 µl of 0.2 mM pyrogallol, and 300 µl testis homogenate. The decrease in absorbance was measured immediately at 420 nm against blank at 10 s intervals for 3 min on a spectrophotometer. Catalase (CAT) was assayed as previously mentioned. 33 Briefly, the assay mixture contained 2.40 ml of phosphate buffer (50 mM, pH 7.0), 10 µl of 19 mM hydrogen peroxide (H2O2), and 50 µl testis homogenate. The decrease in absorbance was measured immediately at 240 nm against blank at 10 s intervals for 3 min on a spectrophotometer. The level of reduced glutathione (GSH) was determined as previously described based on the reaction with Ellman’s reagent (19.8 mg dinitrothiocyanobenzene in 100 ml 0.1% sodium citrate). 34 The absorbance was recorded at 412 nm using a spectrophotometer. The GSH content was expressed as micrograms per milligram protein.
Statistical analysis
Differences between obtained values (mean ± SD, n = 10) were compared by one-way analysis of variance followed by the Tukey–Kramer multiple comparison test. Comparison is made between (i) control (i.e. group I) and other groups and (ii) group V and group VI. Differences were considered statistical significance from at *p < 0.05; **p < 0.01; ***p < 0.001.
Results
Relative testes weight and sperm parameters
Table 1 shows the changes in relative testes weight and sperm parameters. Treatment of male rats (group V) with DEHP (900 mg/kg) significantly decreased the relative testes weight (p < 0.05), while DEHP at doses 300 and 600 mg/kg did not show any significant change as compared to the corresponding control. Pretreatment with quercetin (group VI) reverted the relative testes weight to normalcy (p < 0.05).
Effect of DEHP and/or quercetin on relative testes weight and sperm parameters.

Group I: Control (vehicle), Group II: quercetin (90 mg/kg), Group III: DEHP (300 mg/kg/day), Group IV: DEHP (600 mg/kg/day), Group V: DEHP(900 mg/kg/day), Group VI: quercetin (90 mg/kg/day) + DEHP (900 mg/kg/day). DEHP: Diethylhexyl phthalate. Data are expressed as mean ± SD (n = 10). Comparison is made between: aGroup I and Groups II, III, IV, V, VI; bGroup V and Group VI by one way analysis of variance (ANOVA) followed by the Tukey-Kramer multiple comparison test. The symbol represents statistical significance from control: *p < 0.05; **p < 0.01; ***p < 0.001.
Sperm count was significantly decreased by 23.17 and 34.29% in response to treatment with DEHP (600 and 900 mg/kg) (groups IV and V, respectively), while DEHP at a dose of 300 mg/kg (group III) did not show any significant change in sperm count as compared to the corresponding control. DEHP at a dose of 900 mg/kg (group V) decreased sperm motility by 31.75%, while the other doses did not show any significant change (groups III and IV) as compared to the corresponding control. Moreover, DSP showed significant reduction by 26.43 and 42.81% in response to treatment with DEHP (600 and 900 mg/kg) (groups IV and V, respectively), while it did not show any significant change at a dose of 300 mg/kg (group III) of DEHP as compared to the corresponding control. Pretreatment with quercetin (group VI) reverted the values of sperm count, motility, and DSP to normalcy (p < 0.001, p < 0.05, and p < 0.001, respectively, as compared to group V).
Serum testosterone and ACP (total and prostatic)
Animals treated with DEHP (900 mg/kg; group V) showed significant decrease by 69.46% in serum testosterone content as compared to the related control (Figure 1). DEHP at doses 300 and 600 mg/kg did not show any significant change in testosterone level (groups III and IV). Quercetin treatment (group II) significantly increased testosterone content (p < 0.01) as compared to the corresponding control. Animals pretreated with quercetin (group VI) showed normal testosterone content (p < 0.001, as compared to group V).

Effect of DEHP and/or quercetin on serum testosterone. Group I: control (vehicle); group II: quercetin (90 mg/kg); group III: DEHP (300 mg/kg/day); group IV: DEHP (600 mg/kg/day); group V: DEHP (900 mg/kg/day); and group VI: quercetin (90 mg/kg/day) + DEHP (900 mg/kg/day). Data are expressed as mean ± SD (n = 10). Comparison is made between: (a) group I and groups II, III, IV, V, and VI; (b) group V and group VI by one-way analysis of variance followed by the Tukey–Kramer multiple comparison test. **p < 0.01: statistical significance from control; ***p < 0.001: statistical significance from control. DEHP: di-(2-ethylhexyl) phthalate.
TACP activity showed significant increase by 25.23 and 50.45% in response to DEHP (600 and 900 mg/kg, respectively) treatment as compared to the corresponding control (Figure 2). DEHP at a dose of 300 mg/kg did not show any significant change. Pretreatment with quercetin (group VI) normalized TACP content (p < 0.001, as compared to group V). Animals treated with 900 mg/kg of DEHP (group V) showed significant reduction by 42.86% in PACP content as compared to the corresponding control (Figure 3). DEHP at doses of 300 and 600 mg/kg did not show any significant change in PACP activity (groups III and IV). Pretreatment with quercetin (group VI) reverted PACP content toward normalcy (p < 0.05, as compared to group V).
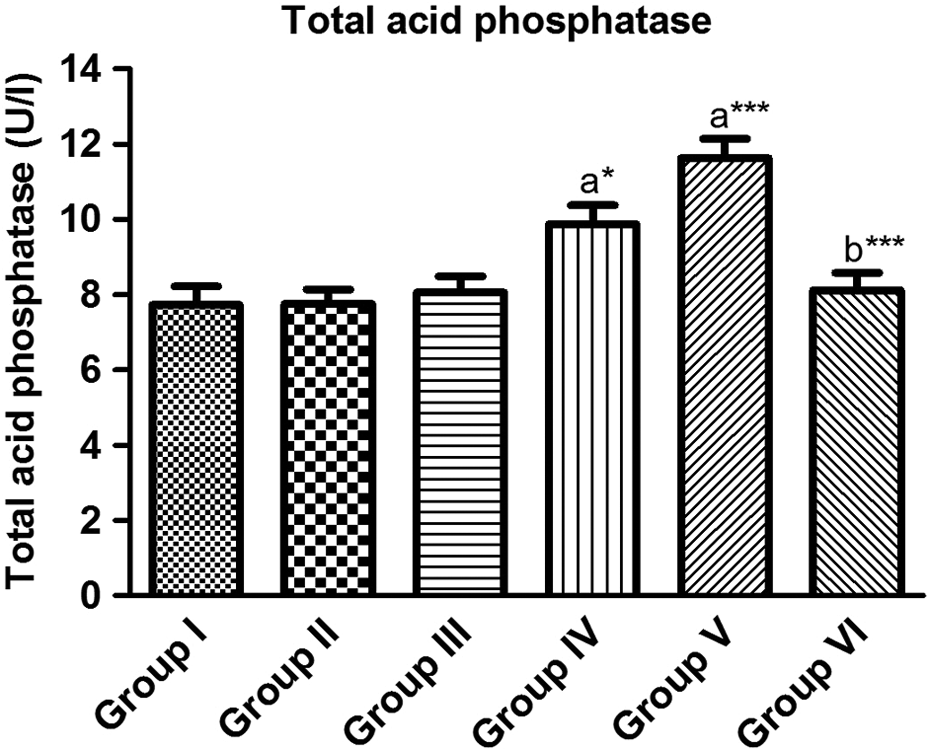
Effect of DEHP and/or quercetin on serum TACP. Group I: control (vehicle); group II: quercetin (90 mg/kg); group III: DEHP (300 mg/kg/day); group IV: DEHP (600 mg/kg/day); group V: DEHP (900 mg/kg/day); and group VI: quercetin (90 mg/kg/day) + DEHP (900 mg/kg/day). Data are expressed as mean ± SD (n = 10). Comparison is made between: (a) group I and groups II, III, IV, V, and VI; (b) group V and group VI by one-way analysis of variance followed by the Tukey–Kramer multiple comparison test. *p < 0.05: statistical significance from control; ***p < 0.001: statistical significance from control. TACP: total acid phosphatase; DEHP: di-(2-ethylhexyl) phthalate.

Effect of DEHP and/or quercetin on serum PACP. Group I: control (vehicle); group II: quercetin (90 mg/kg); group III: DEHP (300 mg/kg/day); group IV: DEHP (600 mg/kg/day); group V: DEHP (900 mg/kg/day); and group VI: quercetin (90 mg/kg/day) + DEHP (900 mg/kg/day). Data are expressed as mean ± SD (n = 10). Comparison is made between: (a) group I and groups II, III, IV, V, and VI; (b) group V and group VI by one-way analysis of variance followed by the Tukey–Kramer multiple comparison test. *p < 0.05: statistical significance from control; **p < 0.01: statistical significance from control. PACP: prostatic acid phosphatase; DEHP: di-(2-ethylhexyl) phthalate.
Testicular LDH-X
Treatment with DEHP at a dose of 900 mg/kg significantly decreased LDH-X activity by 44.87% (group V), while at doses of 300 and 600 mg/kg, DEHP did not show any significant change (groups III and IV) as compared to the corresponding control (Figure 4). Pretreatment with quercetin (group VI) ameliorated LDH-X activity toward normalcy but did not reach normal level (p < 0.05, as compared to the corresponding control).

Effect of DEHP and/or quercetin on testicular LDH-X activity. Group I: control (vehicle); group II: quercetin (90 mg/kg); group III: DEHP (300 mg/kg/day); group IV: DEHP (600 mg/kg/day); group V: DEHP (900 mg/kg/day); and group VI: quercetin (90 mg/kg/day) + DEHP (900 mg/kg/day). Data are expressed as mean ± SD (n = 10). Comparison is made between: (a) group I and groups II, III, IV, V, and VI; (b) group V and group VI by one-way analysis of variance followed by the Tukey–Kramer multiple comparison test. *p < 0.05: statistical significance from control; ***p < 0.001: statistical significance from control. DEHP: di-(2-ethylhexyl) phthalate.
Oxidative stress status
As shown in Table 2, animals treated with 900 mg/kg of DEHP (group V) showed increase in LPO by 66.4%, and reduction in the activities of SOD and CAT by 39.24 and 69.84%, respectively (group V), as compared to the corresponding control. DEHP at doses 300 and 600 mg/kg (groups III and IV) did not show any significant change in LPO or in the activity of either SOD or CAT. Pretreatment with quercetin (group VI) significantly suppressed (p < 0.001, as compared to group V) LPO. Pretreatment with quercetin (group VI) also reverted the reduction in SOD and CAT activities induced by treatment with DEHP (900 mg/kg/day) to normal value. GSH level was significantly decreased by 33.54 and 40.37% in response to DEHP treatment (600 and 900 mg/kg; groups IV and V, respectively) as compared to the related control. Pretreatment with quercetin (group VI) normalized (p < 0.01, as compared to group V) GSH level. Notable, animals treated with quercetin (group II) showed significant increase (31.06%) in GSH level as compared to the related control.
Effect of DEHP and/or quercetin on oxidative stress status.
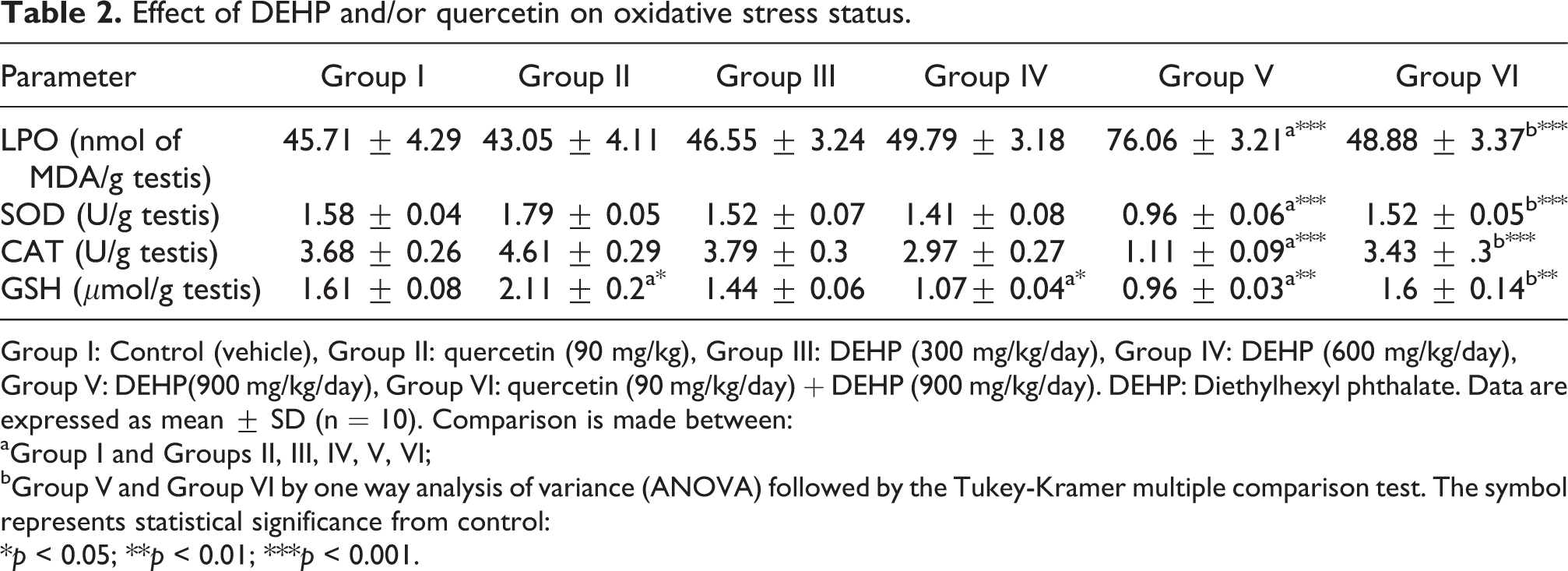
Group I: Control (vehicle), Group II: quercetin (90 mg/kg), Group III: DEHP (300 mg/kg/day), Group IV: DEHP (600 mg/kg/day), Group V: DEHP(900 mg/kg/day), Group VI: quercetin (90 mg/kg/day) + DEHP (900 mg/kg/day). DEHP: Diethylhexyl phthalate. Data are expressed as mean ± SD (n = 10). Comparison is made between: aGroup I and Groups II, III, IV, V, VI; bGroup V and Group VI by one way analysis of variance (ANOVA) followed by the Tukey-Kramer multiple comparison test. The symbol represents statistical significance from control: *p < 0.05; **p < 0.01; ***p < 0.001.
Histopathological examination
Specimens from control animals showed normal seminiferous tubules and spermatogenesis (Figure 5(a)). Similarly, quercetin showed virtually almost the same histologic architecture as control animal (Figure 5(b)). DEHP, however, induced degeneration of spermatogonial cells in some seminiferous tubules (300 mg/kg; Figure 5(c), arrow), degeneration and necrosis with pyknosis in the nuclei of spermatogonial cells and spermatids (600 mg/kg; Figure 5(d)), and azospermia in some seminiferous tubules (900 mg/kg) (Figure 5(e)). Prior administration of quercetin a head of DEHP challenge apparently ameliorated the histologic alterations in the seminiferous tubules compared to DEHP-treated rats (Figure 5(f)).

Representative illustrations of histological morphology of rat testes. (a) Testicular cross section from control rats showing normal seminiferous tubules and spermatogenesis (H and E, ×40). (b) Testicular cross sections from quercetin-treated rats showing normal histologic architecture of seminiferous tubules and spermatogenic stages (arrow) (H and E, ×40). (c) Testicular cross sections from DEHP (300 mg/kg/day)-challenged rats showing degeneration of spermatogonial cells (arrow) in some seminiferous tubules (H and E, ×40). (d) Testicular cross sections from DEHP (600 mg/kg/day)-treated rats showing degeneration and necrosis with pyknosis in the nuclei of spermatogonial cells and spermatids (arrow) (H and E, ×160). (e) Testicular cross sections from DEHP (900 mg/kg/day)-challenged rats showing azospermia in some seminiferous tubules (arrow) (H and E, ×80). (F) Testicular cross sections from rats pretreated with quercetin prior to DEHP (900 mg/kg/day) challenge showing normal intact histological structure of the seminiferous tubules with complete spermatogenic series in the lumen (arrow) (H and E, ×40). H and E: hematoxylin and eosin; DEHP: di-(2-ethylhexyl) phthalate.
Discussion
The significant decrease in relative testis weight observed in this study, after treatment with 900 mg/kg of DEHP, may be explained by the significant decrease in absolute testes weight and unchanging of body weight. The weight of the testis is largely dependent on the mass of the differentiated spermatogenic cells, and the reduction in the weight of the testis may be due to decreased number of germ cells, inhibition of spermatogenesis and steroidogenic enzyme activity. 35 The testicular sperm counts and DSP are important indicators of spermatogenesis. 36 In this study, administration of DEHP reduced sperm count and motility and DSP. Sperm count and motility correlated with testis weight and plasma testosterone concentration, so that the motility of sperm was only affected in rats treated with 900 mg of DEHP. This might be due to the low testosterone level in these animals. Another explanation is that when sperm production is drastically reduced, fewer sperm pass from the testis to the cauda, resulting in decreased spermatozoa in the cauda epididymis. The observed decrease in the epididymal spermatozoa numbers may therefore indicate that DEHP affects the early stages of spermiogenesis, which is well supported by the observed decrease in daily spermatozoa production. These results come in accordance with 37 who showed an obvious reduction in the total sperm count and number of sperm heads after DEHP treatment. Moreover, disturbed testicular histology and spermatogenesis, diminished testosterone, and sperm motility were reported after DEHP treatment. 38 Further, Ge et al. 39 and Noriega et al. 9 reported that the mechanisms by which DEHP exerts its toxic effects in reproductive system is related to its anti-androgenic potential. However, pretreatment with quercetin ameliorated the DEHP-induced sperm toxicity by maintaining the sperm quality near normalcy.
The testis is an androgen-dependent organ and testosterone is essential to maintain the structure and function of the testis and accessory sex glands. 40 It is interesting to note that decreased testosterone secretion would be a result of oxidative stress that is related to insufficiency in the activity of key enzymatic and nonenzymatic antioxidants inside Leydig cells. 41 Available data suggest that Leydig cells are one of the main targets of phthalates. 39 Leydig cells are the primary source of testosterone production in males, and differentiation of Leydig cells in the testes is one of the primary events in the development of the male body and fertility. 42
ACP is considered to be functional indicator of spermatogenesis. The increased activity of TACP in DEHP-administered animals reflects the release of this phosphatase from the lysosomes of the degenerating cells and rapid catabolism of the injured germ cells. 43 The decreased activity of ACP in the prostate might be due to the increased permeability of plasma membrane or cellular necrosis, thereby showing the stress condition of the treated animals. 44
LDH-X is one of the best characterized germ cell-specific isozyme that plays an important role in the process of spermatogenesis and has been shown to be vital for sperm survival and motility. 45 LDH-X is the predominant isozyme in the pachytene spermatocyte and round and condensing spermatids, whereas spermatozoa contain only LDH-X. 46 The significant decrease of LDH-X activity, a consequence of enhanced LPO in DEHP-treated animals, may be due to disintegration of the mitochondrial membrane ultrastructure which in turn affects the membrane-bound LDH-X function. LDH-X plays an important role in transferring hydrogen from cytoplasm to mitochondria by redox-coupling α-hydroxy acid/α-keto acid related to spermatozoal metabolism. 47 This could be one of the contributory factors leading to reduced male sperm concentration and sperm motility on DEHP exposure. 48 The reduction in LDH-X activity in rats treated with 900 mg/kg indicates a suppression of essential sperm maturational processes that precede the penetration of the oocyte by the sperm, such as capacitation and acrosome reaction, which can result in decline of fertility. 49 The alteration in these enzymes activity may lead to destruction of seminiferous epithelium and loss of germinal elements, resulting in reduction in the number of spermatids associated with decrease in the DSP in the testes. 50 The results obtained in this study indicate that pretreatment with quercetin influences the activities of these enzymes to an appreciable extent and suggests the cytoprotective action of quercetin in preventing DEHP-induced testicular damage.
Oxidative stress has been implicated as a major causative factor in male reproductive dysfunction. 51 Studies showed that there is a correlation between reproductive toxicity induced by environmental contaminants and oxidative stress. 52 Previous data have also shown that phthalates were able to produce free radicals by several pathways in germ cells, suggesting the possibility that oxidative stress and mitochondrial dysfunction in germ cells may contribute to phthalate-induced disruption of spermatogenesis. 53,54 DEHP treatment, indeed, was reported to provoke oxidative stress as measured by increases in ROS in subsequently isolated rat spermatocytes 10 and in MA-10 mouse Leydig tumor cell line. 55 Thus, at least one of the mechanisms underlying the reproductive toxicity of DEHP might be the induction of intracellular ROS and/or to cause alterations on intracellular enzymatic and nonenzymatic antioxidants, thereby to produce oxidative stress. The spermatozoa membranes are rich in polyunsaturated fatty acids, so they are susceptible to ROS attack and LPO. 56 LPO causes membrane damage that leads to a decrease in sperm motility, presumably by a rapid loss of intracellular ATP, and an increase in sperm morphology defects. 57 Antioxidants play a major role by continuously inactivating ROS to keep only a small amount necessary to maintain normal cell function. 58 In the present study, the activities of testicular SOD and CAT were significantly decreased in rats treated with 900 mg/kg of DEHP. The existence of a mutually supportive relationship between enzymatic antioxidants, SOD and CAT, against accumulation of ROS inactivates the superoxide anion and peroxide radicals by converting them into water and oxygen. In this study, the observed decrease in SOD activity suggests inactivation of the enzyme possibly due to increased superoxide radical production or an inhibition by the H2O2 as a result of corresponding decrease in the activity of CAT, which selectively degrades H2O2. 59 For more explanation, significant depletion of the testicular antioxidant enzymes and overproduced toxic free radicals leads to a more pro-oxidant environment which per se can significantly increase the susceptibility of spermatogenic cells to oxidative stress. 40 In the present study, DEHP (600 and 900 mg/kg) administration depleted GSH content in testicular tissues that made spermatogenic cells more susceptible to oxidative damage, especially during increased free radical production. 60 GSH is an important intracellular antioxidant that spontaneously neutralizes several ROS.
Quercetin is reported to ameliorate the carbon tetrachloride induced oxidative toxicity in rat testis. 61 Also, cytotoxicity in Sertoli-germ cell cultures due to atrazine is countered by quercetin intervention. 19 Similarly, oxidative damage induced by diethylstilbestrol to hamster spermatogenic cells is also prevented by simultaneous treatment with quercetin. 62 Our study showed that quercetin significantly reduced LPO and provided protective effect against testicular oxidative damage in testes of animals treated with DEHP. The testicular toxicity induced by DEHP is further confirmed by the abnormal histologic findings. DEHP-treated rats showed degeneration of spermatogonial cells in some seminiferous tubules (300 mg/kg), degeneration and necrosis with pyknosis in the nuclei of spermatogonial cells and spermatid (600 mg/kg), and azospermia in some seminiferous tubules (900 mg/kg). These results suggest that DEHP could induce injury of spermatogonia and spermatids, and DEHP can decrease spermatogenesis by adversely affecting spermatogonia followed by depletion of spermatids and spermatozoa. Quercetin-pretreated animals showed normal intact histological structure of the seminiferous tubules with complete spermatogenic series in the lumen, thereby highlighting its protective role in countering the testicular toxicity induced by DEHP.
In conclusion, the findings of this study demonstrate that DEHP impairs testicular function at least, in part, by inducing oxidative stress, and quercetin has a potent protective effect against DEHP-induced testicular toxicity in rats.
Footnotes
Acknowledgments
The authors thank Dr Adel M. Bakeer, Prof. of Pathology, Faculty of Veterinary Medicine, Cairo University, for his help in performing the histopathological examination. The authors acknowledge with thanks Faculty of Pharmacy, Al-Azhar University, for technical and financial support.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
