Abstract
Background
Lung cancer is a notorious disease due to its high malignancy and complex metabolic adaptations, often regulated by the Akt/mTOR pathway.
Objectives
This study investigates the anti-cancer potential of scopoletin, a naturally derived bioactive compound, on A549 lung cancer cells, focusing on its effects on cancer cell viability and metabolic state.
Materials and Methods
A549 cells treated with scopoletin were assessed using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assays and morphological analysis to evaluate cytotoxic effects. Dual staining and metabolic assays analyzed apoptosis induction and metabolic shifts, including lipid peroxidation and antioxidant enzyme (superoxide dismutase (SOD) and catalase (CAT)) activity. Real-time polymer chain reaction (RT-PCR) confirmed gene expression changes, and molecular docking using AutoDock evaluated scopoletin binding affinity to key targets within the Akt/mTOR-mediated pathways. Additionally, colony formation and cell migration assays demonstrated scopoletin’s ability to suppress lung cancer malignancy.
Results
Our results showed that scopoletin effectively stops A549 cell proliferation through apoptosis and disrupts aerobic glycolysis, a characteristic feature of lung cancer’s metabolic state. RT-PCR results showed the downregulation of genes associated with glycolysis and apoptosis, consistent with the observed cellular effects. Furthermore, scopoletin inhibited colony formation and cell migration, indicating its potential to limit lung metastasis. Docking studies confirmed the strong binding interactions between scopoletin and targets within the Akt/mTOR signaling pathway, suggesting a mechanism by which scopoletin modulates lung cancer cell metabolism.
Conclusion
These findings indicate scopoletin’s potential to inhibit A549 cell growth and induce significant metabolic alterations, making it a viable therapeutic candidate for lung cancer. Further research is required to substantiate scopoletin clinical applicability and enhance therapeutic approaches for lung malignancies.
Introduction
Cancer, a critical and deadly disease, ranks as the top reason for death globally. The increasing prevalence of cancer cases globally has raised substantial public health concerns, emphasizing the urgent need for innovative solutions and advanced research approaches (Mohanprasanth & Saravanan, 2024). Now, lung cancer continues to be among the worldwide health issues, being both highly prevalent and deadly. Lung cancer statistics underscore its global burden and the challenges faced in curbing its impact. The World Health Organization (WHO) expects that there will be roughly 2.2 million instances of lung cancer in 2020, making it one of the most commonly diagnosed cancers after breast cancer. The global age-standardized mortality rate for lung cancer is approximately 18 per 100,000 people, with significant variations across different regions. High-income countries, despite lower incidence trends, still report high lung cancer mortality rates due to aging populations and historical smoking patterns. Asia, which harbors nearly 60% of the global population, has seen rising lung cancer cases, particularly in China and India, driven by increasing industrialization and smoking rates. These data highlight the critical need for both greater early detection and more effective therapies. The 5-year survival rate for lung cancer is still low despite improvements in detection and therapy, particularly for patients who are identified at an advanced stage, highlighting the urgent need for novel therapeutic strategies (Li et al., 2023).
The leading cause of lung cancer is tobacco smoking, which accounts for 80%–90% of all cases. Many carcinogens included in tobacco smoke, including nitrosamines and polycyclic aromatic hydrocarbons, cause DNA alterations that result in the transformation of malignant cells. Additional risk factors include being exposed to asbestos, radon, and air pollution, which contribute to DNA damage and inflammation in lung tissues (Xue et al., 2014). Genetic factors also contribute to lung cancer susceptibility, as evidenced by mutations in genes like TP53, EGFR, and KRAS (Foggetti et al., 2021). Moreover, some hormones are involved in cancer progression, such as growth, metastases, and invasion. For instance, the androgen hormone regulates the TMPRSS2 expression in prostate cancer invasion and metastasis (Ramachandran et al., 2024). Similarly, the reported overexpression of androgen hormones is observed in lung cancer (Berardi et al., 2016). Inflammation has also been implicated as a contributor to lung cancer, with chronic respiratory diseases like chronic obstructive pulmonary disease (COPD) increasing the risk of lung malignancies. Thus, both environmental and genetic factors play critical roles in lung cancer etiology, highlighting the need for personalized prevention and treatment strategies (Forder et al., 2023).
Disruptions in key cellular signaling pathways largely drive the progression and metastasis of lung cancer. Among the most affected are the phosphoinositide 3-kinase (PI3K)/AKT/mTOR, Ras/Raf/MEK/ERK, and JAK/STAT pathways, which regulate essential cellular functions such as proliferation, survival, and metabolism. Dysregulation of these pathways promotes cancer cell survival, resistance to apoptosis, and metastasis (Steelman et al., 2011). The PI3K/AKT/mTOR pathway, in particular, is frequently altered in lung cancer, often due to mutations in upstream regulators like EGFR or PTEN loss-of-function mutations, leading to its persistent activation (Sanaei et al., 2022). Hyperactivation of this pathway drives uncontrolled cell proliferation, invasion, and resistance to chemotherapy, making it a critical focus in lung cancer research. The AKT/mTOR signaling pathway plays a central role in regulating metabolism, cell growth, and survival (Pungsrinont et al., 2021). In lung cancer, aberrant activation commonly results from mutations or amplifications in receptor tyrosine kinases (RTKs), such as EGFR or ALK, which trigger PI3K activation and subsequent phosphorylation of AKT. Once activated, AKT promotes cell survival by inhibiting apoptotic proteins, while mTOR, a downstream effector, regulates cell growth and metabolism through mRNA translation and autophagy. Together, these molecular events sustain cancer cell survival and progression, making the AKT/mTOR pathway a promising therapeutic target. Inhibiting this pathway has the potential to suppress tumor growth and enhance chemotherapy sensitivity, offering new avenues for lung cancer treatment (Tomuleasa et al., 2024).
Plant-derived remedies hold significant potential for treating various diseases while sparing normal cells from harm. The unique properties of phytochemicals, such as their ability to regulate the cell cycle, prevent apoptosis, and influence key processes like angiogenesis and metastasis, have drawn considerable research interest. Over the past two decades, the use of complementary and alternative therapies has risen substantially in countries like the USA and India. Notably, nearly 60% of anti-cancer drugs are derived from medicinal plants and other natural sources (Aruchamy & Duraisamy, 2021). We hypothesize that scopoletin (6-methoxy-7-hydroxycoumarin) can inhibit the AKT/mTOR pathway in lung cancer cells, thereby suppressing cancer progression. In multiple cancer models, scopoletin, a naturally occurring substance obtained from a variety of medicinal plants, has demonstrated anti-cancer properties, such as apoptosis induction, cell growth suppression, and signaling pathway modification. By targeting the AKT/mTOR pathway, scopoletin may impede cancer cell survival and proliferation, representing a promising therapeutic strategy for lung cancer. The study aims to evaluate the efficacy of scopoletin in modulating AKT/mTOR pathway activity and its effects on lung cancer progression.
Materials and Methods
Material
Scopoletin (CatLog No.: S2500-50MG; Brand: Sigma-Aldrich), Micro Lipid Peroxidation (MDA) Kit (CatLog No.: KTB1050), Micro Superoxide Dismutase (SOD) Activity Assay Kit (CatLog No.: KTB1030), and Micro Catalase (CAT) Activity Assay Kit (CatLog No.: KTB1040) were all obtained from Abbkine, USA.
Data Mining
We conducted analyses on the dataset utilizing the compound target prediction database (CTD) (
Functional Annotation by Search Tool for Interacting Chemicals (STITCH)
Progress in unraveling the intricate relationships between pharmaceutical compounds and proteins largely depends on computational approaches that identify specific protein targets for drug molecules. The STITCH database (
Cell Lines
A549 human lung cancer cells were cultured and maintained under controlled conditions in a CO2 incubator set at 37°C. The cells were propagated using Dulbecco’s Modified Eagle Medium (DMEM) growth medium supplemented with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin antibiotics.
Cell Viability Assay
To evaluate the cytotoxic effects of scopoletin on the A549 cell line, we employed the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay as described by Jayaraman et al. (2023). The experiment began by seeding 5,000 cells and incubating them overnight with varying concentrations of scopoletin. After incubation, MTT was added to the cells, followed by solubilization with dimethyl sulfoxide (DMSO), and the optical density (OD) value was observed at 590 nm. Furthermore, the morphology of A549 cells was observed and documented at 10× magnification using an Olympus inverted microscope.
Glucose Uptake Assay
A549 cells (1 × 104) were seeded into 96-well plates and incubated for 24 h to allow for cell attachment and growth. The cells were then exposed to scopoletin for 48 h. Following the treatment, they were rinsed twice with Krebs–Ringer-Phosphate-HEPES (KRPH) buffer. Glucose uptake was evaluated using a colorimetric assay kit, adhering to the manufacturer’s guidelines. Cell lysates were prepared and treated with a 2-deoxy-d-glucose (2DG) enzymatic mixture, and the absorbance was recorded at 450 nm after 1 h of incubation.
Lactate Dehydrogenase (LDH) Assay
After incubation periods, cells were collected and lysed, and then the LDH enzyme activity was evaluated by using an LDHA enzyme assay kit (Catalog: MAK066; Brand: Sigma).
Apoptosis Analysis by Staining Methods
After the incubation period, the six-well plate was washed with 1X phosphate buffer saline (PBS). Acridine orange/ethidium bromide (AO/EtBr) staining and 4′,6-diamidino-2-phenylindole (DAPI) staining were added. Incubate for 30 min, and the excess dye was removed by rinsing with 1X PBS.
Evaluate the Reactive Oxygen Species (ROS) and Antioxidant Activity
After the incubation period, the cells were fixed with 4% paraformaldehyde, followed by dichlorofluorescein diacetate (DCF-DA) staining and an additional 15-min incubation. Subsequently, proteins were extracted using a protein lysis buffer. Lipid peroxidation levels and antioxidant enzyme activities, including SOD and CAT, were analyzed immediately.
RNA Isolation and mRNA Expression Analysis
After the incubation period, RNA was extracted using the TRIzol reagent (TAKARA, Japan) and quantified using a NanoDrop spectrophotometer. The isolated RNA was promptly reverse-transcribed into cDNA using the SuperScript IV Reverse Transcriptase kit (TAKARA) and stored at −20°C for subsequent analysis. The mRNA expression of target genes was assessed using specific primers listed in Table 1. Quantitative analysis of mRNA expression was performed using the SYBR Green Master Mix (Thermo).
Real-time Polymer Chain Reaction (RT-PCR) Primers.

Cell Migration Assay
A549 cells were seeded at a density of 1 × 106 cells per well in six-well plates, and they were left to proliferate until they achieved 95% confluency. In the middle of each well, a scratch was made with a sterile 200 µL pipette tip. To get rid of any debris, the wells were washed with PBS after the medium was aspirated. The wells were then filled with 1.5 mL of serum-free media, either with or without scopoletin. Using a 10× magnification, pictures of the scratch area were obtained at the beginning (t0) and 24 h into the incubation period (t24). Using ImageJ software, the wound distances at t0 and t24 were measured, and the wound closure % was computed (Vang Mouritzen & Jenssen, 2018).
Colony Formation Assay
Each 60 mm petri dish had 200 cells in total, and the media were changed on a regular basis. The cells were treated with or without scopoletin after 3–4 days, and they were then given another 4 days to grow. The media were disposed of at the conclusion of the incubation time, and 1X PBS was used to rinse the cells. After adding 1 mL of ice-cold methanol to each dish for fixation, the dishes were promptly put in a freezer set at −20°C. Following fixation, 1 mL of a 0.5% crystal violet solution was applied to each dish, and the dishes were then left in the dark for half an hour. After that, the extra crystal violet was gently washed away with tap water.
Molecular Docking
Ligand and Protein Docking Analysis
The study investigates how scopoletin (CID: 5280460) binds to several proteins involved in the regulation of cancer cells. Crystal structures of the targeted proteins, which were obtained from the Protein Data Bank (PDB) (
Statistical Analysis
The statistical results of the study were expressed as mean values with their corresponding standard deviations. The software program GraphPad Prism 8 was used to analyze the data. The significance thresholds for t-tests were .05, .01, and .001 (p values).
Results
Scopoletin Efficiency in Lung Cancer by Bioinformatic Analysis
This study utilized a range of bioinformatic tools to identify key genes involved in lung cancer regulation. Drug-based databases were initially employed to determine differentially expressed genes (DEGs). A Venn diagram (Figure 1a) highlighted 17 significant genes that showed notable interactions with scopoletin in lung cancer cells. Further analysis of the interactive networks and nodal strength of these genes was conducted using the STITCH server, as shown in Figure 1b, to find the closure interlinked with scopoletin using Cytoscape (Figure 1c). Figure 1d demonstrates that the six hub genes involved in the scopoletin and lung cancer list are AKT1, PI3K, LDHA, GLUT1, mTOR, and EIF4EBP1.
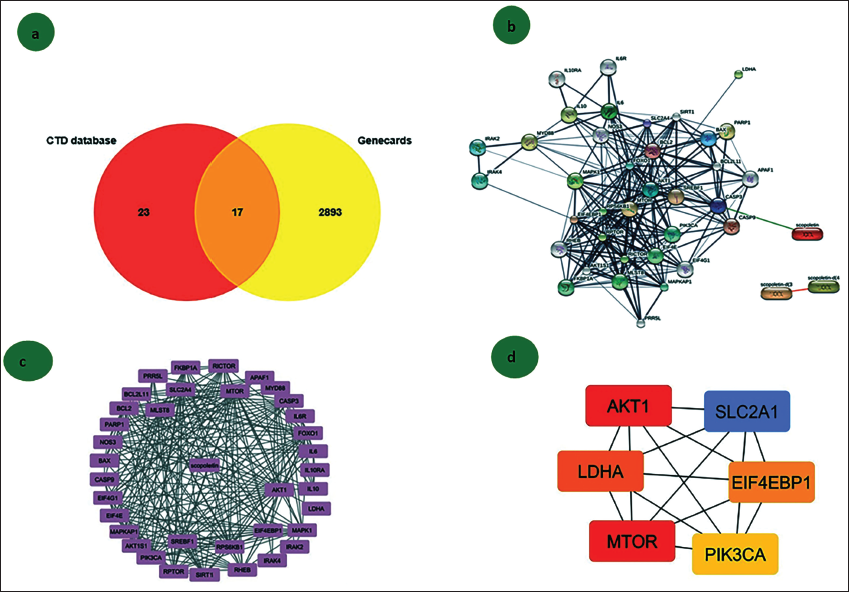
Cytotoxicity Effect of Scopoletin in Lung Cancer A549 Cell Line
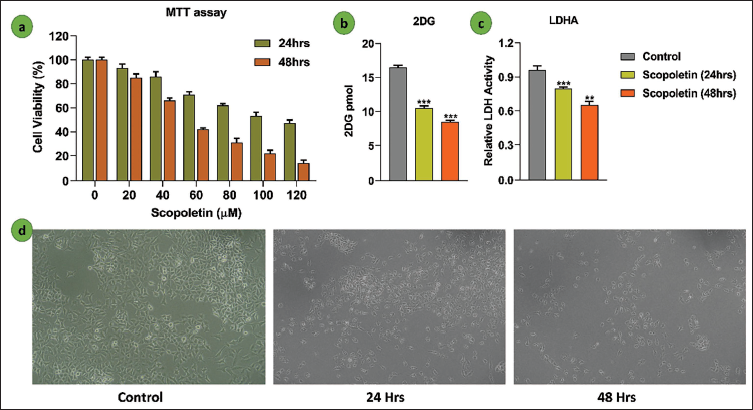
The MTT assay was used to assess scopoletin’s effect on cell proliferation. Scopoletin significantly reduced the growth of A549 cells; this effect became noticeable after incubation for 24 and 48 h. The variations in cell viability percentages between cells treated with scopoletin and control cells are displayed in Figure 2a. Subsequent studies were conducted using concentrations of 110 and 48 µM/mL of scopoletin, which decreased cell viability by 50% at two separate time intervals. Additionally, the glucose uptake assay using 2-DG demonstrated that scopoletin inhibits glucose uptake, as shown in Figure 2b. LDHA, a key enzyme in cancer cell survival, was suppressed by scopoletin, as represented in the graph in Figure 2c. Figure 2d reveals that the scopoletin-treated group exhibited a reduced cell population and altered A549 cell morphology compared to the untreated group.
(a) The Effectiveness of Scopoletin (0–120 µM) for 24 and 48 h of Treatment was Evaluated Using the 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) Test. The Mean ± Standard Deviation (SD) is Used to Represent the Data. Software Called GraphPad Prism 8 was Used to Calculate the Half Maximal Inhibitory Concentration (IC50) Value. (b) 2DG Analysis of the Glucose Uptake Assay. The Mean ± SD of Three Separate Experiments is Used to Represent the Data. Statistically Significant as Shown by ***p < .001. (c) The LDHA Enzyme Assay is Used to Compare the Aerobic Glycolysis in Cells Treated with Scopoletin to the Control Group. The Mean ± SD of Three Separate Experiments is Used to Represent the Data. A Statistically Significant Difference is Shown by **p < .01 and ****p < .001. (d) Morphological Pictures of the A549 (20× Magnification) were Examined at 24-h Intervals with Control and Silymarin Treatment.
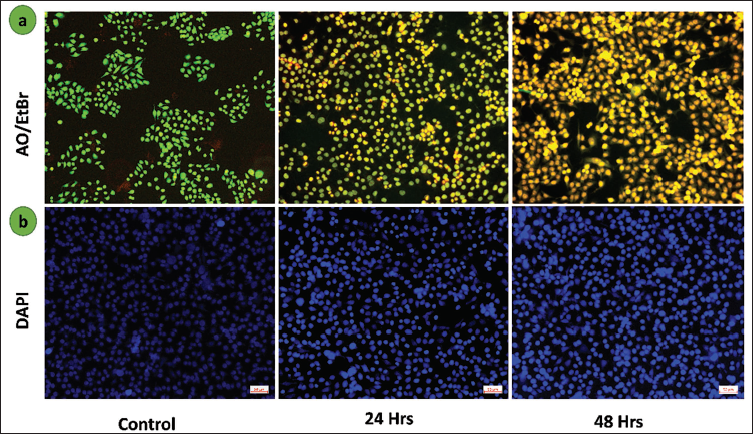
Scopoletin Induces Apoptosis in Lung Cancer A549 Cells
When comparing the scopoletin-treated group to the control group, Figure 3a shows a considerable red fluorescence of dual AO/EtBr fluorescence intensity, indicating enhanced cell death (apoptosis) in lung cancer A549 cells. As seen in Figure 3b, the untreated group had homogenous, intact nuclei, whereas the treated group displayed uneven nuclear morphology and indications of apoptosis. These findings imply that scopoletin changes the nuclear integrity of treated cells and causes apoptosis.
(a) Lung Cancer Apoptosis Induced by Scopoletin Acridine Orange/Ethidium Bromide (AO/EtBr) Dual Labeling was Used to Analyze A549 Cells. (b) A Fluorescence Microscopic Image of 4′,6-Diamidino-2-Phenylindole (DAPI) Staining was Used to Observe the Nuclear Condensation in the Group Treated with Scopletin Against the Control Group. 50 µm is the Scale Bar.
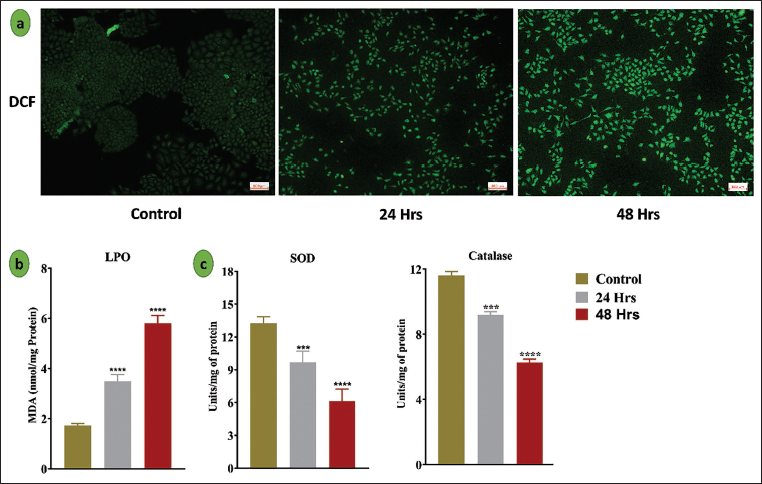
Scopoletin Alters the Intracellular ROS and Anitoxidant Enzyme Activity in Lung Cancer A549 Cell Line
Using DCF-DA (2′,7′-dichlorofluorescin diacetate) labeling, the effect of scopoletin on intracellular ROS production in A549 lung cancer cells was evaluated, as illustrated in Figure 4a. ROS production was much higher in the scopoletin-treated group than in the control group. In addition, malondialdehyde (MDA) levels in control and scopoletin-treated A549 cells were quantified to determine the degree of lipid peroxidation. With MDA levels increasing to 3.4 and 5.8 nM/mg protein at two distinct time intervals, scopoletin administration significantly increased lipid peroxidation, as seen in Figure 4b, suggesting increased oxidative stress in the cells. Additionally, measurements were made of the activities of enzyme antioxidants, such as CAT and SOD (Figure 4c).
Scopoletin Inhibits the A549 Cell Metastasis
The cell migration experiment was used to evaluate scopoletin’s impact on the migratory behavior of A549 lung cancer cells. As shown in Figure 5a, the wound area in the scopoletin-treated group showed a substantial decrease in wound closure compared to the control group (Figure 5b), with only 40% and 67.5% closure seen after 24 and 48 h, respectively. The impact of scopoletin on cell proliferation was also assessed using a colony formation test. As seen in Figure 5c, cells treated with scopoletin generated notably fewer and smaller colonies than untreated cells, indicating a strong inhibitory effect on colony formation. As shown in Figure 5d, the scopoletin-treated group produced 238 colonies in comparison to the control group.
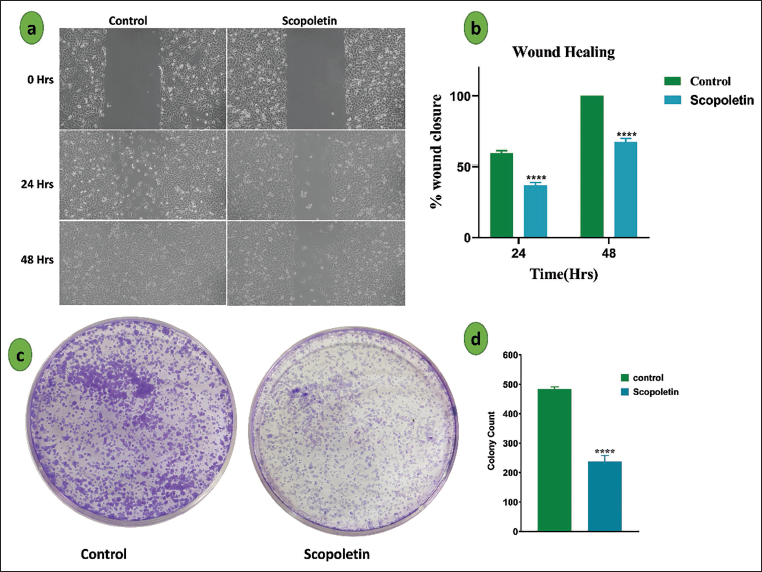
Scopoletin Inhibits the Proliferation of Cancer Cells
Additionally, after treating A549 lung cancer cells with scopoletin at its half maximal inhibitory concentration (IC50) dose, the study measured the mRNA expression levels of many apoptosis-related genes using RT-PCR. The findings, which are displayed in Figure 6, included a number of noteworthy findings. Notably, during 24 and 48 h, scopoletin therapy gradually reduced the mRNA expression of several genes involved in metabolic control, such as PI3K, AKT, EIF4EBP1, GLUT1, mTOR, and LDHA. These genes are linked to the control of apoptosis, cell division, and glycolysis. All of these results point to scopoletin’s modulation of the PI3K/AKT signaling pathway and regulatory effects on A549 cells.
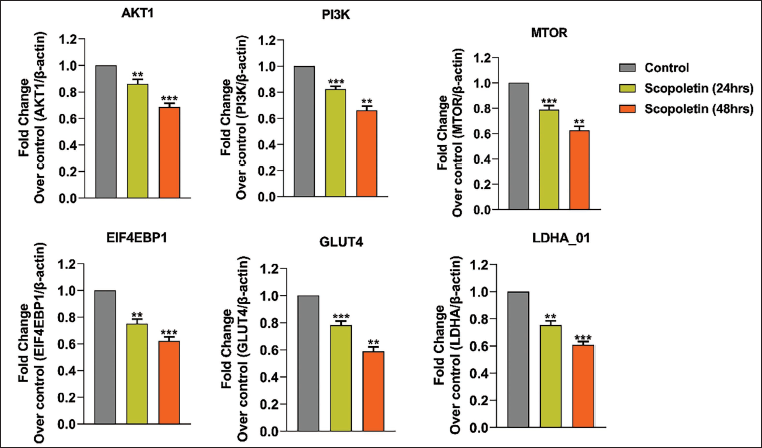
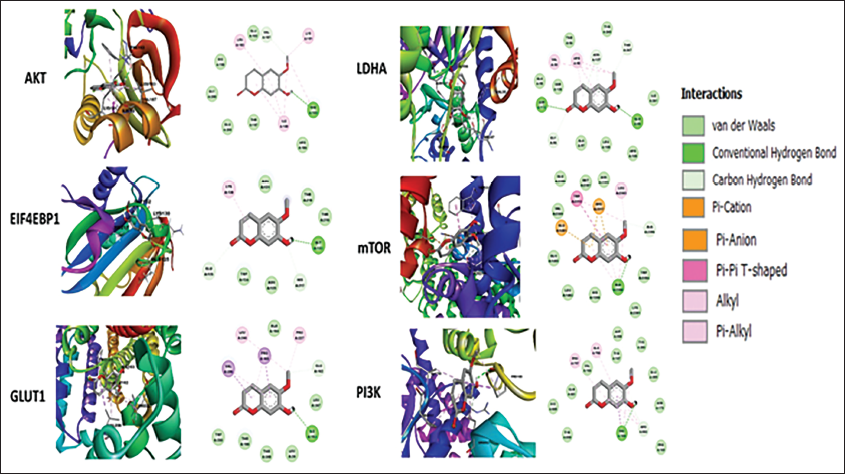
Molecular Docking Scopoletin and Signaling Targets
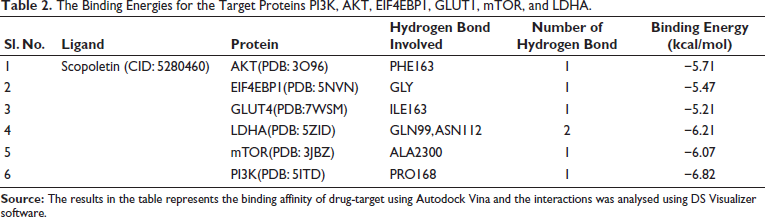
Molecular docking is a vital tool in drug discovery, as it allows for the identification of binding energy and affinity between drugs and target proteins. AutoDockTools version 1.5.6 was used to analyze the binding energy between scopoletin and the target proteins. The 2D and 3D views were visualized using Discovery Studio 2021, as shown in Figure 7. The binding energies for the target proteins PI3K, AKT, EIF4EBP1, GLUT1, mTOR, and LDHA were −6.82, −5.71, −5.47, −5.21, −6.07, and −6.21 kcal/mol, respectively, as presented in Table 2. Notably, PI3K exhibited the lowest binding energy of −6.82 kcal/mol. Scopoletin formed a hydrogen bond with the PRO168 residue of PI3K, and these interactions play a key role in regulating cell death in cancer cells, ultimately inhibiting lung cancer cell proliferation.
The Binding Energies for the Target Proteins PI3K, AKT, EIF4EBP1, GLUT1, mTOR, and LDHA.
Discussion
Cancer tends to rank among one of the biggest causes of death, and issues like metastasis, antibiotic resistance, and the dysregulation of important cellular functions frequently reduce the survival rate (Emran et al., 2022). Scopoletin, a natural coumarin derivative, has recently gained attention for its potential therapeutic effects in cancer treatment. This discussion aims to elaborate on how scopoletin induces ROS production and inhibits cancer cell survival signaling pathways, with particular focus on its modulation of key metabolic genes and its effect on cell migration in cancer cells. Similarly, Natarajan et al. (2024) used a bioinformatics tool to predict FOXM1 gene biomarkers in colorectal cancer.
Initially, we predict the target using bioinformatic tools, followed by picking up the top five hub genes involved in the cancer progression, as well as the scopoletin target, as depicted in Figure 1a–1d. Next, we used the MTT assay method to examine scopoletin’s cell viability against the human lung cancer A549 cell line. The viability of lung cancer A549 cells was reduced by the elevated scopoletin concentration, which also affected the IC50 value. Furthermore, we saw that the cells in the control groups had a distinct morphology and resembled pebbles, while the lung cancer cells in the groups treated with scopoletin displayed the characteristics of apoptosis. Condensed nuclei, cell shrinkage, membrane blebbing, apoptotic bodies, and echinoid spikes are all signs of apoptosis. Compared to the control cells, a greater percentage of cells in the experimental groups displayed floating behavior (Syed Abdul Rahman et al., 2013). Therefore, results from morphological analyses and MTT assays indicate that scopoletin and phyto-compounds have the ability to stop cell division and cause cell death. Scopoletin derivatives have been shown to exhibit cytotoxicity in the hepatocellular carcinoma HepG2 cell line, as previously reported by Zhao et al. (2014) as SC-III3.
ROS are extremely reactive molecules formed during regular cellular metabolism. In cancer cells, ROS are often elevated due to their increased metabolic activity, but these elevated levels can lead to cellular damage, lipid peroxidation, and protein oxidation (Srinivasan et al., 2024). Interestingly, cancer cells exploit ROS for growth and survival, but excessive ROS can overwhelm the cell’s antioxidant defense system, inducing oxidative stress and cell death. Thus, the modulation of ROS levels can provide a therapeutic strategy in cancer treatment. Zhao et al. (2014) reported that scopoletin derivatives, such as SC-III3, show that oxidative stress mediates DNA damage in the hepatocellular carcinoma HepG2 cell line. Anantharaju et al. (2017) indicate that cinnamic acid induces ROS-mediated cell death in cervical and colon cancer. Similarly, scopoletin, a polyphenol, acts as a prooxidant; we observed that it significantly induced ROS production in lung cancer cells, which subsequently triggered oxidative damage within the cells. ROS-induced cellular damage disrupts key cellular functions, including DNA repair, protein synthesis, and apoptosis regulation, thus contributing to the inhibition of cancer cell proliferation and survival. Increased ROS levels lead to the activation of pro-apoptotic signaling pathways, further promoting cell death in cancer cells. Evidently, dual staining by AO/EtBr staining indicates that scopoletin induces apoptosis cell death. The study findings indicate that scopoletin’s ability to generate ROS plays a crucial role in its anti-cancer effects, suggesting its potential as a ROS-inducing agent in cancer therapy.
Moreover, cancer cells often exhibit altered metabolism, a phenomenon known as the Warburg effect, in which cells rely on aerobic glycolysis instead of oxidative phosphorylation to meet their energy demands (Liberti & Locasale, 2016). This metabolic reprogramming is driven by several key signaling pathways and genes that regulate cellular growth, survival, and migration. Scopoletin’s impact on these metabolic pathways, particularly the PI3K/AKT signaling axis, has been a focus of recent studies. The PI3K/AKT pathway is one of the most critical survival signaling pathways in cancer cells. PI3K activation leads to the phosphorylation of AKT, which in turn activates downstream targets involved in cell growth, survival, and metabolism. PI3K/AKT pathway dysregulation is commonly seen in a variety of malignancies and is linked to higher cell proliferation, improved metabolic activity, and resistance to apoptosis (He et al., 2021). In the present study, scopoletin was found to downregulate the expression of PI3K and AKT in A549 lung cancer cells, leading to the inhibition of this survival pathway. The reduced expression of these genes suggests that scopoletin interferes with the PI3K/AKT pathway, impairing cancer cell survival and potentially sensitizing cells to apoptosis. Parra-Perez et al. (2022) demonstrate that hydroxytyrosol, a polyphenol, inhibits the PI3K/AKT signaling pathway in acute human leukemia T cells. Epigallocatechin-3-gallate induces apoptosis by altering the PI3K/AKT/mTOR signaling axis in human pancreatic cancer (Liu et al., 2013). Yang et al. (2023) revealed that p-coumaric acid inhibits osteosarcoma by modulating the PI3K/AKT signaling.
Glucose metabolism is essential for cancer cell survival and proliferation, with GLUT1, a high-affinity glucose transporter, playing a pivotal role by facilitating glucose uptake for glycolysis and adenosine triphosphate (ATP) production. Overexpression of GLUT1 in cancer cells supports their heightened metabolic demands associated with rapid growth (Heydarzadeh et al., 2020). In this study, scopoletin treatment significantly reduced GLUT1 expression in A549 cells, impairing glucose uptake and glycolytic activity, thereby disrupting the metabolic reprogramming that is critical for energy generation and cell division. Similarly, LDHA, which catalyzes the conversion of pyruvate to lactate during anaerobic glycolysis, a key aspect of the Warburg effect, was also downregulated by scopoletin, suggesting its potential to inhibit glycolysis and energy production. This dual inhibition of GLUT1 and LDHA by scopoletin hampers the ability of cancer cells to adapt to metabolic stress, ultimately reducing their survival. Shi et al. (2022) demonstrated that Apigenin alters the glucose transport by PI3L/AKT/mTOR pathway to reduce osteosarcoma. Reyes-Farias and Carrasco-Pozo (2019) listed that Quercetin inhibits the glucose transporter in various cancer models.
From our findings, scopoletin altered the cancer metabolism, which could inhibit the cancer cell migration and colony formation, to inhibit cell proliferation and survival. The ability of cancer cells to migrate and invade surrounding tissues is a key feature of metastasis, which is one of the primary causes of cancer-related mortality. Similarly, previous Zhao et al. (2018), resveratrol inhibits renal cancer migration via modulating AKT/ERK1/2. Migration is regulated by a complex interplay of signaling pathways, including those involved in cell adhesion, cytoskeletal remodeling, and matrix degradation. The inhibition of key metabolic pathways, such as PI3K/AKT, EIF4EBP1, and GLUT1, can disrupt the signaling mechanisms required for cell migration.
Conclusion
In conclusion, scopoletin exhibits potent anti-cancer effects by inducing ROS production and modulating key signaling pathways involved in cancer cell survival, metabolism, and migration. Through the downregulation of metabolic genes such as PI3K, AKT, EIF4EBP1, GLUT1, mTOR, and LDHA, scopoletin effectively inhibits cancer cell proliferation and migration, providing a promising therapeutic strategy for cancer treatment. The ability of scopoletin to target multiple aspects of cancer cell metabolism and signaling pathways highlights its potential as a multifaceted agent for cancer therapy, warranting further investigation in preclinical and clinical settings.
Footnotes
Abbreviations
AKT: Serine/threonine kinase 1; AO/EtBr: Acridine orange/ethidium bromide; CAT: Catalase; CTD: Compound target prediction database; DCF-DA: Dichlorofluorescein diacetate; DEGs: Differentially expressed genes; 2DG: 2-Deoxy-d-glucose; DMEM: Dulbecco’s Modified Eagle Medium; EIF4EBP1: Eukaryotic translation initiation factor 4E binding protein 1; EMBL: European molecular biology laboratory; GLUT4: Glucose transporter type 4; KRPH: Krebs–Ringer-Phosphate-HEPES; LDH: Lactate dehydrogenase; MDA: Malondialdehyde; mTOR: Mammalian target of rapamycin; MTT: 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; PBS: Phosphate buffer saline; PDB: Protein databank; RT-PCR: Real-time polymer chain reaction; SOD: Superoxide dismutase.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
