Abstract
Background
Sortilin 1 (SORT1) has been reported as an oncogene in several human tumors. Nonetheless, the biological functions of SORT1 in hepatocellular carcinoma (HCC) remain poorly understood.
Methods
Western blotting was employed for the determination of protein expression. Hepatocellular carcinoma cell viability, apoptosis, migration, and invasion were measured via CCK-8, flow cytometry, wound healing, and Transwell assays.
Results
Sortilin 1 was upregulated in HCC and closely associated with unsatisfactory outcomes of HCC patients. Furthermore, in vitro and in vivo assays revealed that SORT1 knockdown significantly diminished HCC cell proliferation and metastasis but accelerated HCC cell apoptosis; moreover, SORT1 depletion also restrained the growth of xenografted HCC tumors. Mechanistically, SORT1 activated PI3K/AKT signaling in HCC cells, thereby promoting the malignant behaviors of HCC cells.
Conclusion
This study demonstrated that SORT1 might promote HCC progression by activating PI3K/AKT signaling, indicating that SORT1 might be a promising target and biomarker for HCC treatment and prognosis.
Introduction
Hepatocellular carcinoma (HCC), a prevalent type of primary liver cancer, constitutes about 80% of liver cancer cases 1 and ranks the sixth in cancer-related mortality globally. 2 In recent years, HCC prevalence has been increasing across the world, especially in China. 3 Owing to high recurrence and metastasis rates, many HCC patients die of locally advanced tumors or metastasis in a relatively short period of time. 4 Although great advances have been achieved in HCC therapy, the improvement in the overall survival of HCC patients remains discouraging. 5 Therefore, the identification of the underlying molecular mechanisms in HCC is a critical need.
Sortilin 1 (SORT1; also known as NTR3), one of the VPS10P-domain receptors, was first discovered two decades ago. 6 Sortilin 1 is widely expressed in human cells and tissues 7 and serves as a regulator for protein sorting and trafficking both intracellularly and extracellularly. 8 Previous studies have reported SORT1 dysregulation in several human malignancies, including ovarian cancer, 9 breast cancer, 10 pancreatic cancer 11 and chronic lymphocytic leukemia. 12 As a multifaceted protein, SORT1 assumes cancerogenic or anticancer roles in human cancers depending on the origin of the tumors. 13 For instance, SORT1 silencing retards breast cancer progression and lung metastases. 14 Sortilin 1 knockdown suppresses gastric cancer cell proliferation, invasion, and migration. 15 In addition, SORT1 facilitates neuroendocrine tumor progression by promoting cell proliferation, adhesion, and migration. 16 Therefore, SORT1 recently became a promising target for cancer treatment. As stated in a report by Meroni et al., rs599839 A > G variant might contribute to HCC progression by regulating SORT1 expression 17 implying the involvement of SORT1 in HCC. Nevertheless, the molecular functions and prognostic value of SORT1 in HCC remains to be further investigated.
In our study, we aimed to explore the role and potential mechanism of SORT1 in HCC. Based on in vitro and in vivo assays, it was revealed that SORT1 activated the PI3K/AKT signaling to promote HCC progression. Our findings suggest that SORT1 plays an oncogenic part in HCC progression and might be a promising therapeutic target and prognostic biomarker of HCC.
Materials and methods
Cell culture and transfection
Hepatocellular carcinoma cell lines (HEP3B: BNCC360312, SNU449: BNCC359904, and HUH-7: BNCC337690) obtained from BeNa Culture Collection (Beijing, China) and normal human liver epithelial cell line (THLE-3: CRL-11,233) acquired from ATCC were cultured in DMEM (10% FBS) in a humidified incubator (37°C; 5% CO2). 18 Short hairpin RNA (shRNA) against SORT1 (sh-SORT1#1 or sh-SORT1#2) and negative control (sh-NC) were designed and synthesized by Biomics (Nantong, China). Then, SNU449 and HEP3B cells were respectively transfected with these plasmids Via Lipofectamine™ 2000. 19 For 740 Y-P treatment, transfected HCC cells were treated with 740 Y-P (Sangon Biotech, Shanghai) at 30 μM for 24 h. 20
Western blotting
RIPA buffer (Beyotime) was used for protein extraction from the cells. Total proteins were separated by SDS-PAGE, transferred to PVDF membranes, and then incubated with primary antibodies against SORT1 (ab16640, Abcam), phosphorylated-PI3K (p-PI3K, ab182651, Abcam), total-PI3K (t-PI3K, ab191606, Abcam), phosphorylated-AKT (p-AKT, ab38449, Abcam), total-AKT (t-AKT, ab8805, Abcam), and GAPDH (ab8245, Abcam) overnight at 4°C, followed by incubation with corresponding HRP-conjugated secondary antibody (ab6789 or ab205718; Abcam) for 1 h at room temperature. Subsequently, protein bands were visualized via ECL (Bio-Rad laboratory).
CCK-8
Transfected HCC cells were seeded into 96-well plates (1 × 104 cells/well). After incubation for the indicated time, HCC cells were cultured with CCK-8 solution (Dojindo) for another 2 h. At last, the absorbance was detected with GENios Pro microplate multifunction reader (Tecan) at 450 nm. 21
Flow cytometry
Annexin V-FITC Apoptosis Detection Kit (Beyotime) was applied to assess cell apoptosis. Briefly, transfected HCC cells were treated with trypsin, rinsed with PBS, and resuspended in binding buffer. Next, the cells were cultured with Annexin V-FITC and PI solutions. Finally, flow cytometry was conducted to distinguish apoptotic cells.
Wound healing
The transfected HCC cells were plated into 6-well plates and cultured until 70% confluence. Then, a scratch was made with a sterile pipette tip. Images of the scratch area were captured with a microscope (Olympus, Japan) at 0 and 24 h. ImageJ software program (NIH, USA) was used to evaluate cell migration. 22
Transwell assay
The invasion ability of the HCC cells was analyzed using Transwell chambers (8.0 μm pore size; EMD Millipore) and Matrigel (Corning Inc.).Briefly, HCC cells (4 × 104 cells) were plated in the upper chamber (serum-free medium). The lower chamber was supplemented with complete culture medium (10% FBS). The invaded HCC cells were counted under a microscope after incubation at 37°C for 48 h.
Animal experiment
The animal experiments were approved by the Affiliated Shuyang Hospital of Xuzhou Medical University (approval number: XZ(A)-20–02–6). Six male BALB/c nude mice were applied for the establishment of HCC Xenograft tumor models. Briefly, each mouse was subcutaneously injected with SNU449 cells transfected with sh-NC (n = 3) or sh-SORT1#1 (n = 3) (1 × 106 cells/mouse). The tumor size was detected every 5 days. Finally, the mice were sacrificed 30 days after injection, and the xenografted tumors were harvested and weighed. To assess the effect of SORT1 knockdown on metastasis of HCC, transfected cells (1 × 106) were injected into nude mice through the tail vein. Six weeks later, the lungs were isolated and metastasis was analyzed using H&E staining.
Statistical analysis
Each experiment in this study was conducted in triplicate. Data acquired were exhibited as the mean ± standard deviation (SD). RNA-seq data of TCGA dataset were downloaded from UCSC Xena website (https://xenabrowser.net/) and converted from FPKM (Fregments Per Kilobase per Million) format to TPM format (transcripts per kilobase million). Via Mann-Whitney U test (Wilcoxon rank sum test), SORT1 mRNA expression was analyzed and visualized with ggplot2 package (version 3.3.3) in R program (version 3.6.3). The association between SORT1 expression and the survival time in LIHC was analyzed and visualized with survival package (version 3.2–10) and the survminer package (version 0.4.9) in R software (version 3.6.3). The ROC curve was generated with pROC package (version 1.17.0.1) and ggplot2 package (version 3.3.3) in R program (version 3.6.3). The statistical analysis was performed via GraphPad Prism 6.0 by Student t-test or one-way ANOVA. p value < .05 was deemed significant statistically.
Results
Elevated SORT1 expression in LIHC
Based on data from The Cancer Genome Atlas (TCGA) database, it was found that SORT1 mRNA expression was elevated in several cancer types, including Liver Hepatocellular Carcinoma (LIHC) (Figure 1(a)). Then, we referred to TCGA-LIHC dataset for SORT1 expression in LIHC, and the clinicopathologic features of LIHC patients were presented in Table 1. It was manifested SORT1 level in LIHC tissues was substantially increased, in comparison with that in normal tissues (Figure 1(b)). In addition, IHC results confirmed that SORT1 expression was noticeably increased in HCC tissues compared with normal tissues (Figure 1(c)). Next, Kaplan-Meier method was used to assess the association between SORT1 expression and overall survival (OS) of LIHC patients based on TCGA-LIHC dataset. It was uncovered that LIHC patients with high SORT1 levels presented shorter OS (HR = 1.42; p = .048) (Figure 1(d)). Thereafter, a ROC curve was plotted to calculate the area under the ROC curve (AUC) which was 0.872 (95% confidence interval = 0.823–0.921) (Figure 1(e)). Hence, it was conjectured that SORT1 might be an oncogene and potential prognostic biomarker for HCC. Elevated SORT1 expression in LIHC. (a) SORT1 mRNA expression in pan-cancer based on TCGA database. (b) SORT1 mRNA expression in LIHC tissues and normal tissues based on TCGA-LIHC dataset. (c) IHC showed SORT1 expression in HCC tumor and para-cancerous normal tissues. (d) OS curve of SORT1 in LIHC by Kaplan-Meier method based on TCGA-LIHC dataset. (e) ROC curve of SORT1 in LIHC based on TCGA-LIHC dataset. *p < .05, **p < .01, ***p < .001. Clinicopathologic features of LIHC patients.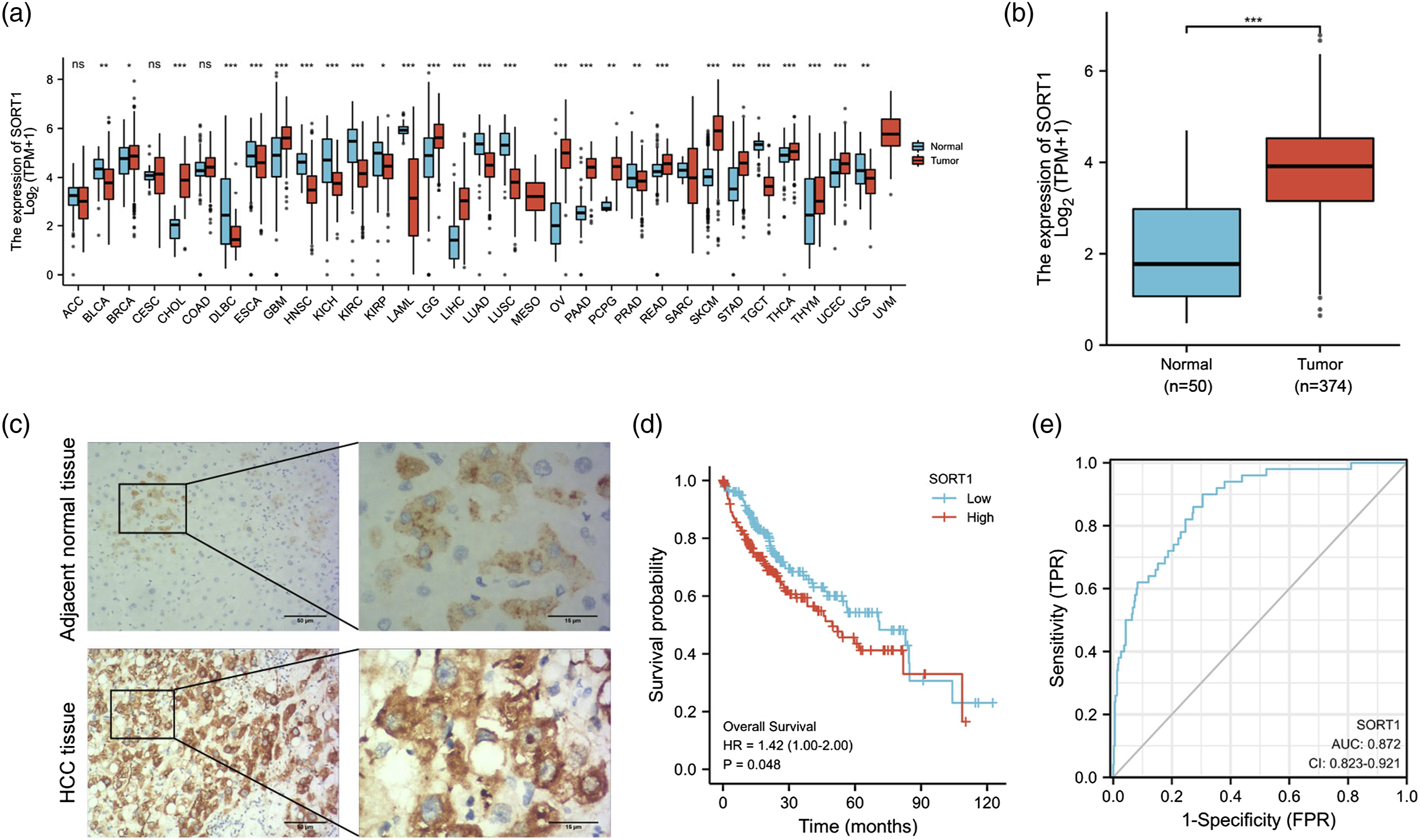

Sortilin 1 upregulation in HCC cell lines in vitro
To explore the putative role of SORT1 in HCC, we measured SORT1 protein levels in normal cells (THLE-3) and HCC cell lines (HEP3B, SNU449, and HUH-7) by Western blotting. Consistently, the SORT1 level was substantially higher in HCC cells, in contrast to normal cells (Figure 2(a)). Besides, since SNU449 and HEP3B exhibited higher SORT1 levels, they were chosen for subsequent experiments. Then, sh-SORT1#1 and sh-SORT1#2 were synthesized to knock down SORT1 in SNU449 and HEP3B cells. Western blotting assays were performed to verify the knockdown efficacies of sh-SORT1#1 and sh-SORT1#2 (Figure 2(b)). As sh-SORT1#1 exhibited higher knockdown efficiency, it was used in subsequent experiments. SORT1 upregulation in HCC cell lines in vitro. (a) SORT1 protein levels in HCC cell lines (HEP3B, SNU449, and HUH-7) and normal cell line (THLE-3) were determined by Western blotting. (b) SORT1 protein levels in sh-NC, sh-SORT1#1, or sh-SORT1#2 transfected SNU449 and HEP3B cells. *p < .05, **p < .01.
Sortilin 1 knockdown suppresses proliferation and induces apoptosis in HCC cells in vitro
To analyze the function of SORT1 in HCC cell proliferation and apoptosis, CCK-8 and flow cytometry assays were performed on HCC cell lines. First of all, SNU449 and HEP3B cells were respectively transfected with sh-NC or sh-SORT1#1. As outlined by CCK-8 assay, SORT1 inhibition reduced the viability of SNU449 and HEP3B cells (Figure 3(a) and (b)). Besides, flow cytometry assay disclosed that SORT1 silencing increased the apoptotic rate of SNU449 and HEP3B cells (Figure 3(c)). Altogether, these data indicated that SORT1 might be a critical regulator of HCC cell proliferation and apoptosis. SORT1 knockdown suppresses proliferation and induces apoptosis in HCC cells in vitro. (a and b) SNU449 and HEP3B cells were respectively transfected with sh-NC or sh-SORT1#1. Cell viability of sh-NC or sh-SORT1#1 transfected SNU449 and HEP3B cells were detected by CCK-8 assay. (c) Flow cytometry was performed to assess the apoptosis of sh-NC or sh-SORT1#1 transfected SNU449 and HEP3B cells. **p < .01.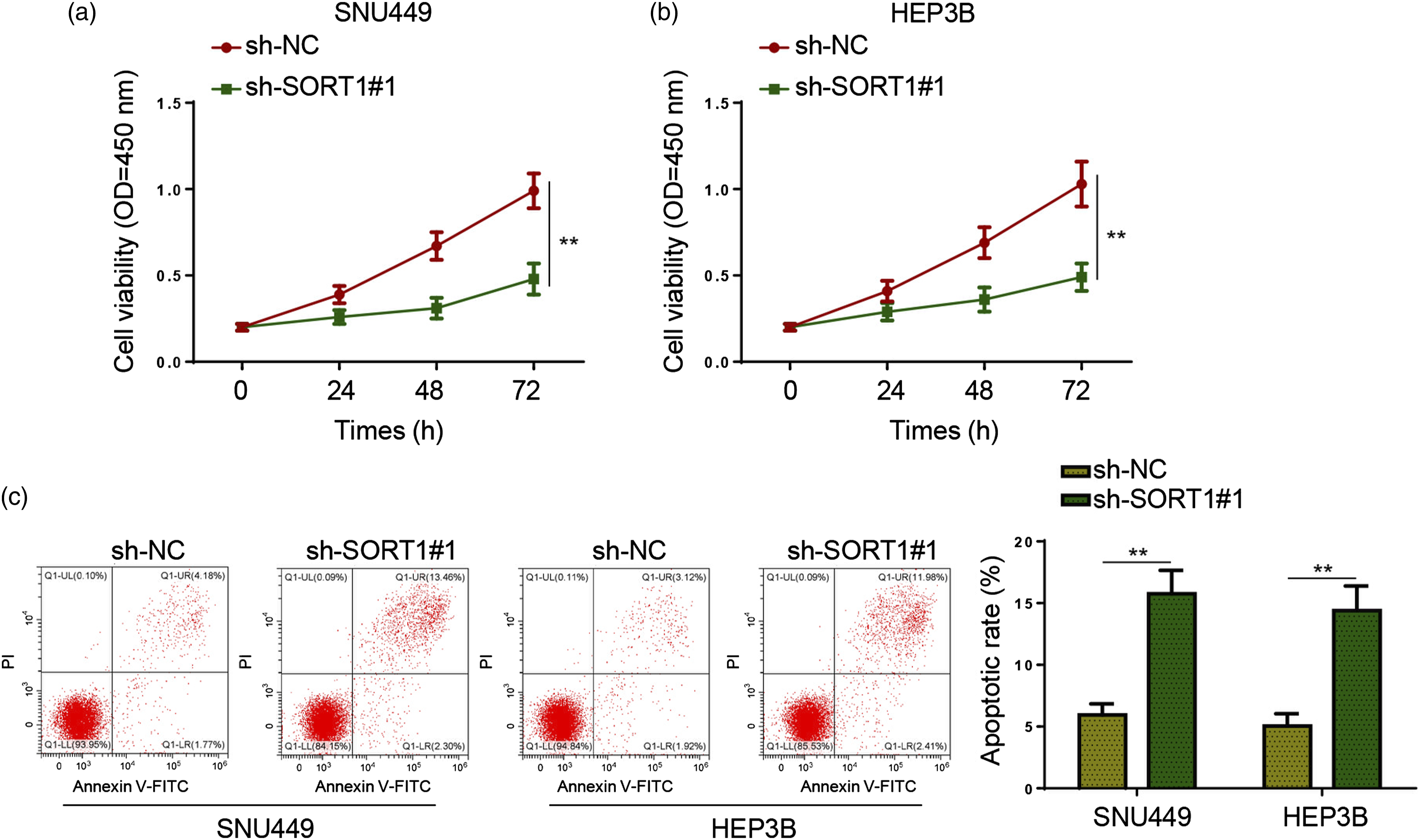
Sortilin 1 deletion restrains HCC cell metastasis in vitro
Subsequently, wound healing and Transwell assays were performed to investigate the functions of SORT1 in HCC cell invasion and migration. As exhibited in Figure 4(a), SNU449 and HEP3B cells in sh-SORT1#1 group exhibited a significant decrease in migrative abilities. Moreover, it was also revealed that SORT1 inhibition dramatically inhibited the invasive capability of SNU449 and HEP3B cells (Figure 4(b)). Therefore, it could be concluded that SORT1 might enhance the migrative and invasive capacities of HCC cells. SORT1 deletion restrains HCC cell metastasis in vitro. (a and b) The migrasive and invasive abilities of sh-NC or sh-SORT1#1 transfected SNU449 and HEP3B cells were evaluated by wound healing (magnification, ×100) and Transwell (magnification, ×200) assays. **p < .01.
Sortilin 1 modulates malignant behaviors of HCC cells via PI3K/AKT pathway in vitro
To explore downstream mechanisms associated with SORT1 in LIHC, Gene Set Enrichment Analysis (GSEA) was performed based on TCGA-LIHC dataset. As illustrated in Figure 5(a), there is a positive association between SORT1 level and PI3K/AKT pathway in LIHC. To verify the association between SORT1 and PI3K/AKT signaling in HCC, p-PI3K, t-PI3K, p-AKT, and t-AKT protein levels were measured in SNU449 and HEP3B cells transfected with sh-NC or sh-SORT1#1. It was shown that p-PI3K and p-AKT levels significantly declined in SORT1-deficient HCC cells, while t-PI3K and t-AKT levels remained nearly unchanged (Figure 5(b)). SORT1 modulates malignant behaviors of HCC cells via PI3K/AKT pathway in vitro. (a) Enrichment analysis of SORT1 in PI3K/AKT signaling pathway by GSEA. (b) p-PI3K, t-PI3K, p-AKT, and t-AKT levels in sh-NC or sh-SORT1#1 transfected SNU449 and HEP3B cells were determined by Western Blotting. (c) SNU449 and HEP3B cells were divided into sh-NC, sh-SORT1#1, and sh-SORT1#1+740 Y-P groups. The p-PI3K, t-PI3K, p-AKT, and t-AKT levels in SNU449 and HEP3B cells from each group were determined by Western Blotting. (d–g) SNU449 and HEP3B cell viability (d), apoptosis (e), migration (f), and invasion (g) in each group. *p < .05, **p < .01, ***p < .001.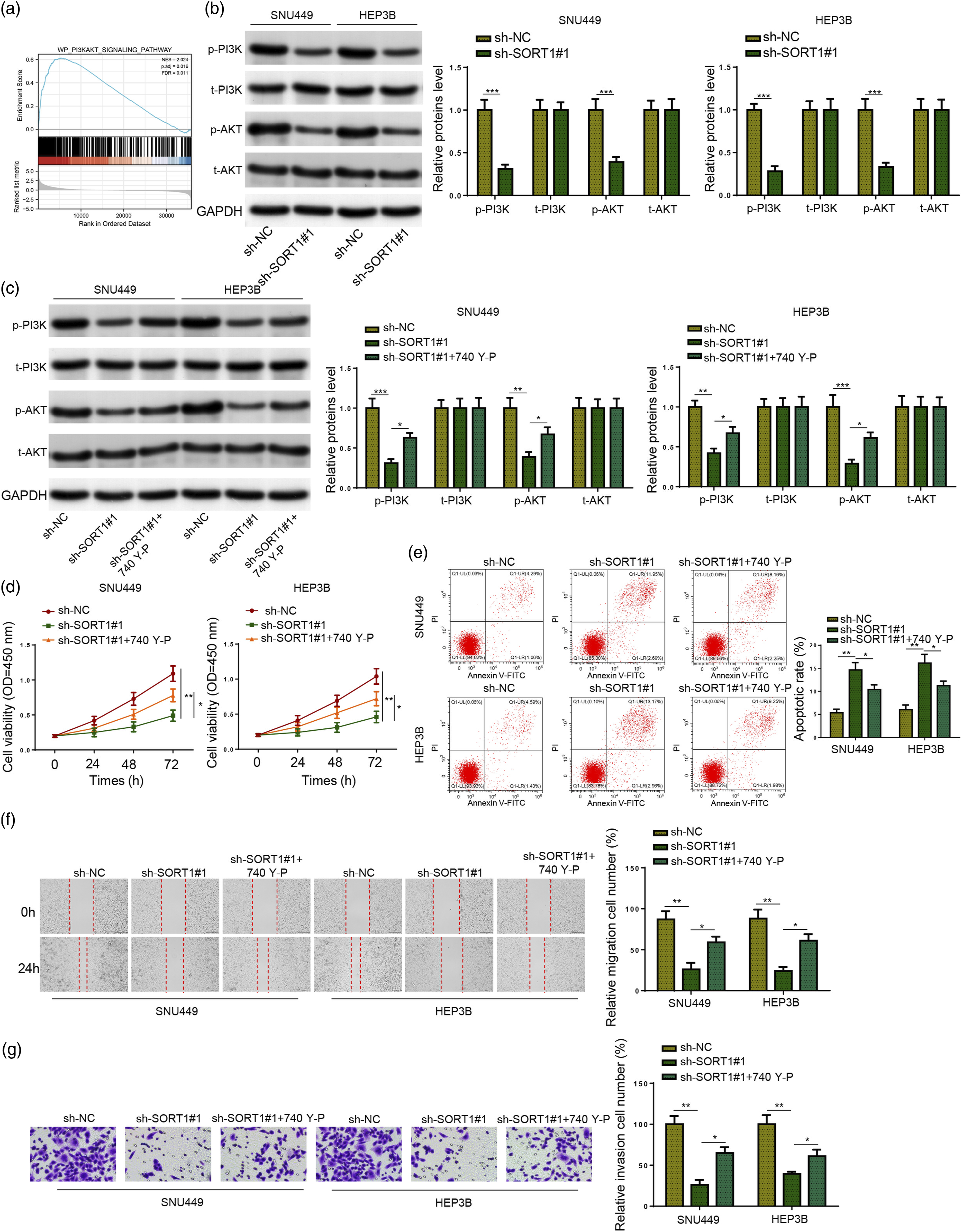
Next, 740 Y-P, a PI3K agonist, 23 was applied to explore the association between PI3K/AKT signaling and SORT1 in the regulation of malignant behaviors of HCC cells. SNU449 and HEP3B cells were divided into three groups: sh-NC, sh-SORT1#1, and sh-SORT1#1 + 740 Y-P. It was shown that 740 Y-P reversed the reduction in p-PI3K and p-AKT levels caused by SORT1 knockdown (Figure 5(c)). Rescue assays further illustrated that 740 Y-P considerably abated the effects of SORT1 inhibition on the viability, apoptosis, migration, and invasion of SNU449 and HEP3B cells (Figure 5(d)–(g)). Taken together, SORT1 contributed to HCC progression in vitro by activating the PI3K/AKT pathway.
Sortilin 1 inhibition impairs HCC tumor growth and lung metastasis in vivo
To verify the functions of SORT1 in HCC in vivo, the xenograft tumor assay was performed. Mice received subcutaneous injection with SNU449 cells transfected with sh-NC or sh-SORT1#1. As shown in Figure 6(a), SORT1 protein level was obviously decreased in sh-SORT1#1 group. In addition, mice in SORT1-deficient group exhibited a remarkable decrease in tumor size and weight (Figure 6(b) and (c)). In addition, H&E staining assays revealed that SORT1 silencing led to a remarkable decrease in the number of metastatic nodules in the lung (Figure 6(d) and (e)). Overall, these data demonstrated that SORT1 promoted tumor growth and lung metastasis in vivo. SORT1 inhibition impairs HCC tumor growth and lung metastasis in vivo. BALB/c nude mice were injected with sh-NC (n = 3) or sh-SORT1#1 (n = 3) transfected SNU449 cells to construct xenograft tumor models. All mice were euthanized 30 days as of injection to harvest tumors. (a) SORT1 protein level was detected in tumor tissues from two groups by Western blotting. (b) Tumor sizes were measured every 5 days (c) Xenografted tumors were weighed after collection. (d and e) Representative H&E-stained sections of lung tissues and the number of tumor nodules on lung surfaces. **p < .01.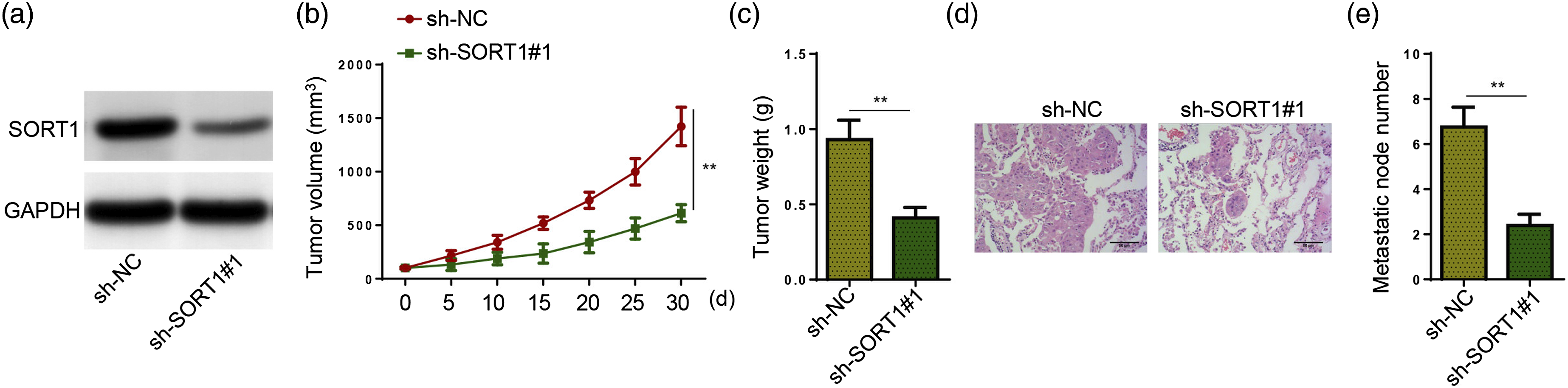
Discussion
Hepatocellular carcinoma is a fatal cancer featured with insidious onset, fast and invasive growth, high malignancy, high recurrence, and high mortality rate. 24 Globally, HCC is the sixth major cause of cancer-associated death. 2 Meanwhile, many HCC patients are of short survival time due to metastasis and early relapse after surgical resection. 25 Hence, HCC remains a threat to public health to this day. 26 As a result, an in-depth understanding of the molecular mechanisms implicated in HCC pathogenesis is needed for the development of novel diagnostic and therapeutic strategies for HCC.
According to earlier studies, patients with higher or positive SORT1 expression exhibited worse prognosis and advanced tumor progression in breast cancer, 14 B acute lymphoblastic leukemia, 27 neuroendocrine tumors 16 and colorectal cancer, 13 suggesting that SORT1 could be a biomarker to predict the survival time of patients with malignant tumors. In this research, after data mining based on the TCGA-LIHC dataset, it was uncovered that SORT1 expression was substantially lifted in LIHC. Additionally, the positive relationship between high SORT1 expression and unsatisfactory survival of LIHC patients, as well as the high AUC level highlighted the potential diagnostic and prognostic values of SORT1 in LIHC. Taken together, it was hypothesized that SORT1 might play an oncogenic role in HCC.
As demonstrated in previous studies, SORT1 inhibition could slow down tumor growth and cancer progression. 28 To cite an instance, SORT1 depletion substantially impedes the proliferative, invasive, and migrative abilities of gastric cancer cells. 15 Besides, SORT1 interference also decreases cell adhesion and invasion in pancreatic cancer. 11 These studies suggest that SORT1 may act as a tumor promoter to promote malignant behaviors of cancer cells and accelerate cancer progression. Consistent with previous studies, it was revealed that SORT1 knockdown may attenuate HCC cell proliferation and increase HCC cell apoptosis. Besides, SORT1 inhibition may attenuate the migrative and invasive functions of HCC cells. Moreover, animal experiments indicated that SORT1 silencing may also inhibit the growth of HCC tumors in vivo. Therefore, it suggests that SORT1 may contribute to HCC tumorigenesis.
Next, GSEA was performed to identify downstream signaling pathways of SORT1 in LIHC. GSEA result disclosed that the PI3K/AKT signaling was closely related to SORT1 expression in LIHC. Moreover, the determination of p-PI3K and p-AKT levels by Western blotting further confirmed that SORT1 positively regulated the PI3K/AKT signaling in HCC. As a major signaling activated in human malignancies, PI3K/AKT regulates cellular processes, including cell proliferation, apoptosis, and migration, of cancer cells. 29 As reported in a prior study, the PI3K/AKT pathway is inaptly activated in almost 50% of HCC. 30 Therefore, the PI3K/AKT signaling is a promising target for HCC treatment. For example, Aloperine mitigates HCC by inducing apoptosis and cell cycle arrest through the PI3K/Akt signaling. 31 PAQR4 contributes to HCC tumorigenesis by activating the PI3K/AKT pathway. 32 PES1 promotes HCC progression via the PI3K/AKT axis. 33 Herein, it was revealed that 740 Y-P, a PI3K activator, could rescue the inhibitory effect of SORT1 knockdown on PI3K and AKT phosphorylation, as well as malignant phenotypes of SNU449 and HEP3B cells. All these results indicated that SORT1 can modulate HCC cell behaviors via the PI3K/AKT signaling.
Conclusion
Our study, for the first time, elucidated that SORT1 could promote HCC progression in vitro and in vivo by regulating the PI3K/AKT signaling, suggesting SORT1 might be a novel target for HCC treatment.
Footnotes
Abbreviations
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
