Abstract
Background
Several studies have demonstrated the anti-inflammatory properties of gastrodin. However, its specific role and mechanism in the context of osteoarthritis (OA) remain unclear.
Objectives
The objective of this study was to explore the function and mechanism of gastrodin in both in vivo and in vitro OA models.
Materials and Methods
In this study, the targets of gastrodin against osteoarthritis were analyzed using bioinformatics methods. The effect of gastrodin on neutrophil infiltration and inflammatory cytokines in OA synovial tissue was evaluated using H&E staining. The levels of inflammatory cytokines were determined using ELISA and Q-PCR. The extracellular matrix degradation-related proteins and PI3K/AKT/FoxO1 signaling pathway were tested using western blotting.
Results
Bioinformatics analysis predicted that the target of gastrodin against OA might act on the FoxO pathway. The use of gastrodin resulted in a significant reduction in the infiltration of neutrophils in the synovial tissue of rats, as observed through H&E staining. Additionally, gastrodin was found to attenuate IL-1β-induced inhibition of chondrocyte viability, pro-inflammatory cytokine production, and extracellular matrix degradation. This effect can be attributed to the suppression of PI3K/AKT/FoxO1 by gastrodin.
Conclusion
These findings indicate that gastrodin is a treatment for OA.
Introduction
Osteoarthritis (OA) is a prevalent degenerative joint disorder that results in disability and pain worldwide, affecting over 300 million people (Safiri et al., 2020). The elderly population is primarily prone to OA, with studies indicating a 50% probability for those over 60 years old and up to 80% for those over 75 years old. OA is more prevalent in women than men, with a disability rate of 53% (Zhang et al., 2020). Pain is the most common clinical symptom of OA, with cartilage degeneration and synovitis being the primary pathological features. Treatment for OA is mainly focused on pain relief, preventing and delaying disease progression (Hill et al., 2007; Klement & Sharkey, 2019; Szebenyi et al., 2006). Non-steroidal anti-inflammatory drugs are commonly used for OA treatment, but their long-term use is limited due to adverse reactions. In severe or advanced cases, surgical treatment such as total knee replacement may be necessary (Skou et al., 2014). However, postoperative pain can affect the quality of life of patients, highlighting the need for safer and more effective treatments.
Gastrodin, the primary active ingredient in Gastrodia elata, has demonstrated anti-inflammatory functions in multiple illnesses (Li & Li, 2022; Liu et al., 2018). Its pharmacological effects include sedative and analgesic properties in the central nervous, cardiovascular, and immune systems (Shu et al., 2013; Sun et al., 2019; Zhang et al., 2020). Additionally, it can regulate cardiovascular function and cut down inflammation (Liu et al., 2018). A former study has revealed that gastrodin may attenuate cerebral ischemic damage in rats by inhibiting inflammatory responses and apoptosis (Xiao et al., 2021). It has also been found to reduce the level of pro-inflammatory factors in lipopolysaccharide-induced acute lung injury (for example, IL-1β, IL-6, and TNF-α). In this experiment, we investigated the effect of gastrodin in both in vivo and in vitro models of osteoarthritis and explored changes in downstream-related pathways.
Materials and Methods
Bioinformatics Analysis
The HERB database (
Grouping and Modeling of Animal Experiments
In this study, we purchased 30 SD rats from the Scientific Experimental Animal Training Center of Chengdu University of Traditional Chinese Medicine. These rats weighed between 175 and 195 g and were aged 4–5 weeks. The ethics committee of Chengdu University of Traditional Chinese Medicine approved the animal care and experimental animal use protocols for this study. The ethics approval reference number for this study is 20230306.
After 1 week of acclimation and feeding, the rats were stochastically divided into three groups: Sham group, OA model, and gastrodin treatment (OA+Gastrodin), with 10 rats in each group. The medial meniscectomy (MMx) method was used for modeling. The modeling methods are as follows: with reference to the previous research methods of the research group, the rat OA model was established by the MMx method (Hao et al., 2022). The medical nursing mat sheet was laid on the animal experimental table. After the rats were anesthetized with 1.25% avertin (Thermo Fisher Scientific, Waltham, MA, USA) intraperitoneally, the rats were shaved in the operation area, fixed on the rat plate in the supine position, and the right knee joint was flexion 45°C and disinfected with iodophor. The incision of the medial parapatellar ligament of the knee joint was used to cut along the inner edge of the patellar tendon. At this time, you can peel off from the inside of the joint. Then take the medial parapatellar incision and cut off the medial collateral ligament. When you see the joint of the femur and tibia, you can use the tip of the scalpel to carefully enter the articular platform from the medial condyle of the knee joint, find the medial meniscus, and remove the medial meniscus. The muscle tissue, connective tissue, and external skin were sutured with the No. 5 surgical line from the inside to the outside. The difference between the Sham group was that only after the joint was opened, the joint capsule, muscle, and cortex were sutured directly, that is, the meniscus was not treated. At the end of the operation, the rats in the OA+Gastrodin group were subcutaneously injected with gastrodin (Shanghai Aladdin Biochemical Technology Co., Ltd. Shanghai, China) 100 µg/kg/day until they were killed (Chen et al., 2018).
Evaluation of Mechanical Pain Abnormality in Rats
The rats were positioned on a barbed wire net, which was elevated and covered with a transparent plexiglass cover. The center of the hind paw of each rat was vertically stimulated with a von Frey filament to determine mechanical hyperalgesia. Moderate force was applied until the filament was bent into an S-shape for 2–3 s. The von Frey test should be repeated 6 times every 2 min from the first paw withdrawal response, and all test results and corresponding filament values should be recorded. Using Chaplan’s method, the above data are statistically calculated to determine the 50% paw retraction rate, which is used to calculate the final pain threshold.
Collection of Rat Knee Joint Specimens and Preparation Before Staining
In anesthetized rats, the hair on their right hind limb was removed with a shaving machine before fixing them on the mouse board in a supine position for perfusion operation. The rat’s operated side leg was cut off, and the surrounding skin and muscle tissue were peeled off to expose the complete knee joint after carefully removing excess muscle tissue post-ankle joint severance with scissors. The knee joints were then mounted in 4% paraformaldehyde (Zhongshan Aobo Biotechnology Co., Ltd. Guangdong, China) for 1 day and subsequently decalcified in 10% decalcification solution at 4°C for about a month. It was then made transparent by soaking in xylene solution (Wuxi Prospect Chemical Reagent Co., Ltd., Jiangsu, China) for an hour. After the wax block was melted, the specimen was placed in an embedding mold and sliced into 6-µm-thick wax slices.
H&E Staining
To stain the slices, first place them in hematoxylin (Beijing Soleibao Technology Co., Ltd. Beijing, China) for approximately 8 min. To differentiate the color, use a 1% hydrochloric acid ethanol solution for 5 s and then immediately place the slices in distilled water for 3 min. Finally, transfer the slices to 0.6% ammonia water (Beijing Soleibao Technology Co., Ltd.) to turn them blue. To stain the tissue, use eosin for 3 min and rinse gently with tap water to avoid tissue detachment. Seal the section with a cover glass, being careful to avoid air bubbles that could affect tissue observation.
Cell Culture and Drug Therapy
Human chondrocytes (CHON-001 cells) were purchased from the typical Culture Preservation Center in the United States (Massachusetts, Virginia, USA). Streptomycin + penicillin solution (Shanghai Zeye Biological Co., Ltd. Shanghai, China), fetal bovine serum (Gibco Scientific, Waltham, MA, USA), and F12K medium (Thermo Fisher Scientific) were mixed according to the ratio of 1:10:90 to form a complete medium for cell culture. According to the previous research experience, chondrocytes were stimulated by 10 ng/mL IL-1β (Baiaolaibo Technology Co., Ltd., Beijing, China) for 1 day to induce the inflammatory environment of chondrocytes (Xu et al., 2021). Different concentrations of gastrodin (i.e., 0.1, 1, 10, 50, and 100 µM) were added to the chondrocytes with a fusion degree of 80%–90% in a six-hole plate and were stimulated with or without IL-1β for 1 or 2 days.
Detection of Cell Viability by CCK-8 Method
The treated cells of each group were inoculated into a 96-well plate, serum-free F12K culture medium, and CCK-8 solution (Beibo Biology Co., Ltd., Jiangsu, China) was added to the well. The absorbance of each well was tested through the microplate reader at 420 nm (Thermo Fisher Scientific).
Analysis of Cleaved-Caspase 3 Activity
The cells of each group in the 6-well plate were digested with 0.25% trypsin (Gibco Scientific), and the relative activity of cleaved-caspase 3 in each group was tested using the kit (Abcam, Shanghai, China). The OD value was obtained at 405 nm.
Q-PCR
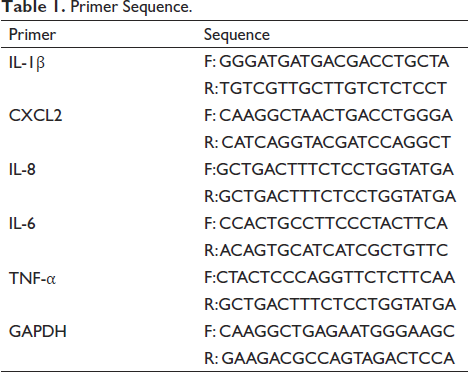
The RNAs were obtained from experimental samples using TRIzol reagent (Tiangen, Beijing, China). To obtain cDNA samples according to the requirements of the reagent kit. The CT value was tested using a fluorescent quantitative PCR instrument (Tiangen Biochemical Technology Co., Ltd.). The primers used were obtained from Guangzhou Ribobio Biotechnology Co., Ltd., (Table 1).
Primer Sequence.
ELISA
Chondrocytes were dispersed in 24-well plates, and the supernatants were collected. The level of inflammatory factors secreted by chondrocytes was tested using an ELISA kit (Thermo Fisher Scientific). The absorbance was obtained at 450 nm. The secretion level of inflammatory factors was calculated according to the corresponding standard curve.
Western Blot
The cell precipitates of each group were digested and collected, and then a certain amount of lysate (Beyotime) was put to the ice to cleave for 30 min. After lysis, total cellular proteins were collected by 12000 r/min centrifugation and 15 min at 4°C. After adding protein sample buffer (Beyotime) for boiling denaturation for 15 min. After electrophoresis, the protein was transferred to a 4.5-µm PVDF membrane (Sigma-Aldrich, St. Louis, MO, USA). The PVDF was put into 5% skimmed milk under 25°C for an hour and incubated with an antibody (TNF-α, IL-6, IL-8, p-AKT, AKT, Collagen II, FoxO1, Aggrecan, ADAMTS5, MMP13, p-PI3K, PI3K, p-FoxO1, and β-actin) (Cell Signaling Technology, Inc., USA) for 8 h. Following incubation, the PVDF membrane underwent three washes with TBST. Subsequently, the membrane was incubated with the secondary antibody. The strips were again washed three times with TBST buffer (Thermo Fisher Scientific), following which the Odyssey infrared laser imaging system was used to scan the membrane (Thermo Fisher Scientific).
Statistical Analysis
Data were presented as mean ± standard deviation. The data were processed using SPSS. The data met the requirements of normal distribution and homogeneous variance. Multiple groups comparison was performed using ANOVA, while two groups comparison was performed using Student’s t-test, and p < 0.05 was considered statistically significant.
RESULTS
The Target of Gastrodin in the Treatment of Osteoarthritis
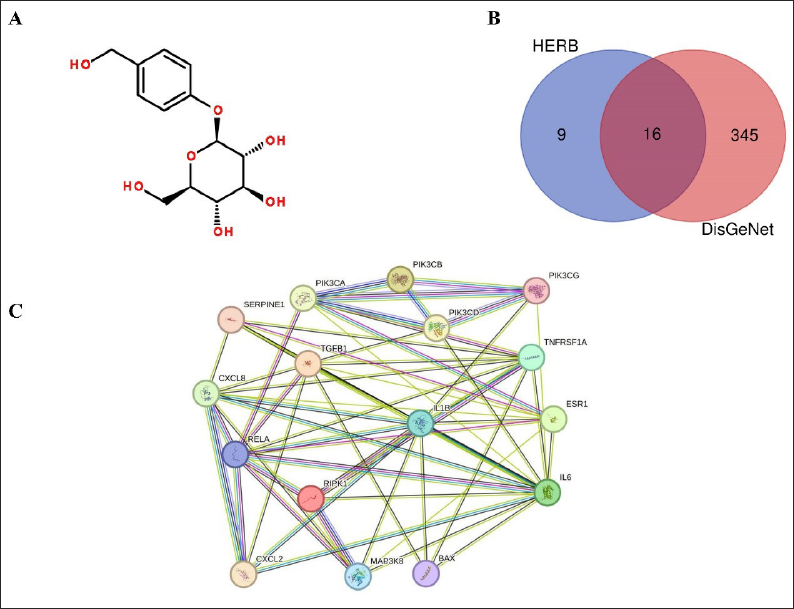
The pharmacological action of gastrodin in osteoarthritis was studied by analyzing its potential targets. The chemical structure of gastrodin is depicted in Figure 1A. HERB was utilized to predict the targets of gastrodin, while DisGeNet software was used to analyze the targets of OA. The Chinese medicine group had 25 targets, while the DisGeNet group had 361 targets. By comparing the two sets of targets, a total of 16 overlapping targets were identified (Figure 1B). Moreover, the interaction network of these targets is displayed in Figure 1C.
Analysis of Targets of Gastrodin and Osteoarthritis. (A) Chemical Structure of Gastrodin. (B) Overlapping Targets of Gastrodin and Osteoarthritis. (C) The Interaction Network of These Overlapping Targets.
Possible Mechanism of Gastrodin Against Osteoarthritis
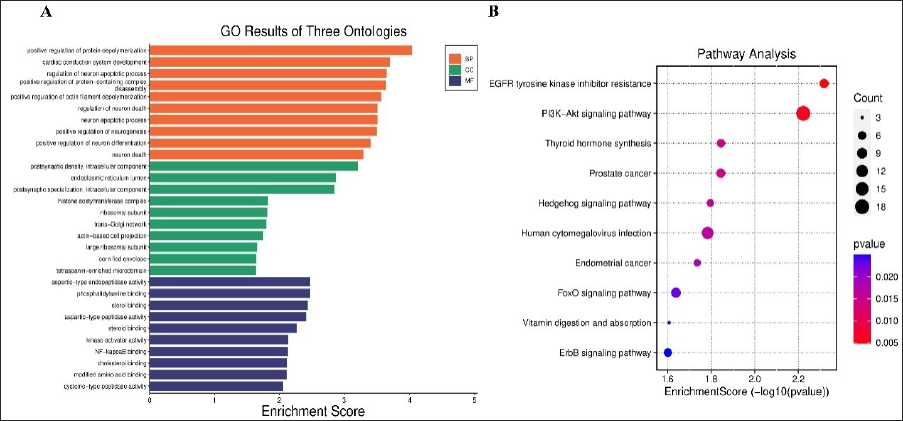
The possible mechanism of gastrodin against osteoarthritis was analyzed by GO annotation analysis (Figure 2A) and KEGG (Figure 2B) of 16 overlapping targets. KEGG clearly states that these genes were primarily located in EGFR tyrosine kinase inhibitor resistance, PI3K-Akt, thyroid hormone synthesis, prostate cancer, Hedgehog signaling pathway, human cytomegalovirus infection, endometrial cancer, FoxO signaling pathway, vitamin digestion, and absorption and ErbB signaling pathway. We speculate that gastrodin may prevent the development of osteoarthritis by regulating the FoxO pathway.
Enrichment Analysis of Overlapping Targets. (A) GO Analysis. (B) KEGG Analysis.
Gastrodin Relieves Mechanical Hyperalgesia in Rats With Osteoarthritis
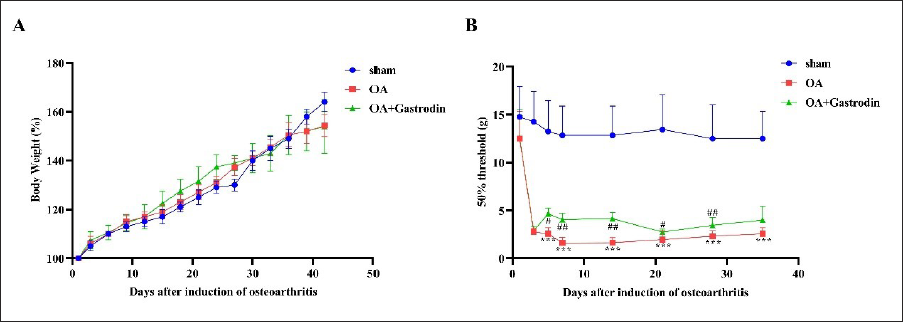
This study evaluated the effect of gastrodin on osteoarthritis rats by recording their body weight every week and testing them on different days after modeling mechanical pain threshold. The results showed a steady upward trend in the weight of rats as non-significant (p > 0.05) (Figure 3A). Interestingly, the rats in the OA model group showed a significant and continuous decrease in the 50% paw retraction threshold, indicating mechanical allodynia which continued during the observation period (p < 0.05). Daily subcutaneous injection of gastrodin after the operation alleviated mechanical hyperalgesia, with a significant reduction observed on the fifth day and maintained until the end of the observation period (p < 0.05) (Figure 3B).
Gastrodin Reduces Neutrophil Infiltration and Proinflammatory Cytokine Expression in the Synovium of Rats With Osteoarthritis
In Figure 4A, the neutrophil infiltration in the synovial tissue of rats in the OA model group was higher compared with the Sham group (p < 0.05). However, the gastrodin treatment could downregulate the number of neutrophils (p < 0.05). Similarly, the synovial score in the OA model group was higher compared with the Sham group, but the gastrodin treatment group showed a lower synovial score (p < 0.05) (Figure 4B). To explore the molecular mechanism of gastrodin, we utilized the Q-PCR method to evaluate the level of IL-1β and CXCL2 in synovial tissue. Our findings revealed that the level of IL-1β and CXCL2 was elevated in the OA model group (p < 0.05). However, the gastrodin treatment group showed a low level of IL-1β and CXCL2 in synovial tissue when compared with the OA model group (p < 0.05) (Figure 4C and D).
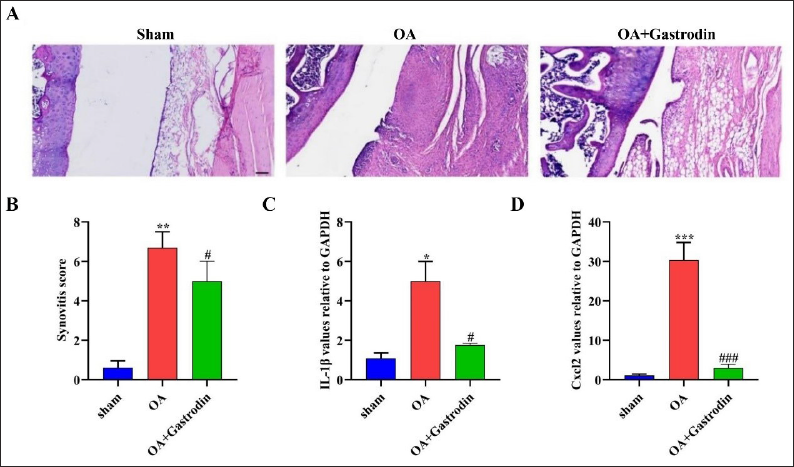
Gastrodin Inhibits the Release of Pro-inflammatory Cytokines in the Synovium of Rats With Osteoarthritis
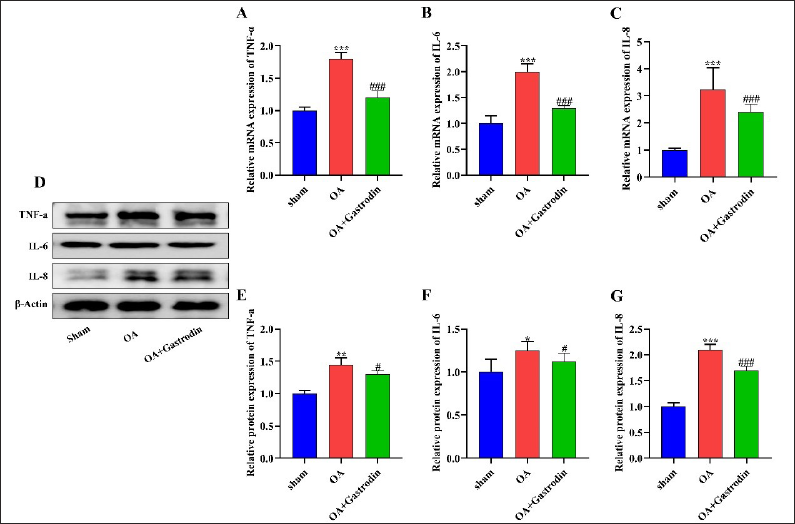
Q-PCR and Western blot were used to detect the expressions of TNF-α, IL-6, and IL-8 in the synovial tissue of rats. The results indicated that the levels of pro-inflammatory cytokines were enhanced in the synovial tissue of rats after modeling (p < 0.05), whereas the rats treated with gastrodin showed lower expressions of the three cytokines in comparison with the OA model group (p < 0.05) (Figure 5A–G).
Effect of Gastrodin on Cell Viability
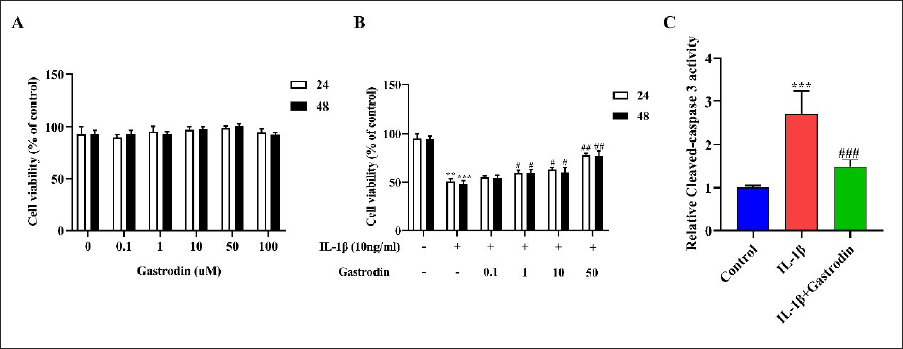
The CCK-8 experiment conducted in vitro found that different concentrations (i.e., 0.1, 1, 10, 50, 100 µM) of gastrodin did not have a significant impact on chondrocyte viability after 24 or 48 h (p > 0.05) (Figure 6A). However, chondrocytes treated with IL-1β (10 ng/mL) had significantly lower cell viability compared with the normal group (p < 0.05). Gastrodin-treated chondrocytes revealed higher cell viability (p < 0.05) (Figure 6B). Additionally, gastrodin was found to attenuate the increase of IL-1β-induced chondrocyte cleaved-caspase 3 activity (p < 0.05) (Figure 6C).
Gastrodin Attenuates Chondrocyte Inflammation Induced by IL-1β
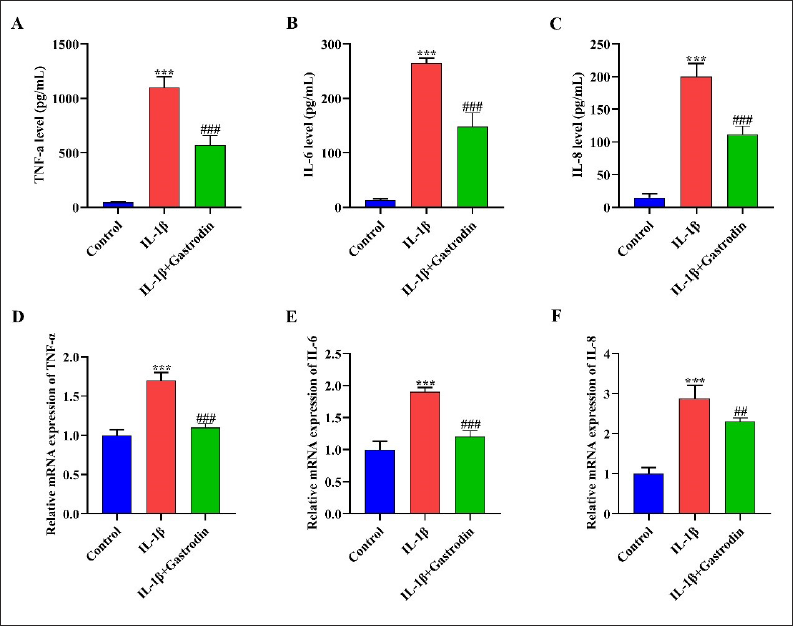
ELISA assay suggests that the expression of TNF-α, IL-6, and IL-8 in chondrocytes increased through treatment with IL-1β (p < 0.05). Gastrodin treatment attenuates the upregulation of IL-1β on its secretion (p < 0.05) (Figure 7A–C). Similarly, the Q-PCR experiment also confirmed the above results (p < 0.05) (Figure 7D–F).
Gastrodin Inhibits Extracellular Matrix Degradation Triggered by IL-1β
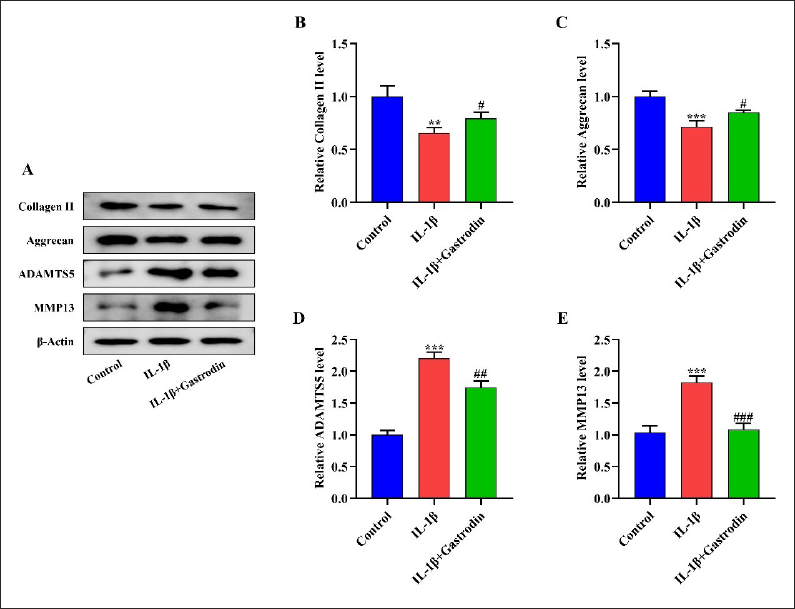
These experiments also demonstrated a significant decrease in the content of Collagen II and Aggrecan in chondrocytes stimulated by IL-1β (p < 0.05). Additionally, there was an increase in the protein expressions of ADAMTS5 and MMP13 (p < 0.05). However, the administration of gastrodin effectively reversed the effect of IL-1β on the aforementioned proteins in chondrocytes (p < 0.05) (Figure 8A–E).
Gastrodin Attenuates the Activation of PI3K/AKT/FoxO1 Signal Pathway in Chondrocytes Stimulated by IL-1β
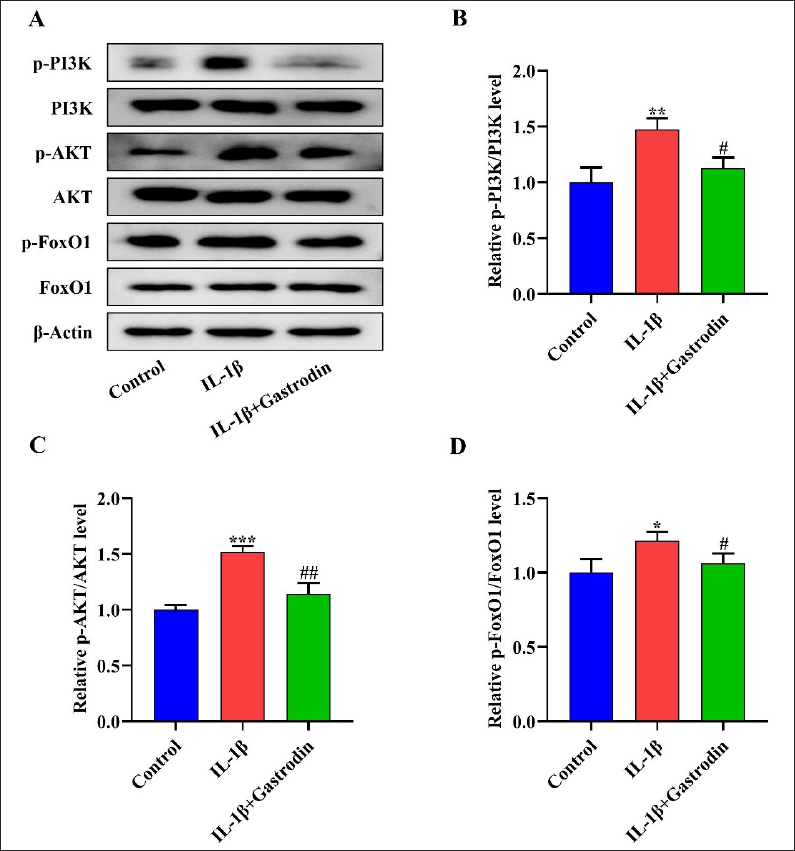
To investigate the influence of gastrodin on the PI3K/AKT/FoxO1 signal pathway in chondrocytes induced by IL-1β, a Western blot assay showed that IL-1β significantly increased the phosphorylation levels of PI3K, AKT, and FoxO1 in chondrocytes (p < 0.05). However, gastrodin attenuated the increase of PI3K, AKT, and FoxO1 phosphorylation induced by IL-1β in chondrocytes (p < 0.05) (Figure 9A–D).
Discussion
The MMx model is a widely used animal model in the study of osteoarthritis pathogenesis (Ogawa et al., 2016). The potential protective function of gastrodin in osteoarthritis was assessed by establishing an osteoarthritis rat model. As expected, the MMx-induced OA model was successfully established. The study observed the anti-inflammatory and analgesic effects of gastrodin on MMx-induced OA rats. Gastrodin was found to effectively attenuate MMx-induced inflammatory infiltration of OA rat knee articular cartilage. The study also found that gastrodin could restrain the release of IL-1β, IL-6, IL-8, and TNF-α in the knee joint tissue of MMx-induced OA rats and reduce the mechanical hyperalgesia of MMx-induced OA rats. These results suggest that gastrodin effectively attenuated inflammation and aches in OA rats.
The cytokine IL-1β plays a crucial role in the progression of osteoarthritis by causing chondrocyte inflammation, which leads to cartilage degeneration (Chen et al., 2018; Defois et al., 2023). Gastrodin has been found to have anti-inflammatory effects not only in IL-1β-treated chondrocytes but also in lipopolysaccharide-treated alveolar epithelial cells, doxorubicin-treated cardiomyocytes, and advanced glycation end products-treated human umbilical vein endothelial cells (Chen et al., 2018; Zhang et al., 2020). Former reports have indicated that IL-1β can cause chondrocyte inflammation and extracellular matrix degradation (Li et al., 2020), which we confirmed in our study. Other findings also showed that gastrodin relieved the inflammatory response triggered by IL-1β in chondrocytes, as well as in doxorubicin-treated cardiomyocytes, cerebral hemorrhage-induced neuroinflammation, and LPS-stimulated Raw264.7 cell inflammation (Huang et al., 2015). The response was unanimous. According to this study, gastrodin has been found to have a protective function on IL-1β-triggered chondrocyte injury by inhibiting extracellular matrix degradation and upregulating the protein amount of Collagen II and Aggrecan, while downregulating the protein expression of ADAMTS5 and MMP13. The study also suggests that the therapeutic potential of gastrodin in osteoarthritis is similar to that of nobility and glycyrrhizin (Fang et al., 2019). Glycyrrhizin treatment significantly inhibited IL-1β-stimulated PI3K/AKT phosphorylation and NF-κB mobilization in human OA chondrocytes. In vivo, glycyrrhizin treatment prevented the destruction of cartilage in mice OA models (Jiang et al., 2020).
Based on our analysis of the KEGG pathway, it was discovered that gastrodin has the potential to regulate the FoxO1 pathway in osteoarthritis. The FoxO1 transcription factor is responsible for regulating bone formation, and its absence can lead to cartilage loss in the later stages of fracture healing (Xiong et al., 2022). Furthermore, FoxO1 plays a critical role in maintaining cartilage homeostasis and extracellular matrix in osteoarthritis (Chen et al., 2018). It is important to note that the transcriptional activity of FoxO1 is reduced by the action of IL-1β (Cantu et al., 2022). The PI3K/AKT pathway is known to be the upstream signaling pathway of FoxO1 (Ren et al., 2020). Several studies have demonstrated that this pathway may contribute to the degradation of cartilage by regulating downstream pathways such as NF-κB and mTOR. This can promote apoptosis, inflammation, and degradation of the extracellular matrix in chondrocytes (Li et al., 2020). This study examined the effects of gastrodin on the PI3K/AKT/FoxO1 signaling pathway in chondrocytes when exposed to IL-1β. The findings indicate that IL-1β stimulation resulted in increased phosphorylation levels of PI3K, AKT, and FoxO1 in chondrocytes. However, treatment with gastrodin showed a decrease in phosphorylation levels of these proteins in IL-1β-stimulated chondrocytes. Subsequent studies will further investigate the effects of gastrodin on the PI3K/AKT/FoxO1 signaling pathway by utilizing signaling pathway inhibitors for intervention.
Conclusion
This research indicates that gastrodin has a pharmacological activity that can alleviate inflammation and related pathological changes in osteoarthritis in rats. Additionally, it has been shown to significantly reduce the occurrence of mechanical hyperalgesia. Gastrodin can prevent IL-1β-triggered inflammation and degradation of extracellular matrix in chondrocytes by regulating the PI3K/AKT/FoxO1 pathway. These findings suggest that gastrodin has therapeutic potential in treating osteoarthritis. In our follow-up research, our group will further investigate the pharmacological effects of gastrodin on OA. We aim to delve into the underlying mechanisms and endeavor to apply these findings in clinical research to explore the potential role of gastrodin in treating OA in humans.
Summary
Bioinformatics analysis predicted that gastrodin may act on the FoxO1 pathway and have 16 overlapping targets against osteoarthritis. In vivo experiments, observed through H&E staining, showed that gastrodin treatment significantly reduced neutrophil infiltration in the synovial tissue of osteoarthritic rat joints. In vitro studies have demonstrated that gastrodin can attenuate the inhibition of apoptosis, proinflammatory cytokine production, and extracellular matrix degradation induced by IL-1β. This effect is due to the inhibitory effect of gastrodin on the PI3K/AKT/FoxO1 signaling pathway. These findings suggest that gastrodin has therapeutic potential for the treatment of osteoarthritis.
Abbreviations
OA: Osteoarthritis; IL-1β: Interleukin-1β; GO: Gene ontology; KEGG: Kyoto encyclopedia of genes and genomes; CCK-8: Cell counting kit-8; ELISA: Enzyme-linked immunosorbent assay; Q-PCR: Quantitative real-time polymerase chain reaction; PI3K: Phosphatidylinositol 3-kinase; AKT: Protein kinase B; PLA2: Phospholipase A2: COX-2, Cyclooxygenase-2; PGE2: Prostaglandin E2; IL-6: Interleukin-6; TNF-α: Tumor necrosis factor-α; MMx: Medial meniscectomy; FoxO1: Forkhead box protein O1; LPS: Lipopolysaccharide.
Footnotes
Acknowledgments
We thank all the researchers from the Chengdu University of Traditional Chinese Medicine for their assistance in completing this experiment.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the National Natural Science Foundation of China (Number: 81973743) and the Research Foundation of Chengdu University of TCM (Number: 030/030029084).
Statement of Informed Consent and Ethical Approval
The ethics committee of Chengdu University of Traditional Chinese Medicine approved the animal care and experimental animal use protocols for this study. The ethics approval reference number for this study is 20230306.
