Abstract
Objective
To assess the effects of resveratrol (RSV) on expression of adhesion molecules in endothelial progenitor cells (EPCs) following tumor necrosis factor-α (TNF-α) stimulation.
Methods
EPCs were treated with RSV and stimulated with TNF-α. A mononuclear cell (MNC) adhesion assay was used to assess the effects of RSV on TNF-α-induced MNC adhesion. Vascular cell adhesion molecule-1 (VCAM-1), intercellular adhesion molecule-1 (ICAM-1) and E-selectin expression levels and nuclear factor κB (NF-κB) activation were assessed by immunoblotting.
Results
MNC adhesion to TNF-α-treated EPCs and VCAM-1/ICAM-1/E-selectin levels in EPCs were increased following TNF-α stimulation and decreased following RSV treatment. TNF-α enhanced NF-κB inhibitor α (IκB-α) phosphorylation in the cytosol as well as nuclear NF-κB p65 levels, both of which were decreased by RSV.
Conclusions
These findings provide new insights into RSV’s anti-inflammatory and anti-atherosclerotic effects. RSV’s mechanism of action might involve downregulation of VCAM-1, ICAM-1 and E-selectin by partial blockade of TNF-α-induced NF-κB activation and IκB-α phosphorylation in EPCs.
Keywords
Introduction
Light to moderate red wine consumption reduces the risk of cardiovascular disease. Intake of red wine markedly reduced the incidence of coronary artery disease (CAD) in the populations of certain areas of France despite their high-fat diets, sedentary lifestyles, and widespread cigarette smoking. This phenomenon has been termed the “French paradox” and has been attributed to the beneficial effects of resveratrol (RSV). RSV is naturally found in foods such as grapes and berries. 1 RSV protects the cardiovascular system, reducing platelet adhesion and aggregation, inhibiting neointimal hyperplasia of damaged vessels, and protecting myocardial cells from ischemia-reperfusion injury.2–4 In addition, RSV exerts anti-inflammatory and anti-atherosclerotic effects. 5 , 6
Atherosclerosis, the primary cause of most common cardiovascular disorders, is a slow and progressive inflammatory process. Atherosclerosis involves increased expression of cellular adhesion molecules, 7 glycosylated proteins found on activated endothelial cells. 8 Cellular adhesion molecules are required for monocyte and lymphocyte adhesion to endothelial tissue.9–11 The current study focused on vascular cell adhesion molecule-1 (VCAM-1, upregulated in acute myocardial infarction), intracellular adhesion molecule-1 (ICAM-1, elevated in coronary circulation in unstable angina cases), and E-selectin (a biomarker of atherosclerosis and coronary heart disease).12–14
Endothelial progenitor cells (EPCs) can circulate, proliferate and differentiate into mature endothelial cells (ECs), but are devoid of markers that characterize mature ECs and do not form a lumen. VCAM-1, ICAM-1, and E-selectin are expressed by EPCs and have essential functions in the atherosclerotic process. 15 , 16 Blood EPCs might adhere to mononuclear cells (MNCs) via VCAM-1, ICAM-1 and E-selectin, contributing to the pathogenesis of atherosclerosis. 17
We hypothesized that RSV might exert its anti-inflammatory and anti-atherosclerotic effects by downregulating expression of adhesion molecules in EPCs. Therefore, RSV was assessed for its capacity to regulate EPC adhesion molecule expression induced by tumor necrosis factor-α (TNF-α). In addition, we investigated whether the effects of RSV were controlled by nuclear factor κB (NF-κB) activation.
Materials and methods
Cell culture
Experiments involving animals were carried out in accordance with the principles laid out in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. All animal studies were approved by the Ethical Board of the First Affiliated Hospital, School of Medicine, Zhejiang University (Zhejiang, China). Male Sprague-Dawley rats (6–7 weeks old, 200 g) were fed a standard diet of rodent chow.
Rat bone marrow-derived EPCs were cultured according to previously published methods. 18 , 19 In brief, MNCs from rat femurs were purified by density gradient centrifugation and cultured in EBM-2 medium (Lonza, Basel, Switzerland) containing 10% fetal bovine serum (FBS) (Gibco, Carlsbad, CA, USA). Rapidly adhering mature ECs and hematopoietic cells were removed by aspirating non-adherent cells and transferring them into new culture dishes after 24 and 48 hours. The resulting cells were grown in EBM-2 medium supplemented with EGM-2 MV, 10% FBS, vascular endothelial growth factor, epidermal growth factor, fibroblast growth factor-2, insulin-like growth factor-1 and ascorbic acid. Non-adherent cells were washed away after 4 days. The medium was refreshed at 3 day intervals.
Immunofluorescence
EPCs were detected among adherent MNCs after 14 days of culture. Cells were treated at 37°C with 2.4 μg/mL 1,1-dioctadecyl-3,3,3,3-tetramethylindocarbocyanine-labeled acetylated low-density lipoprotein (diI-LDL) (Molecular Probes, Eugene, OR, USA), fixed with 2% paraformaldehyde for 10 minutes, and treated with 10 μg/mL fluorescein isothiocyanate-conjugated
Cell proliferation assay
The effects of RSV and TNF-α on EPC proliferation were determined using a 3-(4, 5-dimethylthiazol -2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assay. After culturing for 7 days, cells were digested with 0.25% trypsin and then cultured in EBM-2 medium containing 10% FBS in 96-well culture plates (200 μL/well). After culturing for 48 hours, the supernatants were discarded by aspiration and serum-free EBM-2 medium was added. RSV (0, 1, 10, 25, and 50 μM) was added (200 μL/well). After incubating for 18 hours, EPCs in each well were treated with TNF-α (10 ng/mL) and cultured for 6 hours. Each well received 20 μL of MTT (5 g/L, Fluka Co., Buchs, Switerzerland) and the cells were incubated for another 4 hours. The supernatants were aspirated and the EPCs was shaken with 150 μL of dimethyl sulfoxide for 10 minutes. The absorbance was measured at 490 nm.
MNC adhesion assay
EPC monolayers were pretreated for 18 hours with RSV at various concentrations, then stimulated with TNF-α for another 6 hours. Meanwhile, MNCs were treated with 10 mM 2',7'-bis-(2-carboxyethyl)-5-(and-6)-carboxyfluorescein, acetoxymethyl ester for 1 hour. After washing, 2 × 105 MNCs were incubated with EPCs for 30 minutes at 37°C under a humidified atmosphere containing 5% CO2. Following two washes with phosphate-buffered saline (PBS), five randomly selected high power fields (100×) were observed to assess cell adhesion.
Nuclear protein isolation
EPCs treated with RSV (0, 1, 10, 25, and 50 μM for 18 hours) and TNF-α (10 ng/mL for 6 hours) were used for nuclear protein extraction. After EPC washing (PBS containing 5 mM NaF and 1 mM Na3VO4), a hypotonic buffer (20 mM HEPES, pH 7.9, containing 20 mM NaF, 1 mM Na2P2O7, 1 mM Na3VO4, 1 mM egtazic acid, 0.5 mM phenylmethylsulfonyl fluoride, 1 mM dithiothreitol and 1 μg/mL leupeptin) was used to lyse cells. Cell nuclei were resuspended in a high salt hypotonic buffer supplemented with 20% glycerol and 420 mM NaCl for 30 minutes at 4°C. The samples were centrifuged to yield nuclear extracts in supernatants. Finally, the samples were dialyzed for 5 hours at 4°C into 20 mM HEPES, pH 7.9, containing 100 mM KCl, 0.2 mM ethylenediaminetetraacetic acid , 0.5 mM dithiothreitol, 0.5 mM phenylmethylsulfonyl fluoride and 20% glycerol.
Immunoblotting
Protein amounts in cell lysates were measured using a bicinchoninic acid assay kit (Beyotime, Beijing, China). Equal amounts of total protein were resolved by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and electro-transferred onto polyvinylidene fluoride membranes (Millipore, Burlington, MA, USA). The blocked membranes were incubated with primary antibodies targeting VCAM-1, ICAM-1, E-selectin, β-actin (1:1000, Santa Cruz Biotechnology, Dallas, TX, USA), NF-κB p65, phosphorylated inhibitor of NF-κB (IκB)-α, and IκB-α (1:1000, Cell Signaling Technology, Danvers, MA, USA) for 1 hour. After washing, the membranes were incubated with horseradish peroxidase-conjugated goat anti-mouse IgG antibody (1:3000, MultiSciences, Hangzhou, China) for 1 hour. The blots were developed using enhanced chemiluminescence substrate (Thermo, Waltham, MA, USA) and X-ray film. ImageJ software (National Institutes of Health, Bethesda, MD, USA) was used to quantitate protein bands.
Statistical analysis
Data were presented means ± standard deviations (SDs). Differences among multiple groups were assess using analysis of variance. All statistical analyses were conducted using SPSS version 16.0 (SPSS Corp., Chicago, IL, USA). Values of P < 0.05 were considered statistically significant.
Results
EPC characterization
Following 14 days of culture, MNCs showed a spindle-shaped, EC-like morphology (Figure 1a). EPCs were adherent cells positive for both DiI-LDL uptake and lectin binding by fluorescence microscopy (Figure 1b–d).
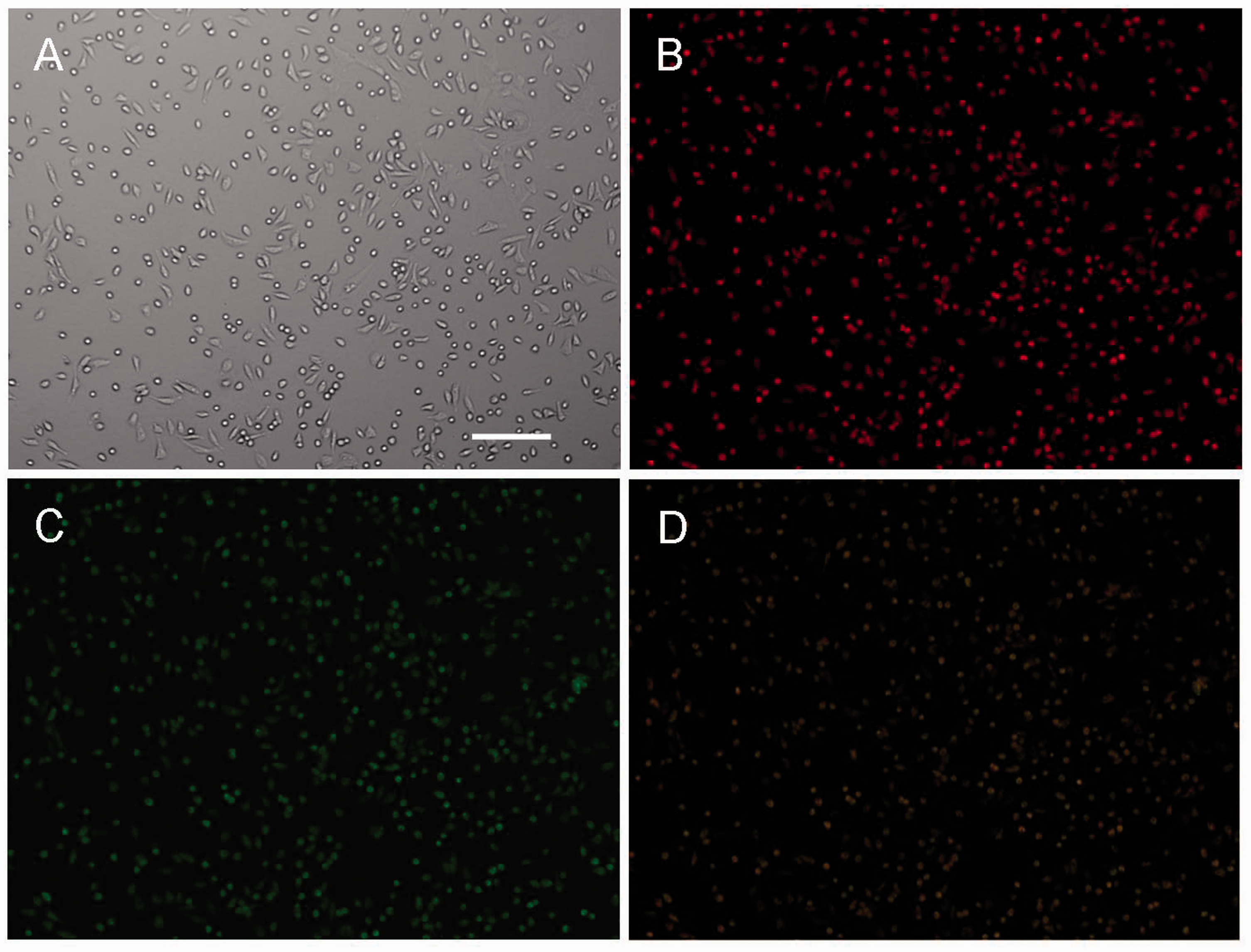
Immunofluorescence microscopy and immunophenotyping of bone marrow-derived EPCs.
Effects of RSV on TNF-α-induced proliferation of EPCs
To rule out potential cytotoxic effects of RSV and TNF-α on EPCs, cell viability was assessed by incubating EPCs with various concentrations of RSV and TNF-α for the indicated durations. Treatment with RSV (0, 1, 10, 25, and 50 μM) for 18 hours and with TNF-α (10 ng/mL) for 6 hours had no significant impact on cell viability (data not shown). These results showed that RSV and TNF-α did not have cytotoxic effects on EPCs.
Effects of RSV on TNF-α-induced MNC-EPC adhesion
To investigate whether RSV altered EPC adhesion following TNF-α treatment, EPCs were treated with RSV (0, 1, 10, 25, and 50 μM) for 18 hours and with TNF-α (10 ng/mL) for 6 hours. Thereafter, MNCs and EPCs were co-cultured to assess adhesion. In the absence of TNF-α, MNCs barely adhered to EPCs, while TNF-α treatment markedly enhanced MNC adhesion to EPCs. RSV pretreatment reduced TNF-α-induced MNC adhesion to EPCs in a concentration dependent fashion. Overt reductions in adhesion were observed at 10 μM RSV (TNF-α vs. 10 μM RSV + TNF-α: 191.8 ± 25.0 vs. 131.2 ± 12.4, P < 0.05) and minimal adhesion was observed at 50 μM RSV (TNF-α vs. 50 μM RSV + TNF-α: 191.8 ± 25.0 vs. 66.6 ± 7.4, P < 0.01) (Figure 2a, b).
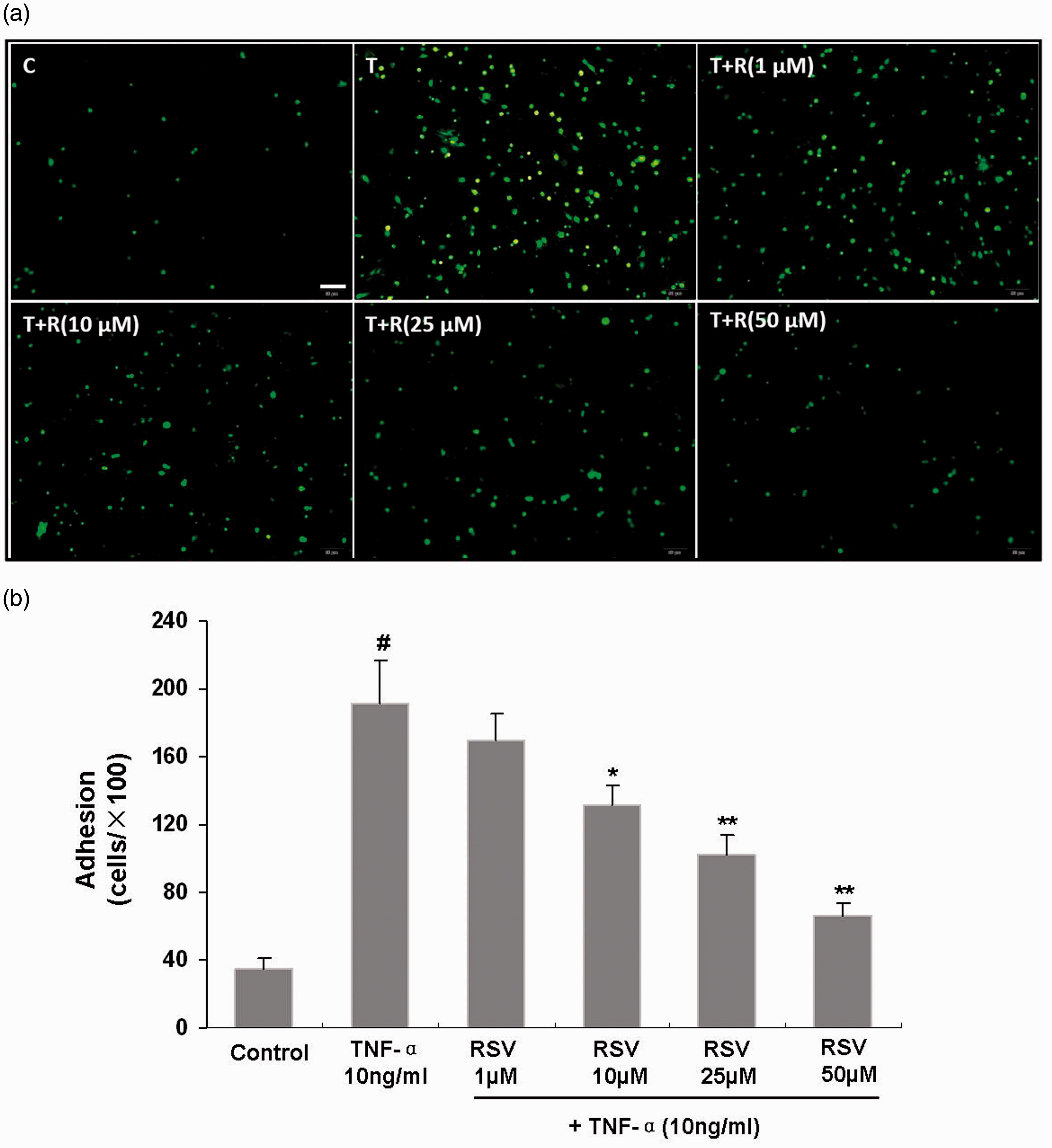
Effects of RSV on TNF-α-induced adhesion of EPCs and MNCs.
Regulatory effects of RSV on VCAM-1, ICAM-1 and E-selectin expression in TNF-α-treated EPCs
Expression of the adhesion molecules VCAM-1, ICAM-1, and E-selectin was assessed by immunoblotting. VCAM-1, ICAM-1, and E-selectin levels were low in unstimulated EPCs, but became starkly elevated following TNF-α stimulation for 6 hours (514.8 ± 54.0% increase in VCAM-1 expression compared with control cells, P < 0.01; 261.0 ± 39.3% increase in ICAM-1 expression compared with control cells, P < 0.01; 280.3 ± 30.3% increase in E-selectin expression compared with control cells, P < 0.01). RSV pretreatment (0, 1, 10, 25, and 50 μM) for 18 hours inhibited TNF-α-induced upregulation of VCAM-1, ICAM-1, and E-selectin expression in EPCs in a concentration-dependent fashion. Overt reductions were observed at 10 μM RSV for VCAM-1 (TNF-α vs. 10 μM RSV + TNF-α: 514.8 ± 54.0% vs. 413.5 ± 39.1% increase in VCAM-1 expression compared with control cells, P < 0.05), 25 μM RSV for ICAM-1 (TNF-α vs. 25 μM RSV + TNF-α: 261.0 ± 39.3% vs. 206.9 ± 22.6% increase in ICAM-1 expression compared with control cells, P < 0.05), and 10 μM RSV for E-selectin (TNF-α vs. 10 μM RSV + TNF-α: 280.3 ± 30.3% vs. 232.3 ± 28.9% increase in E-selectin expression compared with control cells, P < 0.05). Minimal TNF-α-induced upregulation was observed at 50 μM RSV (VCAM-1, TNF-α vs. 50 μM RSV + TNF-α: 514.8 ± 54.0% vs. 246.1 ± 23.6% increase in VCAM-1 expression compared with control cells, P < 0.01; ICAM-1, TNF-α vs. 50 μM RSV + TNF-α: 261.0 ± 39.3% vs. 124.4 ± 16.2% increased ICAM-1 expression compared with control cells, P < 0.01; E-selectin, TNF-α vs. 50 μM RSV + TNF-α: 280.3 ± 30.3% vs. 130.3 ± 14.3% increased E-selectin expression compared with control cells, P < 0.01) (Figure 3a, b).

Effects of RSV on VCAM-1, ICAM-1, and E-selectin expression in EPCs.
Effects of RSV on NF-κB p65 translocation in TNF-α-treated EPCs
To evaluate whether NF-κB activation and nuclear translocation contributed to the regulatory effects of RSV on adhesion molecule expression, nuclear protein amounts of NF-κB p65 were quantitated in TNF-α-stimulated EPCs by immunoblotting.
Elevated nuclear NF-κB p65 protein amounts were observed in TNF-α-treated EPCs compared with control cells (680.6 ± 63.6% compared with control, P < 0.01). Furthermore, RSV pretreatment significantly decreased NF-κB p65 protein amounts, with overt reductions observed at 1 μM RSV (TNF-α vs. 1 μM RSV + TNF-α: 680.6 ± 63.6% vs. 560.8 ± 84.6% compared with control, P < 0.05) and minimally increased NF-κB p65 amounts observed at 50 μM RSV (TNF-α vs. 50 μM RSV + TNF-α: 680.6 ± 63.6% vs. 136.1 ± 12.7% compared with control, P < 0.01) (Figure 4a, b). In addition, TNF-α decreased cytoplasmic NF-κB p65 protein amounts in EPCs while RSV alleviated this effect. These findings indicated that TNF-α induced NF-κB p65 translocation from the cytoplasm into the nucleus, while RSV inhibited TNF-α-associated NF-κB p65 translocation.
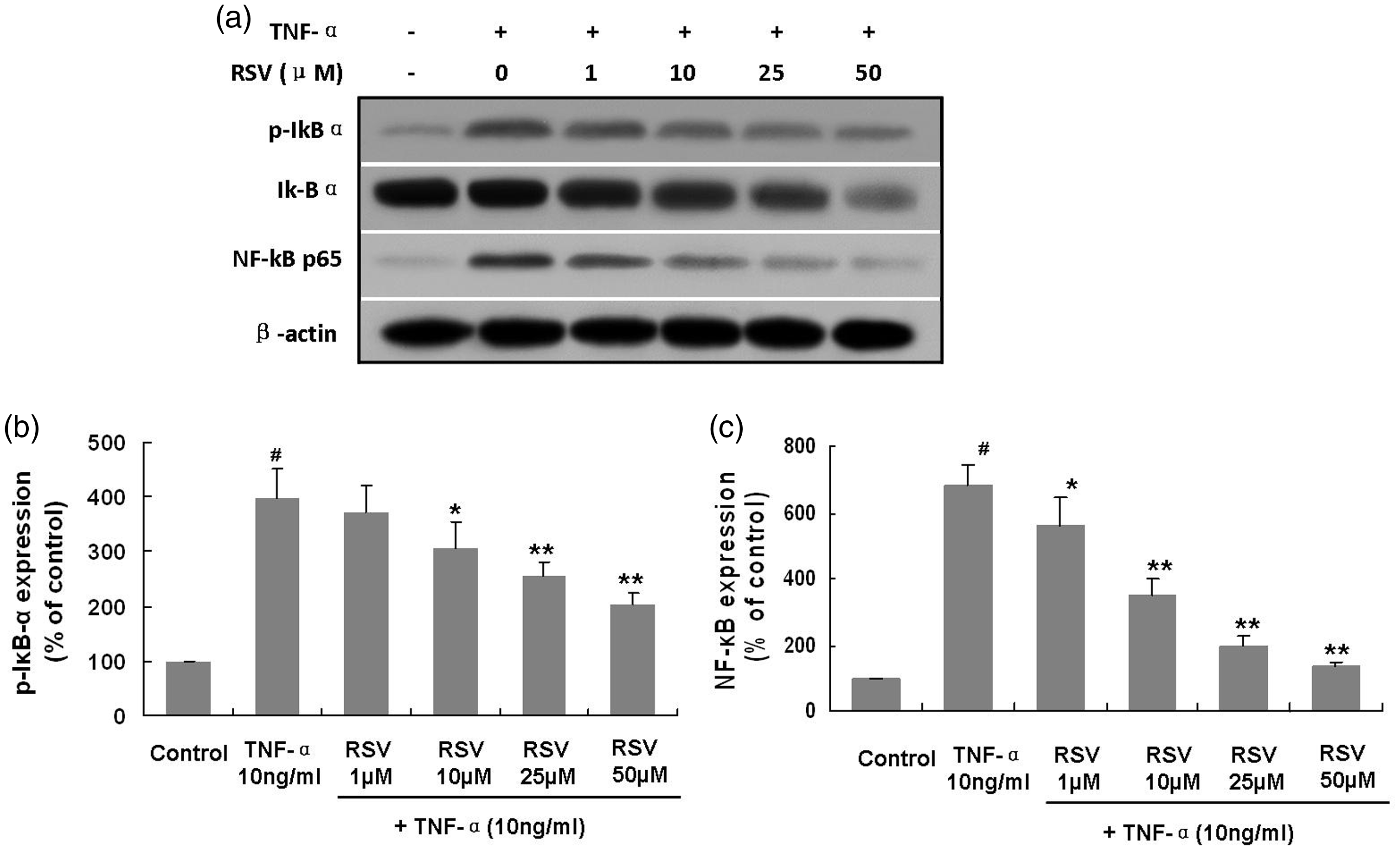
Effects of RSV on IκB-α activation and NF-κB translocation into the nucleus in TNF-α-treated EPCs.
Effects of RSV on IκB-α activation in TNF-α-treated EPCs
Next, the mechanism through which RSV reduced nuclear translocation of NF-κB in TNF-α-treated EPCs was explored by assessing IκB-α activation. Interestingly, TNF-α administration induced significant IκB-α phosphorylation (399.0 ± 52.8% compared with control cells, P < 0.01). RSV pretreatment for 18 hours inhibited phosphorylation of IκB-α in TNF-α-treated EPCs. Overt reductions in phosphorylation were observed at 10 μM RSV (TNF-α vs. 10 μM RSV + TNF-α: 399.0 ± 52.8% vs. 306.2 ± 49.0% compared with control cells, P < 0.05) and minimal phosphorylation was observed at 50 μM RSV (TNF-α vs. 50 μM RSV + TNF-α: 399.0 ± 52.8% vs. 204.0 ± 20.8% compared with control cells, P < 0.01) (Figure 4a, b).
Discussion
Epidemiological findings have indicated that CAD incidence in France is markedly reduced compared with other developed countries with similar fat-rich diets. The so-called “French paradox” may result from moderate consumption of red wine containing RSV. 20 Therefore, RSV represents a potent candidate therapeutic for preventing and treating CAD.
The primary cause of CAD is atherosclerosis, which is initiated by leukocyte adhesion to the endothelium via multiple adhesion molecules. Leukocytes stick to damaged endothelial tissue when EPCs are incorporated into the endothelium. EPCs may adhere to leukocytes in circulation, contributing to the pathogenesis of atherosclerosis. The pro-inflammatory cytokine TNF-α is elevated under atherosclerotic conditions, aggravating atherosclerosis. 21 Here, we assessed RSV’s capacity to regulate adhesion of TNF-α-treated EPCs.
EPCs were pretreated with RSV (0, 1, 10, 25, and 50 μM) for 18 hours and stimulated with TNF-α (10 ng/mL) for 6 hours. TNF-α markedly enhanced EPC adhesion, and administration of RSV overtly inhibited TNF-α-induced EPC adhesion in a concentration-dependent fashion. A previous study found that TNF-α inhibited adhesion of EPCs. However, RSV (20 μM) alleviated the effects of TNF-α. 5 In rats with damaged aortas, low RSV doses (10 mg/kg) increased the adhesion of EPCs, while a higher dose (50 mg/kg) did not. 22 Similarly, RSV (1 mM) promoted EPC adhesion, while higher amounts (60 mM) suppressed adhesion. 22 Differences in RSV concentration and the origins of EPCs may explain these discrepancies.
The primary cause of CAD is atherosclerosis, a chronic inflammatory process in which adhesion molecules are upregulated at the endothelial membrane. 7 Adhesion molecules including VCAM-1, ICAM-1 and E-selectin induce the adhesion of leukocytes as well as their migration into the intima. 7 EC VCAM-1 represents a pro-atherogenic adhesion molecule essential for both atherosclerosis initiation and progression to plaque instability. 23 Serum VCAM-1 levels likely control the degree of coronary injury. 7 Patients with intermediate and major lesions tend to have high VCAM-1 concentrations. 7 VCAM-1 levels on ECs are directly correlated with inflammation during myocardial infarction. 24 In addition to atherosclerotic plaque formation, adhesion molecules might also be involved in the generation of restenotic neointima. 25 VCAM-1 was overtly upregulated in neointimal smooth muscle cells in an animal model of wire-induced vascular injury. 26 In genetically hypercholesterolemic mice, VCAM-1 antibodies protect from neointima generation. 27 Levels of VCAM might be of diagnostic value in detecting restenosis after stent implantation. 25 Nakashima et al. demonstrated that VCAM-1 and ICAM-1 have pathogenic functions in inflammatory and atherosclerotic processes. 28 Bayata et al. showed significantly increased ICAM-1 levels in cases of early restenosis after primary angioplasty for acute myocardial infarction. 25 E-selectin is a glycosylated protein expressed on vascular ECs and is induced by several cytokines, including TNF-α, interleukin-1, and lipopolysaccharide. 29 , 30 Circulatory E-selectin levels reflect endothelial dysfunction, 31 and are important in early detection of atherosclerosis. 31 VCAM-1 and E-selectin levels are also increased in CAD. 8
We found that exposure of EPCs to TNF-α upregulated levels of adhesion molecules, including VCAM-1, ICAM-1 and E-selectin, compared with control cells. RSV markedly inhibited upregulation of VCAM-1, ICAM-1 and E-selectin induced following TNF-α treatment. In a previous study, Chu et al. also demonstrated that TNF-α upregulates ICAM-1, an effect that was reversed by administration of RSV. 32 RSV also reduced TNF-α-induced VCAM-1 and ICAM-1 upregulation in ECs. 5 RSV’s inhibition of TNF-α-induced cell adhesion molecule expression involve suppression of pro-inflammatory NF-κB signaling. 33 , 34 NF-κB is a transcription factor whose translocation into the nucleus during inflammation is important for inducing subsequent immune response. 35 Therefore, we assessed whether RSV’s effects depended upon NF-κB.
Here, we showed for the first time that RSV alleviated TNF-α-induced upregulation of VCAM-1, ICAM-1, and E-selectin in EPCs via regulation of IκB-α/NF-κB signaling. NF-κB dimers remain in the cytoplasm, interacting with IκB family members under normal conditions. 36 Meanwhile, IκB phosphorylation results in its degradation, followed by NF-κB release. 36 NF-κB activation can be measured by p65 translocation from the cytoplasm to the nucleus, where it upregulates transcription of genes such as VCAM-1, ICAM-1, and E-selectin. 37 , 38 Our results also demonstrated that cytoplasmic levels of phosphorylated NF-κB and IκB-α in TNF-α-treated EPCs were dose-dependently reduced by RSV. TNF-α-stimulated EPCs showed high nuclear NF-κB p65 amounts, but these were reduced by RSV pretreatment. These findings showed that RSV’s suppressive effects on NF-κB translocation into the nucleus depended on NF-κB phosphorylation.
We cultured healthy rat bone marrow-derived EPCs to study the effects of RSV on TNF-α-induced adhesion molecule expression in this study. Our findings showed that RSV might exert therapeutic effects on atherosclerosis by down-regulating expression of cell adhesion molecules. However, to further investigate the anti-atherosclerotic effects of RSV, we should use EPCs cultured from atherosclerotic animal models. Furthermore, changes in IκB-α/NF-κB upstream signaling pathways, such as the IκB kinase (IKK)α/β pathways, should be assessed to more fully understand the effects of RSV on NF-κB-dependent signaling and EPC adhesion molecule expression.
In conclusion, this study unveiled a new mechanism through which RSV exerts anti-inflammatory and anti-atherosclerotic effects. The mechanism likely involves VCAM-1, ICAM-1, and E-selectin downregulation via partial blockade of TNF-α-induced IκB-α phosphorylation and NF-κB activation in EPCs. These findings provide additional evidence supporting the beneficial effects of RSV on the cardiovascular system.
