Abstract
Background and Objectives
Alginate and gelatin have biocompatibility and biodegradability for drug formulations or carriers, but the roles and mechanisms in cartilage injury (CI) repair remain unclear. This research investigated the application of alginate-collagen hydrogel (ACH) in rat CI repair and its related mechanisms through in vitro and in vivo experiments.
Materials and Methods
Alginate and modified collagen solutions were mixed to prepare the ACH. The obtained samples were characterized, and their sustained release properties were determined. L929 mouse fibroblasts were assigned into a blank control group (BC group, untreated), a positive control group (PC group, treated with 6.4% phenol), a negative control group (NC group, treated with tissue culture polystyrene), and an ACH group (treated with ACH). The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay assessed cell viability. Meanwhile, the cytotoxicity of the ACH and its impact on cell proliferation and cell attachment capabilities were analyzed. Thirty 4-week-old specific pathogen-free Sprague-Dawley rats were randomly grouped into a chondrocyte group (chondrocytes collected and cultured), a cPAGE group (chondrocytes treated with ACH), a model group (CI model), and an mPAGE group (CI model treated with ACH). The influence of ACH on chondrocyte proliferation and type II collagen (COL-II) expression were assessed via cell counting kit 8 (CCK-8) assay and immunofluorescence staining. Furthermore, the impacts of ACH on chondrocyte tissue and COL-II expression were analyzed using Safranin O-Fast Green staining, the International Cartilage Repair Society (ICRS) score, and COL-II tissue staining. Immunoblotting was utilized to detect tumor necrosis factor alpha (TNF-α), nuclear factor kappa-B (NF-κB), matrix metalloproteinase 13 (MMP-13), and COL-II proteins in cartilage tissue.
Results
The release rate of the constructed ACH reached over 85% within 72 h and achieved degradation equilibrium within 5 days with a weight loss rate of over 60%. L929 fibroblast culture results showed that the ACH posed no significant cytotoxicity, and cell attachment results were not obviously different from those on tissue culture plates (p < .05). MTT assay results demonstrated that the ACH exerted no adverse effects on L929 cell proliferation and growth, indicating good biocompatibility. CCK-8 and COL-II results indicated that chondrocyte proliferation in the cPAGE group was markedly higher versus that in the chondrocyte group (p < .05). Besides, animal experiments unveiled that, relative to the CI group, the mPAGE group had more chondrocytes, upregulated COL-II, and higher ICRS scores (p < .05). MMP-13, NF-κB, and TNF-α were downregulated, and COL-II was upshifted in the mPAGE group, with remarkable differences (p < .05).
Conclusion
ACH exhibited excellent biocompatibility and low cytotoxicity, promoted cell proliferation and growth, and facilitated chondrocyte proliferation in rat CI repair, offering a valuable research foundation for CI treatment.
Introduction
Cartilage injury (CI) and tissue degeneration are common clinical problems, often caused by trauma, diseases, or aging, significantly impacting patients’ quality of life (Patel et al., 2019). The limited self-repair capacity of cartilage and its lack of blood supply make its repair complex and challenging (D’Amico et al., 2022). Therefore, finding effective treatment methods to promote CI repair has become one of the focal points of medical research (Gherghina et al., 2023). Hydrogel is a potential carrier material of interest due to its excellent biocompatibility and controllable drug-release capabilities.
Hydrogel, possessing a three-dimensional network, exhibits highly adjustable physical and chemical properties, enabling it an ideal carrier in various fields (Guo et al., 2020; Liu et al., 2020). Alginate is a type of natural polysaccharide composed of long-chain polymers formed by (1↓4)-β-crosslinked
Alginate and collagen, due to their elasticity, unique biocompatibility, high porosity, water absorption and retention capabilities, hydrophilicity, and favorable physical, chemical, and biological characteristics, have been extensively studied for cartilage reconstruction (Li et al., 2021; Liu et al., 2020). Alginate-collagen hydrogel (ACH) is not limited to providing mechanical support and filling defects; it can also accelerate cartilage regeneration by promoting cell adhesion, proliferation, and differentiation through the provision of bioactive substances and drugs (Chang et al., 2020; Dutta et al., 2021). Phatchayawat et al. (2022) reported that bacterial cellulose-chitosan-ACH can effectively support cell proliferation while providing glucosamine sulfate. Research has shown that ACH can mimic the biological characteristics of cartilage, promoting the proliferation and differentiation of chondrocytes while inhibiting inflammatory responses (Morshedloo et al., 2020).
This research investigated the application of ACH in rat CI repair and its related mechanisms. ACH would be prepared, and its biocompatibility as well as its impact on chondrocytes would be assessed. Additionally, the molecular mechanisms underlying the promotion of rat chondrocyte proliferation by the ACH-containing hydrogel will be evaluated. The ultimate aim was to yield new therapeutic strategies and a deeper understanding of CI treatment to improve the quality of life for patients.
Materials and Methods
Preparation and Detection of ACH
5 g of collagen was dissolved in a 0.1 M phosphate-buffered saline (PBS) solution (Sigma-Aldrich, USA). After complete dissolution, 14 g of ethylenediamine was mixed, and the resulting solution was homogeneously mixed using a Hei-PLATE Mix 20 magnetic stirrer (Haldolf Instruments and Equipment Co., Ltd., China). Subsequently, the pH was adjusted to 5 by adding 6 moles of concentrated hydrochloric acid (Nanyang Xinde Chemical Co., Ltd., China). Afterward, 2.675 g of carbonyldiimidazole (Suzhou Keshengtong New Material Technology Co., Ltd., China) was added, and the solution was diluted to 250 mL with PBS for a reaction of 1 h in a CellXpert C170i constant-temperature cell culture incubator (Eppendorf, Germany) at 37°C. Afterward, it was cooled to room temperature, loaded into a dialysis bag, and dialyzed for 3 days, with the water being changed four times daily. Once dialysis was complete, the product was transferred to plastic petri dishes, placed in a freezer for pre-freezing for 24 h, and subsequently subjected to drying. Besides, quantitative collagen was dissolved in deionized water (Suzhou Jiazhou Purification Equipment Co., Ltd., China) to generate a solution of a specified concentration. Initially, it was placed in an electric constant-temperature forced air-drying oven (Agmate Drying Equipment Co., Ltd., China) at 50°C until completely dissolved and was transferred to an incubator at 37°C for 0.5 h. Quantitative sodium alginate (Sigma-Aldrich, USA) was dissolved in PBS solution with the pH adjusted to 7.4 and stirred until completely dissolved. Subsequently, it was positioned in an incubator at 37°C for 0.5 h. A mixture of a 10% alginate solution and a 20% collagen solution was stirred for 15 s. Using a dual syringe, the resulting mixture was injected into a culture plate and left to gel for 10 min at 37°C, resulting in the formation of ACH.
The hydrogel samples, once prepared, were cut into circular specimens with a thickness of 1 cm, which were immersed in PBS solution at 37°C. Every 12 h, the specimens were removed from the solution and put in a refrigerator for 12 h of freezing. After freeze-drying, samples were weighed, and weight loss was calculated by comparing it to the dry weight. The weight loss rate, indicating the extent of hydrogel degradation (Stillman et al., 2020), was computed.
Varying concentrations of sodium alginate solutions were measured for absorbance at 450 nm, and a standard curve was constructed. Subsequently, 5 mg of hydrogel loaded with 100.0 µmol/L of ACH was put in a 50 mL centrifuge tube, mixed with 40 mL of PBS solution at 37°C, and the constant-temperature water bath shaker was set to an oscillation speed of 15 rpm. The release test was initiated by placing the centrifuge tube into the constant-temperature water bath shaker. At distinct time points (1, 2, 3, 4, 5, 6, 12, 24, 48, 72, and 96 h), 1 mL of solution was obtained from the centrifuge tube, and PBS solution with the same volume was added simultaneously. The absorbance at the corresponding time points was measured. Additionally, the ACH release curve was calculated using the standard curve obtained from the sodium alginate solution.
The prepared ACH samples were poured into a 96-well cell culture plate processed with a 15-min incubation at 37°C, and put in the refrigerator for subsequent use. Sodium alginate and collagen (Fujianpu Collagen Co., Ltd., China) were ground into powder and mixed with P2O5, then dried for more than 72 h. The samples were mixed with potassium bromide (1:100 ratio), pressed into pellets, and subjected to Fourier-transform infrared spectrometer (FTIR) (Nicolet iS50, Thermo Fisher, USA) analysis.
In Vitro Cell Assay
The thawed L929 mouse fibroblasts (ATCC, USA) were resuscitated and cultured with 10% fetal bovine serum (FBS) (Sigma-Aldrich, USA) at 37°C under 5% CO2 conditions. When the fibroblasts reached confluence, 0.25% trypsin solution was introduced to detach the fibroblasts and prepare a cell suspension, which was then sub-cultured. The fibroblasts were assigned into a control group and an ACH group. The ACH group was treated with ACH, while the control group was further sub-grouped into three: blank control group (no additional reagents), control (+) group (6.4% phenol), and control (–) group (tissue culture polystyrene, Aladdin, USA). The toxicity of ACH was evaluated using L929 mouse fibroblasts, assessing cell attachment, cell proliferation, and metabolic activity using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay.
Cytotoxicity and Cell Proliferation
In the ACH group, ACH was added to the 1640 medium (Sigma-Aldrich, USA) at 6 cm2/mL and subjected to a 24-h culture at 37°C under 5% CO2 to obtain an extract. L929 cell suspension was seeded at 5 × 103 cells/mL. Then, 1 mL of the ACH extract was introduced and cultured for 6 h. The fibroblasts were carefully and slowly washed with PBS to discard the residual medium. Crystal violet staining (0.5%, 0.1 mL, 3 min) was applied, followed by rinsing off excess dye with PBS. Sodium dodecyl sulfate solution (1%, 1 mL) was added, mixed, and left to stand for 1 min. Absorbance at 588 nm was quantitated on the 2nd, 4th, and 6th days using a UV spectrophotometer (Shanghai Third Analytical Instrument Factory, China). The relative proliferation rate (Saberian et al., 2021) of the cells was computed.
Cell Attachment Test
0.5 mL of L929 cell suspension was seeded into a plate at 5 × 103 cells. In the ACH group, 1 mL of ACH extract was applied and cultured for 6 h at 37°C under 5% CO2 conditions. The fibroblasts were gently rinsed with PBS to eliminate the residual medium. Crystal violet staining (0.5%, 0.1 mL, 3 min) was applied, followed by rinsing off excess dye with PBS. After that, 0.5 mL of dimethyl sulfoxide (DMSO, Sigma-Aldrich, USA) was added, and samples were taken to measure the absorbance at 570 nm at 0.5, 1, 2, and 4 h.
MTT Assay
Again, 0.5 mL of L929 suspension was seeded at 5 × 103 cells. The fibroblasts in the ACH group were cultured with 1 mL of ACH extract for 6 h at 37°C under 5% CO2 conditions. These fibroblasts were then carefully rinsed using PBS to remove the residual medium. After 50 µL of MTT (5 mg/mL) reagent was introduced, these fibroblasts were performed with a 4-h culture at 37°C under 5% CO2. Afterward, the fibroblasts were gently again subjected to a PBS washing, and 0.5 mL of DMSO was mixed. Samples were taken, and the absorbance of the samples was determined at 570 nm on days 2, 4, and 6.
Animals
Thirty male Sprague-Dawley (SD) rats with SPF grade (approximately 4 weeks old and weighing between 60 and 90 g; Chengdu Dashuo Experimental Animals Co., Ltd., China) were selected. Prior to modeling, the rats were housed under standard conditions, which included a temperature of 23 ± 2°C, relative humidity of 50 ± 5%, low-intensity lighting with a 12-h light-dark cycle, good ventilation, and noise levels below 80 decibels. Meanwhile, they were fasted for 12 h and denied access to water for 6 h prior to the modeling procedure. This research received approval from the hospital’s ethics committee.
Furthermore, the enrolled 30 SD rats were assigned to the chondrocyte group (5 rats, where chondrocytes were collected and cultured), the cPAGE group (5 rats, where chondrocytes were treated with ACH), the CI group (10 rats, which served as the CI model group), and the mPAGE group (10 rats, where the CI model was combined with ACH treatment). Rats in the chondrocyte group and cPAGE group were euthanized 1 week after surgery, and their chondrocytes were collected for cell counting kit 8 (CCK-8) (Abcam, UK) experiment. The animals were euthanized using an intravenous injection of 3% sodium pentobarbital (Sigma-Aldrich, USA) at a dosage of 30 mg/kg sodium pentobarbital. The animal grouping and treatments are shown in Figure 1.
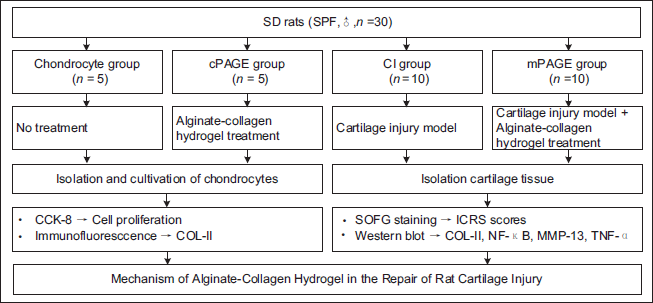
Construction of Rat CI Model
These rats were secured on the operating table, and a 2% sodium pentobarbital solution (2.5 mg/kg) was administered via intraperitoneal injection to ensure that they were in an unconscious and painless state. The right hind limb knee joint area of the rat was cleaned. A longitudinal arc-shaped incision of approximately 1.5 cm in length was cut on the inner side of the patellar ligament of the right hind limb knee joint. Careful dissection of various tissue layers was performed to expose the weight-bearing area of the ankle bone. A circular hole (1 mm) was drilled in the weight-bearing area using a drill. In the ACH group, ACH was injected at the site of CI. In the control group, after the same exposure time, the surgical area was rinsed with saline, and layer-by-layer suturing was performed to ensure wound closure. Penicillin (Sigma-Aldrich, USA) was injected within 1 week postoperatively (100,000 U/day), and the rats were euthanized by CO2 inhalation 4 weeks later. The damaged cartilage specimens from both the control group and the ACH group were fixed in 4% paraformaldehyde, decalcified in a 10% ethylenediaminetetraacetic acid solution (Nanjing Gutian Chemical Co., Ltd., China), and prepared as specimen slides. These slides were saved at –80°C for subsequent use.
Safranin O-Fast Green (SOFG) Staining
The cartilage sections were subjected to SOFG staining as follows. They were stained with 0.02% SOFG (3–5 min) and washed with deionized water to eliminate excess dye until cartilage appeared colorless. Subsequently, they were stained with 0.1% Fast Red (1–2 min) and again subjected to washing with deionized water for the purpose of eliminating excess dye. The tissue sections were then immersed in acidic alcohol (ethanol:acetic acid = 95:5) for 1 min and followed by washing using deionized water. Dehydration of the tissue sections was performed sequentially using 95% and 100% ethanol and xylene. Additionally, the sections were mounted, sealed, and allowed to dry. The recovery of CI in different groups was observed, and scoring was performed regarding the International Cartilage Repair Society (ICRS) cartilage repair score for comparative evaluation.
Western Blotting
Cartilage sections were collected for protein immunoblotting analysis. Specimens were incubated in RIPA buffer at 4°C for 1 h. BCA protein assay determined protein concentration, based on which, the samples were loaded onto a 12% polyacrylamide gel for electrophoresis for their separation. Subsequently, the proteins were transformed to a polyvinylidene fluoride (PVDF) membrane, which was put in a blocking solution (1% FBS, 20 mmol/L Tris saline (TS) buffer (Sigma-Aldrich, USA), 0.05% Tween-20) and incubated at 4°C. Then, it was rinsed with 20 mmol/L TS buffer with 0.05% Tween-20. Membrane incubation was realized with specific primary antibodies (tumor necrosis factor alpha (TNF-α) (1:500), nuclear factor kappa-B (NF-κB) (1:500), matrix metalloproteinase 13 (MMP-13) (1:500), COL-II (1:500)) at 4°C, followed by rinsing. Horseradish peroxidase (HRP)-conjugated secondary antibodies (1:500) were applied and incubated at 4°C, followed by washing. Both the primary and secondary antibodies were purchased from Abcam (UK). Furthermore, the membrane was subjected to imaging analysis employing iBright gel imaging system, with β-actin as an internal reference, to quantify levels of the target proteins.
Chondrocyte Counting
In the chondrocyte group, 10 rats that had undergone successful modeling were euthanized using CO2 after 1 week. They were rolled into two groups: control group (cultured in serum-free Dulbecco’s Modified Eagle Medium (DMEM)-F12 medium (Aladdin, USA), n = 5) and ACH group (cultured with ACH, n = 5). The knee joint tissues from the right hind limbs of these rats were removed, cut into small pieces (2 mm3), washed with physiological saline, and then placed in culture bottles. These tissues were treated with 0.25% trypsin (37°C, 30 min) and subsequently digested with 0.2% type II collagen (COL-II). After filtration to remove residual tissue fragments, the filtrate was centrifuged (1,800 rpm, 10 min) (CR22N high-speed centrifuge, Eppendorf, Germany) for discharging the supernatant. The pellet was then resuspended in DMEM-F12 medium consisting of 10% FBS and cultured (37°C, 5% CO2). Cells were collected for experiments.
The cell suspension prepared in DMEM-F12 was seeded into a 96-well plate, which was pre-incubated in a cell culture incubator for 24 h (37°C, 5% CO2) for supernatant removal, and 10 µL of CCK-8 solution was mixed. This plate was then incubated for another 4 h (37°C, 5% CO2), and absorbance at 450 nm was quantitated. Cell viability and inhibition rate were calculated (Mirrahimi et al., 2019).
Immunohistochemical Staining of Chondrocytes COL-II
The logarithmic-phase chondrocytes prepared in section 2.4.5 were rinsed with PBS (3 times, 3 min each), followed by fixation with 4% paraformaldehyde (20 min). After fixation, the chondrocytes were again washed with PBS (3 times, 3 min each). Subsequently, 0.1% polyethylene glycol octylphenyl ether was added to enhance cell membrane permeability, followed by another wash with PBS (3 times, 3 min each). The fibroblasts were then incubated with 10% FBS for 1 h, followed by an incubation all night with the primary antibody (COL-II, 1:500) at 4°C. Following incubation, cells were rinsed with PBS 3 times for 3 min each and then incubated with the PCSA (1:500). The staining was observed until chondrocytes were clearly visible, after which the chondrocytes were again subjected to a PBS washing to stop the staining reaction. Finally, these chondrocytes were air-dried, mounted, and observed.
Statistical Analysis
Data were analyzed employing SPSS 26.0. Continuous data, denoted as mean ± standard deviation, were compared via t-test. Categorical data indicated as frequencies or percentages were compared with the χ2 test. p < .05 was considered statistically significant.
Results
Characterization and Performance Test Results
Performance testing was conducted on the prepared ACH samples, as demonstrated in Figure 2. As illustrated in Figure 2A, the oxidized ACH reached degradation equilibrium in about 5 days, with a weight loss rate exceeding 60%. Results (Figure 2B) revealed that the samples achieved a release rate of over 75% within 24 h and maintained a stable release of over 80% within 72 h, demonstrating a stable high release rate.
Alginate-Collagen Hydrogel (ACH) Detection Result. (A) Degradation, (B) Release Rate, and (C) Fourier-transform Infrared Spectrometer (FTIR) Results of ACH ((a) Oxidized Sodium Alginate, (b) Modified Collagen, and (c) ACH).
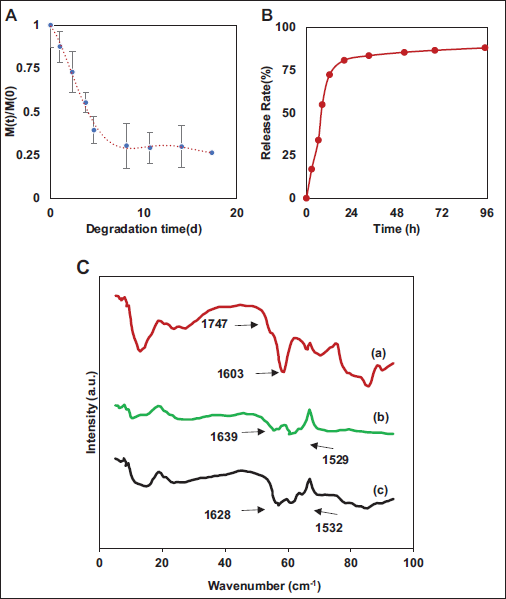
FTIR results of the prepared samples are displayed in Figure 2C. Comparing the spectra with modified collagen and oxidized sodium alginate, it can be observed that in contrast to the peak for the aldehyde group at 1,747 cm–1 in oxidized sodium alginate, the peak in the hydrogel prepared effectively weakened, consistent with the degradation capacity experiment results, indicating a noticeable reduction in aldehyde groups. On the other hand, modified collagen exhibited amide bonds (1,636 cm–1 and 1,529 cm–1), and similar peaks were observed in the sample when comparing the hydrogel with modified collagen.
Results of In Vitro Cell Experiment
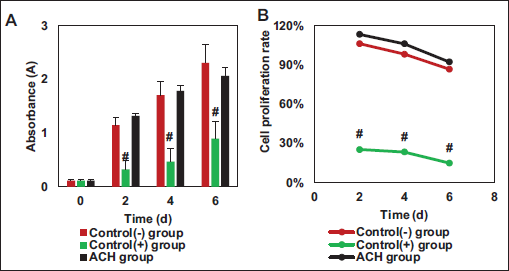
Cytotoxicity and Cell Proliferation: Further verification of the safety of the prepared ACH was conducted through experiments with L929 rat fibroblasts, as explicated in Figure 3. The cytotoxicity experiment results indicated no remarkable difference in absorbance data between the fibroblasts in the ACH group and the control (–) group (p > .05), while a substantial difference was indicated in contrast to the control (+) group (p < .05), as detailed in Figure 3A. The relative proliferation rates based on absorbance values were calculated, as presented in Figure 3B. The average cell relative proliferation rate in the ACH group was 104.1%, whereas it was 97.2% in the control (–) group and only 21.3% in the control (+) group. The ACH group and control (–) group exhibited neglectable difference (p > .05), but a considerable difference was found in comparison to the control (+) group (p < .05).
Cell Attachment Performance
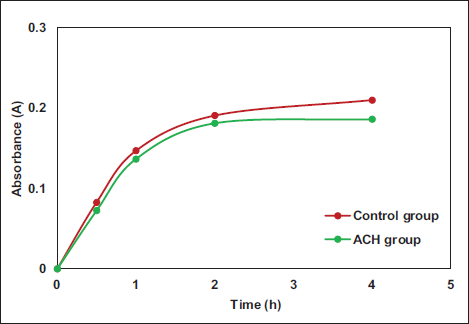
The experimental results revealed that the cell attachment efficiency in the ACH group was slightly inferior to that in the control group but without great significance (p > .05), as illustrated in Figure 4.
Comparison of Cell Attachment Performance.
MTT Assay
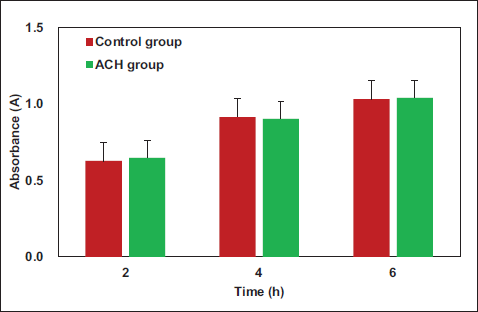
The MTT assay results (Figure 5) demonstrated no visible difference in cell absorbance between the ACH group and the control group (p > .05). In fact, on the 6th day, absorbance in the ACH group was even superior to the control group.
Comparison of Cell Metabolic Activity and Survival State.
Results of Animal Experiment
Results of CCK-8 on Chondrocytes: The CCK-8 assay results for SD rat chondrocytes were demonstrated in Figure 6. Cell activity in the cPAGE group was much higher than that in the chondrocyte group and exhibited a remarkable difference (p < .05). Furthermore, cell proliferation data, as depicted in Figure 6B, indicated that cell proliferation in the cPAGE group was notably superior to that in the corresponding chondrocyte group data at 24 h (p < .05).
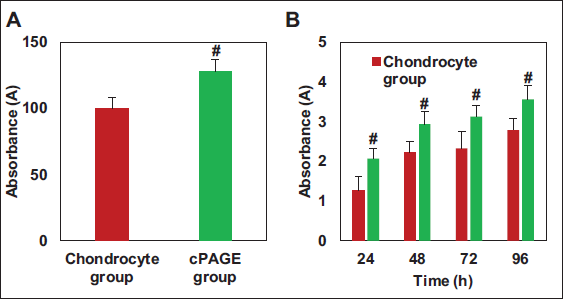
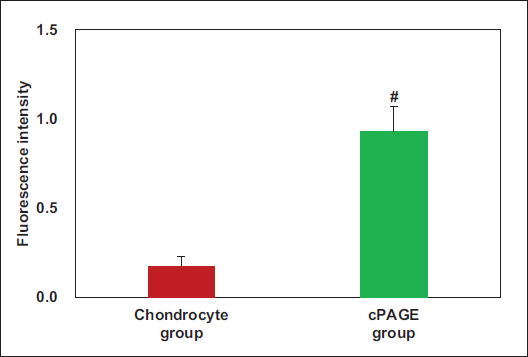
COL-II Staining Results of Chondrocytes: The fluorescence intensity values are presented in Figure 7. As given in Figure 7, more COL-II was observed in the damaged chondrocytes of the cPAGE group relative to the chondrocyte group, with a great difference observed (p < .05).
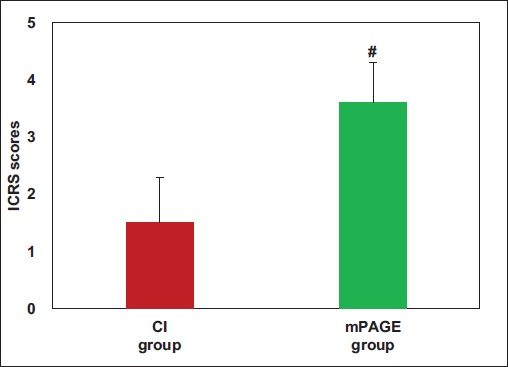
SOFG Staining Results of Cartilage Sections: The ICRS scores for both groups are displayed in Figure 8, with the mPAGE group scoring greatly higher based on the CI group (3.6 ± 0.7 vs. 1.5 ± 0.8), demonstrating an obvious difference (p < .05).
Western Blot Determination of Cartilage Tissue Sections
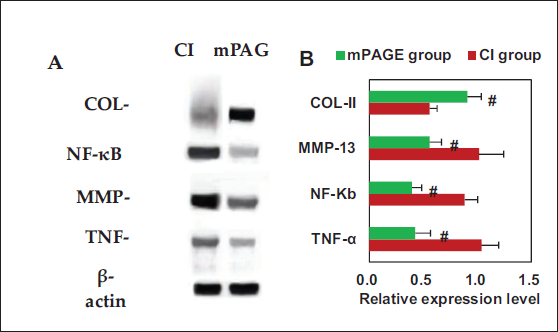
Protein immunoblotting results for NF-κB, MMP-13, TNF-α, and COL-II in rat cartilage specimens are illustrated in Figure 9. NF-κB, MMP-13, and TNF-α in mPAGE group were greatly reduced compared to those in CI group, with obvious differences observed (p < 0.05), while COL-II exhibited a higher expression in contrast to CI group (p < 0.05).
Discussion
Alginate itself possessed properties that were difficult to degrade, and previous research has indicated that the relative molecular weight of sodium alginate remains relatively unchanged over approximately 20 days. To address this challenging degradation property, alginate was oxidized in this experiment. The results in the article showed that oxidized ACH reached degradation equilibrium in about 5 days, with a weight loss rate exceeding 60%, indicating excellent degradation efficiency. The improvement in degradation efficiency was associated with an enhancement in the number of aldehyde groups in oxidized alginate (Brown et al., 2023). According to the FTIR results, compared with modified collagen and oxidized sodium alginate, the spectra of the prepared samples showed a gradual weakening of the aldehyde peak at 1,747 cm–1, consistent with degradation experiment results, indicating a significant reduction in aldehyde groups. On the other hand, modified collagen exhibited characteristic peaks of amide bonds (1,636 cm–1 and 1,529 cm–1), which were similarly observed in the samples compared with modified collagen. They suggest the presence of amide bonds in the hydrogel, conferring properties similar to collagen (Skopinska-Wisniewska et al., 2021).
The importance of cytotoxicity experiments lies in evaluating the impact of biomaterials on cell growth and health. Through these experiments, it can be determined whether materials are harmful to cells, thereby verifying their suitability for biomedical applications. In the study, the absorbance data of fibroblasts in the ACH group showed marked differences versus the control (+) group, and the average cell relative proliferation rate in the ACH group exhibited notable differences versus the control (+) group. This indicates that the prepared ACH exhibits good cell compatibility, suggesting that the addition of an appropriate amount of collagen can ensure a complete reaction of aldehyde groups, thereby avoiding cytotoxicity caused by excessive sodium alginate. Excessive aldehyde groups, when present due to excess alginate, can carry cytotoxicity, but the addition of an appropriate amount of collagen can ensure a complete aldehyde group reaction (Liu et al., 2022; Zhang et al., 2022). The experimental results demonstrate that the prepared sample exhibits good biocompatibility. Cell attachment performance refers to the ability of cells to adhere, spread, and grow on the surface of biomaterials or substrates. It is also an essential indicator for evaluating the biocompatibility of hydrogels (Neves et al., 2020; Xiong et al., 2021). The cell attachment efficiency in the ACH group was slightly inferior to the control group, indicating that ACH can effectively adhere to the surface of damaged cells for cell repair (Bao et al., 2020). Cell attachment performance involves how cells attach to the surface of biomaterials or cell culture substrates and how they extend their shape to adapt to new environments. This is the first step in cell growth and differentiation, and it can impact the survival and functionality of cells. Surfaces with good cell attachment properties typically promote cell growth and proliferation. When cells can effectively attach and interact with a surface, they are more likely to increase their numbers through cell division and proliferation (Han et al., 2021; Lee et al., 2022; Thangprasert et al., 2019). The MTT results revealed that the cell activity in the cPAGE group was greatly higher than that in the chondrocyte group. Additionally, the cell proliferation data observed at 24 h also indicated that the cell proliferation in the cPAGE group was markedly better than in the chondrocyte group. This suggests that ACH can promote the repair of damaged rat cartilage cells after 24 h. The physical and chemical properties of the hydrogel may create a microenvironment that supports cell adhesion and expansion, thereby increasing the contact area between cells and the hydrogel, promoting cell activity and proliferation (Ravichandran et al., 2021; Tong et al., 2021).
COL-II is a type of collagen protein primarily found in tissues such as cartilage and the eyes. The results showed that the COL-II content in the damaged chondrocytes of the cPAGE group was higher versus the chondrocyte group. This indicates that ACH contributes to the recovery of COL-II in damaged cells, thereby promoting the restoration of damaged chondrocytes. ACH may achieve this by activating bioactive substances within the cells, such as growth factors and signaling pathways (transforming growth factor beta (TGF-β), fibroblast growth factor (FGF), and Wnt, among others), which exert a crucial role in collagen synthesis and chondrocyte differentiation, ultimately increasing the synthesis of COL-II (Gu et al., 2022; Li & Li, 2023; Qu et al., 2022; Yang et al., 2023). The Western blot results showed that COL-II expression in the mPAGE group was higher versus the CI group. This suggests that the injection of ACH into rats can effectively promote the repair of damaged cartilage tissue. ACH provides a physical matrix that supports damaged chondrocytes (Ramdhan et al., 2020). This matrix offers a platform for chondrocytes to adhere to and extend from, facilitating their localization and growth in the injured area. The characteristics of the hydrogel aid in cell adhesion and migration, speeding up the positioning of damaged chondrocytes in the injured area, which helps increase cell density at the injury site and promotes the repair process (Cao et al., 2023; Sharma et al., 2020). The hydrogel also promotes the synthesis of COL-II and helps maintain the structural integrity of the cartilage. At the same time, we found that NF-κB, MMP-13, and TNF-α were drastically elevated in the mPAGE group versus the CI group. NF-κB crucially involves in inflammation and immune responses (Capece et al., 2022). Its increase reflects the inflammatory activity in the ACH group, which is a common occurrence in CI repair. The activation of NF-κB may trigger the release of inflammatory mediators, such as TNF-α, to stimulate immune cell responses (Wu et al., 2023). TNF-α with increased level may be associated with the post-CI inflammatory response. MMP-13 participates in the degradation of the extracellular matrix; its increase may be related to tissue remodeling during cartilage repair (Kong et al., 2024). In CI repair, MMP-13 may help clear damaged tissue and create space for the growth of new tissue. COL-II is a major component of cartilage tissue, and its increase indicates that chondrocytes in the ACH group are more actively synthesizing and secreting COL-II, which contributes to cartilage repair (Ao et al., 2022). This may be due to ACH promoting the synthesis and secretion of COL-II by providing a supportive physical and biochemical environment for damaged chondrocytes (Jahangir et al., 2020; Sheng et al., 2023).
The strengths of this study lie in its demonstration of the significant potential of ACH for CI repair, providing valuable insights for the exploration of underlying mechanisms and the development of therapeutic agents for cartilage repair. However, the study also has certain limitations: (a) the degradation rate and mechanism of the alginate component in ACH remain unclear, (b) the mechanical properties of the alginate-gelatin hydrogel for cartilage repair require further validation, and (c) in rodent models, such as rats, the open growth plates during maturation may enhance intrinsic healing capacity. This could lead to overly optimistic repair outcomes in rat models, which may not be fully replicable in humans. Therefore, future studies should focus on the preparation of ACH to assess its in vitro and in vivo degradation and mechanical properties and employ animal models that more closely resemble human physiology to better evaluate the efficacy of ACH in promoting cartilage repair.
Conclusion
This research successfully prepared ACH and demonstrated its significant potential for application in rat CI repair. In vitro experiments unveiled that the ACH exhibited excellent biocompatibility and promoted the proliferation and growth of chondrocytes. In the rat CI model, there was a remarkable increase in the number of chondrocytes, an obvious elevation in COL-II in cartilage tissue, and higher ICRS scores. Meanwhile, the molecular biology analysis unveiled the potential molecular mechanisms of ACH in CI repair. Consequently, the constructed ACH positively influenced the cartilage repair process by hampering TNF-α, NF-κB, and MMP-13, as well as enhancing the expression of COL-II. Therefore, ACH could be a potentially effective therapeutic approach. Future research can further explore the safety, long-term effects, and more detailed molecular mechanisms of ACH to ensure its success in clinical applications.
Footnotes
Abbreviations
ACH: Alginate-collagen hydrogel; CI: Cartilage injury; COL-II: Type II collagen; DMSO: Dimethyl sulfoxide; FBS: Fetal bovine serum; FTIR: Fourier-transform infrared spectrometer; ICRS: International Cartilage Repair Society; MMP-13: Matrix metalloproteinase 13; MTT: 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; PBS: Phosphate-buffered saline; SOFG: Safranin O-Fast Green
Acknowledgments
None.
Authors’ Contributions
Conception and study design: Yezhou Li and Chengjian Tang.
Data acquisition and analysis: Yezhou Li and Chengjian Tang.
Manuscript draft, editing and revision: Yezhou Li and Chengjian Tang.
All authors wrote and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All animal experiments were approved by the Animal Ethics Committee of The First Hospital of Hunan University of Chinese Medicine, in compliance with Chinese national guidelines for the care and use of animals.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Research Project of Hunan Provincial Health Commission (20201807).
Informed Consent
Not applicable.
