Abstract
Background
Spinal tuberculosis involves many complex mechanisms. Motherwort has anti-inflammatory, antioxidant, and anti-tumor activities, but its effect on spinal tuberculosis is unclear.
Objectives
In this study, motherwort was prepared to explore the therapeutic effect and regulatory mechanism of this nano-complex on spinal tuberculosis.
Materials and Methods
0.1 mL of H37Rv standard strain suspension (concentration: 108 CFU/mL) was used to establish a spinal tuberculosis model. The model mice were orally administered 50 mg kg–1 Ajugol to form the Ajugol group, and the same doses of mesoporous silica nanoparticle (MSN) and Ajugol were orally administered to form the Ajugol group. After continuing to feed for 8 weeks, four groups of mice were used for experiments. To analyze whether the mechanism of Ajugol in treating spinal tuberculosis is related to its regulation of phosphoinositide 3-kinase/protein kinase (PI3K/Akt) pathway activity through microRNA-432-5p (miR-432-5p).
Results
No mice died during the modeling period of this study, and the modeling success rate was 100%. An animal model of spinal tuberculosis was successfully established. The bone tissue pathology, bone trabecular number, and thickness of mice were found to be affected by Ajugol and can delay the progression of spinal tuberculosis. This process is related to changes in miR-432-5p and the PI3K/Akt pathway. Under the intervention of Ajugol+miR-432-5p mimic, the number and thickness of bone trabeculae were significantly increased. The above phenomenon was significantly reversed after combining Ajugol with the miR-432-5p inhibitor. Verification test of PI3K/Akt pathway using LY294002 and 740 Y-P further confirmed that Ajugol inhibits the PI3K/Akt pathway by promoting miR-432-5p expression.
Conclusion
Ajugol promotes the high expression of miR-432-5p and inhibits the PI3K/Akt pathway, thereby reducing the activity of this pathway, inhibiting inflammatory responses, reducing cell proliferation and survival, and thus helping to treat spinal tuberculosis, which is essential for clinical practice and has a potential positive impact on public health. Further future research may focus on developing drugs or new treatments.
Introduction
Spinal tuberculosis is a disease caused by infection with Mycobacterium tuberculosis and mainly affects the spinal bones and adjacent soft tissues (Ruparel et al., 2022). Spinal tuberculosis often causes patients to experience severe back pain, especially at night or during activity (Wang et al., 2021). If left untreated, the pain may gradually worsen, causing patients to be limited in their activities of daily living, and may even lead to disability (Na et al., 2023). Effective administration of anti-tuberculosis drug regimens can kill or inhibit the growth of pathogens, thus preventing the spread of infection (Wang, Gao et al., 2020). It also helps relieve compression on nerve structures and prevent nerve damage, thereby reducing the risk of neurological symptoms and complications. However, it has serious side effects and may cause permanent damage to the body, so it is very important to find new treatment approaches (Yong et al., 2021).
Studies have shown (Sun et al., 2023) that in spinal tuberculosis, there may be regulatory mechanisms related to the phosphoinositide 3-kinase/protein kinase (PI3K/Akt) pathway, which affects the survival and apoptosis of infected cells. Some studies have pointed out (Wang, Hu et al., 2022) the PI3K/Akt pathway may regulate the proliferation, differentiation, and bone reconstruction of bone cells, thus affecting the bone damage of spinal tuberculosis, so it may become a potential target for drug treatment. It should be noted that although the role of the PI3K/Akt pathway in spinal tuberculosis has been extensively studied, the specific regulatory mechanisms and mechanisms of action are still a complex area and require further experimental and clinical studies to understand in depth.
MicroRNA (miRNA) has received increasing attention in the research on spinal tuberculosis, and it regulates gene expression (Bahreini et al., 2021). In spinal tuberculosis, miR-155 was found to be upregulated in tuberculosis infection and may regulate the immune response of macrophages and T cells (Hawez et al., 2019). In addition, some miRNAs may affect the survival and apoptosis of alveolar macrophages and other cells by regulating target genes, thereby affecting the infection and growth of M. tuberculosis (Hill & Tran, 2021). The PI3K/Akt signaling pathway plays a crucial role in various biological processes such as cell survival, proliferation, differentiation, and apoptosis, particularly in the nervous system, where it significantly impacts neuronal survival and synaptic plasticity. MicroRNA-432-5p (miR-432-5p), a type of small noncoding RNA molecule, participates in gene regulatory networks by targeting mRNA transcription and inhibiting its expression. Given the importance and wide range of functions of the PI3K/Akt pathway, miRNA can directly or indirectly affect key components of this pathway. Other studies have shown (Li, Zhou et al., 2022) that miR-432-5p can affect the activation status of the pathway through targeted regulation of key molecules such as PI3K and Akt in the PI3K/Akt pathway. miR-432-5p in certain tumors is associated with the activity of the PI3K/Akt pathway (Zhang et al., 2021). On the other hand, miR-432-5p and the PI3K/Akt pathway may be involved in neurological diseases. However, the role of miR-432-5p and the PI3K/Akt pathway in spinal tuberculosis is unclear.
Motherwort is one of the active ingredients extracted from traditional Chinese medicine. Studies have shown (Wang, Zeng et al., 2020) that motherwort glycosides may have the ability to inhibit inflammatory responses, thereby alleviating inflammatory symptoms by regulating inflammatory cytokines and signaling pathways. Moreover, motherwort may have a protective effect on bones, possibly by promoting the proliferation and differentiation of bone cells, thereby promoting the growth and repair of bone tissue (Pan et al., 2020). In addition, motherwort glycosides may also affect the function of the immune system, possibly affecting the immune response by regulating the activity of immune cells and the production of cytokines (Hua & Zhang, 2019). Although motherwort can treat many diseases, such as inflammation and immunity (Gisbert-Garzaran et al., 2020), its mechanism of action in treating spinal tuberculosis needs further exploration. Encapsulating motherwort in mesoporous silica nanomaterials has some potential advantages and applications (Escriche-Navarro et al., 2022). Moreover, motherwort can enhance the stability of the drug and make the drug better distributed and absorbed in the body, thereby improving the efficacy and duration of the drug and the bioavailability of the drug (Laskowski et al., 2019). This study explored the potential of motherwort to treat spinal tuberculosis by regulating the PI3K/Akt pathway through miR-432-5p. This study aims to reveal the synergistic effect of Ach, miR-432-5p, and PI3K/Akt pathways to slow the progression of spinal tuberculosis and its complex molecular mechanisms, and open up new avenues for more effective and personalized treatment.
Materials and Methods
Experimental Materials
Motherwort (Ajugol, specification: 20 mg, purity: high-performance liquid chromatography (HPLC) ≥98%, molecular formula: C42H72O14, purchased from Beijing ETA, molecular structure shown in Figure 1), trap staining kit (Hubei Aimeijie), LY294002 and 740 Y-P (Shanghai Yuanye), interleukin (IL)-6, IL-17, IL-1β, tumor necrosis factor-alpha (TNF-α) antibodies (Wuhan Huamei), miR-432-5p (Beijing Yuhengfeng Technology Ltd.).
Molecular Structural Formula of Motherwort.
Establishment and Grouping of Spinal Tuberculosis Animal Models
A spinal tuberculosis model was established with 0.1 mL of H37Rv standard strain suspension (concentration: 108 CFU/mL). Preparation method of strain suspension: Inject 10% chloral hydrate intraperitoneally at a dose of 3 mL/kg. After the mice are anesthetized, take the lateral decubitus position and shave the left quarter rib area of the mouse to prepare the skin. Insert the needle along the iliac crest perpendicular to the direction of the vertebral body. After the needle enters the vertebral body, push the needle. Inject 0.1 mL of mixed glue into the mouse vertebral body. The needle stays for 30 s. After the glue solidifies, rotate and pull out the needle.
The mice that were successfully modeled were included in the Model group. The model mice were orally administered 50 mg kg–1 Ajugol and designated as the Ajugol group. The model mice were administered the same dose of MSN and Ajugol and were designated as the MSN group and Ajugol–MSN group. After continuing to feed for 8 weeks, four groups of mice were used for experiments. In addition, set up miR-432-5p mimic, LY294002, Ajugol+miR-432-5p mimic, Ajugol+miR-432-5p inhibitor, Ajugol+LY294002, and Ajugol+740 Y-P group, and analyze Ajugol—whether the mechanism of MSN in treating spinal tuberculosis is related to its regulation of PI3K/Akt pathway activity through miR-432-5p. This study was approved by the Ethics Committee of The Affiliated Shuyang Hospital of Xuzhou Medical University.
Trap Staining to Detect Bone Tissue Pathology
Mouse bone tissue was fixed, decalcified, dehydrated, paraffin-embedded 48 h later, and then cut into sections (3 µm thickness). After rehydration, the sections were incubated with Trap culture medium for 1 h and stained with methyl green for 5 min. Finally, rinse the sections twice with pure water, perform gradient dehydration with 95% alcohol, use xylene as the cleaning agent, and seal the sections with transparent resin.
Peripheral Blood Enzyme-linked Immunosorbent Assay (ELISA) Detection of Inflammatory Factor Levels
Collect 0.5 mL of blood from the orbits of mice in each group, separate the plasma, add enzyme-labeled antibodies, react at 37°C for half an hour, add substrate solution at 100 µL/well at 37°C in the dark, and react for 5 min. Add the stop solution, develop the color, then add the reaction stop solution at 50 µL/well, and immediately detect the absorbed OD by ELISA (Wuhan Jinkarui).
Polymerase Chain Reaction (PCR) Detection of miR-432-5p and Western Blot Detection of PI3K/Akt Expression Levels
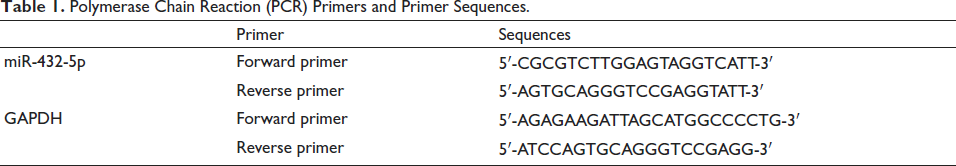
Grind and homogenize bone tissue using liquid nitrogen, extract total mRNA, and transcribe it into cDNA. The analysis used a real-time PCR system (Shanghai Unocal) with glyceraldehyde-3-phosphate dehydrogenase (GAPDH) as a reference. Use the 2–∆∆Ct method to estimate the relative level. Table 1 lists the miR-432-5p primers and primer sequences. The Western blot method was used to detect the phosphorylation level of p-PI3K and p-AKT. After the total protein was extracted from bone tissue and its concentration was determined by the Bradford method, it was electrophoretically separated on sodium dodecyl sulfate (SDS)–polyacrylamide gel and transferred to a polyvinylidene fluoride (PVDF) membrane. 5% skimmed milk was used to block the non-specific binding site in Tris-buffered saline with Tween 20 (TBST) buffer solution for at least 1 h, and rabbit anti-mouse p-PI3K, P-AKT, and GAPDH antibodies were incubated overnight at 4°C, with GAPDH used as an internal reference to standardize protein loading. The membrane was washed three times with TBST for 5 min each time to remove unbound primary antibodies, followed by the addition of horseradish peroxidase (HRP)-labeled goat anti-rabbit immunoglobulin G (IgG) secondary antibodies. After incubation at room temperature for 1 h, the membrane was washed three more times with TBST and developed using ECL chemiluminescent reagent. The band intensity was quantified using ImageJ software, and the relative expression levels of p-PI3K and p-AKT relative to GAPDH were calculated.
Polymerase Chain Reaction (PCR) Primers and Primer Sequences.
Western Blot Detection of IL-6, IL-17, IL-1β, and TNF-α Protein Expression Levels
Use TriZol reagent to extract total protein from bone tissue and detect its concentration, followed by separation on SDS–polyacrylamide gel for Western blot using rabbit anti-mouse IL-6, IL-17, IL-1β, TNF-α, and GAPDH antibodies.
Statistical Analysis
The data obtained in the above experiments were analyzed using SPSS 21.0 and GraphPad Prism software. If no special requirements exist, p < .05 is used as the test standard.
Results
Ajugol can delay the progression of spinal tuberculosis. This process may be related to changes in miR-432-5p and the PI3K/Akt pathway.
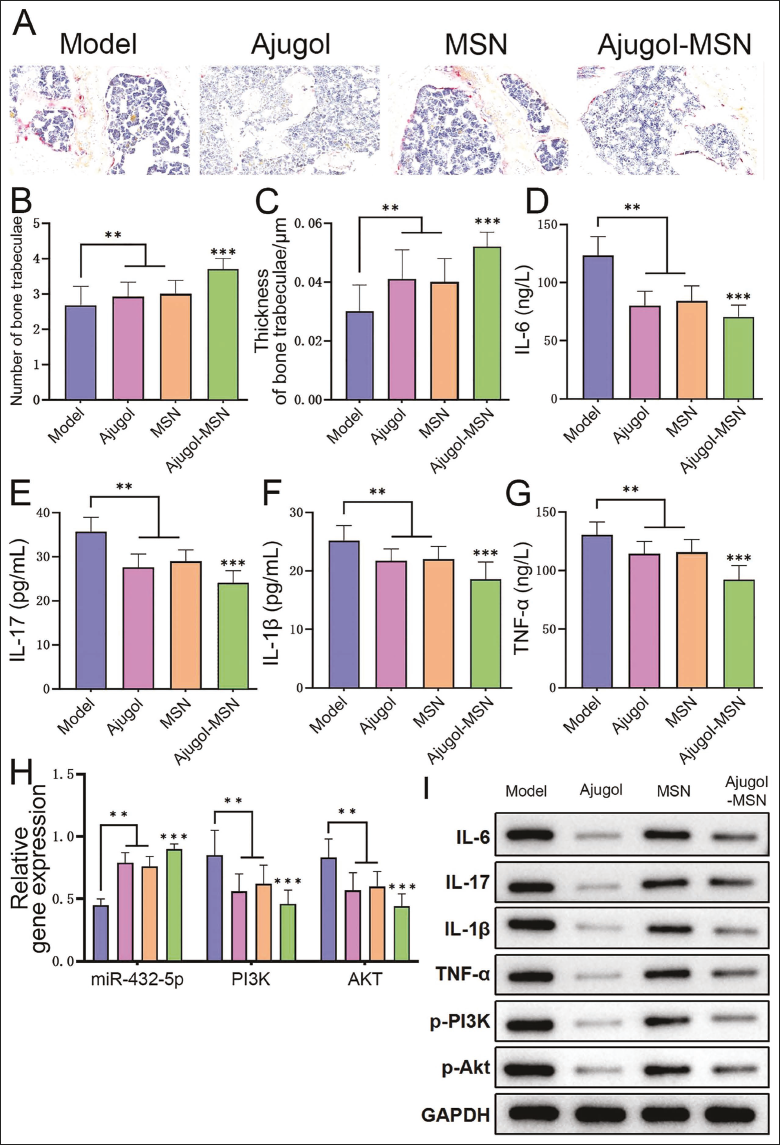
There were no mouse deaths during the modeling period, and the success rate was 100%. Analysis of mouse bone tissue pathology revealed that the Trap staining results of the Model group showed disordered bone trabecular arrangement and loose structure, accompanied by a small amount of bone fractures and an increased number of osteoclasts; the staining results of the other three groups showed all the above phenomena were alleviated, most significantly in the Ajugol group (Figure 2A). Comparing the number and thickness of bone trabeculae, it was found that the Model group was at a low level, and it improved after the intervention of Ajugol, MSN, and Ajugol, with the Ajugol group being the most significant (Figure 2B and 2C). In addition, inflammatory factors (IL-6, IL-17, IL-1β, TNF-α) levels (Figure 2D–2G) and protein levels of PI3K and AKT (Figure 2I) in the Model group were higher, which were significantly reduced after the corresponding intervention. Finally, after Ajugol intervention, the expression of miR-432-5p was the highest. The expression of PI3K and Akt was the lowest (Figure 2H), which was contrary to the Model group, indicating that the effect of Ajugol on delaying the progression of spinal tuberculosis may be related to the effect of miR-432-5p is related to changes of the PI3K/Akt pathway. Therefore, Ajugol significantly improves spinal tuberculosis models, not only delaying the development process of spinal tuberculosis but also improving bone damage and reducing inflammatory response by regulating miR-432-5p and its downstream PI3K/Akt signaling pathway.
Ajugol Can Delay the Progression of Spinal Tuberculosis (n = 5). (A) Trap Staining to Observe Histopathology; (B) Number of Trabeculae in Each Group; (C) Thickness of Trabeculae in Each Group; (D)–(G) Enzyme-linked Immunosorbent Assay (ELISA) Detection of Inflammatory Factor Levels in Each Group; (H) Polymerase Chain Reaction (PCR) Detects the Expression of MicroRNA-432-5p (miR-432-5p) and Phosphoinositide 3-Kinase/Protein Kinase B (PI3K/Akt) mRNA in Each Group; (I) Western Blotting (WB) Detects the Protein Expression of Inflammatory Factors and PI3K and AKT in Each Group; Compared with the Model Group, **p < .01, ***p < .001.
Ajugol Can Increase miR-432-5p Levels in Bone Tissue and Delay the Progression of Spinal Tuberculosis
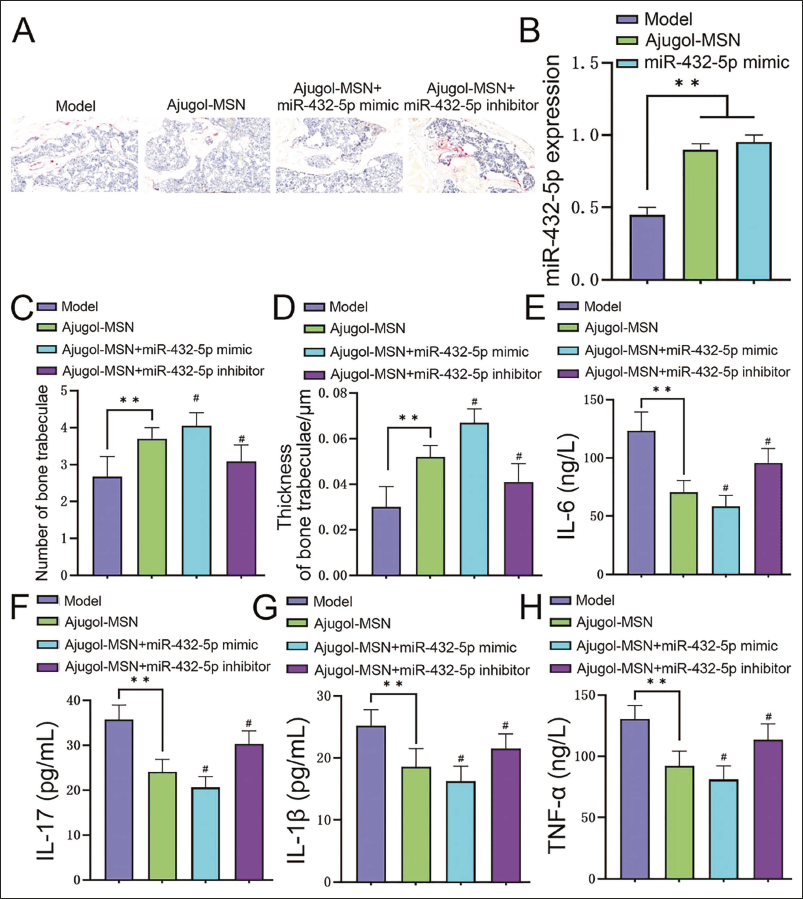
In the Trap staining results, under the intervention condition of Ajugol+miR-432-5p mimic, the disordered arrangement and loose structure of trabecular bone were further alleviated compared with the Ajugol group, and the number of osteoclasts was further reduced (Figure 3A). We tested the miR-432-5p level again and found that it was at a high level in Ajugol (vs. Model group, p < .05), which was consistent with the trend of the miR-432-5p mimic group, reflecting that Ajugol can mediate the increased miR-432-5p levels (Figure 3B). In addition, under the intervention of Ajugol+miR-432-5p mimic, the number and thickness of trabecular bone significantly increased (both p < .05, Figure 3C and 3D), and the levels of inflammatory factors significantly decreased (both p < .05, Figure 3E–3H). However, combining Ajugol with the miR-432-5p inhibitor significantly reversed the above phenomenon.
Ajugol Can Increase the Level of MicroRNA-432-5p (miR-432-5p) in Spinal Tuberculosis and Thereby Delay the Progression of Spinal Tuberculosis (n = 5). (A) Trap Staining to Observe Histopathology; (B) Expression Levels of miR-432-5p in Both Groups; (C) Number of Trabeculae in Each Group; (D) Thickness of Trabeculae in Each Group; (E)–(H) Enzyme-linked Immunosorbent Assay (ELISA) Detects the Levels of Inflammatory Factors in Each Group; Compared with the Model Group, **p < .01, Compared with the Ajugol Group, #p < .05.
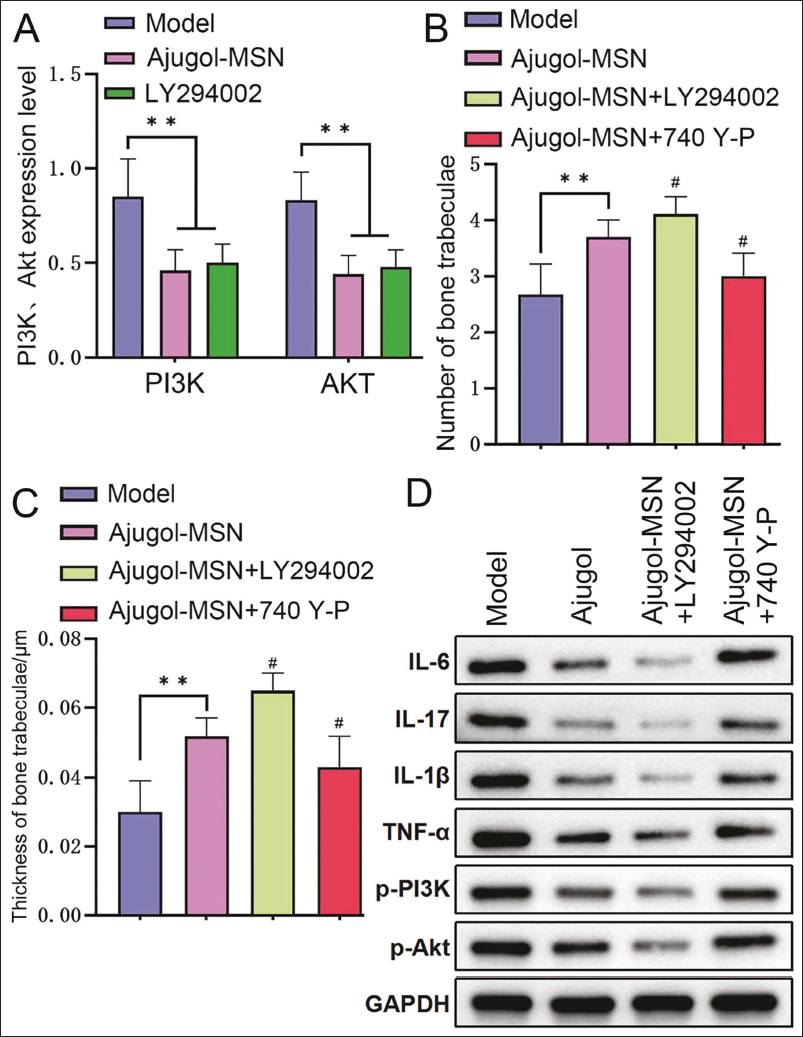
Ajugol Can Inhibit the Activity of the PI3K/Akt Pathway in Bone Tissue and Delay the Progression of Spinal Tuberculosis
We tested the expression of PI3K and Akt again. We found that they were at low levels in Ajugol (vs. Model group, p < .05), which was consistent with the trend of the LY294002 group, reflecting that Ajugol can mediate a reduction in PI3K/Akt pathway activity (Figure 4A). In addition, under the intervention of Ajugol+LY294002, the number and thickness of trabecular bone were significantly increased (all p < .05, Figure 4B and 4C), and levels of inflammatory factor proteins and protein expressions of PI3K and AKT were significantly reduced (all p < .05, Figure 4D). After combining Ajugol with 740 Y-P, the above phenomenon was significantly reversed.
Ajugol Can Inhibit the Activity of the Phosphoinositide 3-Kinase/Protein Kinase B (PI3K/Akt) Pathway in Spinal Tuberculosis and Thereby Delay the Progression of Spinal Tuberculosis (n = 5). (A) PI3K and Akt Expression Levels; (B) Number of Trabecular Bones in Each Group; (C) Thickness of Trabecular Bone in Each Group; (D) Western Blotting (WB) Detection of Inflammatory Factor, PI3K, and Akt Protein Expression in Each Group; Compared with the Model Group, **p < .01, Comparison with the Ajugol Group, #p < .05.
Discussion
Spinal tuberculosis infection destroys the stability of the spinal bones, which may lead to abnormal spinal curvature and even obvious spinal deformity, which not only affects the appearance but also has an impact on the patient’s physical function and quality of life (Garg & Goyal, 2020; Na et al., 2023). Early diagnosis and treatment of spinal tuberculosis can effectively prevent the further spread of infection and deterioration of the condition. Studies have shown (Angeloni et al., 2021; Garran et al., 2019) that motherwort can significantly improve insulin resistance, protect liver damage, reduce serum triglycerides (TG), total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), and low-density lipoprotein cholesterol (LDL-C)/high-density lipoprotein cholesterol (HDL-C) levels in high-fat mice, inhibit mTOR phosphorylation, promote transcription factor EB (TFEB)-mediated lysosome biogenesis and the generation of autophagic lysosomes, promote lipid autophagy, reduce liver lipid accumulation, thereby improving liver steatosis. In addition, the production of TNF-α can be reduced by inhibiting the activation of nuclear factor kappa B (NF-κB) and the release of IL-6, thereby inhibiting the inflammatory response (Li, Zhu et al., 2022; Park et al., 2023). However, motherwort is easily affected by environmental conditions and biological metabolism in its exposed state, leading to degradation and failure, and its solubility is low, affecting its absorption and bioavailability. Encapsulating motherwort can provide protection from premature degradation and prolong its stability. It increases its solubility, promotes its dissolution and absorption, and improves the drug’s biological activity. Therefore, to improve motherwort’s shortcomings, this study successfully prepared Ajugol composite materials. In order to prove whether Ajugol has an inhibitory effect on spinal tuberculosis, we found through Trap staining results that Ajugol can alleviate the sparse trabecular bone structure, reduce the number of osteoclasts, and reduce IL-17, IL-1β, and TNF-α levels in mice, thereby treating spinal tuberculosis, and Ajugol showed a more consistent effect. In addition, during the experiment, miR-432-5p showed high expression, and PI3K and Akt showed low expression, suggesting that miR-432-5p, PI3K, and Akt are involved in treating spinal tuberculosis by Ajugol.
Studies have found (Ferragut Cardoso et al., 2021) that miRNA can affect the differentiation and function of immune cells, thereby regulating the immune system’s response. Under the dual intervention of Ajugol and miR-432-5p mimic, it was found that the number and thickness of trabecular bone in this group were significantly increased, and the levels of IL-6, IL-17, IL-1β, and TNF-α were significantly reduced. In contrast, the miR-432-5p inhibitor showed consistent and opposite results. It shows that Ajugol can reduce miR-432-5p levels in spinal tuberculosis, thereby delaying its progression. Analysis suggests that miR-432-5p can bind to β-catenin, inhibiting the expression of β-catenin. This further helps to reduce abnormal bone proliferation and degeneration. In addition, transforming growth factor-beta (TGF-β) receptor activation can cause the phosphorylation and activation of Smad protein, thereby promoting the high expression of miR-432-5p, inhibiting the production and release of IL-6 and IL-1β, and reducing the inflammatory response in the development of spondylosis. Ajugol strengthened the effect of this gene by promoting miR-432-5p expression.
Another study found (Wang et al., 2022) the role of PI3K/Akt signaling in cell proliferation and bone metabolism in spinal tissue by regulating mTORC1 and 2 signaling. In addition, the use of mTOR inhibitors can inhibit the activity of the mTOR signaling pathway, thereby inhibiting PI3K/Akt signaling and reducing the release of vascular endothelial growth factor (VEGF), affecting the expression of bone formation factors, RANKL, and OPG, thereby regulating bone tissue regeneration and bone metabolism. To explore the specific mechanism of action of Ajugol in inhibiting spinal tuberculosis, we detected the levels of PI3K and Akt. We found that their levels were significantly reduced under the intervention of Ajugol. On this basis, LY294002 and 740Y-P were used, and it was found that under the intervention of LY294002, the number and thickness of trabecular bone increased, and the levels of related inflammatory factors decreased, while the Ajugol+740Y-P group showed the opposite change. It has been proven that Ajugol can inhibit the PI3K/Akt pathway’s activity in spinal tuberculosis, thereby delaying its progression. However, the current study still has issues, such as whether the treatment is affected by other factors and the specific dosage is unknown, which requires further exploration.
Conclusion
In summary, Ajugol promotes the high expression of miR-432-5p and inhibits the PI3K/Akt pathway, thereby reducing the activity of this pathway, inhibiting inflammatory responses, reducing cell proliferation and survival, and thus helping to treat spinal tuberculosis. Further future research may focus on developing drugs or new treatments.
Footnotes
Abbreviations
miR-432-5p: MicroRNA-432-5p; PI3K/Akt: Phosphoinositide 3-kinase/protein kinase B.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not Applicable
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not Applicable
