Abstract
Background
There is an abnormal enhancement of androgen receptor activity in prostate cancer cells, leading to increased sensitivity to androgens and the promotion of cancer cell proliferation.
Objectives
This study aimed to investigate the effects of
Materials and Methods
Kaempferol was isolated and extracted from the hydrolysate of
Results
The recovery rate of kaempferol from
Conclusion
Introduction
Recently, a consistent increase is evident in prostate cancer (PC) (Cicero et al., 2019). Typically, androgens have the capacity to promote cell growth and proliferation by binding to androgen receptors within the body (Linder et al., 2022). In PC cells, there is an abnormal enhancement of androgen receptor activity, leading to increased sensitivity to androgens and the promotion of cancer cell proliferation (Shiota et al., 2022; Sena et al., 2022). Targeting the androgen signaling pathway is a critical therapeutic strategy for managing androgen-dependent PC.
This work involved extracting kaempferol from
Materials and methods
Extraction of Kaempferol from E. ulmoides Leaves
20 mg of kaempferol reference standard (SK8030, purity ≥ 98%, Beijing Solarbio Technology Co., Ltd., China) was dissolved in a brown volumetric flask containing anhydrous methanol to prepare a reference standard stock solution at 0.1 mg mL−1. Subsequently, it was diluted to a series of concentrations at 0.5, 1.0, 2.0, 4.0, 8.0, 12.0, 16.0, and 20.0 µg mL−1 and saved at 4°C. 0.5 g of
Determination of Free Radical Clearing Rate (CR) in Kaempferol
According to the instructions of the free radical scavenging reagent kit (LM80023TK, Shanghai LMAI Biotechnology Co., Ltd., China), 3 mL of Tris–HCl buffer (pH = 8.2) and 0.1 mL of extraction solution with concentrations of 0.0, 0.5, 1.0, 2.0, 4.0, and 8.0 µg mL−1 were taken. The mixed solution was equilibrated in a water bath for 20 min. Subsequently, the addition of 0.3 mL of 7 mmol L−1 pyrogallol was performed to allow a reaction for 5 min. Afterward, 1.0 mL of 10 mol L−1 HCl was introduced to terminate this reaction. The absorbance at 420 nm was assigned to calculate the O2− CR. Subsequently, 0.15 mmol L−1 FeSO4, 2.0 mmol L−1 sodium salicylate, extraction solution, and 6.0 mmol L−1 H2O2 were sequentially added to generate ·OH through a reaction. The reaction proceeded at 37°C for 1 h. The absorbance value at 510 nm was measured to calculate the ·OH CR.
Culture of LNCaP Cells
LNCaP cells (ORC0499, Shanghai Aoruisai Biological Cell Bank, China) were subjected to a culture in RPMI 1640 culture medium (11875093, Thermo Fisher Scientific, USA) containing 10% heat-inactivated fetal bovine serum (26010074, Thermo Fisher Scientific, USA) and 1% penicillin–streptomycin, incubated in a DHP-9160-ST saturated humidity incubator (Wuxi Marit Technology Co., Ltd., China) at 37°C and containing 5% CO2 for 48 h before the medium was changed. Afterward, the medium was changed, and the cells were digested using 0.25% trypsin for subculturing. The cells were grouped into four: The control (Ctrl) group, low-concentration kaempferol (LCK) group, medium-concentration kaempferol (MCK) group, and high-concentration kaempferol (HCK) group. LNCaP cells were harvested, digested, and seeded for an additional 24 hours of culture. Cells in Ctrl, LCK, MCK, and HCK groups were cultured in a complete medium consisting of 0.0, 0.5, 1.0, and 2.0 µmol L⁻¹ of kaempferol extract from
Examination of LNCaP Proliferation Inhibition Rate (PIR)
According to the instructions of the 3-(4,5)-dimethylthiahiazo(-z-y1)-3,5-di-phenytetrazoliumromide (MTT) cell proliferation detection kit (11465007001, Sigma–Aldrich, USA), LNCaP cells were grouped and subjected to different treatments. Subsequently, they were cultured under standard conditions for varying durations of 6, 12, 24, 36, and 48 h. Following these incubation periods, 10 µL of a 5 g L−1 MTT reagent was introduced. The cells were then incubated again under the standard culture conditions for an additional 6 h. After discarding the original culture medium, 100 µL of dimethyl sulfoxide (DMSO) reagent was placed and agitated for 10 min. HM-SY96S enzyme-linked immunosorbent assay (Shenzhen Antongda Equipment Technology Co., Ltd., China) was employed to detect the absorbance of each well at a wavelength of 490 nm, and PIR was calculated: PIR (%) = (1 − absorbance in sample group/absorbance in Ctrl group) × 100%.
Detection of Apoptosis Rate (AR) and Cell Cycle of Cells
LNCaP cells were grouped and processed. After routine culture for 24 h, they were digested with trypsin and subjected to a 10-min centrifugation at 1,000 rpm to discharge the supernatant, rinsed three times for 10 min with 0.1 mol L−1 pH 7.4 phosphate buffer, and subsequently divided into two portions. One portion was fixed by overnight incubation in prechilled 80% ethanol at 4°C. After washing off the ethanol with phosphate buffer, 10 µL of RNAse and 10 µL of propyridine iodide (PI, ST512, Shanghai Beyotime Biotechnology Co., Ltd., China) were mixed. Subsequently, LNCaP cells were incubated at 4°C without light for 30 minutes. Cell cycle alterations were assessed utilizing a CytoFLEX flow cytometer (Beckman Kurt, USA), with a coefficient of variation correction below 3%. According to the instructions of the Annexin V-FITC cell apoptosis detection kit (C1062M, Shanghai Beyotime Biotechnology Co., Ltd., China), the remaining portion of the cells was resuspended in Annexin V-FITC binding solution. Annexin V-FITC and PI Staining Solution were introduced, thoroughly mixed, and the cells were incubated without light for 15 min. Cell apoptosis was subsequently determined using flow cytometry.
Western Blotting
After routine culture for 24 h, the LNCaP cells were digested with trypsin and subjected to a 10-min centrifugation at 1,000 rpm for discharging its supernatant. Afterward, they were performed with three 10-min washes with 0.1 mol L−1 pH 7.4 phosphate buffer and lysed using Radio Immunoprecipitation Assay lysis buffer (R0010, Beijing Solarbio Technology Co. Ltd., China) for an extract of total cellular proteins. Protein concentration in the extracts was quantified using the bicinchoninic acid (BCA) assay method. Subsequently, the proteins underwent SDS-PAGE gel electrophoresis, followed by their transfer onto a PVDF membrane, which was incubated all night at 4°C with Bcl-2 (1:1,000), Bax (1:1,000), caspase-3 (1:1,000), caspase-9 (1:1,000), and GAPDH (1:1,000) protein primary antibody (ab182858, ab32503, ab32351, ab32539, and ab8245, Abcam, UK). Following primary antibody incubation, the membranes were exposed to HRP and IgG (1:1,000) secondary antibodies (ab6759, Abcam, UK) labeled with horseradish peroxidase for 2 h. Signal hybridization was performed using a chemically enhanced luminescence detection kit (P0018FM, Shanghai Beyotime Biotechnology Co., Ltd., China), and bands were visualized and photographed employing a WD-9413A gel imaging system (Beijing Liuyi, China). The grayscale values of the bands were measured using ImageJ, and levels of the target proteins were calculated.
Statistical Analysis
Data were expressed as mean ± standard deviation (SD) and statistically analyzed with SPSS 22.0. Group differences were compared using
Results
Detection of Kaempferol Extract
This work suggested that the original kaempferol content was 0.451 µg mL−1, and the recovered kaempferol content was 0.443 µg mL−1, with a recovery rate of approximately 98.23% and a relative SD of approximately 1.980%. Furthermore, this work compared the HPLC chromatograms of the standard sample and the kaempferol extract from
High-performance Liquid Chromatography (HPLC) Spectrogram of Kaempferol Extract from Eucommia ulmoides Leaves.
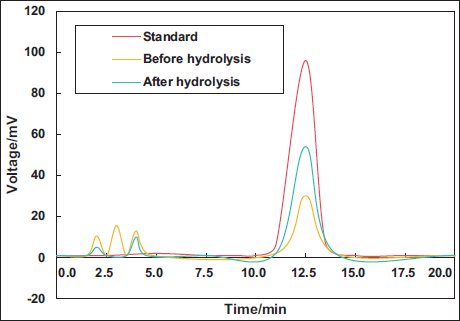
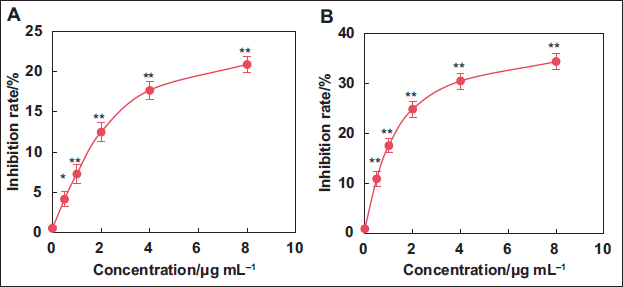
Antioxidant Effect in vitro of Kaempferol Extract from E. ulmoides Leaves
This work evaluated the impact of varying concentrations of kaempferol extract on the CR of O2− and ·OH free radicals. As presented in Figure 2, with the increase in kaempferol concentration, both O2− and ·OH CR gradually increased. In contrast to the 0 µg mL−1 concentration, the O2− and ·OH CR at 0.5, 1.0, 2.0, 4.0, and 8.0 µg mL−1 sharply increased, showing considerable differences (
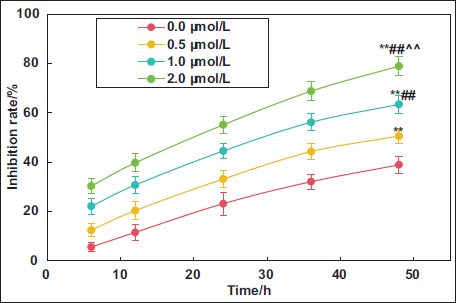
Impacts of Kaempferol Extract from E. ulmoides Leaves on LNCaP Proliferation
This work examined the impact of kaempferol extract at various concentrations on the inhibition of LNCaP cell proliferation. As demonstrated in Figure 3, prolonged time resulted in an enhancement for PIR of LNCaP cells in all groups. The PIR of LNCaP cells after treatment utilizing 0.5, 1.0, and 2.0 µmol L⁻¹ kaempferol exhibited great increases with obvious difference to that in the 0.0 µmol L⁻¹ group (
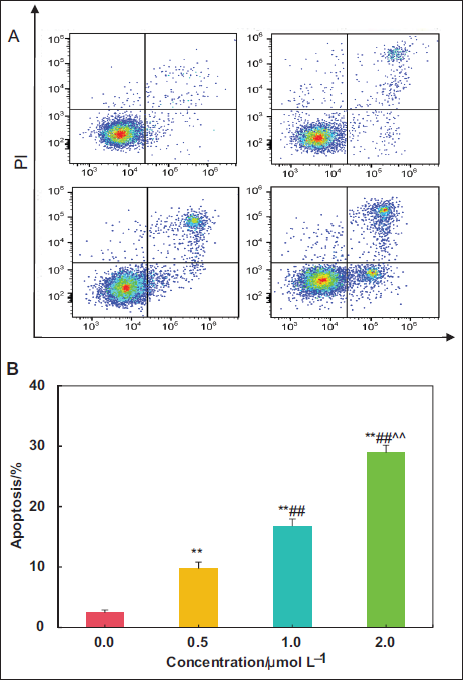
Impacts of Kaempferol Extract from E. ulmoides Leaves on Apoptosis of LNCaP Cells
The impacts of various concentrations of kaempferol extract on apoptosis in LNCaP cells are illustrated in Figure 4. Relative to the 0.0 µmol L⁻¹ group, treatment using 0.5, 1.0, and 2.0 µmol L⁻¹ kaempferol resulted in a highly observable increase in AR of LNCaP cells (
Influence of Kaempferol Extract from E. ulmoides Leaves on the Cell Cycle of LNCaP
This work examined the effects of kaempferol extract with varying concentrations on the LNCaP cell cycle. As explicated in Figure 5, treatment with 0.0, 0.5, 1.0, and 2.0 µmol L⁻¹ kaempferol did not alter the G0/G1 phase of LNCaP cells greatly (
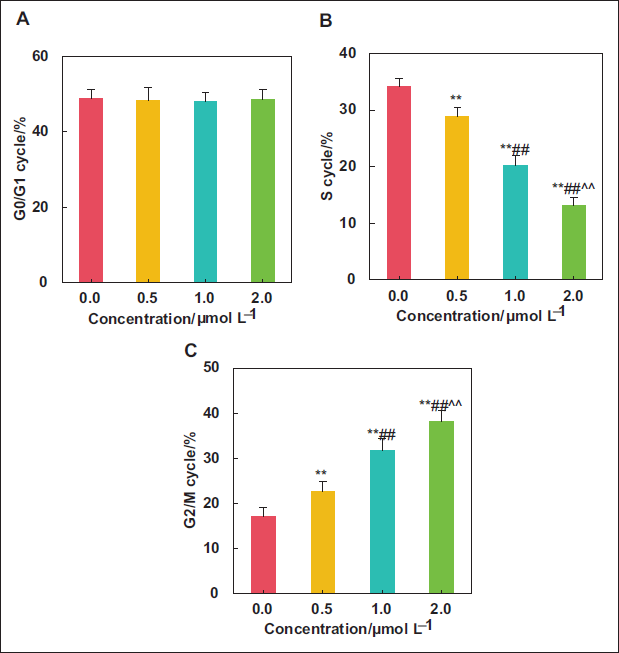
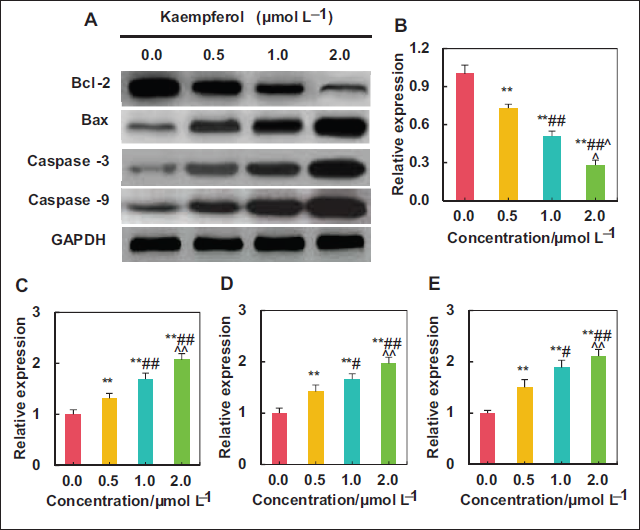
Influence of Kaempferol Extract from E. ulmoides Leaves on LNCaP-related Proteins
This study examined the impacts of kaempferol extract with varying concentrations on levels of ARPs in LNCaP cells, as presented in Figure 6. Treatment using 0.5, 1.0, and 2.0 µmol L⁻¹ kaempferol sharply downregulated Bcl-2 protein in LNCaP cells while upregulated Bax, caspase-3, and caspase-9 proteins, demonstrating obvious differences to the 0.0 µmol L⁻¹ group (
Discussion
Previous studies confirmed that after the isolation and purification of
Under normal circumstances, testosterone promotes the growth and division of cells by binding to intracellular androgen receptors. In PC cells, the abnormal increase in androgen receptor activity leads to enhanced sensitivity to androgens, thereby promoting the abnormal proliferation of PC cells (Hussain et al., 2023; Mekdad et al., 2022). Kaempferol is a plant-derived compound with various pharmacological activities (Yuan et al., 2021). Subsequently, in this study, different concentrations of kaempferol extracts were utilized for the treatment of prostate cancer cells (LNCaP). The results indicated that the proliferation inhibition and ARs of LNCaP cells increased with the concentration of kaempferol. Kaempferol is a natural plant extract, and these results confirm its ability to inhibit the proliferation of LNCaP cells, indicating its characteristics as an anti-PC drug. Inhibition of androgen synthesis or reducing androgen receptor activity is an important strategy for treating androgen-dependent PC (Harris et al., 2022). Apoptosis is crucial for maintaining tissue and organ balance. The apoptosis process in androgen-dependent PC cells is disrupted, leading to cancer cells being unable to be eliminated through normal apoptotic pathways, thereby increasing cancer cell proliferation (Wang et al., 2021). Wang et al. (2021) found that kaempferol can mediate Akt/mTOR pathways to apoptosis in pancreatic cancer cells. This suggests that kaempferol may exert its effects on androgen-dependent PC cell growth, proliferation, and apoptosis through various mechanisms, such as inhibiting androgen receptor activity and activating/inhibiting relevant pathways. To better understand the mechanisms by which kaempferol inhibits proliferation and induces apoptosis in LNCaP cells, this work further investigated its effects on the cell cycle of LNCaP cells. The cell cycle is a fundamental process in cellular life activities, and when the cell cycle at a certain phase becomes abnormal, cells may enter related pathological processes. Drugs induce apoptosis by interfering with the cell proliferation cycle, inhibiting normal transitions between cell cycle phases, and promoting apoptosis development (Arora et al., 2023; Patra et al., 2023; Panjwani et al., 2021). The results demonstrated that treatment with kaempferol significantly shortened the S phase of the cell cycle in LNCaP cells while lengthening the G2/M phase. The S phase in the cell cycle is the DNA replication phase, and G2 is the phase before mitosis (Barnaba et al., 2021). This indicates that kaempferol can arrest LNCaP cells in the G2/M phase, disrupt normal transitions between various growth phases of cells, and exhibit a concentration-dependent slowing of cell cycle progression, suppression of cell proliferation, and apoptosis promotion. This aligns with the results of Zhang, Chen, et al. (2022) and Zhang, Qu, et al. (2022), who confirmed that kaempferol can induce androgen-independent PC cells to stall in S and G2 phases and induce apoptosis.
Various genes or proteins regulate the proliferation, migration, invasion, and apoptosis of cancer cells. Bcl-2 and Bax play opposing roles in the regulation of apoptosis in cells. Bcl-2 prevents cell apoptosis by inhibiting apoptosis signal transduction, regulating mitochondrial membrane potential, and protecting mitochondrial function (Ladokhin, 2020). In contrast, Bax promotes cell apoptosis by increasing mitochondrial membrane permeability and releasing apoptotic factors from the mitochondria (Spitz et al., 2022). Caspase-9 is an initiator protein mainly involved in cell apoptosis initiated through the mitochondrial pathway. When apoptotic signals stimulate cells, caspase-9 is activated (Araya et al., 2021). Activated caspase-3, as an effector protease, regulates key steps in multiple aspects of cell apoptosis, including DNA damage and fragmentation, remodeling of the cell nucleus and membrane, and degradation of intracellular structures and organelles (Asadi et al., 2022; Stefanowicz-Hajduk et al., 2021; Zhang, Chen, et al., 2022; Zhang, Qu, et al., 2022). The results revealed that following treatment with kaempferol, the expression levels of proapoptotic proteins Bax, caspase-3, and caspase-9 in LNCaP cells increased, while the expression level of anti-apoptotic protein Bcl-2 decreased. This indicates that kaempferol can also regulate proteins related to apoptosis, thereby promoting apoptosis in LNCaP cells.
However, this study also has certain limitations, such as the lack of discussion on the effects of
Conclusion
Kaempferol extract from
Footnotes
Abbreviations
AR: Apoptosis rate; ARPs: Apoptosis-related proteins; BCA: Bicinchoninic acid; PC: Prostate cancer; CR: Clearance rate; Ctrl: Control; DMSO: Dimethyl sulfoxide; HCK: High-concentration kaempferol; HPLC: High-performance liquid chromatography; LCK: Low-concentration kaempferol; MCK: Medium-concentration kaempferol; MTT: 3-(4,5)-Dimethylthiahiazo(-z-y1)-3,5-di-phenytetrazoliumromide; PIR: Proliferation inhibition rate; TCM: Traditional Chinese medicine; SD: Standard deviation.
Acknowledgments
The authors are grateful to the Research Plan Project of Hunan Provincial Health Commission for providing financial support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Research Plan Project of Hunan Provincial Health Commission (No. D202304056847).
