Abstract
The objective of this study was to achieve the practical bioapplications of silicon nanowires (SiNWs). In this study, the tumor and normal cell lines were used as models to systematically investigate the cytotoxicity of SiNWs synthesized by HF-assisted etching methods. Morphology observation, Cell Counting Kit 8, real-time polymerase chain reaction, and flow cytometry analysis were used to elucidate the cytotoxicity of SiNWs. The results showed that the cytotoxicity of SiNWs is greatly dependent on cell lines, SiNWs concentration, and incubation time. Particularly, SiNWs show better biocompatibility with tumor cell lines (eg, human epithelial cervical cancer [Hela] cells and human hepatocellular liver carcinoma [HepG2] cells) than normal cell lines (eg, human normal liver [HL-7702] cells and human embryonic kidney [HEK293T] cells). The reasons may be that SiNWs could tightly attach to the cell membrane in the cell medium, which obviously affects cell adhesion and inhibits their cell viability, especially for normal cell lines. From systematical analysis and comparison, we obtain the concentration limits of SiNWs, which may advance SiNWs applications and its toxicological study in vitro.
Introduction
Silicon nanomaterials have been extensively studied due to their many attractive properties, such as excellent electronic/mechanical properties, convenient surface tailorability, and compatibility with conventional silicon technologies. 1 -3 Silicon nanowires (SiNWs), as typical 1-dimensional silicon nanostructures, have drawn intensive attention for their great promise as a platform for various applications ranging from electronics to biology. 4 Biological applications of SiNWs are particularly interesting. For example, Lieber and coworkers develop SiNWs-based field-effect transistors for both spatially resolved, high-sensitivity biomolecular detection and stimulation of neuronal signal propagation. 5 Yang and coworkers demonstrate interfacing of SiNWs with mammalian cells without any external force. 6 Recently, SiNWs are used as bioimaging agent with intrinsic 3D spatial resolution, high photostability, and orientation information, providing new opportunities for cellular interaction studies. 4 Wang et al developed a SiNWs-based system for quantifying mechanical behavior of cells lines, revealing force information of cancer cells. 7
As for any nanomaterials, biosafety assessment of SiNWs is of critical importance for widespread biological/biochemical applications. 8 Cytotoxicity of nanomaterials, such as fluorescent Ⅱ-Ⅵ quantum dots, 9,10 silver/gold nanoparticles, and carbon nanotubes, 11,12 has been extensively studied, providing valuable guidance for bioapplications such as biosensors, bioimaging, disease diagnosis, and therapy. 13 Nevertheless, there has been only scanty and ambiguous information regarding the potential hazards of silicon nanostructures. A recent report revealed that silicon quantum dots with different surface modification could induce different levels of cytotoxicity. 14,15
We herein report a systematic study on the cytotoxicity of SiNWs prepared from HF-assisted etching methods. Four different malignant and normal cell lines, that is, human epithelial cervical cancer (Hela) cells, human hepatocellular liver carcinoma (HepG2) cells, human normal liver (HL-7702) cells, and human embryonic kidney (HEK293T) cells, are used to evaluate the cytotoxicity effect. Our results demonstrate that the tumor cell lines exhibit more favorable compatibility with the SiNWs than the normal cell lines, indicating cell line–dependent cytotoxicity of SiNWs. Based on the experiment results, we discuss the effects of cell lines, incubation time, and SiNWs concentration on cytotoxicity in a detailed way and deduce the thresholds of cytocompatibility concentrations of SiNWs suitable for safe and regulated bioapplication of SiNWs.
Methods and Materials
Synthesis and Treatment of SiNWs
Silicon nanowires were synthesized via HF-assisted etching methods. The SiNWs are produced via a selective etching process by immersing a freshly cleaned Si wafer in AgNO3/HF solution based on electrochemical reaction. The resultant SiNWs are detached from the surface of Si wafer by ultrasonic treatment and collected for cytotoxicity evaluation.
Scanning Electron Microscope Observation of SiNWs
The SiNWs on the surface of Si wafer were detected by a Quanta 400 field emission gun scanning electron microscope (SEM) at an accelerating voltage of 5 keV.
Cell Culture
Hela cells, HepG2 cells, and HEK293T cells are cultured in Dulbecco modified Eagle medium, supplemented with 10% heat-inactivated fetal bovine serum (FBS) and antibiotics (100 μg/mL), streptomycin, and 100 U/mL penicillin at 37°C in the humidified atmosphere with 5% CO2. Human normal liver-7702 cells are cultured in RPMI1640, supplemented with 10% heat-inactivated FBS and antibiotic, streptomycin, and 100 U/mL penicillin, at 37°C in the humidified atmosphere with 5% CO2.
Cell Counting Kit 8 Assay and Cell Morphology Observation
Cells (in culture medium) are dispensed in 96-well plates (90 µL in each well containing 1 × 104). Then 10 µL of SiNWs with serial concentrations (20, 200, 500, 1000, and 1500 µg/mL) are added to each well, respectively. The samples are incubated for different periods (12 hours, 1 day, and 3 days) at 37°C under the humidified atmosphere (5% CO2) and in dark. Established Cell Counting Kit 8 (CCK-8) assays, recognized as one typical method for measuring cell viability, 16 are performed to assess the metabolic activity of cells exposed to the SiNWs. With different incubation time, 10 µL of stock CCK-8 reagents are added to each well, and cells are then incubated for 4 hours at 37°C. Absorbance is measured at 450 nm using microplate reader (Bio-Rad 680; Bio-Rad, Hercules, CA). In addition, morphological observation with laser-scanning fluorescent confocal microscopy (Leica, SP5; Leica) is carried out to assist assessment of cytotoxicity. In this study, cells are dispensed in 96-well plates and treated with the SiNWs as described earlier, with a series of incubation time, 12 hours, 1 day, and 3 days, at 37°C under the humidified atmosphere (5% CO2) and in dark.
Real-Time Polymerase Chain Reaction
Total RNA and protein of cells treated with various concentrations (0, 50, and 150 μg/mL) of SiNWs for 3 days were extracted using TRIzol reagent (Life Technologies) according to the manufacturer’s protocol. 17 The messenger RNA (mRNA) was converted to complementary DNA in 20-µm reaction volumes using a PrimeScript RT Reagent Kit (Takara, Japan). Real-time polymerase chain reaction (RT-PCR) was performed using SYBR Premix Ex Taq(Takara), by following the manufacturer’s instructions, and run on a 7500 Real-Time PCR system (Applied Biosystems). The mRNAs were amplified using gene-specific PCR primer sequences for mouse (n = 5 for each experiment). The control protein in our PCR study is β-actin, and the primers used for quantitative RT-PCR is shown in the Table 1.
Primers Used for Quantitative Real-Time PCR.

Abbreviation: PCR, polymerase chain reaction.
Flow Cytometry Analysis
Cell cycle is assessed by flow cytometry (FACScalibur; BD Corp). 18 In brief, cells seeded on the 6-well plate are treated with 0, 50, and 150 μg/mL SiNWs, for 3 days, respectively. Afterward, 1 × 106 cells are collected, washed twice with phosphate-buffered saline, and then fixed with 70% precooled ethanol and stored at 4°C for 24 hours. Analyses are performed with FACScan flow cytometry software to evaluate the percentage of cells in various phases (subG1, G1, S, G2/ M) of the cell cycle.
Statistical Analysis
All experiments were performed in triplicate, and the data are shown as the means ± standard deviations of 3 separate experiments. Statistical analysis was performed using 1-way analysis of variance followed by Tukey’s test. Differences with P < .05 were considered to be significant, while P < .01 was considered to be highly significant.
Result
Synthesis and Characterization of SiNWs
Vertically aligned SiNWs were prepared on the surface of the Si wafer, and the morphology of SiNWs were characterized by SEM (Figure 1). The cross-sectional SEM images of SiNWs arrays showed that the lengths of SiNWs were about 9.72 ± 1.01 µm (Figure 1A). The vertically aligned SiNWs are detached from the surface of Si wafer by ultrasonic treatment, and the length of the resultant SiNWs was within range of 200 to 1000 nm (Figure 1B).

The characteristics of the silicon nanowires (SiNWs) arrays. A, Side view scanning electron microscopy (SEM) images of SiNWs arrays at high magnification. B, SiNWs well dispersed in culture medium after ultrasonic treatment. Scale bar represents 3 µm and 500 nm, respectively.
Cell Viability
The effects of SiNWs on cell metabolic activities were first examined by CCK-8 assays, which allow sensitive colorimetric assays for the determination of cell viability in cell proliferation and cytotoxicity. Cells were exposed to SiNWs at 6 different concentrations (from 0 to 150 μg/mL) for 12 hours, 1 day, and 3 days, respectively. The Hela cell line, derived from cervical cancer cells, is one of the oldest and most commonly used human cell lines. 19 Figure 2 displays evolution of cell vitality under different exposure concentrations and times. As shown in Figure 2A, SiNWs are noncytotoxic to Hela cells during 1-day incubation, even at the highest concentration (150 μg/mL). When the incubation time was up to 3 days, most of the cells had good viability when the SiNWs concentrations were less than 100 μg, but the cell viability decreased to 86% at 150 μg/mL of SiNWs. Similar results could be seen in another tumor cell line, HepG2, the cell viability retains only 80% for 3-day exposure time (Figure 2B). In order to further study the cytotoxicity of SiNWs, 2 normal cell lines (HL-7702 cells and HEK293T cells) were exposed in SiNWs suspension, respectively. Figure 2C and D shows the evolution of cell viability with different SiNWs concentrations and exposure times for the 2 kinds of normal cell lines. Human normal liver -7702 cells retain 69% cell viability during 1-day incubation at SiNWs concentration from 0 to 150 μg/mL. Moreover, the cell viability is only 60% when the incubation time is 3 days with 150 μg/mL SiNWs (Figure 2C). For HEK293T cells, an obvious decrease in metabolic activity is observed at 3-day incubation time when the concentration of SiNWs is highest (Figure 2D). Those results demonstrated that SiNWs have a better biocompatiblity for tumor cells.

Cytotoxicity of HF-silicon nanowires (SiNWs) with different concentrations and incubation times with Hela (A), HepG2 (B), HL-7702 (C), and HEK293T (D) cells and were detected by CCK-8 assay. The results are means ± standard deviation (SD) from 3 independent experiments, *P < .05, **P < .01. HL indicates human normal liver; HEK293T, human embryonic kidney; Hela, human epithelial cervical cancer; HepG2, human hepatocellular liver carcinoma.
Effect on Cell Morphology
The second significant effect following exposure of cells to toxic materials was the alteration in cell shape or morphology. Morphology change in the 4 kinds of cell lines treated with SiNWs at different concentrations (0, 50, and 150 μg/mL) at 3-day incubation time is illustrated in Figure 3. As shown in the Figure 3A and B, cells kept normal spreading patterns and healthy shape in the untreated control, and the morphology of Hela cells and HepG2 cells incubated with 50 μg/mL SiNWs for 3 days remains the same as that of untreated cells. With 150 μg/mL SiNWs, we observe that cells become slightly swollen and a portion of cells had no SiNWs attached. For the normal cell lines, they exhibit undefined nuclei and granular cytoplasm when incubated with 150 μg/mL SiNWs of 3-day exposure time (Figure 3C and D).
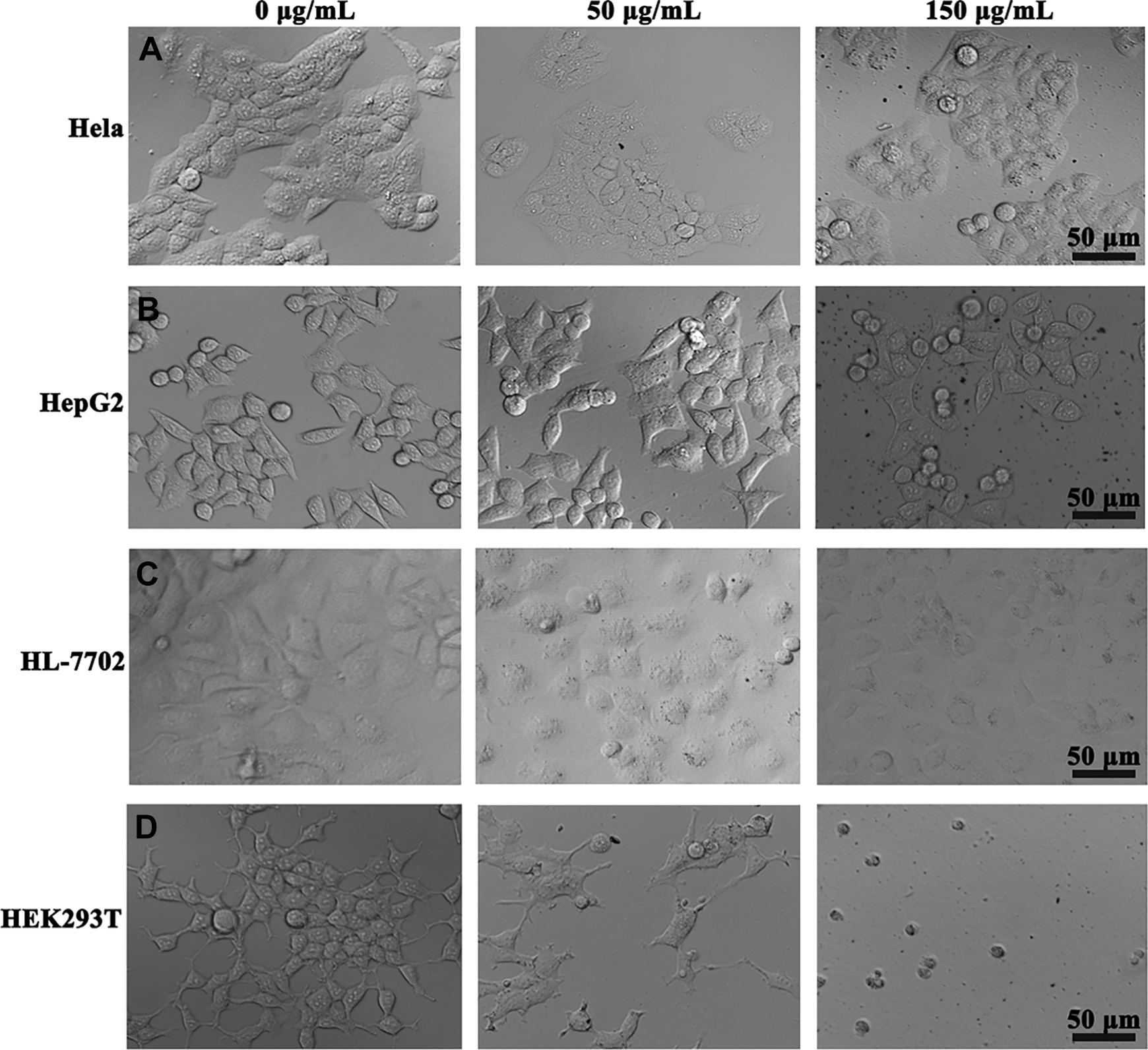
Morphology of Hela (A), HepG2 (B), HL-7702 (C), and HEK293T (D) cells after incubation with different concentrations of silicon nanowires (SiNWs) for 3 days, respectively. Scale bar corresponds to 50 μm. HL indicates human normal liver; HEK293T, human embryonic kidney; Hela, human epithelial cervical cancer; HepG2, human hepatocellular liver carcinoma.
The Quantitative Analysis of Ki67 mRNA
For a quantitative analysis, we measured the mRNA expression of Ki67. Ki67 is a kind of a nucleoprotein and involves in cell cycles, its expression generally exhibits cell proliferation activity. 20 Cells were first harvested and subjected to RT-PCR and when the cells cultured for 3 days in the medium with different SiNWs concentrations. Real-time-PCR analysis of the normal cells (HL-7702 and HEK293T) at third day showed a large and significant decrease (approximately 1.25-fold and 4.56-fold, respectively) in the expression of mRNA level for Ki67 (Figure 4). Moreover, the Ki67 mRNA expression of the tumor cells (Hela and HepG2) at third day has no obvious difference from the untreated groups.
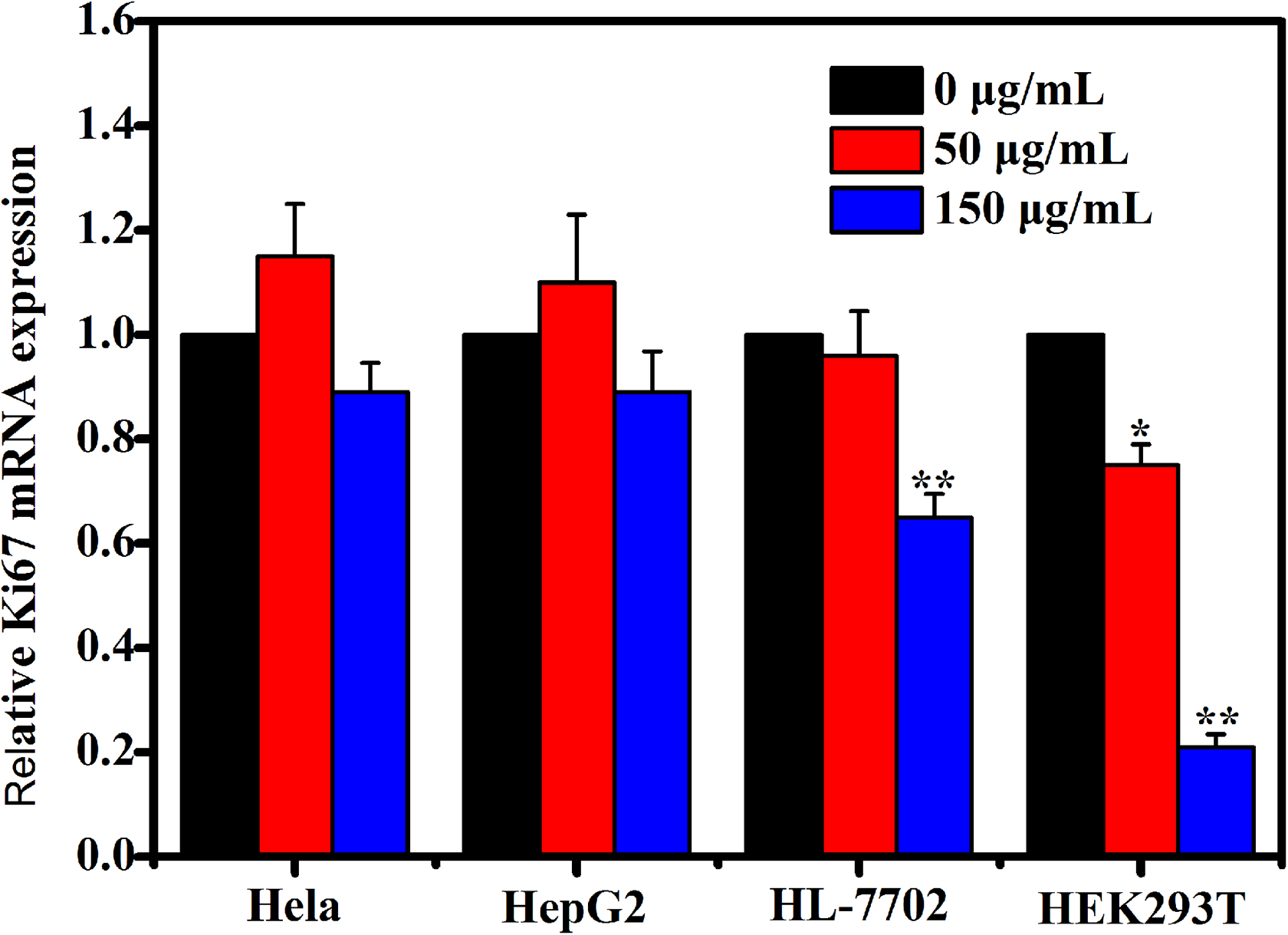
The quantitative analysis of Hela, HepG2, HL-7702, and HEK293T cells viability after incubated with different concentrations of silicon nanowires (SiNWs) for 3 days. The data are presented as the means ± standard deviation, *P < .05, **P < .01. HL indicates human normal liver; HEK293T, human embryonic kidney; Hela, human epithelial cervical cancer; HepG2, human hepatocellular liver carcinoma.
Effect of SiNWs on Cell Cycle
DNA content and cell cycle phase distribution were analyzed by flow cytometry based on propidium iodide staining. Apoptotic cells with degraded DNA appeared in the area left to the G0/G1 peak was called “sub-G1” phase. 21 The percentage of the 4 cell lines in each phase of cell cycle was compared to the 0 μg/mL SiNWs. As shown in Figure 5, tumor cells (Hela and HepG2) treated with 50 and 150 μg/mL SiNWs for 3 days showed no obvious difference compared to the untreated groups. However, normal cells (HL-7702 and HEK293T) treated with 50 and 150 μg/mL SiNWs for 3 days showed a significant increase in sub-G1 population, especially when the concentration of SiNWs was 150 μg/mL, indicating that cell proliferation was inhibited.

The stack column representing cell cycle changes of Hela, HepG2, HL-7702, and HEK293T cells treated different concentrations of silicon nanowires (SiNWs) at different concentrations (0, 50, and 150 μg/mL) for 3 days, P < .01. HL indicates human normal liver; HEK293T, human embryonic kidney; Hela, human epithelial cervical cancer; HepG2, human hepatocellular liver carcinoma.
Discussion
Silicon nanowires show high promise as a novel platform for wide-ranging biological applications due to their various attractive properties. Although the effects of SiNWs to cell morphology, adhesion, and differentiation have been the preliminary studies, the toxic effect of SiNWs on cells is unknown. In this study, our systematic data demonstrate that SiNWs cytotoxicity is greatly dependent on cell lines, SiNWs concentration, and incubation conditions. In comparison to normal cell lines, tumor cell lines are more cytocompatible with the SiNWs. For instance, Hela cells and HepG2 cells preserve >85% metabolic activities when they are cultured with 150 μg/mL SiNWs for 3 days; comparatively, cell viability of HEK293T cells and HL-7702 cells largely reduces more than 30% at the same conditions. This remarkable difference is ascribed to distinctly different morphophysiological properties between tumor cell lines and normal cell lines. Compared to normal cells, tumor cells have a faster cell cycle expressed as an increased rate of growth, which is favorable for enhanced resistance to cytotoxicity of the SiNWs. 22,23 In addition, membranes of tumor cells are usually more rigid than those of normal cells, which may also increase their survival rates with respect to the SiNWs. It is also worth noting that the most severe cytotoxicity is observed for HEK293T cells in comparison to other 3 kinds of cells. Nearly all HEK293T cells are dead when they are cultured with SiNWs at concentration of 150 μg/mL and incubation time of 3 days. It is in sharp contrast to high metabolic activities (>80%) of Hela, HepG2, and HL-7702 cells under the same conditions. One possible reason lies in their relatively poor adhesive ability, leading to large surface contact with SiNWs. 24,25 Besides, as an experimentally transformed cell line, HEK293T cells are sensitive to interference of foreign species. Thus, growth inhibition of HEK293T cells is more easily induced by the SiNWs. 26
Based on the abovementioned results, we further determine the concentration limits of SiNWs as the cytocompatible thresholds. Particularly, SiNWs of high concentrations (100-150 μg/mL) could be safely used for tumor cells but would cause severe cell death when labeling normal cells. Instead, relatively low concentration of SiNWs (≤50 μg/mL) is recommended for use in cell-relevant studies.
Conclusion
In conclusion, our present study showed that the cytotoxicity of SiNWs is greatly dependent on cell lines, surface SiNWs concentrations, and incubation conditions. Consistently, tumor cell lines are more biocompatible with SiNWs than normal cell lines. Moreover, we finally obtain the threshold concentrations of cytocompatibility SiNWs, which are valuable for the design criteria for the use of SiNWs in various bioapplications. Therefore, our study provides a potential SiNWs applications in various biological fields.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
