Abstract
Background
Thyroid cancer is a malignancy that arises from the thyroid gland. While it is considered an uncommon condition, cases of thyroid cancer have been steadily rising in recent years worldwide.
Objectives
The present work assesses the apoptotic-inducing potential of diosmetin against thyroid cancer cells.
Materials and Methods
The impact of diosmetin on the growth of cells was evaluated using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) test. Apoptosis in the experimental CAL-62 cells was assessed using dual staining and 4′,6-diamidino-2-phenylindole (DAPI) staining. Oxidative stress marker levels were quantified in the cell lysates of CAL-62 cells exposed to diosmetin. PI3K/AKT/mTOR signaling protein levels were also assessed using cell lysates from both control and diosmetin-treated CAL-62 cells.
Results
The present results exhibited that diosmetin efficiently decreased the viability of both CAL-62 and BHT-101 cells. Additionally, diosmetin considerably induced apoptosis in CAL-62 cells, as evidenced by the fluorescent staining assay results. Diosmetin treatment remarkably increased TBA reactive species (TBARS) levels and subsequently decreased antioxidant levels in CAL-62 cells. Moreover, diosmetin treatment effectively blocked PI3K/AKT/mTOR signaling in CAL-62 cells.
Conclusion
The findings of this work exhibit that diosmetin efficiently inhibits cell proliferation and promotes oxidative stress-mediated apoptosis in thyroid cancer CAL-62 cells. Consequently, it possesses the potential to serve as an anti-cancer agent in treating thyroid cancer.
Introduction
Thyroid cancer, a rare malignant tumor originating from the thyroid gland, is a rapidly growing disease with an annual incidence and mortality rate that are constantly increasing worldwide, with cases reported to have tripled in the past 30 years (Kaliszewski et al., 2023). Thyroid cancer’s exact cause is unclear, but it can be caused by inherited genetic disorders, lifestyle habits, non-medical radiation exposure, and viral infections like human papillomavirus, and may advance thyroid tumors (Kitahara & Schneider, 2022). Thyroid cancer is more common in women aged 15–49 than in men aged 50–69, indicating a gender disparity. Factors affecting patient survival include tumor pathophysiology, age, initial lesion size, distant metastases, surgical extent, and radioiodine therapy use. The disease impairs vital physiological activities, making treatment challenging due to its indolent nature (Moleti et al., 2023).
Thyroid cancer is a complex and multidimensional condition that has undergone substantial research in recent years. Comprehending the fundamental processes that govern the etiology and advancement of thyroid tumors is essential for formulating successful treatment methods (Lebrun & Salmon, 2024). Cancer’s adaptive characteristics include dysregulated pathways, metabolic flexibility, immunosuppressive conditions, and immune responses. Targeted therapy focuses on the dynamic interactions between the tumor microenvironment and malignant cells (Chiba, 2024). Emerging research highlights indicate that extracellular vesicles play a crucial role in tumor microenvironment communication, facilitating information transfer and tumor progression, and offering potential therapeutic targets (Harahap et al., 2024).
Current treatment options for thyroid cancer face challenges, prompting exploration of alternative interventions like plant-derived bioactive compounds, which have been crucial in radionuclide diagnosis and therapy (Ratajczak et al., 2021). The sodium iodide symporter plays a crucial role in radioactive iodine uptake, but its non-selective nature can cause unwanted side effects, necessitating more targeted approaches (Capdevila et al., 2022). The utilization of plant-derived bioactive compounds is one of the promising avenues for treating thyroid cancer.
Plant-derived compounds have shown potential in treating thyroid cancer by modulating sodium-iodide symporter activity and exhibiting direct cytotoxic effects on thyroid cancer cells (Kaczmarzyk et al., 2024). Despite the promising nature of these plant-derived therapies, significant challenges remain. Optimizing the delivery and bioavailability of these compounds, as well as understanding their precise mechanisms of action, are critical area of ongoing research (Di Dalmazi et al., 2024).
Diosmetin, a naturally occurring bioactive flavonoid found in plants like Caucasian vetch and citrus, has been linked to potential anti-cancer properties due to its ability to modulate key signaling pathways. It has already been reported that diosmetin demonstrated numerous pharmacological properties, including anti-diabetic (Gong et al., 2021), nephroprotective (Wang et al., 2020), anti-sepsis (Liu et al., 2018), cardioprotective (Guo et al., 2022), and anti-inflammatory and antioxidant (Boisnic et al., 2023) properties.
Furthermore, diosmetin has induced apoptosis and inhibited cell viability in various tumor cells, including colon cancer (Koosha et al., 2019), liver cancer (Qiao et al., 2016), ovarian cancer (Zhao et al., 2021), and gastric cancer (Pan et al., 2023) cells. However, the apoptotic-inducing potential of diosmetin against thyroid cancer cells has not been assessed yet. Therefore, the present work has assessed the apoptotic-inducing potential of diosmetin against thyroid cancer cells.
Diosmetin, a flavonoid found in citrus fruits, has been studied for its pharmacological effects due to its polyphenolic structure. It has antioxidant properties, inhibiting pro-inflammatory mediators and suppressing NF-κB activation. It also shows anti-cancer activity, demonstrating significant anti-proliferative effects across various cancer cell lines (Lee et al., 2020). Diosmetin also exhibits anti-microbial and anti-viral effects and contributes to cardiovascular health by improving endothelial function and reducing vascular inflammation. Its neuroprotective effects, including attenuating oxidative stress and neuroinflammation, are being explored for potential applications in neurodegenerative diseases like Alzheimer’s and Parkinson’s. It also has the potential for metabolic regulation, improving insulin sensitivity, and regulating lipid metabolism, potentially affecting metabolic syndrome and type 2 diabetes (Fang et al., 2024; Garg et al., 2022).
In this study, we investigate the effects of diosmetin on CAL-62 thyroid cancer cells, focusing on its ability to induce apoptosis through the modulation of oxidative stress and suppression of the PI3K/AKT/mTOR signaling pathway—a critical axis involved in cell survival, proliferation, and resistance to apoptosis. Our findings provide new insights into the mechanistic basis of diosmetin’s anti-cancer activity and suggest its potential as a promising agent in the treatment of aggressive thyroid cancers.
Materials and Methods
Chemicals
Diosmetin, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), 4′,6-diamidino-2-phenylindole (DAPI), Dulbecco’s modified Eagle medium (DMEM), antibiotics, dimethyl sulfoxide (DMSO), doxorubicin (DOX), acridine orange/ethidium bromide (AO/EB), and others, were acquired from Sigma–Aldrich, USA. The test kits for quantifying the biochemical markers were acquired from Elabscience, USA, and Abcam, USA, respectively.
Cell Culture Maintenance
The thyroid cancer CAL-62 and BHT-101 cells were purchased from ATCC, USA, and cultivated in DMEM with 10% fetal bovine serum (FBS) and 1% anti-mycotic combinations in a 5% CO2 chamber. Cells were harvested upon reaching 80% confluence and utilized for subsequent fluorescence staining and biochemical analyses.
MTT Cytotoxicity Assay
The impact of diosmetin on the growth of the CAL-62 and BHT-101 cells was assessed using the MTT test. The cells were individually cultivated on a 96-well plate for 24 h. Subsequently, the cells underwent diosmetin treatment for an additional 24 h at varying dosages of 5, 10, 20, 40, 80, and 160 µM/mL. After the treatment, an MTT reagent (20 µL) was combined with DMEM (100 µL) and incubated for 4 h. Absorbance was taken at 570 nm after dissolving the formazan crystals with DMSO (100 µL), utilizing an enzyme-linked immunosorbent assay (ELISA) reader (Sarathbabu et al., 2020).
Dual Staining Analysis
Apoptosis in the experimental CAL-62 cells was assessed utilizing a dual-staining technique. The CAL-62 cells were cultured in a 24-well plate for 24 h before exposure to the half-maximal inhibitory concentration (IC50) of diosmetin and/or 2 µg of DOX. Subsequently, AO/EB (100 µg/mL) dyes were introduced to the wells and maintained in the absence of light for 5 min. The cells were subsequently examined using a fluorescent microscope to identify apoptosis in the CAL-62 cells (Sarathbabu et al., 2020).
DAPI Staining
The nuclear morphology of apoptotic cells was analyzed using the DAPI staining technique. The CAL-62 cells were placed on a 24-well plate and subsequently treated with the IC50 of diosmetin and/or DOX (2 µg) for 24 h. Following exposure, cells were fixed in paraformaldehyde (4%) and consequently stained with DAPI (200 µg/mL) for 15 min. The impact of diosmetin on apoptosis in CAL-62 cells was examined utilizing a fluorescent microscope (Sarathbabu et al., 2020).
Analysis of Oxidative Stress Marker Levels
The CAL-62 cells from both control and treatment groups were harvested, and cell lysates were generated using cell lysis buffer to quantify the levels of oxidative stress markers. The concentrations of TBA reactive species (TBARS), superoxide dismutase (SOD), catalase (CAT), and glutathione (GSH) were quantified in the cell lysates of CAL-62 cells exposed to diosmetin, utilizing the appropriate test kits by strictly adhering to the manufacturer’s manual (Abcam, USA).
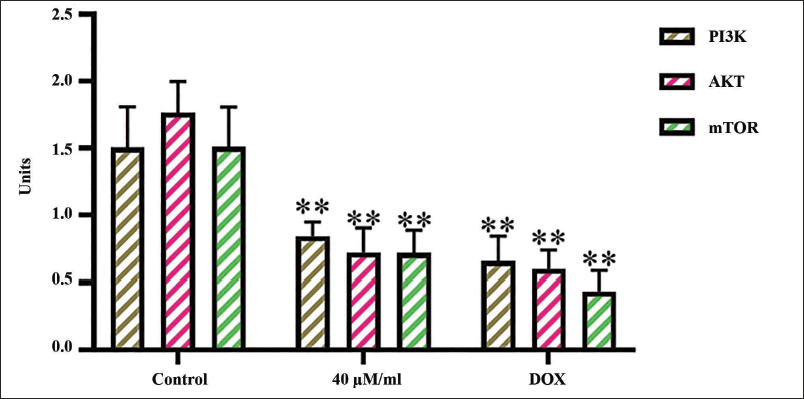
Analysis of PI3K/AKT/mTOR Signaling Protein Levels
To examine the amounts of PI3K, AKT, and mTOR proteins, cell lysates were collected from both control and diosmetin-treated CAL-62 cells. The concentrations of PI3K, AKT, and mTOR were assessed using commercially available test kits, adhering to the manufacturer’s instructions (Elabscience, USA).
Statistical Analysis
Statistical tests were done using GraphPad Prism, and results are shown as the Mean ± Standard Deviation (SD) of triplicate assays. The values are studied utilizing one-way analysis of variance (ANOVA) and Duncan’s multiple range test (DMRT), with p < .05 as significant.
Results
Effect of Diosmetin on Thyroid Cancer CAL-62 and BHT-101 Cell Viability
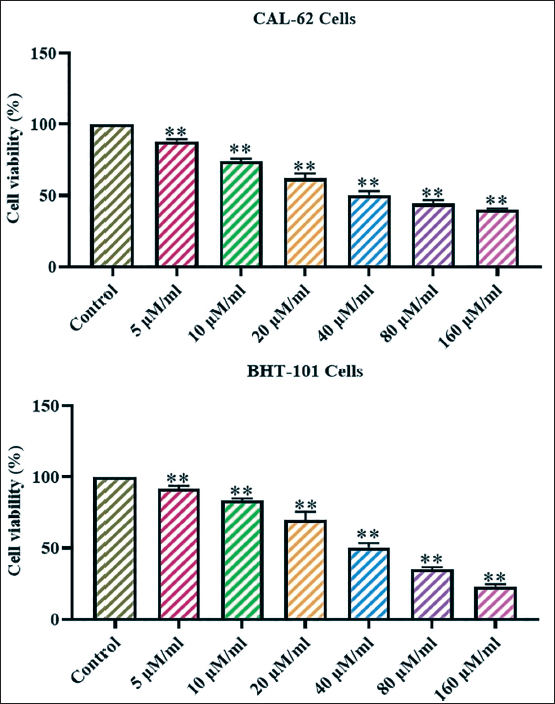
The effect of diosmetin on the thyroid cancer CAL-62 and BHT-101 cells was assessed using the MTT test, with the results illustrated in Figure 1. The diosmetin treatment at several concentrations (5–160 µM/mL) markedly suppressed the growth of both CAL-62 and BHT-101 cells. More specifically, the CAL-62 cells demonstrated high sensitivity to the diosmetin treatment. The present results demonstrate the cytotoxic effects of diosmetin on the growth of both CAL-62 and BHT-101 cells. The IC50 value of diosmetin for both cell lines was noted to be 40 µM/mL (Figure 1). This dosage was chosen for further tests.
Effect of Diosmetin on the Thyroid Cancer CAL-62 and BHT-101 Cell Viability. The Growth of the Thyroid Cancer CAL-62 and BHT-101 Cells was Markedly Diminished by the Varying Concentrations of Diosmetin (5–160 µM/mL). The Half-maximal Inhibitory Concentration (IC50) Concentration of Diosmetin Against Both Cells was Determined to be 40 µM/mL. The Values are Shown as a Mean ± Standard Deviation (SD) of Triplicate Measurements. The Data are Analyzed Statistically Using One-way Analysis of Variance (ANOVA) and Duncan’s Multiple Range Test (DMRT) Assays with GraphPad Prism Software.
Effect of Diosmetin on Apoptosis in CAL-62 Cells
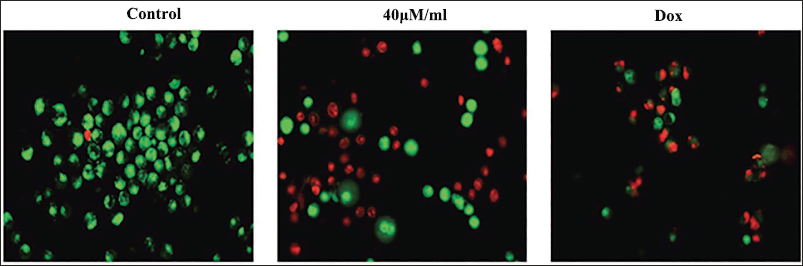
Figure 2 illustrates the outcomes of a dual-staining study conducted to assess apoptosis in diosmetin-treated CAL-62 cells. Treatment of CAL-62 cells with 40 µM/mL of diosmetin led to enhanced yellow/orange fluorescent cells, highlighting the occurrence of both early and late apoptosis, respectively. A greater number of cells displaying strong yellow/orange fluorescence was also found following standard drug DOX exposure, corroborating the apoptosis-inducing capabilities of the diosmetin.
Effect of Diosmetin on the Apoptosis in the CAL-62 Cells. The Increased Yellow and Orange Fluorescence was Found in the CAL-62 Cells Treated with 40 µM/mL of Diosmetin Compared to the Control Cells. The Augmented Yellow/Orange Fluorescence is Positively Linked with the Onset of Early and Late Apoptosis in the CAL-62 Cells.
Effect of Diosmetin on Apoptosis in CAL-62 Cells
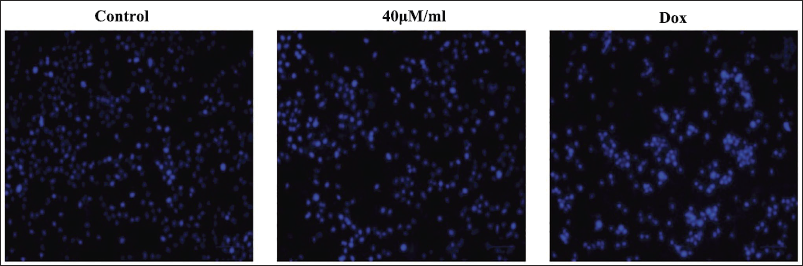
Figure 3 presents the results of a DAPI staining conducted on the diosmetin-treated CAL-62 cells. A greater number of apoptotic events, such as chromatin shrinkage, nuclear impairment, membrane damage, formation of apoptotic bodies, and a reduction in cell count, was noted in CAL-62 cells following treatment with 40 µM/mL of diosmetin. Comparable apoptotic events were also observed in DOX-exposed CAL-62 cells, demonstrating the apoptotic-enhancing capabilities of diosmetin on thyroid cancer cells.
Effect of Diosmetin on the Apoptosis in the CAL-62 Cells. The Treatment of Diosmetin at a Concentration of 40 µM/mL Led to an Increase in Blue Fluorescence and a Subsequent Decrease in Cell Volume, Signifying the Onset of Apoptotic Cell Death in the CAL-62 Cells.
Effect of Diosmetin on Oxidative Stress Indicators in the CAL-62 Cells
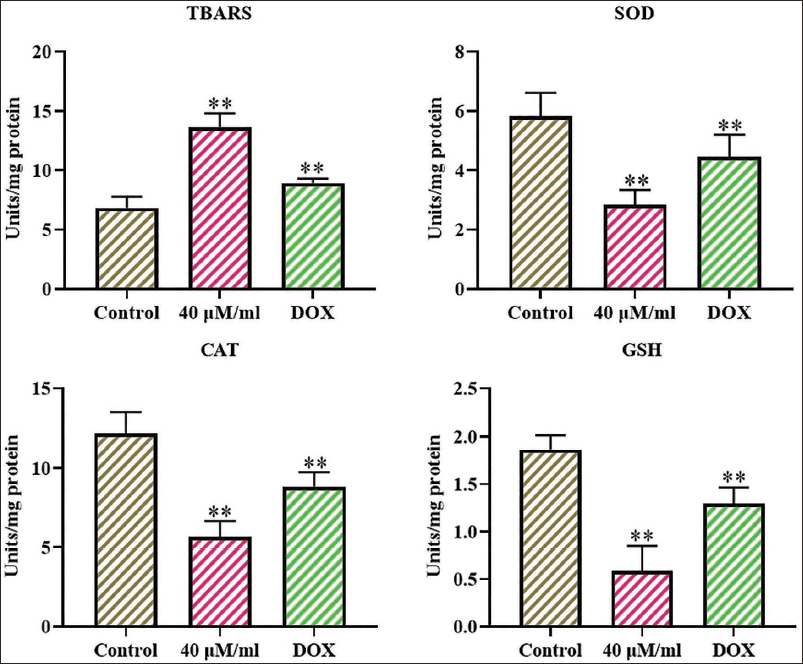
The oxidative stress indicators assessed in control and diosmetin-exposed CAL-62 cells are represented in Figure 4. The control cells had elevated levels of GSH, CAT, and SOD, accompanied by a reduction in TBARS levels. Simultaneously, treatment of cells with 40 µM/mL concentrations of diosmetin led to a remarkable elevation in TABRS concentration and a diminution in GSH, CAT, and SOD concentrations, which is in contrast to the control cells (Figure 4). Similar findings were also found in DOX-exposed CAL-62 cells, which further supported the findings of diosmetin treatment.
Effect of Diosmetin on PI3K/AKT/mTOR Protein Levels in CAL-62 Cells
Figure 5 illustrates the influence of diosmetin treatment on the concentrations of PI3K/AKT/mTOR in experimental CAL-62 cells. The administration of diosmetin at a dosage of 40 µM/mL markedly reduced the PI3K/AKT/mTOR protein concentrations in the CAL-62 cells relative to the control. Furthermore, DOX exposure also effectively diminished the PI3K/AKT/mTOR protein levels in CAL-62 cells. Consequently, it was evident that diosmetin can inhibit cell viability and induce apoptosis in CAL-62 cells by obstructing the PI3K/AKT/mTOR protein levels.
Discussion
Thyroid cancer is a tumor that starts in the thyroid gland. While it is considered an uncommon condition, the cases of thyroid cancer have been steadily increasing in recent years worldwide. The increase in thyroid cancer incidence is especially alarming, as it might interfere with essential biological systems governed by the thyroid gland (Fagin et al., 2023). Researchers have found various potential risk factors for thyroid cancer, including hereditary genetic disorders, lifestyle choices such as obesity and smoking, and environmental exposure to non-medical radiation.
However, the exact cause of thyroid cancer remains largely unexplored. Despite the challenges posed by thyroid cancer, advancements in diagnostic and treatment techniques offer promising avenues for improving patient outcomes (Alhejaily et al., 2023). By understanding the definition, risk factors, and the multifaceted burdens of thyroid cancer, healthcare professionals and researchers can work toward developing more effective strategies for the prevention, early detection, and management of this disease, ultimately reducing the impact on patients (Zhao et al., 2023).
One crucial aspect of thyroid cancer research is the analysis of the cytotoxic effects of bioactive compounds, which can potentially be used as therapeutic agents. Diosmetin, a naturally occurring flavonoid found in citrus fruits, has demonstrated anti-proliferative and pro-apoptotic effects in several cancer types. However, its therapeutic potential and underlying mechanisms in thyroid cancer, particularly in anaplastic thyroid cancer (ATC) cell lines such as CAL-62, have not been fully elucidated.
The MTT cytotoxicity test is an extensively utilized technique for evaluating the in vitro cytotoxic effects of various compounds on cancer cells (Kumar et al., 2024). The MTT assay relies on the transformation of the yellow tetrazolium salt MTT into purple formazan crystals by the action of mitochondrial enzymes in viable cells. This process correlates directly with the quantity of live cells, as overall mitochondrial activity is often proportional to the number of metabolically active cells. The absorbance of formazan crystals can be quantified using a spectrophotometer, offering a precise evaluation of the cytotoxicity of the examined substances. By using the MTT test, researchers can quickly and efficiently assess the cytotoxic effects of a wide range of compounds, identifying those with the most potent and selective activity against thyroid cancer cells (Ali et al., 2021). In this work, the effect of diosmetin on the thyroid cancer CAL-62 and BHT-101 cells was assessed using the MTT test. The results proved that diosmetin treatment at several concentrations markedly suppressed the proliferation of both CAL-62 and BHT-101 cells, which proves the cytotoxic nature of diosmetin against thyroid tumor cells (Figure 1).
Apoptosis is a crucial process in the onset and progression of cancer. The induction of apoptosis has become a central focus in anti-cancer strategies, as cancer cells often evade the normal cell cycle checkpoints and the progression to apoptosis. The two main pathways of apoptosis, the extrinsic and intrinsic pathways, both involve the stimulation of caspases, a family of cysteine proteases that are sequentially cleaved and activated (Lim et al., 2019). Malfunctions in apoptotic pathways can lead to cancer’s resistance to existing treatment methods, rendering the comprehension of the molecular mechanisms governing cell-death programs a vital focus of cancer research. Consequently, strategies that induce apoptosis in tumor cells have the potential to overcome cancer resistance and offer new prospects for drug development (Lee et al., 2023). To evaluate the apoptosis-inducing potential of bioactive compounds, researchers have utilized techniques such as dual staining and DAPI staining assays. Dual staining with AO and EB allows for the differentiation between viable, apoptotic, and necrotic cells, providing a comprehensive assessment of the compound’s effects (Figure 2). DAPI staining, on the other hand, specifically labels the deoxyribonucleic acid (DNA) and can be used to visualize nuclear condensation and fragmentation, which are hallmarks of apoptosis (Lin et al., 2022). In this work, the dual staining and DAPI staining assays were performed to assess the apoptosis-inducing potential of diosmetin on thyroid cancer cells. The present findings indicated a greater number of apoptotic events, such as chromatin shrinkage, nuclear injury, membrane damage, formation of apoptotic bodies, and a reduction in cell count in CAL-62 cells after exposure to diosmetin (Figure 3).
Cancer cells often exhibit enhanced metabolic activity and increased production of reactive oxygen species (ROS), leading to a state of elevated oxidative stress. This heightened oxidative state can be leveraged as a therapeutic approach, where exogenously inducing further oxidative damage can push cancer cells beyond their antioxidant capacity, triggering apoptosis (Ju et al., 2024). It has been demonstrated that modulating the redox balance in tumor cells can be a useful technique for cancer therapy. Increased ROS levels can damage critical cellular compartments, ultimately resulting in apoptosis (Nizami et al., 2023). Moreover, it has been observed that tumor cells often exhibit an adaptive response to oxidative stress, upregulating antioxidant systems to maintain redox homeostasis and promote tumor progression (An et al., 2024). To assess the efficacy of this approach, researchers can analyze various biomarkers indicative of oxidative stress and antioxidant status in cancer cells. TBARS are commonly used as a measure of lipid peroxidation, a hallmark of oxidative damage. SOD, CAT, and GSH are essential antioxidants that play an essential role in maintaining redox balance within cells. By examining the levels of these parameters, researchers can gain insights into the oxidative state of cancer cells and the effectiveness of interventions targeting the redox system (Arfin et al., 2021). The induction of oxidative stress-mediated apoptosis in tumor cells is a promising strategy that has gained significant interest in recent times. By leveraging the inherent vulnerability of cancer cells to elevated ROS, researchers can develop novel therapeutic strategies to eliminate malignant cells while minimizing harm to healthy tissues selectively (Di Carlo & Sorrentino, 2024). The present findings evidence that the diosmetin considerably elevated the TBARS and subsequently diminished the antioxidants in the thyroid cancer CAL-62 cells. These findings evidenced the hypothesis that diosmetin can trigger apoptosis in thyroid tumor cells via upregulating oxidative stress (Figure 4).
The PI3K/AKT/mTOR pathway is a vital regulator of cellular growth, survival, and metabolism, with its dysregulation associated with the onset and advancement of multiple cancer types, including thyroid carcinoma. This pathway is essential in the malignant transformation of human malignancies and their ensuing development, proliferation, and metastasis (Rascio et al., 2021). The PI3K/AKT/mTOR pathway is often activated in several cancer types. The PI3K/AKT signaling is relatively underexplored in endocrine tissue tumors, such as thyroid cancer, in comparison to more prevalent malignancies. There is increasing evidence from human tumors and animal models indicating that this route may significantly contribute to the formation and progression of thyroid cancer (Liu et al., 2023). Activation of the PI3K/AKT/mTOR signaling can contribute to the pathogenesis of many tumor types through various mechanisms, including increased cell growth, survival, and metabolism, as well as invasion and metastasis (Wiese et al., 2023). The PI3K/AKT/mTOR pathway has been extensively researched, with inhibitors targeting its components—PI3K, AKT, and mTOR currently in various phases of clinical development for the treatment of multiple cancer types, including thyroid carcinoma (Figure 5). Notwithstanding encouraging preclinical results, the clinical effectiveness of these inhibitors as standalone treatments has thus far been constrained, indicating that medicines aimed at several signaling pathways may be essential for improved clinical outcomes (Davoodi-Moghaddam et al., 2023).
The findings of the current investigation demonstrated that diosmetin administration significantly decreased the concentrations of PI3K/AKT/mTOR proteins in CAL-62 cells. Therefore, it was clear that diosmetin treatment can inhibit cell viability and induce apoptosis in thyroid tumor CAL-62 cells by inhibiting the PI3K/AKT/mTOR pathway.
Conclusion
The present findings indicate that diosmetin treatment successfully suppresses cell proliferation and induces apoptosis in CAL-62 thyroid cancer cells. The treatment with diosmetin considerably reduced cell viability and induced apoptosis by elevating oxidative stress. Moreover, the diosmetin blocked the PI3K/AKT/mTOR signaling in CAL-62 cells. Consequently, it possesses the potential to serve as an anti-tumor candidate to treat thyroid cancer. Additional assays are still needed to comprehend the precise molecular processes involved in diosmetin-induced apoptosis in thyroid cancer cells.
Footnotes
Abbreviations
BC: Bladder cancer; DMEM: Dulbecco’s modified Eagle medium; DMSO: Dimethyl sulfoxide; FBS: Fetal bovine serum; GSH: Glutathione; ROS: Reactive oxygen species; SOD: Superoxide dismutase; TBARS: TBA reactive species.
Data Availability
Data will be made available on request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable as there is no animal or human subjects involved in the experiments.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
