Abstract
Polysaccharides, known for their structural diversity and biocompatibility, have emerged as critical components in advanced drug delivery systems and biomaterials. This review explores the transformative potential of polysaccharide-based systems, focusing on recent innovations in grafting techniques that enhance their functionality and broaden their applications in healthcare and industry. The review points out the different ways natural polysaccharides like cellulose, starch, chitin, hemicellulose, pectin, and glycogen are used in delivering drugs through various forms, such as nanoparticles, hydrogels, films, and coatings. Additionally, the review talks about new methods for attaching materials, like enzymatic and plasma-induced grafting, and how these methods help make drugs more soluble, stable, and targeted. This review offers a way to improve the use of polysaccharides in eco-friendly and safe medical solutions by tackling issues like how to make them on a larger scale, how strong they are, and how well they work for specific purposes. The synergy between natural polymers and innovative modification techniques continues to unlock new possibilities for regenerative medicine, controlled drug release, and therapeutic applications.
Keywords
Introduction
Polysaccharides
Naturally occurring macromolecules known as polysaccharides are made of glycosidic linkages that unite repeating monosaccharide units. Energy storage, structural integrity, intercellular communication, immunological regulation, and a host of other physiological processes rely on them, and they're found in fungus, algae, plants, and mammals everywhere.1,2 Because of their abundance and structural variety, they are being considered as potential candidates for use in tissue engineering, medication administration, and vaccine development, among other pharmaceutical and scientific uses,3,4 scientists demonstrate the interdisciplinary potential of polysaccharides in therapeutic design and scalability for industrial applications. 5 Chemical changes can enhance the physicochemical and biological properties of polysaccharides for medicinal applications, thanks to their many reactive functional groups (-OH, -NH2, -COOH). The glycosidic connections that support the structural plasticity of polysaccharides are highlighted in Figure 1, which demonstrates their basic structure. 6 Polysaccharide drug delivery systems have been the subject of much study as of late. Their chemical composition, physicochemical characteristics, and potential biomedical uses have all been extensively reviewed.7,8 While these works do a good job of setting the stage, they are mostly descriptive and deal with individual polysaccharides or grafting methods without offering much in the way of critical comparison, mechanistic analysis, or practical insights. To illustrate the point, most of the recent publications on xanthan gum, cellulose, pectin, carrageenan, and alginate have focused on traditional oral formulations, rather than how cutting-edge grafting methods enhance responsiveness, specificity, and scalability in contemporary delivery systems.9–11 There is a lack of systematic evaluation, which makes them useless for directing drug development. New developments in grafting chemistry have changed the way polysaccharides work by allowing for targeted structural alterations that improve their solubility, bioadhesion, responsiveness to stimuli, and controlled release. Smart, responsive, and industrially scalable drug delivery systems may now be designed using techniques like pullulan bioconjugation, click chemistry, and alginate thiolation.1,12 By connecting molecular innovation to practical medicinal goals, grafted derivatives of polysaccharides such as carrageenan and gellan gum have shown more economic promise and greater functional diversity than many traditional polymers.

Polysaccharides showing glycosidic bonds.
This review offers a unique and comprehensive look at modern grafting methods for drug delivery systems based on polysaccharides, with an emphasis on how well they scale, how responsive they are to stimuli, and how useful they are in production. 13 This work delves into new grafting strategies for pH/redox-responsive hydrogel systems, including pullulan bioconjugation, click-chemistry conjugation, and alginate thiolation, in contrast to earlier reviews that merely touch on xanthan gum, cellulose, pectin, carrageenan, and alginate in traditional oral formulations. 14 The controlled release profiles, targeted specificity, and drug loading are all much improved by these methods. The functional diversity and economic potential of derivatives grafted with carrageenan and gellan gum are significantly higher than those of many conventional polymers. 15 This review emphasizes the potential of grafting innovations to propel the advancement of biomedical systems based on polysaccharides by bridging the gap between theoretical understanding and practical therapeutic and industrial problems (Table 1).
Comparative Overview of Representative Recent Reviews on Polysaccharides and Grafting Strategies.

Grafting
Grafting is a chemical method where certain parts are permanently attached to the main structure of a polymer, like a polysaccharide. This modification enhances the polymer's properties, such as solubility, mechanical strength, and bioactivity, enabling its application in advanced drug delivery systems and tissue engineering. 22 The term “preservation” often refers to stabilizing polymers or coatings to maintain their structural integrity, but it is not directly related to grafting. Therefore, discussions on preservation and grafting should remain separate to avoid confusion. Grafting techniques like free-radical polymerization, plasma-induced grafting, and enzymatic grafting allow for careful control over the structure and function of polysaccharides, increasing their usefulness in medicine and industry.23,24
This study places an emphasis on primary experimental evidence to assess grafting methods, in contrast to earlier assessments that depend substantially on secondary summaries. The review offers a more thorough and evidence-based evaluation of modern polysaccharide modification techniques by combining mechanistic insights, material-property assessments, and drug-delivery success criteria from the original investigations.
Understanding how these chemicals impact material qualities and biological performance is just as crucial as understanding the many grafting and modification processes that are detailed. To increase grafting density and introduce new physicochemical properties like improved hydrophilicity, charge, or responsiveness, chemically initiated grafting involves the creation of radical sites or activated functional groups, which allow the covalent attachment of synthetic monomers. Enzymatic grafting maintains the integrity of native polysaccharides by oxidatively coupling aromatic or phenolic substrates, resulting in more regulated and biocompatible connections. The azide-alkyne cycloaddition and other click-chemistry processes produce stable triazole linkages with excellent uniformity, enabling the controllable manipulation of chain length and surface architecture. Swelling, solubility, mucoadhesion, mechanical strength, disintegration rate, and drug-release profiles are all impacted by changes in graft density, length, and chemical composition, among other mechanistic variables. Consequently, grafted polysaccharides typically outperform their unmodified counterparts in cellular absorption, burst release, stability in physiological settings, and pharmacological efficacy. To rationally design drug-delivery systems based on polysaccharides with predictable biological behavior, it is crucial to understand these mechanistic linkages.
To provide a coherent foundation for analyzing grafting methods, the following section first discusses the structural classification and physicochemical properties of polysaccharides used in pharmaceutical systems. This materials-focused overview establishes the baseline characteristics that later modification techniques aim to improve.
Polysaccharide Materials Relevant to Drug Delivery
Classification of Organic Polysaccharides
Polysaccharides, classified as the third principal category of biopolymers (carbohydrates), are crucial for immunological function, hemostasis, reproduction, disease prevention, and therapeutic efficacy. 25 Polysaccharides influence various biological processes in cells, including structural assistance, energy preservation, lubrication, and cellular communications. 1
Polysaccharides, the most abundant carbohydrates in nature, are classified according to their chemical composition, which comprises monosaccharide elements connected by glycosidic connections. 26 They may also form covalent connections in conjunction with other entities such as peptides, lipids, and amino acids. Figure 2 presents the classification of polysaccharides based on their chemical composition and structural diversity. It categorizes polysaccharides into homopolysaccharides, heteropolysaccharides, and their derivatives, providing a framework to understand their wide-ranging biological and industrial applications. This classification serves as a reference for the discussion of individual polysaccharides in the subsequent sections.
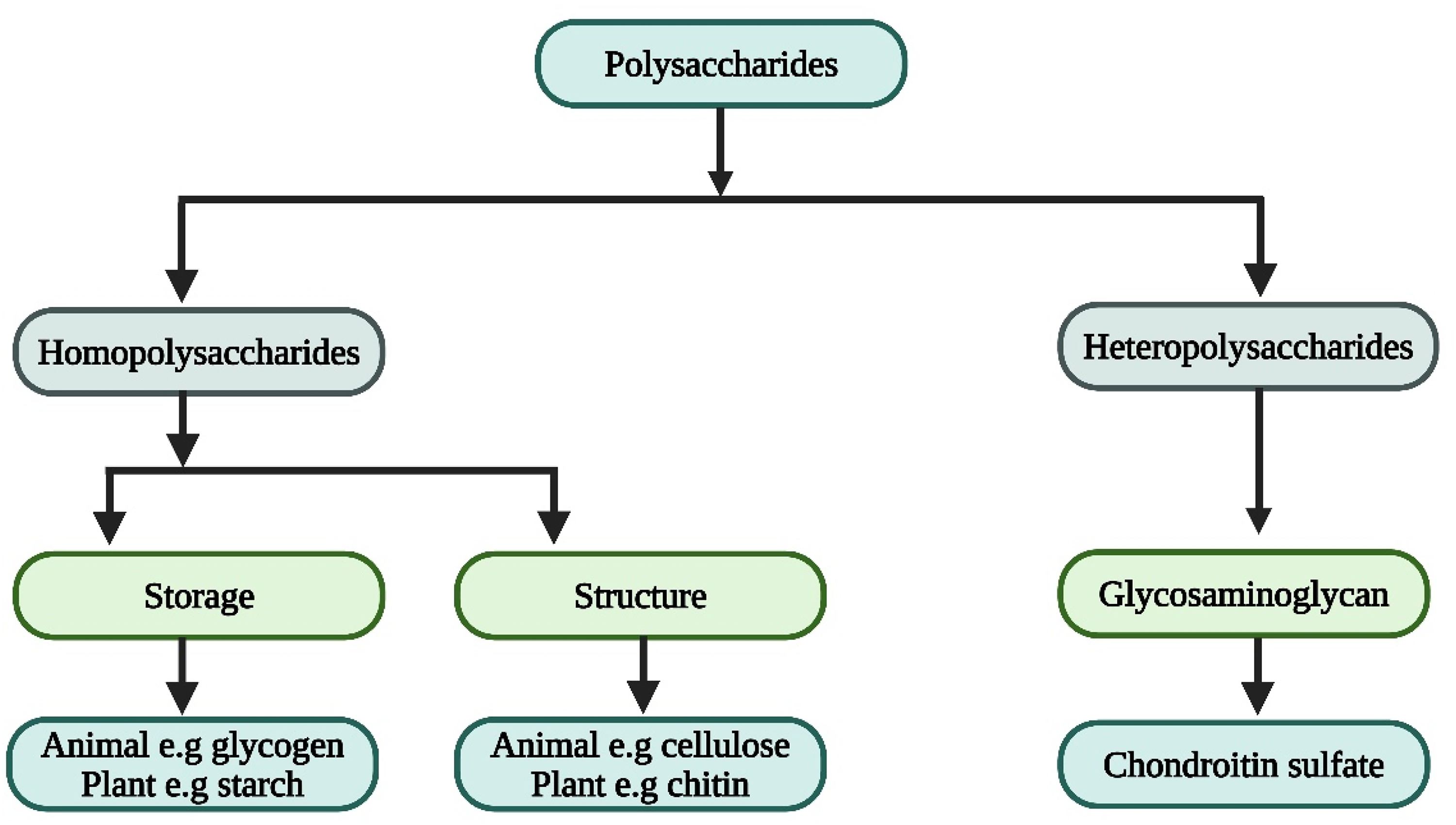
Classification of polysaccharides.
Cellulose
Derived primarily from the cell walls of plants, cellulose is also obtained from bacteria (eg,
The structure of cellulose is shown in Figure 3, which displays its straight chain made of β-D-glucose units connected by β-1,4-glycosidic bonds. The crystalline and amorphous regions of cellulose, shown in the figure, significantly influence its mechanical properties and reactivity. This structural representation underscores cellulose's versatility, particularly its applications as a sustainable material in drug delivery and tissue engineering. The OH groups interact to form intra- and inter-OH H-bonds, leading to a crystalline supramolecular polymer (ordered) region. 29 Randomly arranged cellulose molecules create amorphous regions that link the structured crystalline zones. 30 The configuration of cellulose substantially influences its reactivity. The OH units on the cellulose molecule surface can form the two intramolecular and intermolecular H-bonds. 31 The H-bonds formed among polymer chains, which can enhance their linear coherence, largely determine their chemical and physical properties. However, cellulose possesses some inherent drawbacks, such as little wrinkle opposition, inadequate solubility, and an absence of thermoplasticity. A regulated physical or chemical change in the exterior is essential to enhance the characteristics of cellulose. The synthesized element of cellulose obtained from nature generally can't be treated as a man-made polymer. 22 A suggested option is to work with cellulose molecules by attaching exogenous groups to their interface. This makes it possible to change the surface properties of cellulose without affecting the important natural characteristics of the drug, which include self-assembly in different polymer structures and stronger connections between particles and materials. Various drug delivery systems employ traditional cellulose and its variations, such as cellulose ethers, cellulose esters, and oxycellulose. Carboxymethyl cellulose (CMC) and hydroxypropyl methylcellulose (HPMC) are several cellulose ethers present in nature. 32

Structure of cellulose.
Cellulose is the most widely accepted, naturally occurring, multifunctional polymer, extensively utilized in medication formulation as excipients, controlled-release delivery systems, and nanocarriers. Common derivatives utilized include microcrystalline cellulose (MCC) for tablet formulation. 33 HPMC for controlled medication release and nanocellulose for enhancing drug solubility profiles. 34 Nonetheless, the challenges encompass the inadequate solubility of cellulose in aqueous environments, the scaling issues of nanocellulose mass production, and the constraints of focused drug administration. 35 Future research will focus on improving solubility, scaling up nanocellulose manufacturing, and optimizing drug-polymer interactions to promote bioavailability and targeted drug delivery. Nanocellulose, with its unique properties, finds diverse applications. Drug delivery systems, particularly hydrogels for wound healing, employ nanocellulose. Furthermore, it plays a crucial role in tissue engineering, controlled-release formulations, and in the use of excipients such as microcrystalline cellulose (MCC). However, using it in real life has problems like not dissolving well in water, difficulties in producing nanocellulose on a large scale, and having limited ability to be shaped when heated. Despite its widespread applications, cellulose research faces challenges in developing cost-effective, scalable methods for nanocellulose production. Issues like its low solubility and limited thermoplasticity hinder broader applications. Moreover, there is a need for studies focusing on the long-term biocompatibility and safety of cellulose-based materials in medical applications, particularly in drug delivery systems and tissue engineering. Recent studies highlight the potential for functionalized nanocellulose in precision medicine, but commercialization remains limited due to production costs.
Cellulose is characterized by excellent mechanical strength, biocompatibility, and structural versatility, and thus constitutes a prominent constituent in drug delivery systems and tissue engineering scaffolds. Derivatives like HPMC and CMC are industrially well-established as excipients and controlled-release matrices. Nevertheless, native cellulose is poorly soluble and not thermoplastic, meaning that it cannot be easily processed and is not metabolically compatible without chemical alteration. From the translational perspective, improvements in functionalized nanocellulose manufacturing and scalable derivatization procedures can increase its utility in precision drug delivery and green pharmaceutical formulations.
Hemicelluloses
Figure 4 illustrates the structural diversity of hemicellulose, a polysaccharide that forms a mesh-like network surrounding cellulose fibrils. This network, held together by hydrogen bonding and van der Waals forces, is crucial for hemicellulose's application in hydrogels and nanoparticles for controlled drug delivery.36–38 Chemical reactions such as H-bridging and van der Waals forces are commonly employed to associate them with tiny cellulose fibrils. Hemicellulose serves as a biological reserve and molecular messenger within cells. 39 Exceeding sixty billion tons in yearly worldwide manufacturing, it is the second highest prevalent reusable element of lignocellulosic organic matter, after cellulose.40,41

Structure of hemicellulose.
Hemicellulose, a biocompatible and biodegradable polysaccharide, plays an increasingly significant role in contemporary drug research. 42 Hemicellulose is being utilized in drug delivery systems within hydrogels, films, and nanoparticles for controlled and sustained release mechanisms. Xylan and xyloglucan derivatives are utilized in oral and ophthalmic drug delivery applications. 43 While it is a commendable material, issues such as inadequate mechanical strength, inefficient large-scale extraction, and pharmacokinetic research hinder its broader application. The stability, scalability, and targeted delivery capabilities of hemicellulose structures must be enhanced to fully realize their promise in therapeutics. Hemicellulose-based nanoparticles have been developed for targeted cancer therapy. A recent report highlighted the use of xylan nanoparticles loaded with doxorubicin for site-specific drug delivery, achieving improved therapeutic efficacy and reduced systemic toxicity. Hemicellulose, a natural polysaccharide, finds applications in various fields. It is utilized in hydrogels and nanoparticles for controlled drug delivery and serves as a biodegradable alternative in food packaging. However, its widespread use is hindered by challenges such as inadequate mechanical strength, difficulties in large-scale extraction, and limited research on its pharmacokinetics. Hemicellulose is a promising biomaterial, but its inadequate mechanical strength and scalability restrict its industrial applications. Current research on hemicellulose-derived nanoparticles for drug delivery has demonstrated efficacy, yet pharmacokinetic data remains sparse. Additionally, its interactions with various active pharmaceutical ingredients and long-term stability in formulations are areas requiring further investigation. Recent advancements in eco-friendly extraction methods show promise but need refinement for large-scale implementation.
Hemicellulose is compelling for drug delivery due to its biodegradability, biocompatibility, and capacity for hydrogel and nanoparticle formation for controlled release. It is also a renewable alternative in packaging and biomedical applications. Its mechanical vulnerability, extraction difficulty, and limited pharmacokinetic information, however, hold back wider application. Translationally, enhancements in extraction efficiency, mechanical strengthening, and targeted delivery systems can unleash hemicellulose's full potential for scalable therapeutic applications, especially in oral and mucosal delivery.
Pectin
In addition to pectin, the cell walls of plant cells comprise cellulose, lignin, and hemicelluloses (Figure 5). It is part of an identical category of polysaccharides as agar, which are capable of forming gels. 44 Pectin is a vital hydrocolloid utilized across various food goods in the business. Apple pectin serves as the primary manufacturing origin of high-methoxyl (>50%) pectin, while sunflower mucilage is an inherently low-methoxyl (50%) pectin. 45 Modified pectin has been used in colon-specific drug delivery systems due to its pH-sensitive properties. A 2023 study explored high-methoxyl pectin hydrogels as a delivery platform for probiotics, enhancing their viability in the gastrointestinal tract. While pectin has been extensively explored for colon-specific drug delivery and probiotic applications, challenges persist in optimizing its properties for biomedical applications. Modified pectins, such as low-methoxyl variants, exhibit potential in targeted drug delivery, but stability under physiological conditions needs further investigation. Emerging studies on pectin-based hydrogels reveal promising results, yet comprehensive trials assessing their bioavailability and scalability are lacking.

Structure of pectin.
Pectin's pH sensitivity and gel-forming capability make it an attractive polysaccharide for probiotic and colon-targeted delivery systems. Its safety profile and natural abundance also ensure industrial uptake. However, pectin tends to lack mechanical stability and source- and degree-of-methoxylation-dependent variability. Improving its stability in physiological conditions and stabilization of modification techniques will be important for clinical acceptance. Its scalability and regulatory familiarity would ensure sound building blocks for biomedical products of the future.
Starch
Starch is a free-of-scent white powder. The polysaccharide consists of amylose and amylopectin compounds. The proportions vary by origin and type of starch, with amylose typically comprising 20%–25% and amylopectin 75%–80% of overall starch. 46 Figure 6 showcases the dual composition of starch, highlighting amylose's linear structure and amylopectin's branched configuration. This composition is pivotal to starch's functionality in drug formulations as a binder, disintegrant, and vehicle for controlled-release delivery systems.47–49 Like wheat and cassava, it comprises the predominant source of carbohydrates in the dietary regimen of humans. 50

Structure of starch.
Starch is a biodegradable natural polysaccharide; this readily accessible natural polymer is often utilized in medication formulation due to its compatibility with biological systems and biodegradability. 51 Its uses encompass functioning as a binder, disintegrant, and filler in medicinal tablets. Cross-linked starch is utilized in sustained-release medication delivery systems. Nanosized starch may soon facilitate targeted therapeutic delivery for poorly soluble medicines such as paclitaxel. 52
Nevertheless, starch exhibits disadvantages such as inadequate mechanical strength, inconsistent gelation characteristics, and a lack of systematic control over drug delivery kinetics. The study was deficient in optimal starch derivatives for improving stability. 53 It also investigated the complete potential of the material in nanoparticle-based drug delivery systems to achieve improved therapeutic results. Starch nanoparticles have been employed for the encapsulation of poorly soluble drugs like paclitaxel. The study demonstrated enhanced drug bioavailability and sustained release profiles, particularly useful in cancer therapy. Starch's potential as a biodegradable material for drug delivery and food packaging is well recognized. However, limitations in its mechanical strength and inconsistent gelation properties pose significant barriers. Research on nanosized starch has shown improvements in bioavailability for poorly soluble drugs, yet the efficiency of targeted drug delivery systems remains underexplored. Improving the accuracy of starch-based products and how well they break down in complicated settings is important for making better use of them.
Starch is a low-cost, biodegradable, and biocompatible polysaccharide with extensive pharmaceutical application as a binder, disintegrant, and drug release modifier. Starch derivatives at the nanoscale have potential in increasing drug solubility and bioavailability, especially for poorly water-soluble drugs. It has, however, irregular gelation character, poor mechanical strength, and fast enzymatic degradability, which hinder controlled delivery. Translationally, it might be enhanced by starch modification using cross-linking and grafting approaches to enhance stability and render it fit for targeted delivery purposes.
Glycogen
Figure 7 illustrates glycogen's highly branched structure, which facilitates its role as an energy reserve and a promising candidate for drug delivery systems. The figure emphasizes glycogen's capacity to carry substantial pharmaceutical payloads, especially in cancer therapy and gene delivery. It resembles other storage polysaccharides such as amylopectin. However, it appears more compact and branching. The cytoplasm of cells from animals generally contains glycogen, a sort of glucose. While glycogen is crucial for neurological metabolic processes, it lacks manufacturing applications and is mentioned here solely for thoroughness. 54

Structure of glycogen.
Glycogen is an aliphatic polysaccharide that is increasingly attracting attention for therapeutic development owing to its biocompatibility and non-immunogenic attributes, in addition to its ability to hydrolyze into glucose. 55 Nanoparticles have been employed in several medication delivery applications, particularly in cancer therapy and gene transfer, owing to their enhanced solubility and controlled release characteristics. Glycogen's highly branching structure facilitates substantial payloads of pharmaceuticals or genetic material. 56
Despite the numerous commendable challenges associated with glycogen extraction, including limited scalability and the absence of comprehensive
Glycogen's densely branched structure makes it capable of holding large therapeutic payloads, and its biocompatibility and lack of immunogenicity make it an attractive option for next-generation gene and cancer treatments. Limited supply, high cost of extraction, and limited long-term safety data, however, limit its practical utility. Translationally, the invention of affordable production strategies and thorough in vivo research can validate glycogen as a dedicated carrier for gene therapy and precision medicine.
Chitin
Chitin is a long chain of molecules found in animals, known for being water-repellent and having -COCH5 and -NH2 in its structure. It does not mix well with water and common plant-based solvents; however, it can dissolve in a solution like dimethylacetamide when combined with 5% lithium chloride and chloroalcohols in water with acidic minerals. Figure 8 depicts the linear polymer structure of chitin, with its characteristic acetyl and amine groups. This structure underpins chitin's unique properties, such as biodegradability and mucoadhesion, making it highly suitable for drug delivery applications and wound dressings.57,58 Chitin is a form of chitosan that has had its acetyl groups removed, comes from the shells of crustaceans, and is being studied in the pharmaceutical industry because it is safe for the body, breaks down naturally, and is not toxic. 59 The derivative chitosan is utilized in many drug administration methods, combining orally, topical in nature, and nasal routes, owing to its mucoadhesive characteristics. 60 Chitosan nanoparticles are used to dissolve and make it easier for the body to absorb medicines that don't dissolve well, like insulin and cancer treatments.

Structure of chitin.
Associated concerns include fluctuation in quality due to inconsistent sources, inadequate mechanical strength, and limited specificity for medications. 61 The current deficiencies are advanced modification methods, scalability, and the further utilization of chitin in gene delivery and tissue engineering applications. Chitosan, a derivative of chitin, has been explored for its mucoadhesive properties in nasal drug delivery systems. Recent work reported its use in enhancing the absorption of insulin via the nasal route, achieving better glycemic control compared to subcutaneous delivery. Despite extensive research, chitin and its derivative chitosan face challenges in quality consistency due to variations in their natural sources. Large-scale extraction and refinement methods are underdeveloped, limiting their commercial viability. Recent work on chitosan's mucoadhesive properties has highlighted its potential in nasal drug delivery systems, yet long-term studies on its safety and efficacy are insufficient. Advancements in chitosan modification techniques are essential to overcome mechanical strength limitations and broaden its application scope in drug delivery and tissue engineering. A summary of polysaccharides, their nanomaterial forms, biomedical applications, and associated challenges is presented in Table 3.
Chitin and its derivative chitosan show high biocompatibility, mucoadhesion, and biodegradability, which have prompted immense research for the application of these in oral, nasal, and transdermal drug delivery. Nanoparticles of chitosan are especially promising agents for drug absorption enhancement of macromolecules like insulin. Yet, inhomogeneity of natural sources, variable quality, and mechanical restrictions continue to be barriers for industrial implementation. Translationally, streamlining extraction procedures and enhancing chemical alterations can enhance reproducibility and extend clinical relevance to mucosal vaccines and tissue engineering.
Self-Build
Self-build is a process wherein polysaccharides spontaneously assemble into ordered structures due to intrinsic molecular interactions, such as hydrogen bonding, hydrophobic interactions, or van der Waals forces. 62 This natural assembly often requires minimal external intervention, relying on the inherent chemical properties of the polysaccharides. For example, alginate turns into a gel when it comes into contact with calcium ions, which link the polymer chains together to form a stable structure that can hold drugs and release them slowly. Similarly, chitosan can self-assemble into nanoparticles in response to pH changes, enhancing its functionality in sustained drug delivery systems. Other examples include the formation of cellulose nanofibrils or hemicellulose-based films, which exploit self-assembly to improve mechanical strength and barrier properties. Recent advancements have demonstrated the potential of self-assembled polysaccharide hydrogels in wound healing and tissue engineering, offering biocompatibility and tunable properties. 63 These developments highlight the utility of self-build processes in creating functional materials without the need for extensive chemical modifications.
Alginate
Brown seaweeds are the primary source of alginate, an anionic polysaccharide that occurs naturally. The structure consists of homopolymeric or heteropolymeric blocks that arrange linear copolymers of β-D-mannuronic acid and α-L-guluronic acid residues. Medications, wound dressings, and tissue engineering all take advantage of alginate's gelling ability in the presence of divalent cations, such as calcium. Because of its hydrophilicity, biocompatibility, and ability to form stable hydrogels, it is an excellent carrier for encapsulating therapeutic chemicals that are either hydrophobic or hydrophilic. 64 Because of its delayed gelling time, natural breakdown, and potential ability to prevent the degradation of bioactive components, alginate has found extensive usage in formulations based on nanotechnology.
Alginate's potential to create stable hydrogels at room temperature makes it a prime candidate for encapsulating responsive therapeutics and wound dressing and tissue engineering applications. Its biocompatibility and availability have already resulted in numerous approved medical devices. Alginate gels are, however, mechanically unstable and acidic sensitive. Translational research efforts are directed towards strengthening gel structures and modulating degradation profiles to enhance performance in oral and injectable delivery systems.
Carrageenan
Red seaweeds, namely those belonging to the Rhodophyceae family, are the primary sources of carrageenan, a sulfated linear polymer. The presence of varying degrees of sulfuration in the alternating units of galactose and anhydrogalactose results in the formation of diverse kinds such as ƙ-, ɩ-, and λ-carrageenan. These structural alterations impact their solubility, gel strength, and functionality. Carrageenan is a typical stabilizer, thickener, and gel-maker in the food and pharmaceutical industries. Due to its ability to produce gels that can be heated and cooled, it has demonstrated potential in biomedical applications as a matrix for controlled drug delivery and as an ingredient in hydrogel systems. 65 Carrageenan is an excellent material for the development of complex therapeutic systems due to its biological activities, which include antiviral and immunomodulatory effects.
Carrageenan has good gelling properties and biological activities like antiviral and immunomodulatory activities and is, therefore, an excellent candidate for controlled-release matrices and mucoadhesive systems. However, inconsistency in sulfate content and batch-to-batch gel strength are issues of concern in formulation. Translational success will hinge on material standardization and maximizing cross-linking approaches to guarantee consistent behavior in pharmaceutical applications.
Xanthan Gum
Xanthomonas campestris ferments carbohydrates to generate xanthan gum, a polysaccharide that is not synthesized inside cells. The backbone resembles cellulose, while the side chains consist of mannose and glucuronic acid, which are trisaccharides. Its exceptional stability over a wide range of temperatures and pH levels is due to its unique arrangement, which also imparts a high viscosity at low concentrations. Xanthan gum is a popular rheology modifier, stabilizer, and suspending agent in pharmaceutical formulations due to its numerous useful properties. Its compatibility with other natural polymers opens up several potential applications, including mucoadhesive formulations, topical gels, and sustained-release drug delivery systems. 66 Xanthan gum's non-toxicity, biodegradability, and compatibility with living organisms make it an ideal tool for studies in the pharmaceutical and biomedical fields. Figure 9. Mechanism of polyphenol grafting onto chitosan via free-radical processes, where ascorbic acid generates •OH and ascorbyl radicals that initiate chitosan radical formation, enabling the creation of macro-radicals on the polysaccharide backbone. These reactive sites subsequently interact with phenolic compounds, facilitating their covalent incorporation into the chitosan matrix. Nucleophilic addition and coupling reactions then stabilize the grafted structures by forming strong covalent bonds between the polyphenols and radicalized chitosan chains.

Mechanism of polyphenol grafting onto chitosan via free-radical processes.
Stability of xanthan gum over pH and temperature ranges and the capacity of the gum to create viscous solutions are the reasons for its widespread use as an excipient and sustained-release matrix. Its natural origin and safety profile justify regulatory acceptance. Yet, its high viscosity and inconstancy in rheology can be a hindrance for processing and reproducibility. Xanthan gum is industrially established and can aid in the development of strong, mucoadhesive, and controlled-release drug delivery systems with proper formulation strategies. Table 2 summarizes the quantitative comparison of the grafted polysaccharides systems.
Quantitative Comparison of Representative Grafted Polysaccharide Systems.

Novel Polysaccharides
In recent years, novel polysaccharides have been explored for their unique structural properties, biocompatibility, and versatility in advanced biomedical and industrial applications.
75
These polysaccharides, derived from unconventional sources, exhibit promising potential in drug delivery systems and other therapeutic uses.
Tamarind Seed Polysaccharide (TSP) Tamarind seed polysaccharide, a branched xyloglucan, has been extensively studied for its mucoadhesive properties and thermal stability.
76
It has been employed in sustained-release formulations, particularly for ophthalmic and oral drug delivery. Recent studies highlight its application in nanoparticle synthesis for targeted drug delivery, demonstrating enhanced bioavailability and controlled release profiles. Taro Stolon Polysaccharide Taro-stolon polysaccharide, a lesser-known material, exhibits significant water absorption capacity and gel-forming ability.
77
Its structure, primarily composed of glucose, galactose, and mannose, enables its use in wound healing applications and hydrogels for tissue engineering. Emerging research has demonstrated its potential in encapsulating hydrophobic drugs, ensuring effective delivery to target sites. Cassia Fistula Seed Galactomannan Cassia fistula seed galactomannan is a plant-derived polysaccharide with a high galactose-to-mannose ratio.
78
It has demonstrated utility as a thickening agent in pharmaceutical formulations and as a matrix for sustained drug release. Novel formulations utilizing this polysaccharide have shown potential in improving the solubility and stability of poorly water-soluble drugs. Okra Gum Okra gum, extracted from the pods of
After we covered the basic structures and properties of important polysaccharides, we will move on to the ways that are utilized to change these materials. To further understand how these three grafting methods chemicals, enzymatic, and plasma-induced improve particular functional qualities pertinent to medication delivery, we present them with mechanistic details (Table 3).
Summary of Polysaccharides, Their Nanomaterial Forms, Biomedical Applications, and Associated Challenges.

Control Parameters in Nanoparticle Synthesis
Precise control over nanoparticle synthesis parameters is crucial to achieving desired characteristics such as size, morphology, and surface charge. Temperature and pH levels play pivotal roles in determining the physical and chemical properties of polysaccharide-based nanoparticles. 93 For example:
Conventional Applications of Polysaccharides
Polysaccharides have a long history of applications in various domains, driven by their biocompatibility, non-toxicity, and versatile physicochemical properties. Their use spans food technology, pharmaceuticals, cosmetics, and agriculture. Below, scientists summarize key conventional applications supported by recent findings from the referenced studies.
Pharmaceutical Excipients Polysaccharides such as starch, cellulose, and alginate are widely utilized as excipients in drug formulations. These materials act as binders, disintegrants, and fillers, ensuring the stability and efficacy of tablets and capsules.
94
Microcrystalline cellulose, for instance, remains a gold standard in tableting due to its excellent compressibility and stability. Controlled Drug Release Systems The ability of polysaccharides to form hydrogels under specific conditions has been exploited for controlled drug release applications. Alginate and chitosan hydrogels, for example, provide sustained release of therapeutic agents over extended periods, reducing dosing frequency and improving patient compliance.
93
Conventional polysaccharides like guar gum, xanthan gum, and carrageenan are extensively used in the food industry as thickening, gelling, and stabilizing agents. These materials enhance the texture, viscosity, and shelf life of food products, with minimal impact on nutritional value.
95
Polysaccharides such as hyaluronic acid and pectin are commonly used in wound dressings and biomedical applications due to their water-retention capacity and bioactivity. Hyaluronic acid-based formulations, in particular, promote tissue regeneration and provide a moist environment conducive to healing.
96
Polysaccharides like chitosan and guar gum have found applications in agriculture as soil conditioners, seed coatings, and biodegradable films. These materials improve soil structure, enhance seed germination, and reduce reliance on synthetic chemicals.
97
Additionally, their use in water treatment as flocculants showcases their potential in environmental sustainability.
98
Polysaccharides as Drug Delivery Tools
Polysaccharides have garnered significant attention in drug delivery due to their biocompatibility, biodegradability, and ability to be chemically modified for specific applications. They provide flexible options for different drug delivery methods, like nanoparticles, hydrogels, micelles, films, and beads, which allow for controlled, long-lasting, and targeted release of medicines. While chitosan-based systems are widely studied, other polysaccharides such as cellulose, starch, hemicellulose, pectin, and glycogen also demonstrate great potential in diverse drug delivery applications.99,100
Nanoparticles
Polysaccharide-based nanoparticles are widely researched as possible vehicles for delivering medicines, improving how well drugs dissolve, and allowing for precise delivery. Chitosan nanoparticles, recognized for their strong ability to stick to mucous membranes, are particularly good for oral, nasal, and eye treatments. Chitosan nanoparticles, well known for their excellent mucoadhesive performance, stand out as more suitable routes for oral, nasal, and ocular administration. 101 These nanoparticles are known for their better ability to be absorbed by the body for large molecules like insulin and have been successfully used to create and deliver cancer treatments like doxorubicin. Similarly, starch-based nanoparticles are also used to surround these hard-to-dissolve drugs to improve their solubility and effectiveness. Indistinguishably, starch-based nanoparticles are also used in the encapsulation of such hydrophobic drugs to enhance their solubility and therapeutic efficacy. 102 The cross-linked variant of starch demonstrated its potential for sustained release in the application of anticancer and anti-inflammatory drugs. Moreover, cellulose nanoparticles in the form of nanocrystals and nanofibers have found their application in wound healing and as drug carriers in controlled release systems, thereby enhancing their relevance in biomedical and pharmaceutical research. 103 Therapeutic exosome platforms demonstrate how cargoes (lncRNAs, miRNAs, proteins) modify immune metabolism and phenotype in situ; these biological mechanisms can be effectively duplicated by polysaccharide nanocarriers that deliver identical payloads with controlled release and targeting ligands. Exosomal lncRNA AK083884 modulates PKM2/HIF-1α signaling and induces M2 polarization in viral myocarditis. This pathway may be addressed using nanoparticles with chitosan or hyaluronan grafts that deliver lncRNA or small-molecule glycolysis modulators in response to cardiac inflammation. 104
Hydrogels
Hydrogels are three-dimensional hydrophilic polymer networks that can absorb and retain large quantities of water. They have created a great deal of impact in drug delivery due to their biocompatibility, tuning of physical properties, and responsiveness to environmental stimuli, such as pH and temperature. 105 Alginate-based hydrogels are commonly used in wound dressings and systems that release drugs in a controlled way, especially for delivering protein-based medicines and in gene therapy. 106 Pectin hydrogels are primarily used in colon-targeted drug delivery systems, as they are sensitive to changes in pH, thereby ensuring site-specific release within the gastrointestinal tract. Chemoembolic therapy, particularly in the application of physics-based drug formulations, has also utilized hemicellulose hydrogels for sustained and controlled drug release. These hydrogel systems show the versatility of natural polysaccharides in forming responsive and effective drug delivery systems. 107
Films and Coatings
Polysaccharide-based films and coatings are crucial for safeguarding active pharmaceutical ingredients and regulating drug release. Among cellulose derivatives, hydroxypropyl methylcellulose (HPMC) and carboxymethyl cellulose (CMC) are common coating agents for tablets and capsules, improving the stability of the formulation and modulating the drug release profile. 108 Chitosan-based films have gained prominence in transdermal drug delivery systems, particularly as patches designed for prolonged release in the treatment of pain and hormone-related conditions. 109 In addition, pectin films with excellent bioadhesion properties are being investigated for use in wound healing and localized drug delivery with targeted therapeutic effects at the site of application. These film-forming polysaccharides provide versatile and biocompatible solutions to enhance drug stability, delivery efficiency, and patient compliance. 110
Micelles and Vesicles
Micelles and vesicles derived from amphiphilic polysaccharide derivatives are emerging as potential carriers in delivering poorly soluble drugs. These tiny structures take advantage of the way the modified polysaccharides naturally come together to trap water-repelling drugs inside them, improving their solubility and stability. 111 Chitosan micelles have also been effective in delivering anticancer agents when it involves improving the drug solubilization and prevention from early degradation. Hydrophobic compounds, like anticancer agents, can mix well with micelles made from hemicellulose, which has shown to be a good way to enhance how well the drug works and deliver it more effectively to the right places. These polysaccharide-based micellar systems can be considered a useful solution to the solubility problems in drug formulations, as well as to widen the therapeutic use of poorly water-soluble drugs. 112
Beads
Polysaccharide-based beads have gained wide acceptance in drug delivery systems since they are easy to fabricate and have a very nice capacity for encapsulating a wide variety of therapeutic agents. Alginate beads, which are often linked with calcium ions, have been widely researched for oral delivery because they protect delicate substances like proteins and probiotics from the stomach's strong acid, allowing these substances to be released effectively in the intestine. 113 On the other hand, researchers have studied those pectin beads for colon-specific drug delivery, taking advantage of their selective degradation by colonic bacteria. The specific release of drugs at the site of action enhances therapeutic efficacy and reduces systemic side effects. These types of bead-based systems are simply a testimony to the versatility and efficiency of polysaccharides in realizing controlled and site-specific drug delivery. 114
Mechanistic Basis of Improved Solubility, Stability, and Targeting
The efficacy of polysaccharide-based carriers is contingent upon their intrinsic structural properties and capacity for chemical modification. Amphiphilic polysaccharides in nanoparticles spontaneously self-assemble into core-shell structures, wherein hydrophobic domains encapsulate poorly soluble drugs, while the hydrophilic outer corona enhances water dispersibility, thereby improving apparent solubility and bioavailability. 115 Hydrogels facilitate drug stabilization through ionic or covalent crosslinking, creating hydrated three-dimensional networks that safeguard sensitive biomolecules, such as peptides, proteins, and nucleic acids, from enzymatic or acidic degradation, while permitting controlled release in response to environmental stimuli, including pH, temperature, or redox conditions. 116 Micelles and vesicles formed from amphiphilic polysaccharides offer a thermodynamically stable environment that encapsulates hydrophobic medicines within the micellar core, hence inhibiting early degradation and prolonging circulation time. Moreover, the accessible functional groups on polysaccharides (–OH, –COOH, –NH2) offer numerous sites for chemical conjugation with targeting ligands, antibodies, or peptides, thus enabling site-specific drug delivery to tumors, inflamed tissues, or mucosal surfaces. 101 These techniques demonstrate how polysaccharide-based systems circumvent solubility challenges, enhance drug stability, and facilitate site-specific delivery, rendering them adaptable platforms for next-generation therapies.
Challenges and Future Directions
Despite the significant promise polysaccharides hold in drug delivery, several challenges continue to limit their widespread application. One major hurdle is scalability and cost, as the large-scale production of polysaccharide-based nanomaterials and hydrogels often requires complex and expensive processes. Additionally, some polysaccharides, such as starch and pectin, suffer from low mechanical strength and limited stability under physiological conditions, which can affect their performance in biomedical applications. Another important problem is improving how well polysaccharide-based drug carriers can target specific areas, since delivering medicine accurately to sick tissues or organs is still a key area of research. Future advancements in this area are likely to come from new chemical changes, creating mixed systems that pair polysaccharides with synthetic polymers, and adding designs that respond to certain triggers. These new ideas will make polysaccharide carriers work better and be more flexible, helping to create more effective and user-friendly treatment systems that can tackle a wide range of diseases, like cancer and infections.
Integration of in Vivo and Clinical Evidence
Several polysaccharide-based systems exhibit robust
Nevertheless, despite these encouraging results, the comprehensive clinical data remain limited. Most polysaccharide systems are still in the early stages of clinical trials or preclinical testing, and there isn't enough information on their long-term safety, chronic use, and how they affect the immune system.
120
Consequently, forthcoming research must prioritize stringent
Translational and Regulatory Considerations
The efficacy of laboratory work is merely one issue of concern. The issues of clinical preparedness, regulatory compliance, and manufacturing scalability are also pertinent. Despite the inclusion of sophisticated polysaccharide formulations like chitosan and nanocellulose in early-stage clinical trials, these products have persistently failed to progress beyond initial phases, underscoring the formidable obstacles that remain. 121 In the industry, producing polysaccharide derivatives in substantial amounts, consistently, and with good quality remains challenging due to the inherent variability of the source, batch-to-batch discrepancies, and the complex extraction or modification processes. 122 Before approval, regulatory bodies like the FDA and EMA mandate comprehensive characterization, substantiated evidence of confirmed reproducibility, and extensive long-term safety data, particularly regarding contaminants, uniformity, and stability. Despite the benefits of biodegradation and consequent non-toxic decomposition of polysaccharides in sustainable pharmaceuticals, systematic solutions are still necessary. This encompasses standardized extraction methodologies, adjustable scalable green synthesis techniques, and synchronized regulatory frameworks that effectively bridge the divide between research and therapeutic or industrial application.
Several regulatory and industrial hurdles prevent grafted polysaccharides from being commercialized, despite their encouraging effectiveness in laboratory-scale drug delivery systems. Enzymatic grafting, ATRP, and RAFT are some of the technologies that can be used for large-scale production, but they can be expensive due to the complexity of the purification procedures and the need for precise control over the grafting density, impurity levels, and raw material variability. Extensive toxicological evaluations, including studies on biodegradation, immunogenicity, hemocompatibility, and grafting degree, as well as residual monomers, catalyst traces, endotoxins, and long-term material stability, are also required by regulatory agencies (FDA/EMA). Changing breakdown routes and producing new metabolites or surface charges that impact biological interactions are two reasons why grafting can be dangerous. In addition, enzymatic or plasma-assisted grafting, which are less harmful to the environment and more sustainable, are being preferred over chemical procedures that rely on solvents. For grafted polysaccharide-based drug-delivery systems to be commercially translated successfully, it is vital to address these industrial, regulatory, economic, and safety constraints.
Emerging Trends and Advanced Technologies
Polysaccharide-based drug delivery systems are at the forefront of pharmaceutical innovation, driven by their biocompatibility, biodegradability, and versatility. Recent advancements have focused on optimizing their functionality for targeted and controlled drug delivery applications. Emerging trends and technologies include:
Stimuli-Responsive Polysaccharides
Stimuli-responsive polysaccharides are increasingly engineered to respond to specific stimuli such as pH, temperature, enzymes, or light. For example, there are products made with chitosan that react to pH changes and are used to deliver drugs specifically to the colon, like chitosan-folate combinations for releasing cancer medications. 123 However, temperature-responsive polysaccharides, such as those grafted with poly(N-isopropylacrylamide), are largely experimental and primarily used in preclinical research for hyperthermia-based cancer therapies. For example, pH-sensitive polysaccharides like chitosan derivatives have been developed for colon-targeted drug delivery systems, where the drug is released in response to the acidic or basic environment. 124 In the same way, temperature-sensitive polysaccharides, like those attached to poly(N-isopropylacrylamide), allow for drug release when it's hot, which often happens in tumor tissues. 125
Nanocarriers for Precision Medicine
Advances in nanoparticle fabrication have enabled the use of polysaccharides like dextran, alginate, and hyaluronic acid for precision medicine. Hyaluronic acid-based nanocarriers for targeted cancer therapies are commercially available, such as HylaMedrix® nanogels for drug delivery to tumor tissues. 127 Similarly, alginate-based hydrogels are widely used in wound healing dressings (eg, Kaltostat® and Algisite®), highlighting their industrial adoption. These nanoparticles can be functionalized with ligands or antibodies for specific targeting, such as directing chemotherapy drugs to tumor cells while sparing healthy tissue. 128 This approach reduces systemic toxicity and enhances therapeutic outcomes.
Hybrid systems combining polysaccharides with synthetic polymers or other biomaterials are gaining popularity. Commercial uses include PLGA-alginate nanoparticle systems for delivering water-repelling drugs, with products like Eligard® (a PLGA-based injection for prostate cancer treatment) showing success in clinical trials. However, the use of hybrid systems involving polysaccharides such as alginate or chitosan is still largely in experimental stages. For instance, alginate-poly(lactic-co-glycolic acid) (PLGA) nanoparticles have shown superior encapsulation efficiency and controlled drug release for hydrophobic drugs. 129 These hybrid systems leverage the benefits of polysaccharides while overcoming limitations like low mechanical strength.
Polysaccharides in Gene Therapy
Polysaccharide-based carriers are increasingly used for gene delivery, with chitosan nanoparticles leading the way. 130 These carriers protect nucleic acids (eg, siRNA, mRNA) from degradation and enhance cellular uptake. Recent advancements include the use of mannan-coated chitosan nanoparticles for targeted delivery to immune cells in vaccine development.
Extracellular Vesicles-Mimetic and Extracellular Vesicles-Complementary Delivery
Engineered vesicles produced from M2 macrophages, equipped with targeting modules (such as chemokine antagonists/peptides), localize to damaged myocardium and reprogram macrophages. Strategies involving extracellular vesicles provide design principles (surface ligands, immune-metabolic payloads) that can be applied to grafted polysaccharide systems to mimic homing and intracellular signaling effects without utilizing biological resources. This convergence facilitates the development of extracellular vesicle-inspired, polysaccharide-based carriers that integrate GMP-compliant polymers with EV-level targeting mechanisms for the immunotherapy of inflammatory heart disease.
3D Printing and Polysaccharide Scaffolds
The integration of polysaccharides into 3D printing technologies is transforming the field of tissue engineering and drug delivery. For example, alginate-based bioinks are commercially available for 3D bioprinting applications, such as CELLINK Bio X, which allows the fabrication of customized drug delivery scaffolds. 131 While cellulose and hyaluronic acid-based bioinks are in development, their use remains experimental. Biodegradable scaffolds composed of alginate or cellulose derivatives are now being 3D printed to deliver drugs in a localized and sustained manner, with potential applications in bone regeneration and cancer therapy.
Utilisation in Drug Coating (Infectious Disease, Oncology, Cardiovascular etc)
Polysaccharides are very promising as better materials for drug coatings because they are naturally available, work well with the body, and affect how drugs are released. Their unique structural features, such as being water-attracting and able to form gels or films, make them perfect for making coatings that protect drugs from breaking down, control how quickly they are released, and help deliver them to specific areas.
Principal Advantages of Polysaccharide-based Pharmaceutical Coatings
Safeguarding of Active Pharmaceutical Ingredients (APIs): Polysaccharide-based coatings can safeguard active pharmaceutical ingredients against enzymatic breakdown or the acidic pH of the gastrointestinal system. 132 Chitosan and alginate are frequently employed as coating materials in the formulation of orally delivered medications to safeguard sensitive compounds until they reach their site of action.
Regulated and Prolonged Release: Polysaccharide coatings can be designed to alter the medication release profile, facilitating continuous or delayed release of the drug. This decreases the dose frequency and enhances patient adherence. 133 Hydroxypropyl methylcellulose (HPMC) coatings can facilitate a prolonged release mechanism by creating a gel-like barrier encasing the medication.
Targeted Delivery: Customizing the characteristics of polysaccharide-based coatings allows for targeted medication delivery to specific organs or tissues. pH-sensitive polysaccharide coatings, like those made from guar gum or xanthan gum, help release medication only in the colon, which is helpful for treating inflammatory bowel diseases. 134
Application in Various Disease Areas
Two recent directions inform polysaccharide oncology delivery strategies. First, a multi-omic HCC study identifies STAMBPL1 as a CS-6 target that modulates amino acid metabolism, mTOR signaling, apoptosis, and migration, demonstrating how metabolic rewiring and deubiquitinase pathways yield potent anti-tumor activity
Second, AKR1C3 promotes HCC by upregulating HMGCR and CYP7A1, which leads to more cholesterol and bile acid synthesis. In mice, genetic or pharmacologic down-modulation stops proliferation and lung metastasis. 138 This highlights the potential for polysaccharide-grafted nanocarriers to simultaneously administer statins or AKR1C3 modulators alongside chemotherapeutics, exploiting the weaknesses in tumor lipid metabolism and employing ligand-directed hyaluronan or galactose-bearing polysaccharides for targeted delivery to the liver.
Drug-Specialised Delivery Tool (Infectious Disease, Oncology)
Polysaccharide Drug Delivery in Infectious Diseases
Polysaccharides have become highly valuable in drug delivery systems for infectious disorders owing to their excellent biocompatibility and biodegradability, together with the benefit of chemical modification. 140 Natural polymers like chitosan, alginate, and dextran are widely used to create systems that deliver treatments in a controlled way, improving results for infections caused by bacteria, viruses, and fungi.
One notable quality that facilitates using polysaccharides in drug delivery for treating infectious illnesses is their application in hydrogels, nanoparticles, and microparticles. Topical applications utilize chitosan nanoparticles directly at the infection site to reduce systemic toxicity and enhance
Polysaccharides are effective in combating antibiotic resistance. Nanoparticles using dextran to help deliver antibiotics have been created to specifically target resistant bacteria, which helps lower the chances of resistance by keeping the right amount of medicine at the infection site.
141
(B) Oncological Drug Delivery
Carbohydrate-based polymers are among the most deeply prevalent small molecules that exist and possess significant potential for therapeutic applications in numerous human disorders.142–144 They are naturally occurring long-chain molecules, usually found as structural or storage units, based on their function and the uniformity of their building blocks. The utilization of shape and heteropolysaccharides has a longstanding past in the pharmaceutical discipline. Traditional therapies previously employed them as immunological modulators, anti-tumor aids, and antioxidants.145,146 Diverse polysaccharides are generating significant interest from researchers for various applications in nanotechnology. When used, they can make systems that release substances slowly, films, nanogels, and coatings for different tiny structures, which provide benefits like better stability, safety, low toxicity, attraction to water, compatibility with living things, and the ability to break down naturally.
147
Recent initiatives have led to the development of polysaccharide-based nanomaterials with highly efficient fundamental structures for the controlled release of therapeutic medicines, particularly as tumor-targeting carriers and for various biomedical applications.148,149
(C) Vaccine development
Vaccination deserves the utmost efficacious and successful medical assistance for the prevention of infectious illnesses, capable of reducing mortality, extending life, or enhancing the standard of living. 150 Creation of a coronavirus immunization is essential for protection; nevertheless, it may prove ineffective against future strains, necessitating preparedness for the next outbreak. 151 Polysaccharide adjuvants can augment the immunological efficacy of vaccines, enhancing adaptive immunity, innate immunity, and cellular immunity. Chitosan effectively stimulates bodily and cellular immunological responses and has a demonstrated security profile in both wildlife and people. It is now utilized as an auxiliary to enhance vaccination effectiveness, particularly for RNA virus vaccines.152,153 Chitosan provides little help to animals fighting RSV infection if given after they are infected, but it significantly lowers RSV disease in mouse models when used with a killed respiratory syncytial virus vaccine before infection. This research proposed that chitosan could function as an effective therapeutic intervention or adjunct for RSV contagion. 154 Chitin-adjuvanted immunizations can boost antibody levels against A- and B-type HIV by four to six times compared to doses without chitosan. Inactivated AIV A/H5N2, when mixed with chitosan and given to mice that were later exposed to the same virus, showed better immunization and protection compared to the antigen without chitosan. 155 A chitosan-adjuvanted vaccine maintained at four °C can retain its auxiliary characteristics for a minimum of 8 months. Chitosan can enhance the regenerative and cytotoxic capabilities of spleen nuclear WBCs in mice. Consequently, chitosan serves as a viable emollient for shots against weakened influenza, establishing a standard for the advancement of vaccines targeting new coronaviruses.
Advancing Targeted Drug Delivery Systems
Recent studies emphasize the potential of polysaccharides in designing targeted drug delivery systems. For example, microbiota-sensitive drug delivery systems based on natural polysaccharides show promise in achieving colon-specific drug release. 12 These systems leverage the pH-sensitive properties of polysaccharides, enabling precise drug release profiles while minimizing systemic side effects.
Integration of Marine-Derived Polysaccharides
Marine polysaccharides, such as alginate and carrageenan, have emerged as robust platforms for drug encapsulation. Recent advancements demonstrate their ability to form biocompatible, stimuli-responsive hydrogels, opening new avenues for wound healing and tissue regeneration. 4
Grafting Innovations
State-of-the-art grafting techniques have enabled the functionalization of polysaccharides for enhanced drug delivery. For instance, free-radical grafting of polyethylene glycol onto cellulose enhances hydrophilicity and drug-carrying capacity, paving the way for sustained drug release systems. 2 Similarly, enzymatic grafting of chitosan enhances its mucoadhesive properties, optimizing nasal drug delivery applications.
Current Status of Nanoencapsulation in Drug Delivery
Nanoencapsulation has evolved into a critical approach to polysaccharide-based drug delivery systems, providing excellent protection and release of the therapeutic agents. These systems now use natural materials like alginate, chitosan, and dextran to create tiny carriers that improve how well drugs work in the body, lower side effects, and make the drugs more stable. Examples from recent clinical and preclinical applications of these systems include the following: alginate-based nanocapsules for oral delivery of protein drugs, chitosan nanoparticles for mucosal delivery of vaccines, and dextran-based carriers for targeted release of antibiotics. 156 These examples indicate the immense usefulness of nanoencapsulation focused on localized and sustained delivery of drug types for the benefit of specific therapeutic needs, especially within the fields of oncology, infectious disease, and chronic inflammation. Yet, true and widespread clinical translation is dependent on further refinement of parameters for their formulations, scale-up, and regulatory standardization.
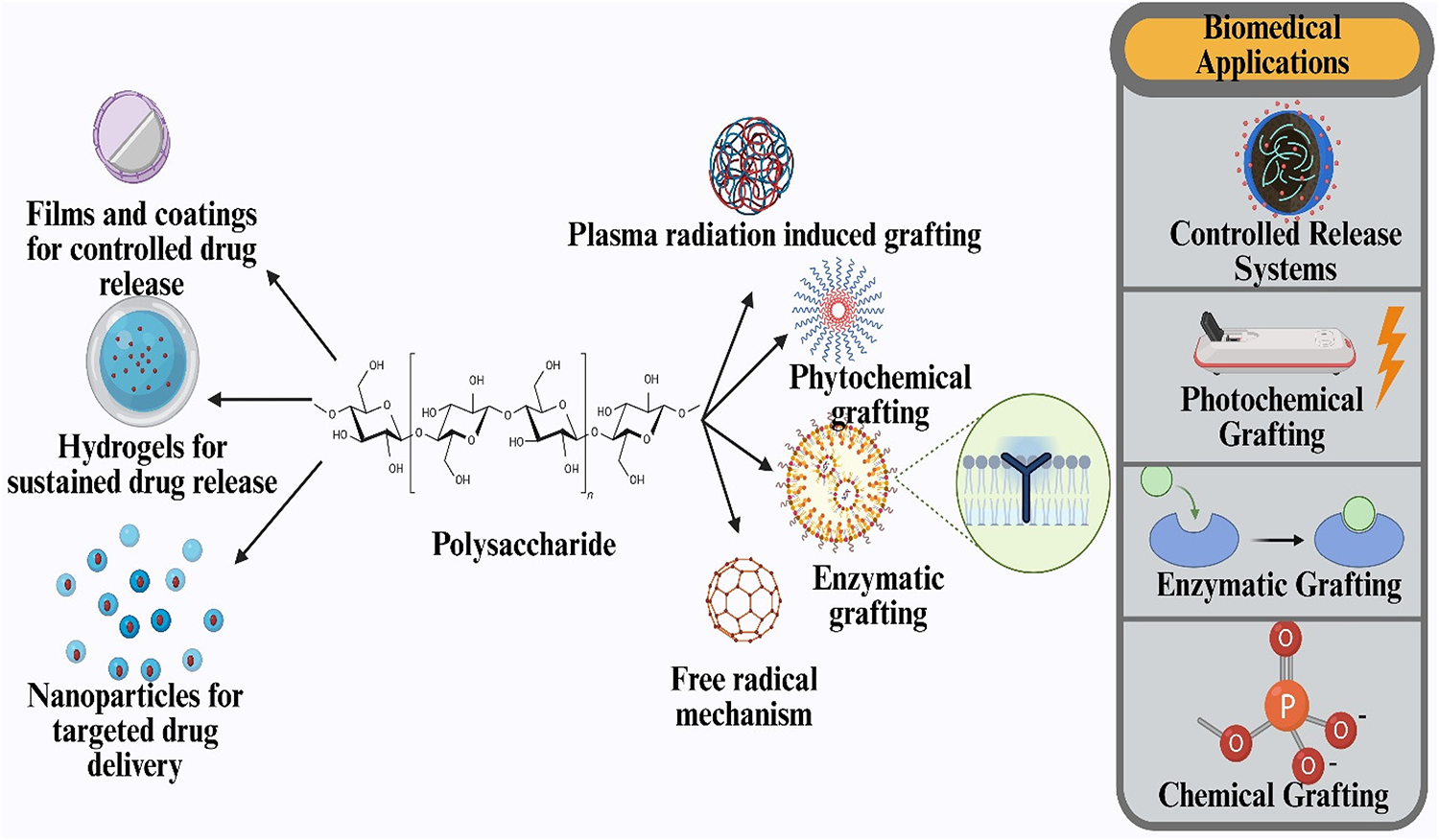
Grafting Technique of Polysaccharides
Grafting is a process in which functional groups or polymers are covalently attached to the backbone of a substrate polymer, such as a polysaccharide. Recent advancements in grafting techniques have enabled the development of materials with enhanced mechanical strength, bioactivity, and targeted functionalities, making them highly suitable for drug-delivery applications. Figure 10 presents an overview of polysaccharide grafting techniques and their key features.

Overview of polysaccharide grafting techniques and their key features.
Chemically Initiated Grafting
The chemical process of grafting can occur via two primary mechanisms: free radical and ionic pathways. The catalyst plays a crucial function in the chemical process since it dictates the trajectory of the grafting procedure. Likewise, with the conventional free-radical process, melt grafting and atom transfer radical polymerization (ATRP) are noteworthy approaches for executing transplantation. ATRP has emerged as a precise and controlled grafting method, allowing for the creation of block copolymers with tailored properties. 157 For instance, ATRP has been used to graft polyethylene glycol (PEG) onto cellulose, enhancing its hydrophilicity and drug-carrying capacity. This technique is particularly beneficial for developing hydrogels for sustained drug release.
Chemically initiated grafting is the most generally applicable and popular method for the modification of polysaccharides, with good monomer compatibility and relatively low expense. It is favorably suited to scale-up production due to its simplicity of setup and clear protocols for execution. Its principal disadvantages are the limited ability to control grafting sites and possible generation of toxic by-products, which can restrict biomedical applications unless strict purification is carried out. Translationally, this method is attractive for industrial-scale applications but may require optimization for clinical use to address biocompatibility concerns.
Primary laboratory investigations demonstrate chemically initiated grafting's practicality. Truong et al (2021) showed that ATRP-grafted cellulose doubled hydrogel modulus and reduced burst release by 45% for encapsulated peptides. Another work by Carlmark and Malmström (2002) grafted styrene onto cellulose fibers with good control, verifying ATRP's mechanistic accuracy. Xu et al (2019) found that free-radical–based N-isopropylacrylamide grafting onto chitosan caused thermo-responsive swelling and regulated protein release. These main data show how regulated grafting chemicals improve function.
Free-Radical Grafting
Graft copolymers are created when initiators produce free radicals during the chemical process. These radicals are then transferred to the substrate and combined with monomers. Both direct and indirect methods can be used to produce free radicals.
Free radicals are generated indirectly via a redox process.158–160 Specific intermediate metal cations can immediately oxidize the spine of a polyamide framework, producing complementary radical sites. The redox force of metallic ions is a crucial factor in assessing patching efficacy. Generally, ionic metals that have minimal potential for oxidation are favored for enhanced grafting efficiency.161–167
Grafting Using Live Polymerization
Recent advancements in ‘living polymerization’ techniques have emerged, offering the possibility of branching reactions. In, 168 A fairly realistic concept of a ‘living polymer’ maintains its capacity for prolonged propagation and can achieve a specified size limit while exhibiting minimal cessation or network transmission. The characteristics of electrostatic and conventional free-radical polymerization are combined in targeted free-radical polymerizations. To create unreactive (or “dead”) polymers and ensure consistent polymer sizes with a broad range of weights, conventional free-radical polymerization requires ongoing initiation, which halts the growth of chain radicals through association or disproportionation reactions. Live polymerization yields live polymers with minimal polydispersities and controlled molecular weights.169–175
On polymeric substrates and colloidal particles, guided radical polymerizing over surface transfer has been reported.176–181 For instance, the work of Carlmark and Malmstroem 182 provides evidence in this regard. Immobilized 2-bromoisobutyryl bromide by interaction from the -OH groups present on a paper filter. Grafting is then carried out by submerging the altered sheet in a chemical reaction mixture that contains ethyl acetate and methyl acrylate.
Photochemical Grafting
UV and visible-light-induced grafting techniques have seen rapid development. These methods have been applied to graft acrylic derivatives onto dextran for use in colon-targeted drug delivery systems, providing pH-responsive behavior. Upon light absorption via a chromophore attached to a macromolecule, it transitions to an elevated energy state, potentially leading to the dissociation of responsive free radicals and initiating the branching reaction. The addition of photosensitive substances like benzoin ethyl ether, colors like Na-2,7 anthraquinone sulfonate, or acrylate azo dye might accelerate the process if light absorption is unable to provide free-radical locations by link cleavage. Grafting using a photochemical approach can take place either alone or in combination with a sensitizer.183–185
Photochemical grafting offers fast, spatiotemporally controlled functionalization under mild conditions and is thus promising for the construction of stimuli-responsive drug delivery systems. It offers localized modification without extensive chemical reagents and can be used to fabricate smart carriers for colon-targeted and controlled-release purposes. Efficiency can be compromised by light penetration, and substrate compatibility restricts its application to some polysaccharides. From a translational point of view, precision and scalability of the method with the right light sources render it promising for next-generation biomedical coatings and responsive systems.
Grafting Produced by Plasma Radiation
Plasma technology has advanced significantly, enabling grafting under environmentally friendly conditions without solvents. Plasma-induced grafting of chitosan has been applied to produce bioactive coatings for wound healing and targeted drug delivery systems.186,187 The fundamental processes of plasmas include electron-induced stimulation, ionization, and dissociation. The excited electrons from the plasma have sufficient energy to sever those chemical linkages in the polymeric structure, leading to the generation of macromolecular electrons that eventually initiate graft copolymerization.
Plasma-induced grafting is a chemical-free, environmentally friendly method that allows polysaccharide surface-specific modification, suitable for biomedical coatings and wound-healing purposes. It does not require chemical initiators, allowing for improved biocompatibility and fewer residual contaminants. The method is more or less restricted to surface modifications and needs specific equipment, which can limit its large-scale industrial application. However, its green profile and capacity to form bioactive surfaces render it of very high value for coating of medical devices as well as scaffolds for tissue engineering.
Enzymatic Grafting
The catalytic grafting approach is rather recent. The concept is that an enzyme facilitates the chemical/electrochemical grafting reaction. 188 Enzymatic methods are gaining popularity due to their specificity and mild reaction conditions. For example, tyrosinase-mediated grafting has been employed to modify chitosan, creating nanoparticles with enhanced mucoadhesive properties for nasal drug delivery. 189 The polyphenol oxidase (PPO) connection to polydicarbazole was documented, and thionine and toluidine blue appeared to be firmly fused to the poly(dicarbazole) backend.
Enzymatic grafting is differentiated by its high selectivity, gentle reaction conditions, and environmentally friendly character, which is especially beneficial for delicate biomolecules. The process provides clean functionalization with no use of toxic reagents, minimizing safety issues and protecting polysaccharide integrity. Challenges to the method include enzyme instability, narrow substrate range, and increased cost, which are a problem for scale-up. Translational applications include stabilizing enzyme systems and creating economical biocatalytic processes that may propel enzymatic grafting to a premier method of nasal, mucosal, and vaccine delivery systems.
Mechanistic Underpinnings of Grafting and Function
To further mechanistic insight, we describe how grafting reaction kinetics and polymer architecture control functional performance. Using controlled/living and radical grafting (eg, ATRP), activation/deactivation versus termination rate ratios control graft length distribution and polydispersity, which control hydrophilicity, chain mobility, and network development. Side-chain chemistry and grafting density control hydrogel mesh size, swelling, and relaxation behavior, advancing drug release from Fickian diffusion to anomalous or erosion-controlled regimes. For amphiphilic graft copolymers, the critical aggregation concentration and core–shell stability of micelles are adjusted by hydrophobe length and corona charge, directly impacting solubilization of hydrophobic payloads and serum stability. Cationic or hydrogen-bonding grafts (quaternary or catechol/phenolic motifs) increase mucoadhesion and paracellular transport by way of enhanced interfacial interaction and transient modulation of the tight junction. Stimuli-responsiveness is attributed to graft-implanted dynamic linkages such as disulfides (redox), imines/acetals (pH), and enzyme-cleavable peptides (proteases) that are susceptible to environment-regulated scission, facilitating on-demand release and intracellular unloading in reductive or acidic tumor/endosomal environments. In general, control of initiation kinetics, graft density, and linker chemistry provides a direct means for the optimization of solubility enhancement, cargo protection, targeting (avidity), and spatiotemporal release, correlating synthesis parameters with translatable drug-delivery outcomes. 138
Critical Evaluation of Grafting Methods
Each grafting method provides distinct advantages and limitations that determine its translational suitability. Atom Transfer Radical Polymerization (ATRP) enables precise control over molecular architecture and is highly efficient for synthesizing well-defined polysaccharide conjugates; however, its reliance on metal catalysts raises biocompatibility and scalability concerns. Plasma-induced grafting, by contrast, offers surface-specific modifications without extensive chemical reagents, making it attractive for biomedical coatings, though the need for specialized equipment limits widespread industrial use. Enzymatic grafting is particularly noteworthy for its eco-friendliness and high selectivity under mild conditions, yet enzyme instability and cost hinder large-scale applications. Photo-induced grafting provides rapid, spatiotemporally controlled reactions, well-suited for stimuli-responsive drug delivery platforms, but its efficiency is often influenced by light penetration and substrate compatibility. Traditional chemical grafting remains the most accessible and low-cost approach, but it often suffers from poor control over reaction sites and potential generation of toxic by-products. Taken together, these techniques demonstrate that the choice of grafting strategy should balance efficiency, scalability, cost-effectiveness, and safety, depending on the desired biomedical or industrial outcome. Table 4 presents a detailed comparison of grafting methods for polysaccharide modification in drug delivery applications.
Comparative Analysis of major Grafting Methods for Polysaccharide Modification, Highlighting Their Efficiency, Scalability, Biocompatibility, Advantages, Limitations, and Translational Feasibility in Drug Delivery Applications.

Pharmaceutical Utilization of Organic Polysaccharides
Organic polysaccharides, which come from renewable sources, have many benefits compared to synthetic polymers, including being safe for health, breaking down naturally, being compatible with living tissues, being affordable, and being readily available. These biopolymers are widely used in the pharmaceutical sector in the form of fine particles, microspheres, films, inserts, matrices, and dense fluids. Common examples include dextrin, chitosan, alginate, pectin, guar gum, starch, and xanthan gum. They are applied in drug delivery systems to optimize drug bioavailability, ensure chemical stability, and achieve controlled release rates. Biopharmaceutical products formulated with these polysaccharides are administered via oral, nasal, transdermal, and ocular routes, enhancing both efficacy and ease of administration. Renewable materials are becoming pivotal in the development of advanced drug-delivery systems. 194
Chitosan in the Pharmaceutical Sector
Because they are very compatible with living tissues, break down naturally, and have natural germ-fighting properties, chitosans are used in many medicine and biomedicine products. Chitosan is a polysaccharide obtained from the partial deacetylation of chitin. Chitosan can be considered an effective carrier for delivery routes for various therapeutic agents like insulin, human growth hormone, and plasmid DNA.195–197 Chitosan-based nanoparticles and hydrogels are very promising for delivering drugs directly through the nose, mouth, skin, and eyes because they stick well and help the drugs get absorbed through mucous membranes. For instance, researchers have developed chitosan-polyethylene glycol hydrogels to improve the pulmonary delivery and retention of curcumin, suggesting its potential application in non-invasive drug delivery systems. 198 Though very useful, chitosan poses a challenge for wider application because of its very limited solubility under neutral or basic pH conditions. To overcome this, chemical modifications such as grafting copolymerization have been employed; particularly, the creation of N-trimethyl chitosan chloride (TMC) that gives better solubility and functionality under physiologically relevant conditions. 199 Commercially, chitosan appears in wound dressings such as ChitoFlex® and in drug delivery systems like chitosan nanoparticles from Biomedica. Such examples demonstrate that chitosan has substantial translational relevance. 200
Pectin in the Pharmaceutical Sector
Pectin comes from the walls of plant cells and is a type of complex sugar that is found in large amounts. It is commonly used in medicine because it works well with the body and can serve many purposes, like helping to thicken and stabilize products. Because of its gelling properties and biological tissue interactions, pectin is considered suitable for different dosage forms that include hydrogels, dermal patches, and microparticles.201–205 Pectin is a key type of polymer used to deliver drugs specifically to the colon because it is broken down only by the bacteria in the colon, enabling targeted drug release for treatment in that area. Modified pectin formulations have also shown some considerable promise for use in insulin delivery and applications in gene therapy. Furthermore, established evidence suggests that pectin binds with galectins found on the surface of malignancies. This binding eventually results in apoptosis and substantiates its possible therapeutic use in the treatment of cancer. 206 Commercial prospects with interest include pectin-based colon-targeted tablet developments with PharmaPectin. This indicates a growing trend of clinical interest in this natural polysaccharide.
Gums in the Drug Sector
Compared to other gums, Guar gum and acacia gum find their prime application in pharmaceutical formulations due to their origin, biocompatibility, and functional versatility. Recent improvements in grafting methods, especially acrylamide grafting, have greatly improved how well these polymers work in delivering drugs by adding groups that help with loading and releasing the drugs. 207 A major advancement with Nisopropylacrylamide-grafted acacia gum is that it can create drug delivery systems that respond to temperature changes, allowing for controlled release of medications like diltiazem hydrochloride and nifedipine. 208 These innovations highlight the potential of modified natural gums in creating responsive and efficient drug delivery platforms.
Alginate in the Pharmaceutical Sector
Alginate is a naturally occurring polysaccharide of β-D-mannuronic acid and α-L-guluronic acid, known for excellent biocompatibility, biodegradability, and non-toxic nature. Thus, it is an ideal material for controlled drug delivery and diverse biomedical applications. 209 Its unique ability to form a gel when divalent cations like calcium are present helps to trap and slowly release medicine. 210 Microcapsules made from alginate are commonly used in gene therapy and hormone therapy, where alginate-poly-L-lysine mixtures help protect the cells involved in gene therapy from the immune system. Alginate dressings, such as Kaltostat®, greatly assist in treating exudative wounds due to their high absorption and hemostatic properties. 211 Zinc-enriched alginate fibers have facilitated platelets’ activation and coagulation, further supporting wound healing. Alginate-based hydrogels and wound dressings are commercially available from ConvaTec and Smith and Nephew, affirming alginate's clinical relevance and versatility. 212
Starch in the Pharmaceuticals Sector
Starch is a polymer present in plant storage organs primarily as linear amylose and branched amylopectin chains. It is an excellent polysaccharide in pharmaceutical and industrial applications because of its biocompatibility, biodegradability, and natural availability. Its hydrophilic character is favorable for cell adhesion and growth applications, making it an appropriate material for making wound dressings and scaffolds in tissue engineering. Researchers have developed starch-based nanoparticles to enhance the bioavailability of therapeutic agents through controlled drug release via oral delivery. Researchers recently developed starch hydrogels to sustain the release of anti-inflammatory drugs, showing potential applications in managing chronic diseases. 213 Applications also extend into areas such as transnasal insulin delivery. We demonstrate that starch nanoparticles improve bioavailability and deliver controlled release. We further modify them for specific applications, such as producing high amylose methacrylic acid coproducts in pH-sensitive, colon-targeted delivery systems. 214 Commercially, starch-based carriers like EcoSphere® starch microspheres are employed in both drug delivery and tissue engineering, reflecting their practical relevance and versatility. 215
Future Direction
Polysaccharide drug delivery systems derived from advanced laboratory creations will become patient-ready only when mechanistic design, sustainable manufacturing processes, analytical rigor, and regulatory considerations align within a cohesive framework. Despite remarkable laboratory accomplishments, significant knowledge gaps and translational obstacles persist. At the molecular level, next-generation initiatives must delineate precise design principles governing graft architecture (length, dispersity, density) and functional outcomes, including mesh size, diffusivity, mucoadhesion, protein corona interactions, and release kinetics for diverse therapeutics (small molecules, peptides, nucleic acids). Current grafting methodologies lack the precision necessary for consistent management of these parameters, and orthogonal chemistries that separate ionic charge from stimuli-responsiveness remain underexplored. Likewise, zwitterionic or glyco-inspired grafting could prevent opsonization while maintaining receptor-mediated internalization, but requires thorough exploration. These experimental methodologies must be integrated with multiscale modeling (molecular dynamics, network physics, PBPK modeling) to enhance
From a chemical standpoint, a shift towards genuinely aqueous, metal-free bio-orthogonal grafting processes remains a necessary advancement yet to be realized. Despite the potential of enzyme reactions, photo-initiated reactions under visible light, thiol–ene conversions, and click-like transformations, scalability, repeatability, and depth-controlled patterning are necessary for validation. Additional methods for developing dynamic and reversible bonding for the creation of self-healing or stimuli-responsive materials are still in their early stages.
Industrial translation remains difficult. Continuous photografting platforms, long-lived enzymatic reactors, and high-throughput plasma or LED systems are needed to scale grafting reactions, although major production demonstrations are rare. Production heterogeneity and batch-to-batch variation hinder regulatory certification, especially for chitosan and nanocellulose systems. There are no standard techniques for characterizing network rheology, nanoparticle quality, impurity profiles, or grafting uniformity. To close these gaps, analytical processes must meet FDA and EMA endotoxin control, reproducibility, and extractable/leachable assessment standards.
From a clinical perspective, patient-centered design ideas remain nascent. Future product improvements should incorporate changeable rheology, gelation inducers, mucoadhesion, palatability, and conformability relevant to patient populations (pediatrics, geriatrics, oncology, infectious diseases). The absence of adequate long-term safety data for chronic exposure substantially hinders clinical approval. The discipline lacks shared reference materials, standardized minimum reportable standards, and open-access datasets for structure–property–performance utilities, hindering quick reproducibility and interlaboratory comparisons. Finally, although polysaccharides are promoted as “greener” alternatives, their environmental characteristics are often not systematically evaluated. Future study must encompass traceable biomass feedstocks, cradle-to-gate life cycle assessments, and end-of-life degradation studies to validate reduced carbon footprints and the absence of microplastic persistence. To bolster trust, clinic-oriented examples must demonstrate a thorough integration of scientific innovation with manufacturing and regulatory compliance. Illustrative cases include enzyme-grafted, redox-responsive nanocarriers for concurrent delivery of chemotherapeutics and genes; roll-to-roll photo- or plasma-grafted antimicrobial wound films with verified shelf life and packaging; and long-acting, in situ gelling hydrogels designed for biologics produced via continuous processes. Advancing these systems through established go/no-go criteria related to potency, safety, manufacturability, and affordability will set the benchmark for effective translation.
Conclusion
The integration of natural polysaccharides with advanced grafting techniques continues to open new possibilities in materials science, biotechnology, and drug-delivery research. Grafting-based modifications have helped overcome limitations inherent to native polysaccharides—such as poor solubility, limited mechanical strength, and instability under physiological conditions—thereby enabling their expanded use in drug-delivery systems, regenerative medicine, packaging, and environmental applications. Despite these advancements, several critical research gaps remain.
Future work must prioritize deeper structure–function analysis to clarify how grafting density, chain architecture, and functional group chemistry influence biological performance, including mucoadhesion, cellular uptake, biodegradation, and immunogenicity. Likewise, systematic biocompatibility and long-term toxicity studies are still scarce and are essential for successful clinical translation. More effort is also required to develop scalable, reproducible, and sustainable grafting methods, particularly using green-chemistry approaches such as enzymatic catalysis, solvent-free plasma activation, or energy-efficient photografting. These methods hold promise not only for reducing environmental impact but also for improving regulatory acceptance by minimizing residual reagents and reducing purification requirements.
Translational progress further depends on addressing industrial and regulatory challenges, including batch-to-batch variability of natural raw materials, ensuring consistent grafting outcomes, and meeting stringent FDA/EMA expectations for characterization, impurity profiling, and stability data. Standardizing analytical protocols and extraction/synthesis procedures will be crucial for bridging laboratory innovations with scalable manufacturing.
Polysaccharide-based materials also hold strong potential as sustainable alternatives to petroleum-derived polymers, as demonstrated by biodegradable alginate and chitosan wound dressings, starch- and cellulose-based packaging films, and environmentally benign chitosan flocculants in water treatment. Expanding the use of grafted polysaccharides in such eco-friendly technologies aligns with global sustainability goals and supports the development of greener industrial solutions.
Overall, the transformative capacity of polysaccharides enhanced through precise and innovative grafting techniques offers immense opportunities for medicine, industry, and environmental engineering. Addressing the identified research gaps through interdisciplinary collaborations focused on biocompatibility, scalability, regulatory compliance, and green chemistry will be essential for advancing grafted polysaccharides from promising laboratory materials to robust clinical and commercial products.
Footnotes
List of Abbreviations
Acknowledgements
The authors are thankful to Kampala International University and Galgotias University for providing the necessary facilities to conduct this research work.
Ethical Approval
Not applicable.
Consent to Participate
Not applicable
Consent to Publication
All authors read and agreed to the final copy of the findings as contained in the manuscript.
Author Contribution Statement
J.V., S.Y. and S.P.N.B. designed and performed the experiments, derived the models and analyzed the data. S.P.N.B. assisted S.Y. in measurements. S.P.N.B. and A.K.C. were assisted by J.V. and S.Y. in carrying out simulations. S.P.N.B., A.K.C., C.T., S.K.K. and J.V. wrote the manuscript draft. J.V., S.Y., S.P.N.B., A.K.C., C.T., R.K.K. and S.T.S. critically reviewed the manuscript. Writing—review and editing: All authors, Validation and Approval: All authors.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: I hereby declare that this submission is entirely my own work, in my own words, and that all sources used in researching it are fully acknowledged and all quotations properly identified.
Availability of Data and Materials
Not applicable.
