Abstract
Background:
Momordica cochinchinensis (MC) is shown with high antioxidation and is used as herbal medicine in South-East countries.
Objectives:
This study aims to determine the four parts (aril, fruit, leaf, and seed) of MC extract against human breast cancer cells with underlying mechanisms of actions.
Materials and Methods:
The effects of MC extract were examined by sulforhodamine B, colony formation, wound healing, real time polymerase chain reaction (RT-PCR), and Western blotting method.
Results:
Four parts of MC extracts significantly increased MCF-7 cells death, interestingly, seed extract had the greatest activity with IC50 values of 412.3 ± 49.8 µg/mL for 24 h and 113.3 ± 6.4 µg/mL for 48 h. MC extracts potentiated the anticancer drug effect, doxorubicin, with lower IC50 values when compared with MC treatment alone. Moreover, MC extracts decreased colony formation and accompanied by inhibited cell migration. Furthermore, Seed extract inhibited migration by reducing matrix metalloproteinases (MMP) 2, MMP 9, and vascular endothelial growth factor A (VEGFA) inhibition. The molecular analyses revealed that seed extract inhibited cell viability by decreasing cyclin D1 consistent with activating p21, cytochrome c, and caspase-3 protein levels along with reduction of mevalonate (MVA) pathway, Rac1, and RhoA levels, it appeared to inhibit growth and migration.
Conclusion:
Taken together, MC extract induces MCF-7 cell death and decreases cell migration relatively via attenuation of MVA pathway.
Introduction
Momordica cochinchinensis (MC) belongs to Cucurbitaceae family. MC is one of the most valued food and is used as a herbal medicine1, 2 in East and Southeast Asia including Vietnam, Thailand, Laos, Myanmar, and Cambodia. 3 MC fruits are known as baby jackfruit or “gac”, and several parts of MC, such as aril, seed, leaf, and fruit, have been reported to have pharmacological effects. Several data have been reported that MC has numerous pharmacological effects to treat many disease conditions such as infections, immune-stimulating, antioxidant, anti-inflammatory, and anticancer actions. 3
Focusing on anticancer effects, seeds of MC have been shown to suppress 4 lung cancer 5 and breast cancer.4, 6 The two major compounds of saponins in MC reduce lung cancer cel1 proliferation in all cell types tested, Calu-6, A549, H1299, and H1264. 5 In breast cancer cells, the MC seeds with ethyl acetate extract decreased cell cycle arrest in G2 stage and were associated with reducing cyclin D1 expression. 7 Moreover, MC extract stimulated cell apoptosis via upregulating tumor suppressor gene (p53), Bax and downregulating nuclear factor kappa B (NF-κB), Bcl-2. 7 Consistent with aril MC extract, the data indicated that the extract caused induction cell apoptosis by decreasing Bcl-2 gene along with enhancing caspase 6, 8, and 9 activity. 6 Another effect of MC on cancer cells is inhibiting the cancer cells migration by significantly inhibiting the expression and secretion of matrix metalloproteinases (MMP) 2 and MMP 9 in breast cancer cells. 4 Furthermore, in vivo study of the fruit of MC extract has been indicated that the extract attenuated the colon cancer cells proliferation and suppressed angiogenesis in Balb/c mice. 8 MC extract can cause the induction of cancer cells death and inhibition of cancer cells migration.
As an ongoing investigation of anticancer activities of MC and less information to compare each part of MC including aril, fruit, leaf, and seed, on the human breast MCF-7 cancer cells, this study explored the efficacy of four parts of MC extracts on cell proliferation, cell migration, and effects on mevalonate (MVA) pathway in MCF-7 cells.
Materials and Methods
The Plant Extraction
Four parts of MC including aril, fruit, leaf, and seed, were obtained from Udon Thani Province, Thailand, in April 2020. The species identification of MC was done by Asst. Prof. Pornpimon Wongsuwan, staff of the Applied Thai Traditional Medicine Department, Faculty of Medicine, Mahasarakham University and then kept at the herbarium at the Faculty of Science, Mahasarakham University (Herbarium no. MSUT-7229). For extraction, MC was dried, mashed, and macerating them in 95% (v/v) ethanol for seven days at room temperature. The extract was then filtered, evaporated, and lyophilized, and the crude extract was kept at −20°C. The percentage yield of extract of aril, fruit, leaf, and seed was 5.89%, 9.23%, 5.14%, and 8.87% per dry weight of MC.
The Cell Culture and Cell Cytotoxic Method
The breast MCF-7 cancer cell line was obtained from ATCC (Manassas, VA, USA) and cultured from the instruction. The cell culture and cytotoxic method were operated essentially as described previously. 9 To determine the effect of four parts of MC extract on breast MCF-7, cancer cells were grown by using sulforhodamin B (SRB). Cancer cells were plated onto 96-well culture plates overnight and incubated with MC extracts at the concentration of 0–500 µg/mL for 24 and 48 h or in combination of MC extract with anticancer drug, doxorubicin, for 48 h and measured cell viability by SRB method. 9
Colony Formation Method
To determine the effect of four parts of MC extract on cell growth by using colony formation, 9 the cancer cells were cultured onto 6-well culture plates at the densities of 500 cells/well overnight, incubated with four parts of MC extract (0–100 µg/mL) for 24 h, cultured for another 15 days, fixed with 100% methanol, stained with 0.5% crystal violet, and counted the colony formation.
Wound Healing Method
To determine the effect of four parts of MC extract on MCF-7 cells migration by using wound healing assay 9 cancer cells were plated onto 24-well plates overnight, made the wound, incubated with MC extract for 72 h, and took the wound images.
Gene Expression Method
To determine the effect of seeds of MC extract on cells gene expression (MMP2, MMP9, vascular endothelial growth factor A (VEGFA), ICAMP1, Rac1, RhoA, and ACTB), real time polymerase chain reaction (RT-PCR) was performed. 9 The cancer cells were plated onto six well-plates overnight, treated with seed extract for 24 h, and the total RNA was isolated. Further, 1 µg of total RNA was converted to cDNA and cDNA products were then used as a template in real-time PCR. The primer sequences were as follows and ACTB was used as an internal control (Table 1). The relative expression of each gene was quantified using 2-∇∇Cq method.
Primer Sequences for RT-PCR.
Protein Extraction and Western Blot Method
To determine the effect of seed of MC extract on cells protein expression, Western blotting was performed. 9 The cancer cells were plated onto six well plates overnight and then exposed to the extract for 24 h, lysed, and then the protein concentration was measured. The protein lysates were loaded on 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE), transferred to a polyvinylidene fluoride (PVDF) membrane, blocked with 2.5% albumin, incubated overnight with the primary antibodies at 4°C against Rac1 (1:1000), RhoA (1:1000), Cyclin D1 (1:1000), p21 (1:1000), cytochrome c (1:1000), caspase 3 (1:1000), and β-actin (1:2500), and then incubated with secondary antibody (1:2500). After exposing with substrate, the band densities were visualized, read, and captured.
Statistical Analysis
The Student’s t-test was used to compare the control and treatment groups with *p < 0.05. The data are expressed as mean ± SE.
Results
MC Extracts Decreased Cells Viability and Potentiated the Doxorubicin Effect
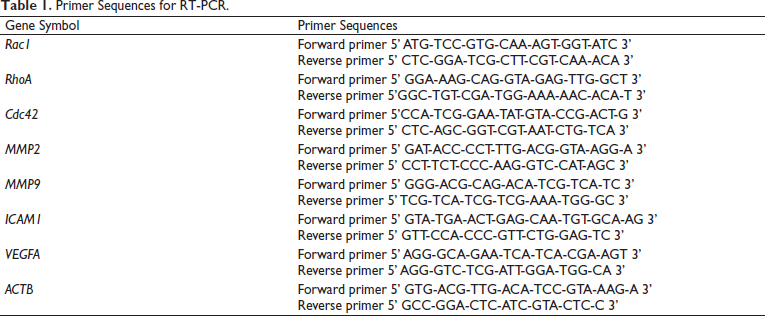
The data indicated that four parts of MC composed of aril, fruit, leaf, and seed, decreased cancer cells viability. Moreover, the seed of MC extract had the highest activity against the cells with the IC50 values of 412.3 ± 49.8 µg/mL for 24 h and 113.3 ± 6.4 µg/mL for 48 h (Figure 1A–E). The cytotoxic activity showed the greatest from seed>leaf>fruit>aril of MC extracts.
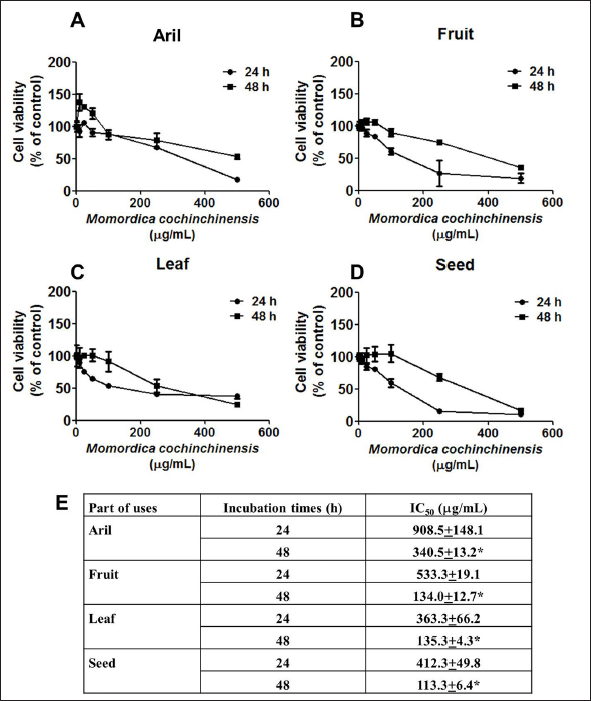
For more information, in combination with MC extracts with anticancer drugs, doxorubicin was used. The results demonstrated that cancer cells had undergone death after exposure to four parts of MC extract with doxorubicin (Figure 2A–E). The fruit extracts of MC had higher activity when combined with the doxorubicin, the IC50 values of fruit MC extract alone was 226.39 ± 41.49 µg/mL and MC extract plus doxorubicin was 25.67 ± 6.67 µg/mL, respectively; however, fruit and seed extract showed similar results.
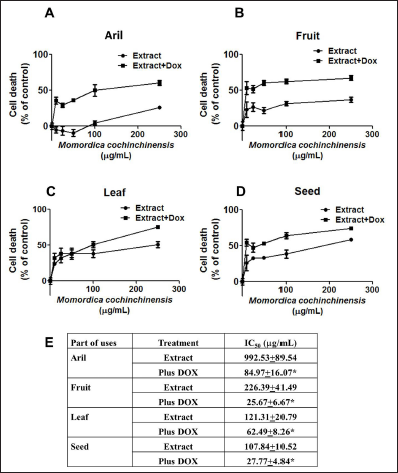
In the colony formation study, four parts of MC extracts inhibited the colony formation by dose-dependent manner with low IC50 values (Figure 3A–D). The extract from aril had the greatest effects to inhibit colony formation than seed>fruit>leaf extract, respectively, with IC50 values was 8.45 ± 2.40, 1078 ± 1.53, 29.99 ± 6.17, 46.83 ± 4.96 µg/mL. The results indicated that MC extract significantly suppresses cell growth, potentiates doxorubicin effects, and decreases the formation of MCF-7 cells colony.
MC Extracts Inhibited Cells Migration by Reducing Gene-Related Migration
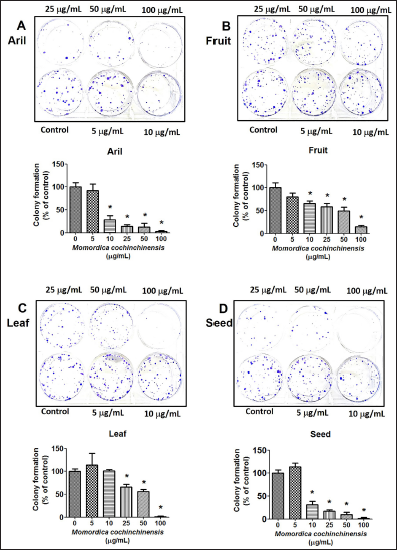
The reduction of migratory effect of four parts of MC extract on cancer cells was examined by using wound healing assay and RT-PCR. After cancer cells were incubated with different doses of four parts of MC extract and the results indicated that all of MC extracts inhibited cell migration by increasing the uncovered area between two sides (Figure 4A–E). Interestingly, seed extract had more efficacy than the three parts of MC extract; therefore, in the next experiments the seed extract was used to explore gene and protein expression which related cell viability and migration.
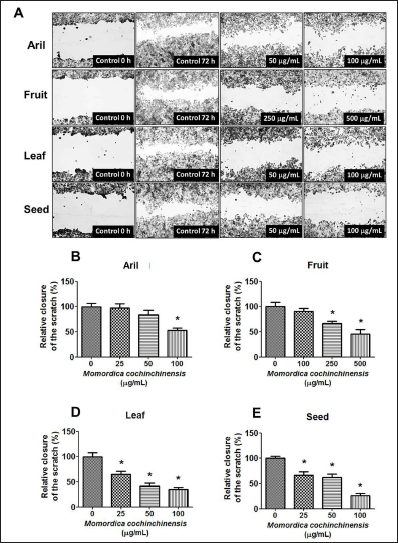
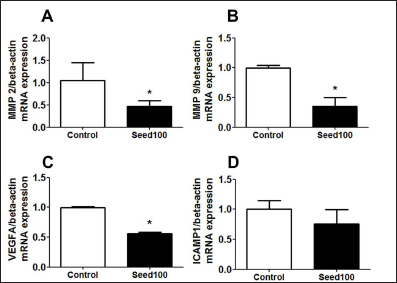
The gene-related migration was examined at the MMP2, MMP9, VEGFA, ICAMP1 levels by using RT-PCR. Seed extract significantly downregulated MMP2, MMP9, VEGFA levels but did not affect the ICAMP1 levels (Figure 5A–D). These data suggested that MC seed extract treatment significantly suppressed cancer cells migration by reducing MMP2, MMP9, VEGFA expression levels in MCF-7 cells.
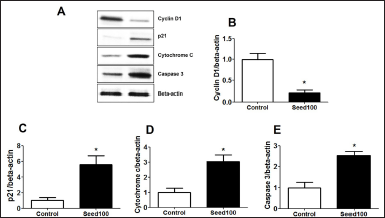
MC Seed Extract Reduced Cell Proliferation by Decreasing Protein-Related Cell Death and Apoptosis
The effects of MC seed extract on MCF-7 cells death were examined by Western blotting method. The results showed that MC seed extract significantly suppressed cyclin D1 accompanied with activating the p21, cytochrome c, and caspase-3 to induce cancer cell death and cell apoptosis (Figure 6A–E).
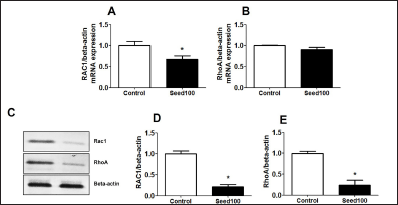
MC Seed Extracts Decreased MVA Pathway in MCF-7 Cells
Subsequently, the important involvement of MVA pathway to control cell proliferation and migration of cancer cells has been testified, the effect of MC seed extract on this MVA cascade in MCF-7 cells was determined in this study. The data indicated that Rac1 gene was significantly suppressed in seed extract-treated cells, suggesting the inhibitory effect of this extract on the MVA pathway (Figure 7A–B) with correlating Rac1 and RhoA protein suppression (Figure 7C–E). The data obtained showed that seed extract significantly inhibits MVA pathway including Ra1 and RhoA expression.
Discussion
A little information is available on the anticancer actions of the parts of MC. The one part of MC which has been explored is the aril extract, indicating the effect to suppress human breast cancer cells proliferation. 6 The data indicated that aril of MC extract which had high levels of lycopene showed high cytotoxic effects and anti-estrogencity to MCF-7 cells inducing apoptosis through enhancing caspase 6, 8, and 9 activity. For non-edible parts of MC such as seed extract, showed quite extensively that seed extract inhibited the breast cancer cells and induced apoptosis by interfering mitochondrial and MAPK pathway. 7 Like this study, the seed of MC extract inhibited the growth by stimulating p21 and suppressing cyclin D1 levels. p21 (CDK inhibitor) is a general cell cycle inhibitor straight controlled by both p53-dependent and p53-independent pathways, it suppresses tumor colony growth by decreasing cyclin protein in the cell cycle. 10 Moreover, the induction of p21 protein activated the cell cycle arrest at G2/M stage along with a reduced polyploid fraction of cells, which implies a functional G2/M arrest. 11 The induction of p21 and reduction of cyclin D1 from seed of MC extract caused activation of cell death or inhibition of cell growth in MCF-7 cells.
The stimulation of apoptosis by MC was shown in the human breast cancer cells as well. MC extract-induced cancer cells apoptosis was focused on the extract from seed involving the caspase cascade, resulting in DNA fragmentation, chromatin condensation, blebbing, organelles disintegrates, and apoptotic bodies.12, 13 This study indicated the mechanism by which MC-induced apoptosis stimulated the caspase cascade by activating the cytochrome c and caspase 3 protein levels after treating with seed extract of MC. Caspase enzymes are key proteins of programmed cell death/apoptosis and caspase 3 is a final step protein in apoptotic pathway to activate protease enzyme, finally apoptosis occurred. 14 Moreover, cytochrome c activates the assembly of apoptosome via association with Apaf 1, which initiates the cascade of caspase step. 15 Therefore, the natural products caused induction of apoptosis on cancer cells by activating caspase cascade.
Further, metastasis of cancer characterizes a progressive step of malignancy and is the leading cause of cancer-associated deaths or inducing the secondary tumor. 16 Several herbal medicines have been reported to show significantly antimigratory effects on many cancer cell types.9, 17, 18 Our results showed that seed extracts attenuated the basal migratory ability of MCF-7 cells with down-regulating of the expression of metastasis-associated genes such as MMP2, MMP9, VEGFA, and ICAMP1. The inhibitory effect on MMP2 and MMP9 levels has also been reported in the extract from the seed of MC extract. 4 Moreover, VEGFA and ICAMP1 were inhibited when treated with the plant extract and correlated with antimigration. 17 For more information, the plant extract inhibited renal cell migration by inhibiting VEGFR-3 pathway 19 and interfering MMPs through ERK, JNK, p38, and PI3K/Akt Signaling Pathways. 20 These results suggest that seed of MC extract may possibly exert this effect via suppression migratory pathway.
Numerous studies have suggested the involvement of MVA pathway in carcinogenesis, aggressive and metastasis of cancers including breast cancer; we now investigate how the extracts modulate the MVA pathway. This pathway produces the metabolites, which is an important role for the activity of Rac and Rho downstream signaling, which affect growth, invasion, differentiation, angiogenesis, migration, and apoptosis. 21 We have indicated that seed extract inhibits Rac1 gene expression; however, it did not show the effect on RhoA gene levels. Furthermore, the extract caused inhibition of MVA products both of Rac1 and RhoA protein expression, which suppresses cancer cell proliferation and migration. Therefore, we need to explore another aspect of the MVA pathway in detail, comprising the upstream and downstream signaling pathways.
Conclusion
MC extracts has a potent cytotoxic, apoptotic, and antimetastatic activity, maybe through modulating the MVA pathway, especially seed extract. This may provide a new approach to human breast cancer prevention and chemotherapy.
Summary
Four parts of MC extract showed a potent cytotoxic induction, apoptotic stimulation, and migratory inhibition through decreasing the Rac1 and RhoA in MVA pathway. The higher effects were found in the seed extract by 95% ethanol. This may provide a new approach to human breast cancer prevention and chemotherapy.
Footnotes
Abbreviations
Acknowledgment
The authors are grateful to Mahasarakham University Faculty of Medicine for equipment support.
Author’s Contributions
BB and RK designed and conducted the experiments. BB analyzed data and wrote the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research project is financially supported by Mahasarakham University (Fast track 2020).
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
