Abstract
Objectives
To study the antioxidant effects of the traditional Mongolian medicine Zha-Chong-Shi-San-Pill ethanol extract (EEZC) on cerebral stroke for future clinical use.
Materials and Methods
Active components and mechanisms of EEZC against cerebral stroke were predicted and analyzed via ultra-performance liquid chromatography-electrospray ionization tandem mass spectrometry (UPLC-ESI-MS/MS), network pharmacology, and molecular docking. Next, an oxidative damage model was established in bEnd.3 cells using H2O2 to simulate cerebral stroke damage. Multiple cellular tests were performed to verify the mechanisms, including cell counting kit-8 (CCK-8), flow cytometry, terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) staining, enzyme-linked immunosorbent assay (ELISA), and Western blotting.
Results
It was found that EEZC conferred protection against cerebral stroke H2O2-treated bEnd.3 cells. Moreover, in vitro experiments revealed that EEZC increased cell survival rates and suppressed cell apoptosis and reactive oxygen species (ROS) release. Moreover, EEZC reduced the levels of interleukin-6 (IL-6) and MMP-9, and increased the levels of interleukin-10 (IL-10), superoxide dismutase (SOD), glutathione (GSH), vascular endothelial growth factor (VEGF), phospho-endothelial nitric oxide synthase (p-eNOS), phospho-phosphatidylinositide 3-kinases (p-pi3k), and phospho-protein kinase B (p-AKT).
Conclusion
These results strongly suggested that EEZC might protect against cerebral stroke via the PI3K/Akt/eNOS signaling pathway.
Keywords
Introduction
Cerebral stroke is a “brain attack” disease, which is caused by cerebral artery occlusion, with high mortality and disability rates (Xiong et al., 2022). In China, an estimated 2 million new cases of cerebral stroke are reported annually, accounting for 70% of all strokes (Herpich & Rincon, 2020). Currently, cerebral stroke is managed using several Western medicines, including thrombolytic drugs, anti-coagulation therapy, defibrate, anti-hypertensive, lipid-lowering medication, and so on (Rabinstein, 2020). However, the combined application of these drugs increases the occurrence of adverse reactions, posing a heavy burden on the family (Herpich & Rincon, 2020). Therefore, there is an urgent need for novel, safe, affordable, and efficacious drugs against cerebral stroke.
According to Mongolian medicine records, various Mongolian formulas have exhibited a positive effect in improving cerebral stroke (Bai & Fu, 2022). Zha-Chong-Shi-San-Pill (ZC-13) is a typical Mongolian representative preparation that has been widely used to treat “Sa bing” (Bao et al., 2021). This formula is composed of Hezi (Fructus Terminalia), Zhicaowu (Wild Aconite, Root), Shichangpu (Acorus tatarinowii), Muxiang (Aucklandia Radix, Aucklandia lappa), Shexiang (Moschus), Shanhu (Linnaeus), Zhenzhu (pearl), Dingxiang (Flos Eugenia caryophyllata), Chenxiang (Lignum, Aquilaria sinensis), Yuliangtu (red claystone), Cishi (Magnetitum), Gancao (Radix Glycyrrhiza uralensis), and Roudoukou (Semen Myristica fragrans), with the main function being to remove wind, invigorate the circulation of blood, and calm the heart and mind (Guo, 2020). A previous study showed that ZC-13 treatment inhibited inflammatory response in cerebral ischemia, exerted anti-coagulant and anti-thrombotic benefits, and prevented cell apoptosis. Moreover, its water extracts reduced lipid peroxidation and eliminated oxygen free radicals (Dong et al., 2022; Shen et al., 2022; Tian et al., 2022). Although ZC-13 has shown good therapeutic effects, its precise mechanisms of action and active constituents remain elusive, limiting its clinical utility. Ethanol extraction, a widely used method in traditional Chinese medicine research, was utilized to investigate the potential mechanisms by which Zha-Chong-Shi-San-Pill ethanol extract (EEZC) alleviates oxidative stress. This study employed ultra-performance liquid chromatography-electrospray ionization tandem mass spectrometry (UPLC-ESI-MS/MS), network pharmacology, and H2O2-treated bEnd.3 cells to elucidate these mechanisms (Lou et al., 2004; Zhang et al., 2015).
Materials and Methods
Materials
ZC-13 was obtained from the Affiliated Hospital of Inner Mongolia University. H2O2 (323381-25ML) was acquired from Merck (American). Fetal bovine serum (FBS) was acquired from Hyclone. The one-step terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) apoptosis assay kit and counting kit-8 (CCK-8) kit were obtained from Beyotime Technology Co. Ltd. (China). Malondialdehyde (MDA), superoxide dismutase (SOD), and glutathione (GSH) enzyme-linked immunosorbent assay (ELISA) kits were purchased from Proteintech Group, Inc. (China). Anti-bodies of PTEN (14796S), PI3K (9532S), phospho-phosphatidylinositide 3-kinases (p-PI3k) (9662S), AKT (9494S), eNOS (9572S), phospho-endothelial nitric oxide synthase (p-eNOS) (9574), vascular endothelial growth factor (VEGF) (50661), MMP-9 (13667S), and phospho-protein kinase B (p-AKT) (9504S), and secondary anti-bodies labeled were obtained from Cell Signaling Technology Inc. (American). Enhanced chemiluminescence (ECL) substrate was purchased from Merck Millipore (American).
UPLC-ESI-MS/MS Analysis
The ZC-13 powder was extracted three times with 70% ethanol at room temperature. The extracts were then filtered and concentrated at 40℃. ZC-13 compounds were analyzed using UPLC-ESI-MS/MS. The chromatographic separation was carried out using a Waters ACQUITY UPLC HSS T3 C18 column (2.1 × 100 mm, 1.8 µm). The diode array detector (DAD) wavelength was set at 254 nm. Compound Discoverer 3.1.0.305 software was used to identify compounds.
Obtaining ZC-13, Oxidative Stress, and Cerebral Stroke-related Targets
Active ingredients were screened for oral bioavailability (OB) ≥30% and drug-likeness (DL) ≥0.18 using the Traditional Chinese Medicine Systems Pharmacology (TCMSP) database based on identified compounds via UPLC-ESI-MS/MS analysis. Targets of these ingredients were obtained from TCMSP and Swiss Target Prediction. Putative targets of oxidative stress and cerebral stroke were acquired from GeneCards (
Protein–Protein Interaction (PPI) Mapping of ZC-13, Oxidative Stress, and Cerebral Stroke
Intersections of active ingredient-related targets, oxidative stress targets, and cerebral stroke-related targets were obtained via Venny 2.1.0 as potential targets of ZC-13 for cerebral stroke. Then, these potential targets were employed to construct PPI networks using STRING software (
Gene ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analyses were performed on core targets using the database for annotation, visualization, and integrated discovery (DAVID) database (
Molecular Docking
The key component structure files of interleukin-6 (IL-6) (PDB ID:4yw7), TP53 (PDB ID:8syg), CAP3 (PDB ID:1cp3), JUN (PDB ID:2h7h), and MMP-9 (PDB ID:5th9) in structure data file (SDF) format were retrieved from the Protein Data Bank (PDB) format. Ligands preparations were downloaded from PubChem (
Cell Culture and CCK-8 Assay
The bEnd.3 cells were cultured in Dulbecco’s modified Eagle medium (DMEM) supplemented with 10% FBS and 0.01% anti-biotics (penicillin and streptomycin) in a 5% CO2 incubator at 37℃. After reaching 80%–90% confluency, cells were sub-cultured in 96- or 6-well plates. Next, adherent cells were treated with 800 µM H2O2 for 3 h in DMEM. Then, the medium was removed and replaced with DMEM containing different concentrations of EEZC for 48 h. Cell viability was determined using the CCK-8 assay and absorbance at 490 nm.
TUNEL Assay
Cells in each group were treated with H2O2 and EEZC. The cells were fixed with 4% paraformaldehyde, then incubated with 0.2% Triton X-100 for 30 min, washed with phosphate-buffered saline (PBS) for 10 min, added 50 µL fluorescein-dUTP, and incubated at 37°C for 60 min. They were washed twice with PBS or Hanks’ balanced salt solution (HBSS) and observed under a fluorescence microscope.
Annexin V-FITC/PI Apoptosis Detection
The apoptotic rate was assessed using the Annexin V-FITC/PI Apoptosis Detection Kit and flow cytometry. bEnd.3 cells were seeded in 6-well plates (3 × 105/well) overnight, treated with EEZC and H2O2, then incubated with binding buffer, Annexin V-FITC, and propidium iodide (PI) for 20 min at 37℃. The cells were analyzed using FlowJo 10.7.1 software.
Intracellular Reactive Oxygen Species (ROS) Production Assay
Intracellular ROS generation was measured using the 2′,7′-dichlorofluorescein diacetate (DCFH-DA) method and flow cytometry. bEnd.3 cells were seeded in 6-well plates (3 × 105/well), treated with EEZC and H2O2, and then collected. DCFH-DA (5 µM) was added for 30 min, and ROS levels were analyzed by flow cytometry.
ELISA Assay
Cells in each group were treated with H2O2 and EEZC, washed with PBS, and lysed with radioimmunoprecipitation assay (RIPA) buffer containing phosphatase and protease inhibitors. They were then scraped and centrifuged at 13,000 rpm for 15 min at 4°C to remove debris, and the supernatant was collected for protein quantification based on the bicinchoninic acid (BCA) assay kit. An 80 µg protein content was quantified in a 30 µL sample volume. The concentrations of MDA, total SOD, VEGF, interleukin-10 (IL-10), and interleukin-1β (IL-1β) were determined by measuring the absorbance values at 500 nm following the manufacturer’s protocols.
Western Blot Assay
H2O2-treated bEnd.3 cells were lysed in cell lysis buffer with protease and phosphatase inhibitors at 4℃ for 30 min. The cells were then centrifuged at 13,000 × g for 5 min to collect the supernatant, and the total protein was quantified to 80 µg using a BCA kit. Proteins were separated using sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinylidene fluoride (PVDF) membranes. After blocking with 5% non-fat milk for 1.5 h, the membranes were incubated overnight at 4°C with primary anti-bodies (PTEN, p-PI3K, PI3K, p-AKT, AKT, p-eNOS, eNOS, MMP-9, and VEGF at the ratio of 1:1,000). They were then washed three times with tris-buffered saline with Tween (TBST) for 5 min and treated with anti-rabbit or anti-goat IgG secondary anti-bodies. After three TBST washes, protein bands were visualized with a Chemiscope and quantified using ImageJ software.
Statistical Analysis
Data are presented as the mean ± standard deviation (SD). One-way analysis of variance (ANOVA) with the Tukey–Kramer post hoc test assessed significant differences between group means. All experiments were conducted in triplicate, and results were visualized using GraphPad Prism 8, with p values < .05 and < .01 deemed statistically significant.
Results
Effect of EEZC on Cell Viability in H2O2-treated bEnd.3 Cells
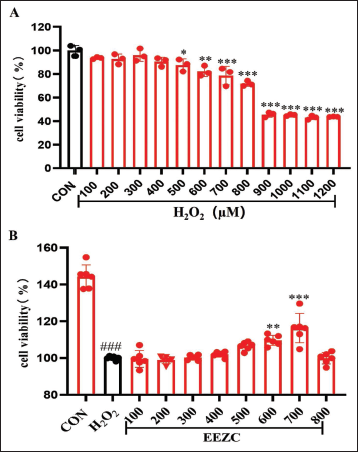
To determine the optimal conditions for modeling, cells were exposed to H2O2 concentrations ranging from 100 to 1,200 µM for 3 and 6 h. Significant cell damage was evident at a concentration of 600 µM after 3 h (Figure 1A), with 30% of cell damage observed at 800 µM. Thus, we selected 800 µM for 3 h for future experiments. Next, we evaluated the therapeutic effects of EEZC on H2O2-induced oxidative damage in bEnd.3 cells. The extract significantly increased cell viability in a dose-dependent manner (100–700 µg/mL). Therefore, concentrations of 175, 350, and 700 µg/mL were chosen for further investigation (Figure 1B).
Chemical Composition of EEZC
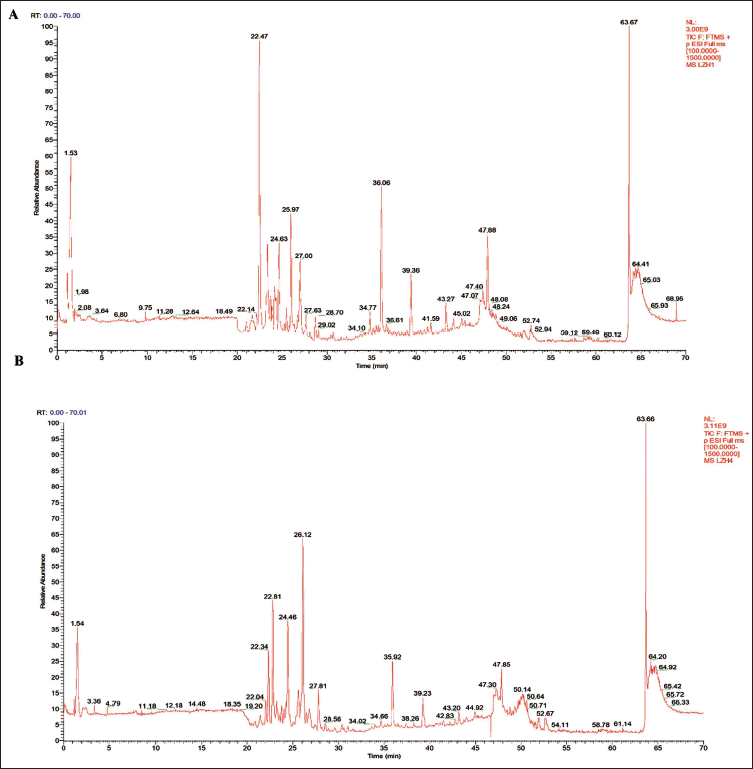
The chromatograms of EEZC in negative and positive ion modes are shown in Figures 2A and B. In total, 319 compounds, including 74 flavonoids, 74 organic acids, 46 terpenes, 21 phenols, 21 alkaloids, 7 coumarins, 5 phenylpropanoids, and 71 others, were identified using Compound Discover 3.1.0.305 software.
Chemical Composition of Zha-Chong-Shi-San-Pill Ethanol Extract (EEZC). Total Ion Chromatogram Monitored in Negative (A) and Positive (B) Ion Modes for EEZC.
Potential Targets and Pathways of EEZC for Cerebral Stroke
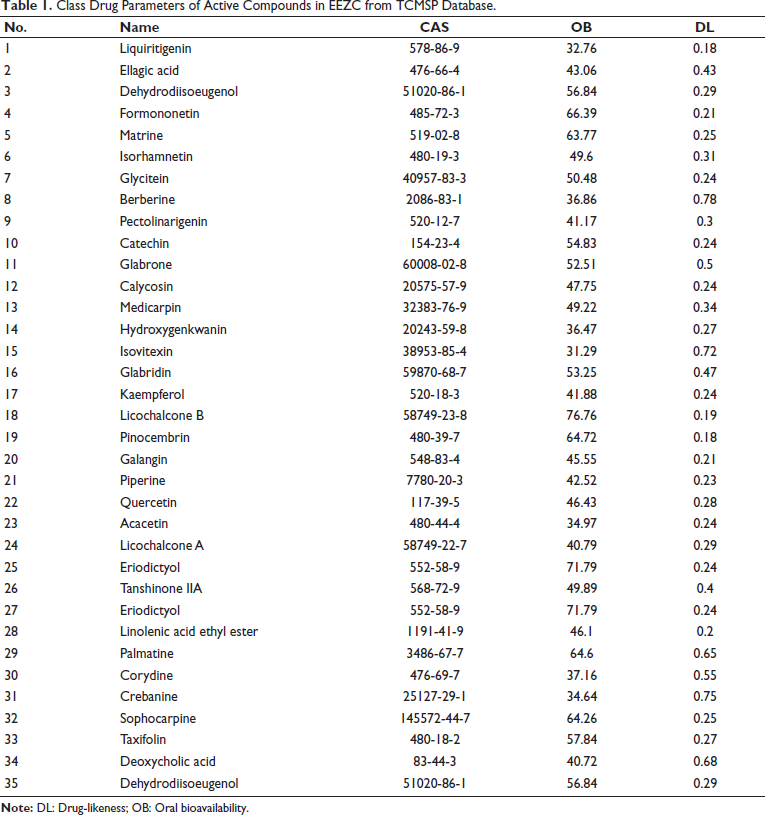
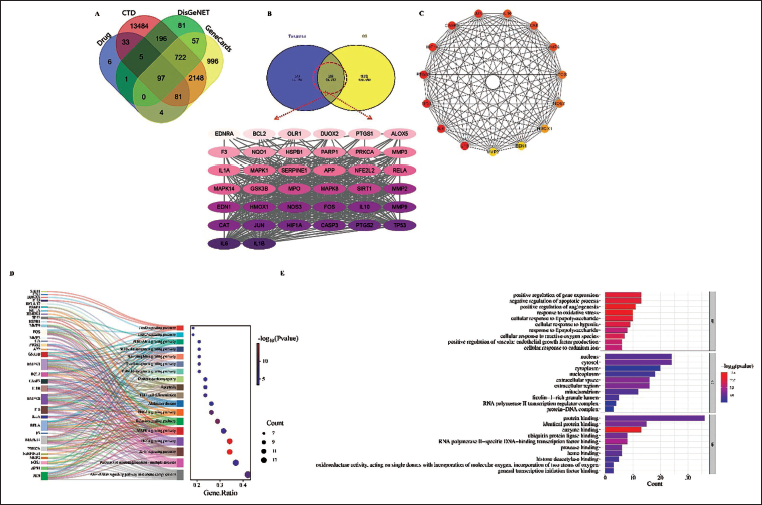
A total of 26 active constituents were screened based on OB and DL criteria (Table 1). We identified 227 drug targets, 2,990 oxidative stress targets, and 18,546 disease targets. Using Venny 2.1, we identified 38 potential targets of the EEZC related to cerebral stroke (Figure 3A and B). The top 10 core targets included IL-1β, IL-6, TP53, PTGS2, HIF-1A, CASP3, JUN, IL-10, catalase (CAT), and MMP-9 (Figure 3C). To systematically understand ZC-13’s physiological and biological functions in cerebral stroke, GO and KEGG enrichment analyses of 38 intersection targets were shown in Figure 3D and E. EEZC mainly focuses on aging, response to oxidative stress, and cell apoptosis, and it mainly acts on cAMP, FoxO, PI3K/Akt, NOD-like receptors, and neurotrophin signaling pathways.
Class Drug Parameters of Active Compounds in EEZC from TCMSP Database.
Potential Targets and Pathways of Zha-Chong-Shi-San-Pill Ethanol Extract (EEZC) for Cerebral Stroke. (A) Venn Diagram for the Intersection of the Active Components-cerebral Stroke Targets. (B) Venn Diagram for the Intersection Targets and Oxidative Stress Targets. (C) Protein–Protein Interaction (PPI) Network of the 38 Potential Therapeutic Targets. Each Node Represents a Relevant Gene, and the Node Color Indicates the Strength of the Degree. (D) Mulberry Diagram of Kyoto Encyclopedia of Genes and Genomes (KEGG) Pathways. (E) Gene Ontology (GO) Functional Terms.
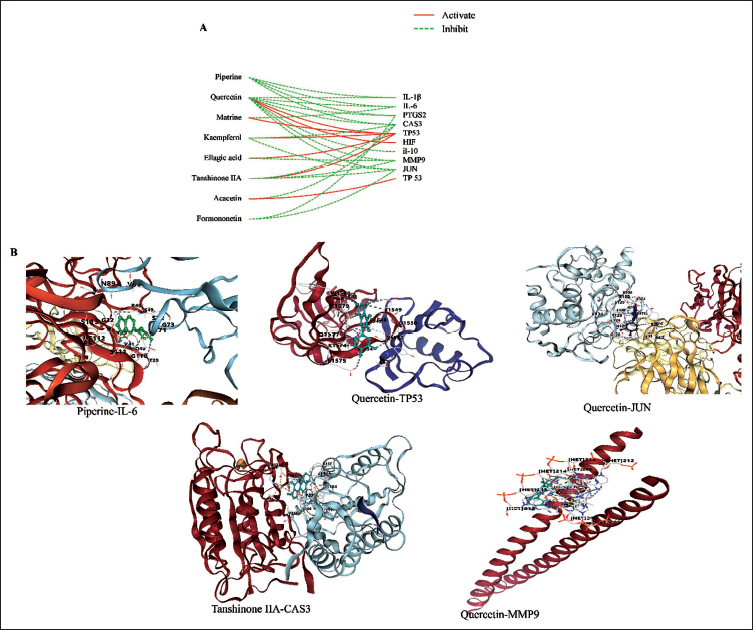
Molecular Docking and Analysis
To identify the primary active ingredients of ZC-13, a chat was established to link the ingredients to targets. It was found that quercetin, piperine, tanshinone IIA, and kaempferol were the key components. Molecular docking is used to identify the most stable conformation of a small molecule ligand with a target protein, with binding energies below −5.0 kcal/mol indicating strong interactions; lower values suggest even stronger binding. Molecular docking analysis showed that quercetin, piperine, tanshinone IIA, and kaempferol exhibited strong binding affinities with key targets IL-6, caspase-3, MMP-9, TP53, and JUN. The lowest binding energy for each ingredient-target pair was visualized using PyMol software 2.6.0 in Figure 4.
Molecular Docking of Active Compounds and Core Targets. (A) The Diagram of Active Compounds and Their Corresponding Core Targets. (B) The Strongest Binding of 3D Docking Molecules in Quercetin, Piperine, Tanshinone IIA, and Kaempferol to IL-6, TP53, CAS3, MMP-9, and JUN.
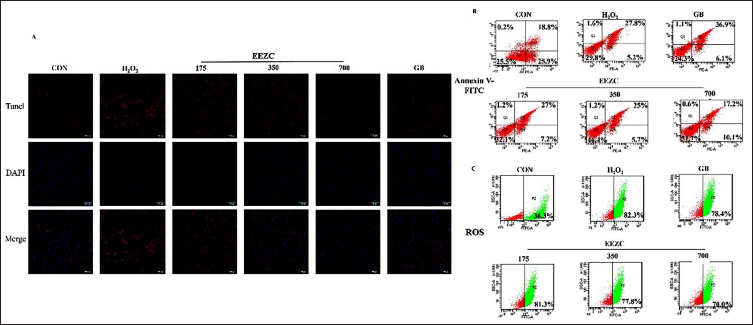
EEZC Exerts Anti-apoptotic and Antioxidant Effects on H2O2-induced bEnd.3 Cells
Further experiments were conducted to investigate the anti-apoptotic and antioxidant properties of EEZC on H2O2-induced oxidative damage in bEnd.3 cells. The TUNEL staining and Annexin V-FITC/PI assays revealed that EEZC treatment reduced H2O2-induced apoptosis, particularly in the late stage (Figure 5A and B). Given that oxidative stress can induce apoptosis, the antioxidant effects of EEZC were also evaluated by measuring ROS levels via flow cytometry. Figure 5C demonstrates that H2O2 exposure for 3 h significantly raised ROS levels in bEnd.3 cells, while EEZC significantly reduced them, indicating that it effectively eliminates ROS.
Zha-Chong-Shi-San-Pill Ethanol Extract (EEZC) Exerts Anti-apoptotic and Antioxidant Effects on H2O2-treated bEnd.3 Cells. (A) Terminal Deoxynucleotidyl Transferase dUTP Nick-end Labeling (TUNEL) Staining Results to Determine the (B) Anti-apoptotic Effects and Flow Cytometry Analysis of (C) Reactive Oxygen Species (ROS) Levels.
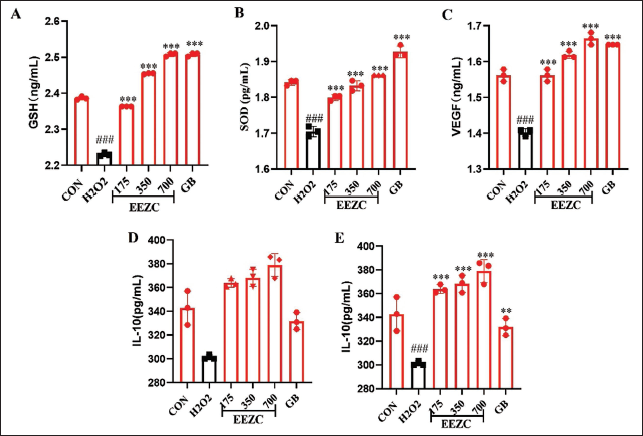
EEZC Inhibits the Release of Inflammatory and Oxidative Stress Indicators
Network pharmacology revealed that the EEZC-improved cerebral stroke was associated with oxidative stress, apoptosis, and inflammatory factors. Therefore, we further explored the expression of associated inflammatory and oxidative stress (including IL-1β, IL-10, GSH-Px, T-SOD, and VEGF). The changes in the cell supernatant related to oxidative stress and inflammatory factors after treatment with EEZC are displayed in Figure 6. Compared with the control group, the groups exposed to H2O2 exhibited significantly increased IL-1β activity (p < .01) and decreased IL-10, GSH, SOD, and VEGF activities (p < .01). Meanwhile, the groups treated with EEZC showed opposite effects.
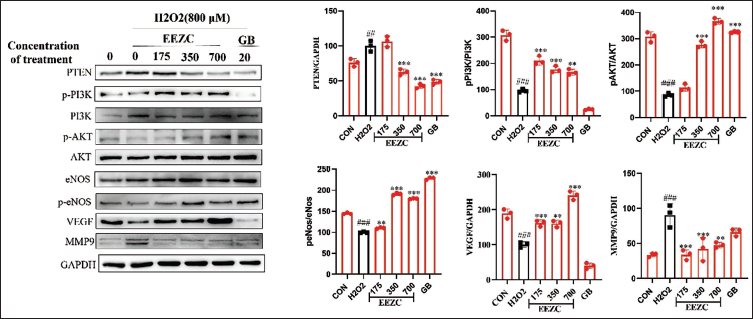
EEZC Activates PI3K/AKT/eNOS Signaling Pathway
Based on the results of network pharmacology, we validated the effect of EEZC on the PI3K/Akt/eNOS pathway and its downstream related genes using Western blot analysis, and the results are depicted in Figure 7. The results indicated that EEZC activated the PI3K, AKT, and eNOS proteins by increasing their phosphorylation, thereby upregulating VEGF expression and significantly inhibiting PTEN and MMP-9 expression.
Discussion
Ischemic cerebrovascular disease presents a significant threat to human health and survival, characterized by high incidence and mortality rates (Jiang et al., 2022). Currently, the primary treatment for ischemic stroke is intravenous thrombolysis within 4.5 h or interventional thrombectomy within 6 h to quickly restore blood flow. However, this approach may trigger biochemical reactions that lead to ischemia-reperfusion injury, worsening neuronal death and impairing neurological function. Clinical treatments for stroke focus on two main strategies: restoring blood flow through intravenous thrombolysis or endovascular therapy and protecting brain cells with neuroprotective drugs like antioxidants and ion channel blockers (Bo et al., 2022; Rabinstein, 2020). ZC-13, as a traditional Mongolian medicine, has been used for many years to alleviate cerebrovascular diseases such as stroke, cerebral ischemia, and encephalorrhagia (Bao et al., 2021). In this study, we found that EEZC significantly reversed H2O2-induced cell damage and rescued endothelial cell impairment at concentrations of 350 and 700 µM. This demonstrated that EEZC reduced vascular oxidative damage and reperfusion injury in cerebral ischemia treatment, suggesting that it has the potential to mitigate adverse reactions associated with polypharmacy.
Utilizing UPLC-ESI-MS/MS and network pharmacology analyses, we found that the pharmacological mechanisms underlying the antioxidant effects of EEZC were likely driven by flavonoid constituents, primarily the quercetin, piperine, tanshinone IIA, and kaempferol. Similarly, Li and Peng et al. showed that quercetin treatment attenuated cerebral ischemic injury by promoting microglia/macrophages M2 polarization and inhibiting ferroptosis through PI3K/Akt/NF-κB signaling pathway and Nrf2/HO-1 signaling pathway, respectively (Li et al., 2023; Peng et al., 2024). Zhang et al. reported that piperine ameliorated ischemic stroke-induced brain injury in rats by regulating the PI3K/AKT/mTOR pathway (Zhang, Yang et al., 2022). Su et al. uncovered that tanshinone IIA treatment resolved traumatic brain injury by reducing ischemia-reperfusion via the miR-124-5p/FoxO1 axis (Su et al., 2024). Zhang et al. showed that kaempferol inhibited the activation and number of neutrophils in the rat peripheral blood and brain by the JAK1/STAT3 pathway (Zhang et al., 2022; Gao et al., 2022). These research findings provide additional empirical evidence supporting our assertion that these four compounds are promising candidates for quality markers of ZC-13. By addressing the disconnect between current research on ZC-13 quality markers and their clinical application, this research provides a solid foundation for future in-depth investigations into the identification of reliable quality markers.
On the other hand, from our network pharmacology analysis, we discovered that EEZC improves oxidative stress during cerebral ischemia and may mainly affect the top 10 genes of IL-1B, IL-6, TP53, PTGS2, HIF-1A, CASP3, JUN, IL-10, CAT, MMP-9 and top five pathways of cAMP, FoxO, PI3K/Akt, NOD-like receptor, and neurotrophin signaling pathways. Nonetheless, it must be acknowledged that the analysis within network pharmacology predominantly relies on data derived from various databases, which often include computationally predicted interactions and effects (Li & Zhang, 2013). Future studies are needed to validate the present findings via empirical experimentation. Numerous studies have indicated that the PI3K/Akt/eNOS signaling pathway can prevent cell death and promote survival, making it an essential pathway with a potential therapeutic target in stroke, diabetes, cardiovascular diseases, and cancer. In a cerebral stroke, it enhances antioxidant defenses, reduces inflammation, supports blood vessel formation, and minimizes blood-brain barrier damage (Ates et al., 2022). Meanwhile, studies showed that activating the PI3K/Akt/eNOS pathway in stroke-affected mice boosted SOD, glutathione peroxidase (GPx), and CAT levels, reducing oxidative stress (Chen et al., 2019). To evaluate the expression of relevant proteins and cytokines, Western blot analysis was utilized to measure p-Akt, p-PI3K, p-eNOS, MMP-9, and VEGF, while ELISA was employed to quantify GSH, SOD, VEGF, IL-10, and IL-1β. These findings showed that EEZC significantly abolished the abnormalities caused by H2O2. This suggests that EEZC may positively impact endothelial cell injury and angiogenesis by inhibiting oxidative stress via the PI3K/Akt/eNOS pathway.
In conclusion, our preliminary investigation suggests that EEZC may ameliorate cerebral ischemia by modulating the Pi3K/AKT/eNOS signaling pathway, with quercetin, piperine, tanshinone IIA, and kaempferol identified as key components. These findings offer foundational data supporting the potential clinical application of ZC-13 and underscore the need for further research into the total flavonoids and specific flavonoid constituents within ZC-13 to enhance its development. Although our current in vitro findings are promising, future studies should validate these results using more rigorous in vivo models, including serum analysis.
Conclusion
In summary, this study shows that EEZC reduces apoptosis and oxidative stress in H2O2-treated bEnd.3 cells through the PI3K/Akt/eNOS pathway. ZC-13 could be a potential treatment for cerebral stroke, but more in vivo research is needed to confirm these results.
Footnotes
Abbreviations
CCK-8: Cell counting kit-8; DL: Drug-likeness; DMEM: Dulbecco’s modified Eagle medium; ECL: Enhanced chemiluminescence; ELISA: Enzyme-linked immunosorbent assay; FBS: Fetal bovine serum; IL-1β: Interleukin-1β; IL-6: Interleukin-6; IL-10: Interleukin-10; GO: Gene ontology; GSH: Glutathione; KEGG: Kyoto Encyclopedia of Genes and Genomes; MDA: Malondialdehyde; OB: Oral bioavailability; p-AKT: Phospho-protein kinase B; p-eNOS: Phospho-endothelial nitric oxide synthase; p-PI3K: Phospho-phosphatidylinositide 3-kinases; SOD: Superoxide dismutase; TCM: Traditional Chinese medicine; TCMSP: Traditional Chinese Medicine Systems Pharmacology; UPLC-ESI-MS/MS: Ultra-performance liquid chromatography-electrospray ionization tandem mass spectrometry; VEGF: Vascular endothelial growth factor; ZC-13: Zha-Chong-Shi-San-Pill.
Acknowledgments
The authors thank Dr. Japhet for its linguistic assistance during the preparation of this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This experiment was approved by the Experimental Animal Ethics Committee of the Affiliated Hospital of Inner Mongolian Minzu University (Ethics Batch Number: NM-LL-2020-04-01-24) and the date (01-4-2020) of this approval. Experimental compliance [The Animal Research: Reporting of in vivo Experiments (ARRIVE) guidelines].
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by the National Natural Science Funding of China (No. 82060816), Inner Mongolia Autonomous Region Education Department Project (NJZY21435, Basic scientific research business fees of colleges and universities directly under the Inner Mongolia Autonomous Region (GXKY22043, GXKY22255), Inner Mongolia Natural Science Foundation (Nos. 2023MS08060, 2024QN08083, 2024QN08028), Inner Mongolia Health and Family Planning Commission (No. 202201461), Open Project of Inner Mongolia Autonomous Region Individualized Medication Engineering Technology Research Center (MDK2021055, MDK2024004). Open Project of Inner Mongolia Autonomous Region Chronic Disease Traditional Chinese and Western Medicine Integration Basic Research and Transformation Innovation Center (2024MBKF004). Project of Traditional Chinese Medicine (Mongolian Medicine) Science and Technology Plan of Inner Mongolia Autonomous Region Health Commission (ZMY-2024107).
