Abstract
Background
Cerebral ischemic stroke, a devastating neurological condition, occurs when the blood supply to a specific region of the brain is interrupted, leading to a cascade of complex physiological and biochemical processes that ultimately result in cellular dysfunction and tissue damage.
Objectives
This study was dedicated to assessing the beneficial activities of kaempferol along with rehabilitation training in improving cerebral ischemic stroke-induced neurological complications in the experimental rat model.
Materials and Methods
The experimental rats underwent middle cerebral artery occlusion to initiate ischemic stroke. The experimental rats subsequently underwent kaempferol treatment and rehabilitation training alone or in combination. The experimental rats’ neurobehavioral, balance beam, and rotary stick scores were evaluated. The concentrations of inflammation-associated proteins and apoptotic protein levels were assessed in the brain hippocampal tissues of the experimental rats using commercial diagnostic kits.
Results
The kaempferol treatment and rehabilitation training alone or in combination significantly decreased the neurobehavioral, balance beam, and rotary stick scores in the rats with ischemic stroke. Furthermore, the kaempferol treatment and rehabilitation training alone or in combination successfully diminished the inflammation-associated protein levels. It regulated the pro- and anti-apoptotic protein concentrations in the brain hippocampal tissues of the rats with cerebral ischemic stroke.
Conclusion
The present research highlighted that kaempferol treatment and rehabilitation training alone or in combination can enhance the regaining of neurobehavioral and motor functions in rats with ischemic stroke. The present findings may facilitate the advancement of kaempferol as a new therapeutic candidate to treat ischemic stroke.
Introduction
Cerebral ischemic stroke, also known as ischemic stroke, is a significant public health complication worldwide. This condition occurs when the brain’s blood supply is blocked, usually due to a blockage in a blood vessel, leading to tissue damage and potentially severe neurological impairments. Ischemic stroke is responsible for the majority of all stroke incidences, comprising approximately 87% of the total stroke burden globally (Majumder, 2024). The global prevalence of ischemic stroke is staggering, with the disease affecting millions of individuals annually. According to the World Health Organization, cerebrovascular diseases, including ischemic stroke, are accountable for 5.4 million mortalities worldwide each year. The burden of ischemic stroke extends beyond mortality, as survivors often suffer from long-term neurological debilities that considerably impact their capacity to integrate into society, posing financial and social burdens for both the individual and their caregivers (Ahmed et al., 2024). The increased stroke burden continues to exacerbate the immense pressure on people affected by the disease, their families, and societies. The public health significance of ischemic stroke is further amplified by the projected rise in its burden over future decades, primarily driven by demographic transitions, particularly in developing countries (Paul & Candelario-Jalil, 2021).
The primary causes of ischemic stroke encompass thrombosis, embolism, and localized hypoperfusion, any of which can result in diminished or halted cerebral blood flow, thus impacting neurological function. The brain is an organ that is highly sensitive to ischemia, as it requires a constant supply of oxygen and glucose to maintain normal cellular function. Even brief periods of ischemia can initiate a complex sequence of events that may culminate in permanent cerebral damage (Capirossi et al., 2023). The onset of cerebral ischemic stroke implicates a multifaceted cascade, including the disruption of the blood–brain barrier, the activation of inflammatory mechanisms, and the induction of oxidative stress and excitotoxicity. The interruption of blood flow results in the depletion of oxygen and glucose, which, in turn, disrupts cellular metabolism and the production of high-energy phosphates, such as adenosine triphosphate. This energy crisis triggers several events, including the release of excitatory neurotransmitters, the activation of calcium-dependent pathways, and the generation of reactive oxygen species (ROS), all of which participate in the progressive damage and death of brain cells (Kuriakose & Xiao, 2020).
Current treatment approaches focus on restoring blood flow through interventions such as intravenous tissue plasminogen activator or mechanical thrombectomy, as well as managing post-stroke complications like cerebral edema. However, the narrow therapeutic time window and limited efficacy of these treatments have prompted the exploration of alternative and complementary approaches (Arnalich-Montiel et al., 2024). One promising avenue is the use of rehabilitation training, which has been shown to enhance neuroplasticity and functional recovery in stroke patients. Rehabilitation therapies, such as physical, occupational, and speech-language therapies, can help rebuild neural connections, promote the reorganization of brain networks, and facilitate the development of compensatory strategies (Robbins et al., 2023). Another emerging area of research is the investigation of plant-derived bioactive compounds (Açar et al., 2023; Alcorta et al., 2024; Ferrarini et al., 2022). These compounds have been found to target multiple pathways involved in ischemic stroke, including inflammation, oxidative stress, and excitotoxicity. By modulating these pathways, plant-based treatments may help mitigate the secondary damage associated with stroke and support the brain’s natural repair mechanisms (Zhu et al., 2022). Kaempferol, a 3,4′,5,7-tetrahydroxyflavone, is a bioactive flavonoid compound that is found extensively in various vegetables, fruits, and medicinal plants. It has already been highlighted that kaempferol has demonstrated numerous biological properties, including anti-cancer (Ju et al., 2021), anti-asthma (Gong et al., 2013), anti-diabetic (Alkhalidy et al., 2018), anti-diabetic nephropathy (Sharma et al., 2019), hepatoprotective (Lee et al., 2023), anti-ulcer (Li et al., 2018), and neuroprotective (Beg et al., 2018) properties. Nonetheless, there is no sufficient scientific data to claim its effects on improving neurological complications along with the rehabilitation methods in the ischemic stroke model. Consequently, this study was dedicated to assessing kaempferol’s beneficial effects along with the rehabilitation training in improving the cerebral ischemic stroke-induced neurological complications in the experimental rat model.
Materials and Methods
Experimental Rats
The current study employed healthy rats who were acclimated to the laboratory environment (temperature of 26 ± 5ºC, humidity of 55 ± 5%, and a 12-h light/dark cycle) for 7 days and housed in pathogen-free cages.
Middle Cerebral Artery Occlusion (MCAO) Model
The rats were anesthetized using 1% pentobarbital sodium and thereafter kept under anesthesia with 1.5% isoflurane and 80% oxygen. A surgical operation was conducted to access the middle cerebral artery (MCA) and insert an 18-mM filament through a 5-mM incision in the common carotid artery (CA). The filament was subsequently advanced to the internal CA to deliberately occlude the MCA. Following 120 min of occlusion, the string was carefully removed, facilitating the re-establishment of blood flow. The identical procedure was conducted on the sham-operated rats (control) without the implantation of the filament. During the MCAO, the rats’ body temperature was meticulously maintained at a stable 37 ± 0.5ºC utilizing a thermostatic surgical tray. Post-operation, the rats were positioned on the surgical tray until they regained consciousness, after which they were returned to their cages.
Treatment Procedures
The experimental rats were distributed into five groups as follows, each consisting of six rats: Group I (sham-operated control rats), Group II (MCAO rats), Group III (MCAO rats + rehabilitation training rats), Group IV (MCAO + kaempferol-treated rats), and Group V (MCAO + rehabilitation + kaempferol-treated rats). Rehabilitation training commenced 24 h post-operation, involving crawling on rotary bars and balance beams that alternated rotation clockwise and anti-clockwise at 3 rpm. The training occurred for 10 min twice every day over a span of 21 days (Bell et al., 2015). Kaempferol was administered via daily abdomen injection at 25 mg/kg concentration for 21 days.
Analysis of Motor Function and Neurological Behavior
Twenty-four-hours post-operation, animals were evaluated for neurological function utilizing the previous scoring system (Longa et al., 1989). Rats with scores ranging from 1 to 3 were randomized for the following studies. They underwent re-evaluation at 2, 7, 14, and 21 days post-operation before initiating rehabilitation training sessions and kaempferol treatment. The animals were evaluated for motor function prior to rehabilitation training and kaempferol treatment, as detailed previously (Gan et al., 2013).
Analysis of Biochemical Markers
The levels of inflammation-associated proteins were assessed in the hippocampal tissues of experimental rats. The hippocampal tissues were mechanically homogenized in a Tris buffer solution containing a blend of protease inhibitors for 20 min. The homogenate was centrifuged at 10,000 rpm for 20 min. The resultant liquid portion was employed to examine the concentrations of inflammation-associated proteins, including toll-like receptor 4 (TLR-4), myeloid differentiation primary response 88 (MyD88), nuclear factor kappa B (NF-κB), tumor necrosis factor alpha (TNF-α), and interleukin 1 beta (IL-1β). The marker concentrations were evaluated using commercially acquired diagnostic kits. The estimations were performed in triplicate according to the technique outlined by the manufacturer (MyBioSource, USA).
Analysis of Apoptotic Protein Levels
The commercially procured diagnostic kits were utilized to assess the quantities of B-cell lymphoma 2 (Bcl-2), Bcl-2-associated X protein (Bax), and caspase-3 in the brain hippocampal tissue homogenates of the rats. The manufacturer’s specified methods were followed to perform the tests in triplicate (Abcam, USA).
Statistical Analysis
The values are illustrated as mean ± standard deviation (SD) of three replicates. The data are studied using GraphPad Prism. The data changes among experimental groups were analyzed using a one-way analysis of variance (ANOVA) and Duncan’s multiple range test (DMRT). A significance of
Results
Effect of Kaempferol and Rehabilitation Training on the Neurobehavioral Scores in Experimental Rats
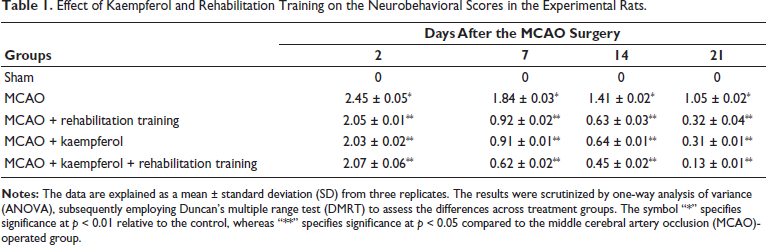
The neurobehavioral scores of experimental rats were evaluated, and the outcomes are presented in Table 1. The rats subjected to MCAO surgery had significantly elevated neurobehavioral scores compared to sham-operated rats. However, the neurobehavioral scores of the kaempferol (25 mg/kg)-treated and/or only rehabilitation-trained rats were decreased than the MCAO-operated rats. Interestingly, the MCAO rats demonstrated significantly reduced neurobehavioral scores when subjected to 25 mg/kg of kaempferol treatment and rehabilitation training (Table 1).
Effect of Kaempferol and Rehabilitation Training on the Neurobehavioral Scores in the Experimental Rats.
Effect of Kaempferol and Rehabilitation Training on the Balance Beam Scores in the Experimental Rats
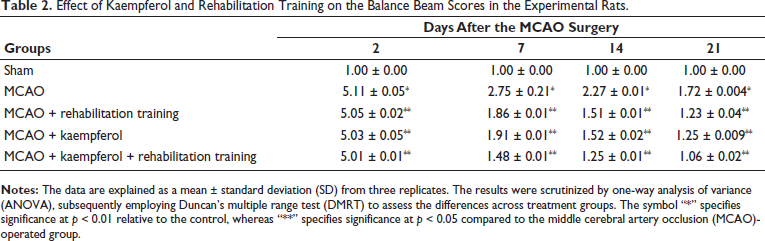
Table 2 illustrates the balance beam scores of experimental rats. A notable increase in the balance beam scores was seen in MCAO-operated rats when compared with sham-operated rats. Contrastingly, the MCAO-operated rats exhibited a considerable reduction in the balance beam scores in response to the kaempferol alone treatment and/or only rehabilitation training when compared with MCAO-operated rats. Captivatingly, the 25 mg/kg of kaempferol treatment along with rehabilitation training demonstrated the effective reduction in the balance beam scores of the MCAO-operated rats.
Effect of Kaempferol and Rehabilitation Training on the Balance Beam Scores in the Experimental Rats.
Effect of Kaempferol and Rehabilitation Training on the Rotary Stick Scores in the Experimental Rats
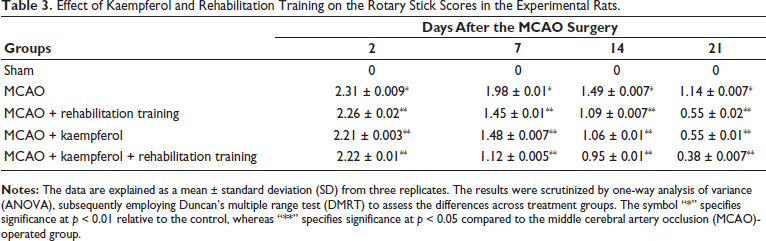
The effect of kaempferol and rehabilitation training on the rotary stick scores of experimental rats was studied, and the results are illustrated in Table 3. As indicated in Table 3, the MCAO-operated rats demonstrated significant elevation in the rotary stick scores when compared with sham-operated rats. Whereas, a considerable reduction in the rotary stick scores was observed after treatment with 25 mg/kg of kaempferol alone and/or subjected to rehabilitation training only. Interestingly, the rotary stick scores of the MCAO-operated rats drastically diminished after treatment with the 25 mg/kg of kaempferol concurrent with the rehabilitation training.
Effect of Kaempferol and Rehabilitation Training on the Rotary Stick Scores in the Experimental Rats.
Effect of Kaempferol and Rehabilitation Training on the Inflammation-associated Protein Levels in Experimental Rats
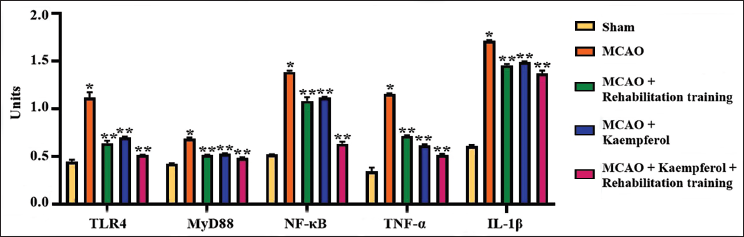
Figure 1 demonstrates the effects of kaempferol and rehabilitation training in the concentrations of inflammation-associated protein levels in the brain hippocampal regions of the experimental rats. The marked increase in the concentrations of inflammatory proteins, including MyD88, TLR-4, NF-κB, TNF-α, and IL-1β, was found in the brain hippocampus of the MCAO-operated rats. In contrast, the treatment with 25 mg/kg of kaempferol alone and/or subjected to rehabilitation training only resulted in a marked reduction in the MyD88, TLR-4, NF-κB, TNF-α, and IL-1β concentrations in the hippocampal tissues of MCAO-operated rats. Nonetheless, the combination of 25 mg/kg of kaempferol treatment concurrent to the rehabilitation training demonstrated the successful reduction of MyD88, TLR-4, NF-κB, TNF-α, and IL-1β concentrations in the hippocampal regions of the MCAO-operated rats.
Effect of Kaempferol and Rehabilitation Training on the Inflammation-associated Protein Levels in the Experimental Rats. The Data are Explained as a Mean ± Standard Deviation (SD) from Three Replicates. The Results were Scrutinized by One-way Analysis of Variance (ANOVA), Subsequently Employing Duncan’s Multiple Range Test (DMRT) to Assess the Differences Across Treatment Groups. The Symbol “*” Specifies Significance at p < 0.01 Relative to the Control, whereas “**” Specifies Significance at p < 0.05 Compared to the Middle Cerebral Artery Occlusion (MCAO)-operated Group.
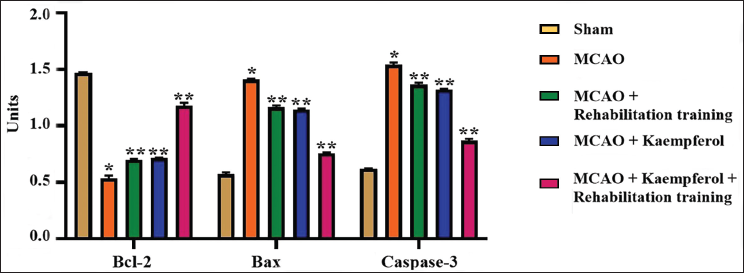
Effect of Kaempferol and Rehabilitation Training on the Apoptotic Proteins in Experimental Rats
The influence of kaempferol and rehabilitation training on the apoptotic protein levels in the hippocampus of experimental rats was assessed, and the findings are demonstrated in Figure 2. The MCAO-operated rats exhibited a drastic increase in the concentrations of Bax and caspase-3. They concurrently diminished Bcl-2 levels in their brain hippocampal tissues compared to the hippocampal tissues of the sham-operated rats. A significant reduction in both Bax and caspase-3 was noted and elevation in Bcl-2 level was observed in the hippocampal tissues of the 25 mg/kg of kaempferol alone-treated and/or rehabilitation-training-only subjected rats. Nevertheless, the 25 mg/kg of kaempferol treatment along with the rehabilitation training remarkably diminished the Bax and caspase-3 concentrations and subsequently elevated the Bcl-2 level in the hippocampal tissues of the MCAO-operated rats.
Effect of Kaempferol and Rehabilitation Training on the Apoptotic Protein Levels in the Experimental Rats. The Data are Explained as a Mean ± Standard Deviation (SD) from Three Replicates. The Results were Scrutinized by One-way Analysis of Variance (ANOVA), Subsequently Employing Duncan’s Multiple Range Test (DMRT) to Assess the Differences Across Treatment Groups. The Symbol “*” Specifies Significance at p < 0.01 Relative to the Control, whereas “**” Specifies Significance at p < 0.05 Compared to the Middle Cerebral Artery Occlusion (MCAO)-operated Group.
Discussion
Cerebral ischemic stroke, a pivotal cause of disability and mortality worldwide, poses a significant burden on both patients and healthcare systems. Despite advancements in medical and surgical interventions, post-stroke therapies remain limited, often leaving patients with severe neurological deficits that severely impact their quality of life. The need for novel, effective, and accessible treatment options has become increasingly apparent, particularly as the prevalence of stroke continues to rise, especially in developing countries (Balch et al., 2020). The current landscape of stroke treatment is oppressed with challenges. While preventive strategies have been advocated for decades, their implementation remains inadequate, leaving many patients vulnerable to the devastating consequences of stroke. Moreover, the therapeutic strategies available for ischemic stroke are accessible to only a small number of patients, often due to significant costs and potential side effects (Cui et al., 2021). In this context, the exploration of plant-based therapies for cerebral ischemic stroke has developed as a favorable avenue to rectify the limitations of current therapies. Despite these obstacles, the transformative potential of neuroplasticity in stroke rehabilitation has garnered increasing attention. Emerging therapies, such as brain stimulation, brain–computer interfaces, and peripheral nervous transfer, have shown promise in improving brain remodeling and restoration, potentially leading to enhanced functional recovery (French et al., 2016). Yet, the implementation of these advanced neurotechnologies remains limited, and a more comprehensive understanding of individual variability in response to interventions, as well as the optimization of intervention timing and duration, is crucial for their successful clinical application. The need for plant-based therapies that can address the unique challenges faced by stroke patients has become increasingly evident (Patil et al., 2022).
Analyzing neurobehavioral scores, which measure various aspects of cognitive, motor, and sensory function, is crucial in understanding the extent and nature of these deficits and guiding the rehabilitation process. Ischemic stroke can trigger a complex sequence of pathophysiological events, including diminished cerebral blood flow, inadequate oxygen and glucose supply, and the activation of various cellular and molecular cascades that can result in permanent brain damage. These processes can result in numerous neurological impairments, which can be captured by assessing neurobehavioral scores (Lynch et al., 2024). Neurobehavioral scores comprehensively evaluate a patient’s functional status and can help identify specific areas of deficit, such as sensory or motor impairments, cognitive dysfunction, or language deficits. By evaluating these scores, clinicians can acquire more insights into the extent and nature of the patient’s stroke-related deficits, which can be used to tailor rehabilitation strategies and set realistic goals for recovery (Martinez-Tapia et al., 2020). Moreover, the analysis of neurobehavioral scores can also have prognostic value. Studies have shown that the severity of initial neurological deficits, as measured by these scores, can predict long-term functional outcomes and the likelihood of achieving meaningful recovery. The careful analysis of neurobehavioral scores can inform clinical decision-making, guide the development of personalized rehabilitation plans, and help patients and their families understand the potential trajectory of recovery (Nalamolu et al., 2021). The current study has demonstrated that the rats subjected to MCAO surgery had significantly elevated neurobehavioral scores. However, the neurobehavioral scores of both kaempferol alone, only rehabilitation trained, and kaempferol-treated along with rehabilitation trained rats were found to be decreased than the MCAO-operated rats. These findings evidence the improvement of neurobehavioral changes in the MCAO rats by the kaempferol treatment and rehabilitation training techniques.
One critical aspect of post-stroke rehabilitation is the assessment and management of balance impairments, which are common among stroke survivors. Balance control is a multifaceted process necessitating precise assessment of sensory data and suitable motor responses. Individuals who have experienced a stroke frequently display several sequelae, including compromised balance, which can profoundly affect their everyday activities, ambulation, and mobility (Pinosanu et al., 2024). Assessing balance control in stroke patients is essential for minimizing hazardous activities and implementing successful treatment strategies. Optimal balance is crucial for stepping patterns, gait velocity, and preliminary gait training in individuals with stroke. Furthermore, cognitive function influences balance, and individuals with cognitive dysfunction have been documented to experience balance deficits and an increased risk of falls (Singh et al., 2022). The analysis of balance beam scores can provide valuable data about the rehabilitation and recovery of individuals with cerebral ischemic stroke. Balance regulation encompasses both voluntary and reactive aspects, activated by the central nervous system in reaction to self-generated or externally generated disturbances. Voluntary adjustments are linked to feedforward systems that aid in the planning and execution of movements while preserving balance control against self-induced disturbances. In contrast, reactive adjustments refer to the automatic correction of balance in response to external distresses that challenge the postural system (Li et al., 2022). A comprehensive assessment of balance control, including both static and dynamic steady-state balance, as well as proactive and reactive balance control, can help identify specific impairments and guide targeted rehabilitation interventions. Balance beam scores can be used to measure these different aspects of balance control, providing a comprehensive understanding of the patient’s balance abilities and the progress made during rehabilitation (Feng et al., 2020). It has been conducted to investigate the importance of balance assessment and rehabilitation in post-stroke patients. It highlights the critical role of early and targeted physical rehabilitation in promoting brain organization, plasticity, and the recovery of balance control (Wang et al., 2024). In this work, the MCAO-operated rats exhibited a considerable reduction in the balance beam scores in response to the kaempferol treatment alone or in combination with rehabilitation training. This is evidence of the modulation of neurobehavioral changes in the MCAO rats.
Rehabilitation efforts to restore motor function and cognitive abilities are crucial for stroke survivors, as they can significantly improve quality of life and reduce the burden on healthcare systems. One promising approach to stroke rehabilitation is the analysis of rotary stick scores, which can provide valuable insights into a patient’s sensorimotor performance and guide the development of targeted interventions. The rotary stick test is a widely used assessment tool in stroke rehabilitation, where patients are asked to manipulate a handheld rotary stick in specific ways to measure their motor control, coordination, and reaction time. This information can be beneficial for understanding the influence of cerebral ischemic stroke on fine motor skills and the potential for neuroplastic changes that can facilitate functional recovery (Trueman et al., 2017). It has highlighted the importance of analyzing rotary stick scores in the context of stroke rehabilitation. These investigations have demonstrated that the analysis of rotary stick performance can reveal the extent of motor impairment, identify areas for targeted intervention, and track the progress of rehabilitation efforts (Choi et al., 2012). It has been shown that the analysis of rotary stick scores can help clinicians distinguish between motor deficits, such as impaired coordination, reduced stability, or delayed reaction times. By understanding the specific nature of a patient’s motor impairments, clinicians can develop more personalized rehabilitation plans that address the unique challenges faced by each individual. Improvements in rotary stick performance may indicate the restoration of neural pathways and the strengthening of sensorimotor connections, which can be crucial for the recovery of motor function (Wayman et al., 2016). The findings of this work indicated the increased rotary stick scores in the MCAO-operated rats. Interestingly, a considerable reduction in the rotary stick scores was observed after treatment with kaempferol alone or along with the rehabilitation training, which highlights the increase in sensorimotor performance of MCAO rats.
Primarily, cerebral ischemic stroke is developed because of the interruption of blood delivery to the brain. This disruption initiates several mechanisms, including inflammation, that ultimately lead to irreversible neuronal damage and brain damage. The primary players in this process are the TLR-4 and the MyD88, which have participated in the pathophysiology of ischemic stroke (Sun et al., 2021). TLR-4 is a member of the toll-like receptor family, which plays a pivotal role in activating immune responses. In the context of cerebral ischemic stroke, TLR-4 has been shown to be implicated in the onset of atherosclerotic lesions and the adverse remodeling that may occur after ischemic injury to the brain. TLR-4 activation leads to the inflammatory cell influx, like microglia and astrocytes, which participate in the inflammatory response and tissue damage observed in ischemic stroke (Ashayeri Ahmadabad et al., 2020). MyD88, on the other hand, is an adaptor protein essential for the signaling pathways of various toll-like receptors, including TLR-4. MyD88 is a key mediator of the inflammatory response triggered by TLR-4 activation, as it facilitates the triggering of transcription factors and subsequent pro-inflammatory cytokine production and chemokines. In the context of cerebral ischemic stroke, MyD88-dependent pathways have been shown to participate in ischemic brain damage and the associated inflammatory response (Gong et al., 2023). The stroke-induced inflammatory cascade involves the release of various harmful compounds, which compromise the integrity of the vascular system, lead to cell death, and ultimately result in secondary brain damage. The activation of the MyD88-dependent pathways plays a crucial role in this inflammatory response, further exacerbating the detrimental effects of ischemic stroke (Mao et al., 2023). Similarly, the present findings also demonstrated a significant increase in the concentrations of TLR-4 and MyD88 in the hippocampus of the MCAO-operated rats. Nonetheless, the treatment with the kaempferol alone or in combination with the rehabilitation training successfully diminished the TLR-4 and MyD88 concentrations in the MCAO rats, which proves its anti-inflammatory effects.
The primary drivers of ischemic stroke are thrombosis, embolism, and focal hypoperfusion, all of which can contribute to a reduction in cerebral blood flow and the subsequent cascade of events. One of the key mechanisms underlying the pathophysiology of cerebral ischemic stroke is the inflammatory response, which is mediated by various cytokines and transcription factors (Pawluk et al., 2020). NF-κB is a pivotal transcription factor essential in regulating the inflammatory response during cerebral ischemic stroke. Various stimuli, including ischemia, activate NF-κB, enhancing the pro-inflammatory cytokine expressions (Jover-Mengual et al., 2021). TNF-α is a potent mediator of inflammation and has been shown to have a crucial role in neuroinflammation. Furthermore, it can contribute to the recruitment of immune cells, the breakdown of the blood–brain barrier, and the exacerbation of neuronal injury. It can also play a role in repairing and regenerating damaged tissue (Lin et al., 2021). IL-1β is another key inflammatory cytokine that participates in the pathophysiology of cerebral ischemic stroke. Activated macrophages and microglia produce it and can also contribute to the inflammatory response, leading to further tissue damage and neuronal death (Alsbrook et al., 2023). The current findings demonstrate the increased levels of NF-κB, TNF-α, and IL-1β in the hippocampal tissues of the MCAO-operated rats, which witnesses the onset of inflammatory response in the brain tissues. Captivatingly, the kaempferol alone or in conjunction with the rehabilitation training effectively reduced those inflammatory biomarkers in the hippocampal tissues of the MCAO-operated rats. These outcomes proved the anti-inflammatory properties of the kaempferol against ischemic stroke conditions.
A key aspect of the pathogenesis of ischemic stroke is neuronal apoptosis, which is mediated by a delicate balance between pro- and anti-apoptotic proteins. The Bcl-2 family of proteins, including the pro-apoptotic Bax and the anti-apoptotic Bcl-2, play a central role in regulating the intrinsic apoptotic pathway (Korshunova et al., 2021). When Bax is activated, it triggers the release of cyt-c from the mitochondria, activating caspase-3, a crucial executioner caspase that drives the apoptotic process. Ischemic injury and reperfusion further exacerbate this imbalance, leading to an abundance of ROS that can directly damage mitochondria and initiate the apoptotic cascade (Zhang et al., 2022). In addition to the Bcl-2 family, other signaling pathways have participated in the onset of cerebral ischemic stroke. The inflammatory response, driven by the upregulation of cytokines and chemokines, can also contribute to the apoptotic process and exacerbate neuronal injury (Pei et al., 2023). The current results exhibited a drastic elevation in the concentrations of apoptotic proteins Bax and caspase-3 and concurrently diminished the Bcl-2 levels in the brain hippocampal tissues of the MCAO-operated rats. A significant reduction in both Bax and caspase-3 was observed, and an elevation in Bcl-2 level was found in the hippocampal tissues of the kaempferol treatment alone or in combination with the rehabilitation training in the MCAO rats. These findings clearly proved that the kaempferol treatment along with rehabilitation training successfully mitigated the neuronal apoptosis in the brain tissues of rats with ischemic stroke via regulating pro- and anti-apoptotic proteins.
Conclusion
In conclusion, the present research highlighted that kaempferol treatment and rehabilitation training alone or in combination can enhance the regaining of neurobehavioral and motor functions in ischemic stroke-induced rats. The kaempferol treatment and rehabilitation training alone or in combination led to a reduction in neurobehavioral scores and motor functions, decreased the inflammation-associated protein concentrations, and regulated the pro- and anti-apoptotic protein levels in the rats with ischemic stroke. The present findings may facilitate the advancement of kaempferol as a new therapeutic candidate to treat ischemic brain injury. Moreover, additional research is necessary to elucidate the therapeutic mechanisms of kaempferol against cerebral ischemic stroke.
Footnotes
Abbreviations
ANOVA: Analysis of variance; Bax: Bcl-2-associated X protein; Bcl-2: B-cell lymphoma 2; CA: Carotid artery; DMRT: Duncan’s multiple range test; IL-1β: Interleukin 1 beta; MCA: Middle cerebral artery; MCAO: Middle cerebral artery occlusion; MyD88: Myeloid differentiation primary response 88; NF-κB: Nuclear factor kappa B; rpm: Rotations per minute; ROS: Reactive oxygen species; SD: Standard deviation; TLR-4: Toll-like receptor 4; TNF-α: Tumor necrosis factor alpha.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This work has been approved by the Institutional Animal Ethical Committee of the Department of Rehabilitation Medicine, Shanghai Pudong Gongli Hospital, Shanghai, China.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Shanghai Pudong New District Health System Excellent Young Medical Talent Training Program, PWRq2023-08.
