Abstract
Background:
Patients with rheumatoid arthritis (RA) have an increased risk of SARS-CoV-2 infection due to intrinsic characteristics of the pathology and the medications used to treat it. The aim was to evaluate the incidence of and factors related to SARS-CoV-2 infection in patients with RA in Colombia.
Methods:
This was an observational study of patients diagnosed with RA who were treated at a health care institution in Colombia. The study evaluated whether the patients presented SARS-CoV-2 infection and other clinical variables. Variables associated with the risk of SARS-CoV-2 infection were identified.
Results:
A total of 2566 patients with RA were identified. They had a median age of 61.9 years, and 81.1% were women. They were mainly treated with synthetic disease-modifying antirheumatic drugs (DMARDs) (85.3%), glucocorticoids (52.2%), and biological DMARDs (26.8%). The incidence of SARS-CoV-2 infection was 5.1%, and the factors that increased the risk included treatment with synthetic DMARDs with or without biological DMARDs but with concomitant systemic glucocorticoids [odds ratio (OR): 2.18, 95% confidence interval (CI): 1.21–3.93 and OR: 1.69, 95% CI: 1.05–2.74, respectively] and receiving antidiabetic drugs (OR: 2.24, 95% CI: 1.27–3.94). A total of 20.8% of patients with COVID-19 required hospitalization and 3.8% died.
Conclusion:
The incidence of COVID-19 is higher among patients with RA who receive DMARDs and glucocorticoids simultaneously or who have diabetes mellitus than among patients with RA not receiving these drug combinations, which should guide treatment strategies.
Keywords
Introduction
In Wuhan, China, at the end of 2019, a series of cases of pneumonia caused by a new coronavirus were reported. 1 The pathogen was named SARS-CoV-2 (severe acute respiratory syndrome coronavirus type 2), and the pneumonia that it produced was named coronavirus disease 2019 (COVID-19) by the World Health Organization (WHO). 2 It quickly spread to different regions and countries, and on 30 January 2020, the WHO declared the COVID-19 epidemic a public health emergency of international concern. 3 This infection has affected tens of millions of people worldwide and has caused millions of deaths. 4 In Colombia, according to the National Institute of Health, the number of confirmed cases exceeds 5 million, and more than 127,000 people have died. 5
The pandemic has generated concerns among patients with autoimmune rheumatic diseases because they have a higher risk of contracting bacterial, viral, and fungal infections than the general population,6–9 and it has been shown that these pathologies had more than double the risk of SARS-CoV-2 infection. 10 Rheumatoid arthritis is one of the most common autoimmune rheumatic diseases; it has a prevalence of 0.24–0.52% in Colombia.11,12 The inherent immunological dysregulation of these patients; their pharmacological management, which often includes systemic glucocorticoids and synthetic and biological disease-modifying antirheumatic drugs (DMARDs); and their chronic comorbidities predispose them toward developing infections.8,9
However, the recommendations of the American College of Rheumatology (ACR) and the European League Against Rheumatism (EULAR) generally suggested maintaining the use of immunomodulatory and immunosuppressive therapies during the COVID-19 pandemic13,14 while reducing glucocorticoid doses to less than 10 mg per day when possible. 14 These suggestions aimed to avoid relapses of the disease, which can occur when treatment is abruptly suspended.6,15 To date, data on the epidemiology of COVID-19 in rheumatic populations are scarce, 9 as are data related to the influence of antirheumatic treatment on SARS-CoV-2 infection. 15 Studies carried out in Colombia on these topics are unknown. For this reason, we conducted a follow-up study of a cohort of patients with the objective of evaluating the incidence of and factors related to SARS-CoV-2 infection in a group of patients with rheumatoid arthritis in Colombia.
Materials and methods
An observational, prospective cohort study was conducted that included all patients with a confirmed diagnosis of rheumatoid arthritis who were older than 18 years and were receiving management and continuous follow-up by the specialized healthcare service provider (Institución Prestadora de Servicios Especializada: IPS-E) in 2019 in the different cities where it provides services. This health service provider cares for approximately 3.2% of patients with rheumatoid arthritis in the country, in 16 different cities, distributed in most geographic regions of Colombia.16,17 These patients were followed from 1 April 2020 to 31 May 2021. Each of the patients was followed by telephone after giving prior verbal informed consent. The patients were called in each of the follow-up months. They were asked whether they had had a confirmed diagnosis of SARS-CoV-2, and this information was used to establish the rate of COVID-19 in this group of patients. All patients had free access to diagnostic tests through the Colombian health system. In addition, for patients who had been diagnosed with COVID-19, possible related complications were evaluated.
Based on the information obtained from the specialized IPS and interviews and on the consumption of medications (systematically obtained from the dispensing company, Audifarma SA), a database was designed to collect the following groups of patient variables:
Sociodemographic: sex, age, city/department of origin, occupation, education, and health system affiliation regime (contributory or paid for employer and subsidized by state).
Clinical: diagnosis of rheumatoid arthritis (seropositive, seronegative, or unspecified), activity level [Disease Activity Score-28 (DAS-28)] at the beginning and end of follow-up, related complications (articular, pulmonary, ocular, renal, digestive, and others).
Pharmacological:
3.1 Medications used for the management and control of rheumatoid arthritis: (a) synthetic DMARDs (methotrexate, leflunomide, chloroquine, hydroxychloroquine, sulfasalazine, and azathioprine); (b) biological DMARDs (etanercept, adalimumab, rituximab, abatacept, certolizumab, tocilizumab, infliximab, and golimumab); (c) JAK inhibitors (tofacitinib and baricitinib); and (d) systemic glucocorticoids (prednisolone, deflazacort, prednisone, and methylprednisolone). 3.1 Comedications, grouped into the following categories: (a) antidiabetics (oral and subcutaneous), (b) antihypertensives and diuretics, (c) lipid-lowering, (d) antiulcer, (e) antithrombotics (antiplatelet and anticoagulant), (f) analgesics/anti-inflammatory medications, (g) thyroid hormones, (h) antidepressants, (i) anti-epileptic medications, (j) bronchodilators and inhaled corticosteroids, (k) anti-osteoporotics, (l) antihistamines, and others.
Outcomes: (a) SARS-CoV-2 infection, (b) hospitalization, (c) need for oxygen supplementation and mechanical ventilation, (d) admission to the intensive care unit, and (e) death related to COVID-19.
The protocol was approved by the Bioethics Committee of the Technological University of Pereira under the category ‘research without risk’ (approval code: 03-110520).
The data were analyzed with the statistical package SPSS Statistics, version 26.0 for Windows (IBM, USA). A descriptive analysis was performed using frequencies and proportions for qualitative variables and measures of central tendency and dispersion for quantitative variables, depending on the parametric behavior established by the Kolmogorov–Smirnov test. The general incidence of SARS-CoV-2 infection and the incidence among the different medication and treatment groups were established. Quantitative variables were compared using Student’s t test or analysis of variance (ANOVA), and categorical variables were compared using the chi-square test. Binary logistic regression models were performed that included variables with a significant association in the bivariate analyses and those that could offer a biologically plausible explanation for the outcome of new SARS-CoV-2 infection (yes/no). The Hosmer–Lemeshow test was performed to describe the model’s goodness of fit. The predictive capacity of the model was determined according to the area under the curve (AUC). Statistical significance was established at p < 0.05.
Results
A total of 2566 patients diagnosed with rheumatoid arthritis were identified. The patients were distributed across 21 different cities. A total of 81.1% (n = 2080) were women, and the median age was 61.9 years (interquartile range: 53.3–69.4 years; range: 21.6–96.3 years). The most common occupation was related to household activities (n = 616; 24.0%). The majority of the patients were from Bogotá (n = 1607; 62.6%) and were predominantly affiliated with the contributory regime of the Colombian health system (n = 2498; 97.3%) (Table 1).
Sociodemographic, clinical, and pharmacological variables of a group of patients diagnosed with rheumatoid arthritis, Colombia.

DAS-28, Disease Activity Score-28; IQR, interquartile range.
Seropositive rheumatoid arthritis was the most frequently identified type (n = 2147; 83.7%). Complications were reported in 20.8% (n = 533) of the patients, especially ocular (n = 532; 20.7%) and joint complications (n = 58; 2.3%). At the beginning of the follow-up period, the patients had a median DAS-28 score of 2.5 (range: 0.0–8.0), and pathological activity was present in 45.8% (n = 1176) of the evaluated subjects (Table 1). The majority of the patients received some type of synthetic DMARDs (n = 2190; 85.3%), followed by systemic glucocorticoids (n = 1340; 52.2%) and biological DMARDs (n = 687; 26.8%) for the management of rheumatoid arthritis (Table 2). The most common treatment regimen was synthetic DMARDs associated with systemic glucocorticoids (n = 922; 35.9%) (Table 3). 6.2% (n = 160) did not receive any medication.
Incidence of SARV-CoV-2 infection according to antirheumatic drugs in patients with a diagnosis of rheumatoid arthritis, Colombia.
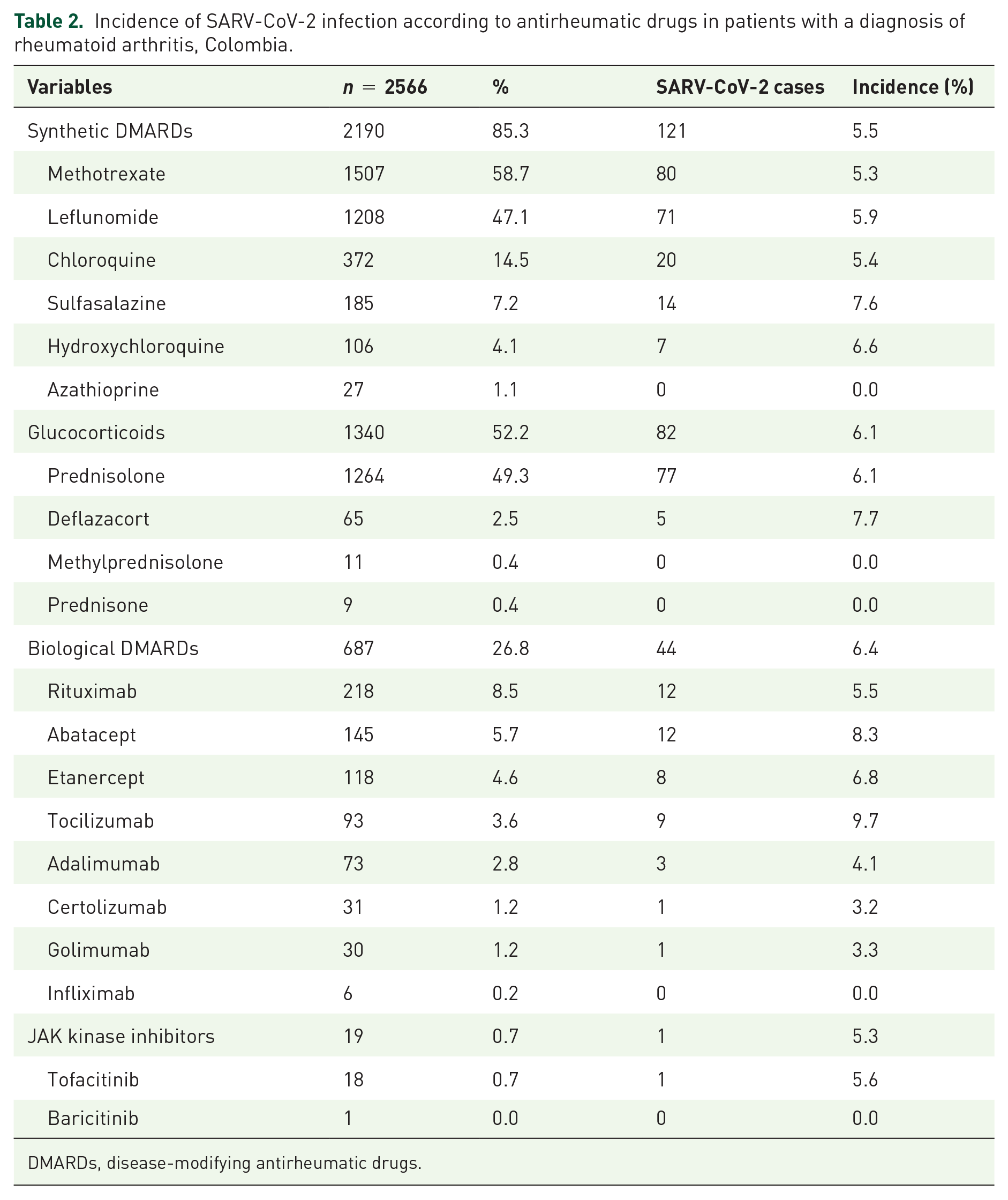
DMARDs, disease-modifying antirheumatic drugs.
Incidence of SARV-CoV-2 infection according to antirheumatic treatment regimens in patients with a diagnosis of rheumatoid arthritis, Colombia.
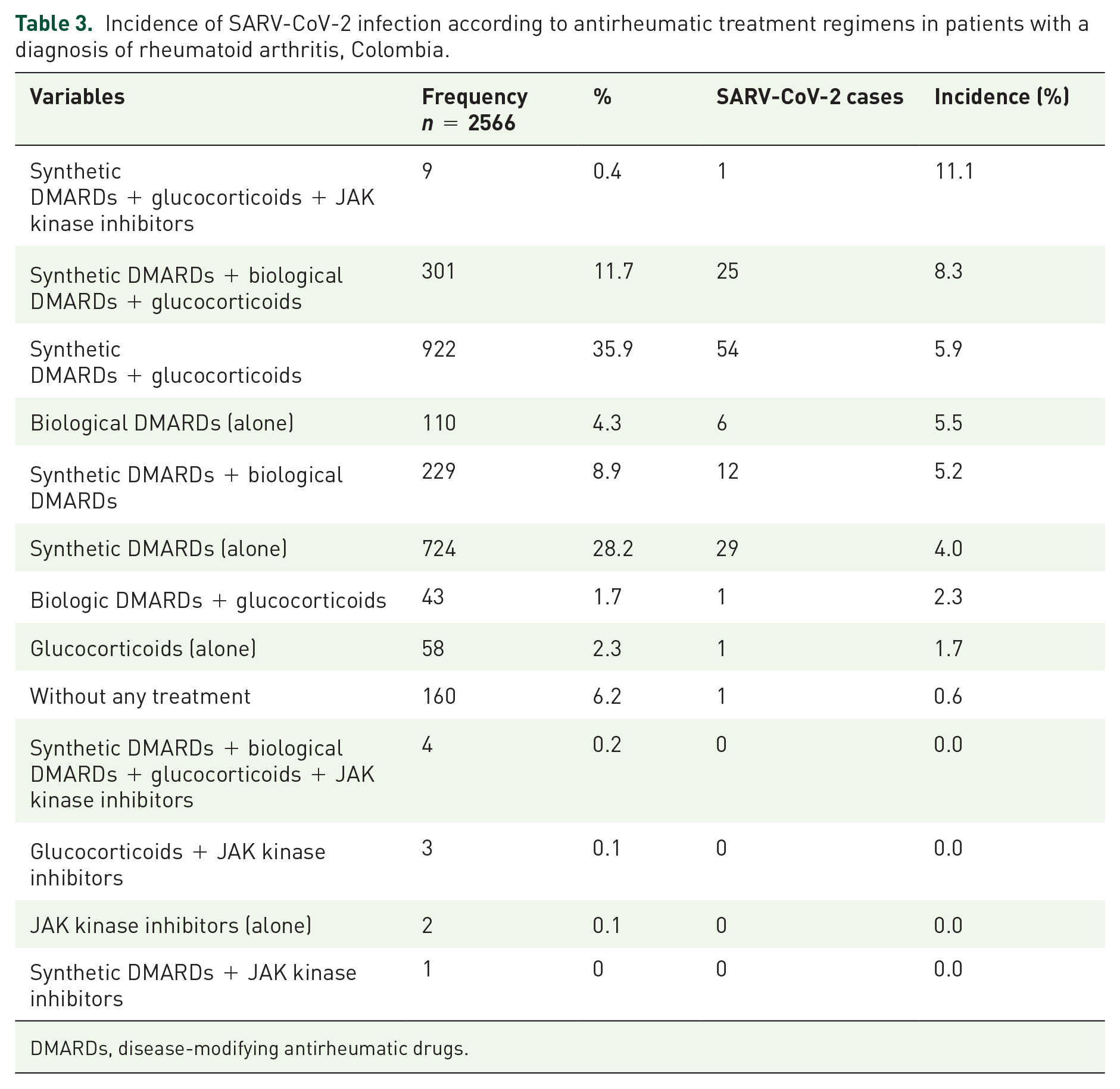
DMARDs, disease-modifying antirheumatic drugs.
SARS-CoV-2
During the follow-up period, the incidence of SARS-CoV-2 infection was 5.1% (n = 130) in the total group of patients with rheumatoid arthritis; it was 5.4% (n = 8) in patients under 40 years of age, 5.5% (n = 78) in patients between 40 and 64 years and 4.4% (n = 44) in patients 65 years of age or older. 40.0% (n = 52) of the cases occurred during the first 7 months of follow-up. Tables 2 and 3 show the incidence of infection according to drug groups and treatment regimens. Of the 130 patients with SARS-CoV-2, 24.6% (n = 32) required hospitalization, 20.8% (n = 27) required supplemental oxygen, and 4.6% (n = 6) were adequately managed. In the intensive care unit, 3.8% (n = 5) of the patients required invasive mechanical ventilation, and 3.8% (n = 5) died as a result of COVID-19.
Multivariate analysis
A binary logistic regression adjusted for sex, age, origin, disease activity, body mass index, and comedications found that patients who were prescribed antidiabetic drugs and those who were managed with synthetic DMARDs plus glucocorticoids or synthetic DMARDs plus biological DMARDs and glucocorticoids had a higher probability of SARS-CoV-2 infection. No variable decreased the risk (Hosmer–Lemeshow test p = 0.961, and AUC = 0.649) (Table 4).
Binary logistic regression on the variables related to presenting a new SARS-CoV-2 infection, in patients with a diagnosis of rheumatoid arthritis, Colombia.

CI, confidence interval; DAS-28, Disease Activity Score-28; DMARDs, disease-modifying antirheumatic drugs; OR, odds ratio; Sig, statistical significance.
Discussion
The present study identified the incidence of COVID-19 and the factors related to an increased probability of SARS-CoV-2 infection in a cohort of patients diagnosed with rheumatoid arthritis and treated at a specialized service in Colombia. The identification of these risk factors could allow the proposal of intervention measures that could reduce the risk of infection in these patients, further prioritizing handwashing, social distancing, and wearing face coverings in public when social distancing is not possible. 14 Similarly, in high-risk patients, when there are significant increases in the number of COVID-19 cases, teleconsultation or telemedicine care models could be implemented. 14 The identification of the patients most susceptible to presenting infections will also allow prioritizing vaccination strategies as well as boosters in this population. 18
The median age of the patients with rheumatoid arthritis was similar to that found in some studies (60.8–67.3 years),6–8,19 but slightly higher than that reported by other studies conducted in the country (53.2–58.0 years).20–23 There was a predominance of women, as reported in the majority of studies (71.3–83.8%).6,7,11,15,19–21,24 However, in two studies, conducted in Italy and the United Kingdom, the authors found a higher proportion of men.8,9 Seropositive rheumatoid arthritis was the most frequent type, which is consistent with what has been found in the literature.8,20–22 Compared with other pharmacoepidemiological studies from Colombia,20–23 disease activity was lower in the present study; however, in a recently published study that included more than 80,000 patients with rheumatoid arthritis, the average DAS-28 score was very similar to that found in our report. 12 This is relevant because the degree of disease activity has been related to the prognosis of patients. 18 Thus, Strangfeld et al. 25 found that patients with moderately and severely active rheumatoid arthritis who became infected with SARS-CoV-2 had an 87% risk of dying from complications of COVID-19 [odds ratio (OR): 1.87; 95% confidence interval (CI): 1.27–2.77].
In terms of the pattern of medications used to manage rheumatoid arthritis, this study found that synthetic DMARDs were the most commonly used therapeutic group, which is consistent with the findings of other international7,15,24,26 and domestic studies,12,20–23 and there was a marked predominance of methotrexate use.9,12,15,20–24,26 In this cohort of patients, just over a one-quarter received biological DMARDs, which was lower than the rate described in other international studies (34.2–57.4%)8,26 but consistent with domestic reports (20.9–28.7%).12,21,23 In comparison, half of the patients in the present study received systemic glucocorticoids, a rate that was higher than that described in several European countries (8.2–38.0%)7–9 and in the United States (28.2%) 15 but lower than that found in France (57.4%) 26 and Iran (64.0%) 24 and even in another study from Colombia (74.0%). 21 In general, the antirheumatic treatment that these patients received was in line with the recommendations of clinical practice guidelines.27,28 The differences in management may be due to the degree of disease activity, the clinical practice guidelines adopted by the institution providing the services, the evolution timepoint, the habits of the prescriber, the prescriber’s university education and degree of continuous education, patient comorbidities, the marketing strategies used by the pharmaceutical industry, and the characteristics of each country’s health systems.21,29,30
The incidence of SARS-CoV-2 infection in this cohort was 5.1%, which is consistent with that found in France by Costantino et al., 26 who reported an incidence of 6.9% in patients with chronic inflammatory rheumatic disease, including ankylosing spondylitis, rheumatoid arthritis, and psoriatic arthritis, and with that found in Italy by Zen et al., 6 who described an incidence of 7.2%; in contrast, in the United States, Spain, and Korea, the incidence of SARS-CoV-2 infection was between 3.9% and 18.9% in patients diagnosed with rheumatoid arthritis, 31 and in England, the incidence was 2.5%. 8 These variations can be explained by the different methodological designs used in the studies, such as the patient identification method, the rheumatological diseases that were considered, the duration of follow-up, and the time at which the study was conducted.6,8,26,31
In addition, it was found that compared with the general population, patients with rheumatological disease have an increased risk of SARS-CoV-2 infection. For example, a cohort study showed that patients with rheumatoid arthritis had a 25% higher risk of COVID-19 than individuals without rheumatoid arthritis. 8 The immunological alterations of patients with rheumatoid arthritis, as well as their pharmacological management with systemic glucocorticoids and synthetic or biological DMARDs and their chronic comorbidities, predispose them toward developing infections.8,9
Several factors were found to increase the risk of SARS-CoV-2 infection in this group of patients with rheumatoid arthritis. For example, those who were prescribed antidiabetic drugs had more than double the risk of infection; this is consistent with the findings of Malek Mahdavi et al., who found that patients with rheumatoid arthritis and diabetes mellitus had an increased risk of infection (OR: 1.77; 95% CI: 1.01–3.12; p = 0.050). 24 On the contrary, patients who were treated with synthetic DMARDs with or without biological DMARDs but with concomitant systemic glucocorticoid use had a higher risk of infection than those treated with only synthetic DMARDs, which is also consistent with other reports.24,32 Authors in Iran found that patients who received glucocorticoids had an increased risk of infection (OR: 2.58; 95% CI: 1.57–4.25; p = 0.001), 24 while a report from South Korea found no increased risk of infection among glucocorticoid users (OR: 1.18; 95% CI: 0.92–1.51; p = 0.19) but reported that those who received more than 10 mg per day had a risk of 47%. 32 In addition, Akiyama et al., 10 in a meta-analysis, found that compared to monotherapy with biological DMARDs, combined therapy with glucocorticoids, synthetic DMARDs, and biological DMARDs increased the risk of hospitalization and death due to COVID-19 by 2–3 times in patients with autoimmune diseases.
Finally, one-quarter of patients who were infected with SARS-CoV-2 required in-hospital management, which is consistent with the findings of studies conducted in the United States (21.0–26.0%)15,19,33 but differs from reports from Iran and Spain, where a higher proportion of cases required hospital management (38.3–43.9%).24,34 On the contrary, the rate of mortality as a result of complications of COVID-19 in the present report was 3.8%, similar to that described in several cohorts of patients in the United States (3.6–5.5%)15,19,33 but lower than that found in Iran (8.6%) 24 and in a group of patients with rheumatological pathologies in Spain (22.2%). 34
Some limitations should be considered when interpreting the results, since access to outpatient or hospital medical records was not obtained to verify the diagnosis of COVID-19 or its severity and complications, among other clinical and paraclinical variables. Similarly, medications that the patients may have used that were prescribed outside the health system or were not provided by the dispensing company were not known. However, the study included a significant number of patients affiliated with the health system in a large number of cities in Colombia.
Based on these findings, it can be concluded that in this cohort of patients with a diagnosis of rheumatoid arthritis, the incidence of COVID-19 is increased in those being treated with synthetic DMARDs with or without biological DMARDs but with concomitant systemic glucocorticoids and in patients using other medications, such as antidiabetic drugs. No variables were identified that reduced the risk of COVID-19. These results can be useful for clinicians who care for patients with rheumatoid arthritis because the recognition of these factors can be used to improve care and the quality of care by focusing on risk factors and encouraging personal protection and vaccination behaviors among these patients.
