Abstract
CaCO3 hydrogel incorporation into Plaster of Paris (POP) formulations decreased the resorption rate of the POP after implantation in the body. Although an inflammatory process is required as part of wound healing, the accumulation and activation of inflammatory cells in the POP–hydrogel CaCO3 implant area needs to be controlled. Therefore, cinnamaldehyde, as an anti-inflammatory agent with a unique α, β-unsaturated aldehyde, was incorporated into the CaCO3 hydrogel. During the incorporation, both the lipophilic and hydrophilic sides of the cinnamaldehyde molecule can influence the physical and mechanical properties of the CaCO3 hydrogel, in which mechanical properties of a tissue engineering scaffold are important to fine tune cellular activity during implantation. On the other hand, as a 3-dimensional polymeric structure, crosslinking is needed for the CaCO3 hydrogel to stabilize and increase its molecular weight for better mechanical strength, and more resistance to heat, wear, and solvent attack. For that purpose, dehydrothermal treatment (DHT) was applied to the crosslink hydrogel system as a favorable crosslinking method to avoid the use of a chemical agent. In this study, 3 groups of hydrogels of CaCO3, namely DHT crosslinked, loaded with cinnamaldehyde, and loaded with cinnamaldehyde followed by DHT crosslinking were developed before being combined with POP in 50 wt%. To evaluate the effect of DHT to the final POP-cinnamaldehyde-loaded CaCO3 hydrogel properties and biocompatibility, scanning electron microscopy, contact angle, surface roughness, hardness, diametral tensile strength, and in vivo biocompatibility studies were conducted. It was observed that cinnamaldehyde with DHT treatment improved the POP–hydrogel CaCO3 properties and had good biocompatibility. Thus, POP-cinnamaldehyde-loaded CaCO3 hydrogel can be a promising bone substitute containing an anti-inflammatory agent.
In the treatment of bone defects, hydroxyapatite, calcium sulfate (CS), coralline, and calcium carbonate have been successfully used. 1,2 Although CS and Plaster of Paris (POP) show a positive effect in bone regeneration, 3 they have a too fast resorption rate and are unable to provide a 3-dimensional (3D) framework for the osteogenesis process. 4 -6 To solve this problem, Dewi and coworkers have incorporated calcium carbonate hydrogel into CS formulations. 7
It must be noted that bone implant surgery may increase infection risk after the wound exposure procedure. In this case, soft tissues usually show a more severe inflammatory response than bone 8,9 as the result of the accumulation and activation of inflammatory cells to the implant area. The human body responds to all implantation procedures by a wound-healing process. An inflammatory process that initiates a series of wound healing inevitably occurs in response to an external wound or lesion. 10 Although this process is required as part of wound healing, the discomfort that occurs in the early stage of inflammation can negatively affect the patient’s general condition. The signs of inflammation can be characterized by the appearance of rubor, calor, dolor, tumor, and functio laesa. 11 Postoperative pain requires treatment that not only can make the patient comfortable, but also can improve tissue repair of the surgical wounds. 12 Doctors usually prescribe oral anti-inflammatory drugs, especially non-steroid anti-inflammatory drugs), to reduce the pain that occurs during the inflammatory phase, in addition to antibiotics. 13
Therefore, an anti-inflammatory substance is needed. As the main component in cassia oil, as well as cinnamon bark oil, cinnamaldehyde (CA) or cinnamic aldehyde (3-phenyl-2-propenal) is known to be an anti-inflammatory 14,15 and antimicrobial agent active against a number of Gram-positive and Gram-negative bacteria. 16,17 The compound is categorized as a GRAS by the Flavoring Extract Manufacturers’ Association for Food Use based on 21 Code of Federal Regulation part 172.515. 18 CA suppresses lipopolysacharide (LPS)-stimulated macrophages to produce nitric oxide. 15,19,20 It is also reported by Chao and coworkers 21 that the essential oil of Cinnamomum osmophloeum Kaneh leaves at a dose of 60 µg/mL exhibited an effective inhibitory effect on interleukin 1β (IL-1β) and IL-6 production in LPS-stimulated macrophages.
With its interesting applicative prospects, CA has a lipophilic character from the hydrocarbon skeleton and a hydrophilic character from its functional groups. 21 Since the compound has both lipophilic and hydrophilic sides, it can affect the mechanical properties of a composite when they were mixed together, as shown in the previous studies when CA was loaded into a calcium carbonate hydrogel 7 before combined with CS. 4 CA is a cyclic terpene alcohol with a low solubility in water. 22 It has a similar effect as glutaraldehyde (GA) as a crosslink agent, since it has an aromatic α, β-unsaturated aldehyde. 23 Babu and co-workers 23 succeeded in the use of CA to crosslink chitosan nanoparticles to enhance the delivery of baicalin. To reduce the crosslink effect capability of CA, we used dehydrothermal (DHT) treatment to generate the formation of intermolecular crosslinks through a condensation reaction in a functional group of gelatin.
A hydrogel is a 3D network composed of a polymer backbone, water, and a crosslinking agent. Two fundamental methods of crosslinking for gelatin are physical treatments such as heat (DHT), ultraviolet radiation, gamma radiation, 24 and chemical such as aldehyde (ie, formaldehyde, GA, or glyceraldehyde), poly-epoxides, and isocyanates. 25 So far, GA is the most widely used crosslinking molecule due to its low cost and effectiveness for collagen-based material. However, GA is cytotoxic. 26 Therefore, DHT treatment was used to improve the properties of collagen because this does not involve the use of any chemical reagent. 27
As a newly developed biomedical device, CA loaded in calcium carbonate hydrogel must be biocompatible. Thus, the overall objective of the current research was to study the effect of DHT treatment to a POP–CaCO3 hydrogel scaffold with CA loaded inside as an anti-inflammatory agent on its properties (surface topography, contact angle, surface roughness, hardness, mechanical strength) and biocompatibility in vivo. The study was conducted with 3 different CaCO3 hydrogels: CaCO3 prepared with the DHT crosslinked (HD), loaded with CA (HC), and loaded with CA followed by DHT crosslinked (HDC).
Results and Discussions
Scanning Electron Microscopy Analysis
The images shown in Figure 1 show different pore sizes among the CaCO3 hydrogels when crosslinked with DHT, CA, and GA. The surface topography of the CaCO3 hydrogel with CA showed a smaller pore size than the others (Figure 1(C)). Since CA has an aldehyde group, it can act as a crosslink like GA. Scanning electron microscopy (SEM) showed that by adding CA to the gelatin polymer, the material becomes denser (Figure 1(C)) and has a pore pattern like when it is crosslinked with either DHT (Figure 1(B)) or GA (Figure 1(D)). When CA is crosslinked with either DHT (Figure 1(E)) or GA (Figure 1(F)), the porosity becomes denser and smaller in size. The pore size and porosity of the scaffold are important to determine the cells to grow into the scaffold and for extracellular matrix formation. 28 The crosslink methods will be an important point to consider in the preparation of a scaffold that functions for guided tissue regeneration with the ability to act as a barrier composite with a certain architectural designed pore size.

Scanning electron microscopy of hydrogel CaCO3 with 50× magnification. It describes (A) without any crosslinker, (B) by DHT, (C) by adding cinnamaldehyde, (D) by GA, and (E) by a combination of cinnamaldehyde and DHT as well as (F) a combination of cinnamaldehyde and GA. DHT, dehydrothermal treatment; GA, glutaraldehyde.
Contact Angle
The contact angle of the specimens was analyzed at 0, 1, and 2 seconds after they were dropped into 6 µL phosphate-buffered saline (PBS). The PBS decreased with time until it was all resorbed into the specimen. The result of the study (Table 1 and Figure 2) showed that POP is very hydrophilic since it has a contact angle of 0° at time 0 after the specimen was dropped into the PBS. The hydrogel system is known to be hydrophilic when it has a contact angle <90o [21]. On the contrary, POP-HD had a contact angle of 101.02° ± 2.48°. CA addition to the CaCO3 hydrogel decreased the contact angle of POP-HD from 101.02° ± 2.48° to 30.92° ± 6.24°, which makes it less hydrophilic. Based on Figure 3, at 2 seconds, POP-HDC had a contact angle of 1.40 ± 3.12, which indicates that it has higher hydrophilicity than POP-HD (43.58 ± 11.64) and POP-HC (6.73 ± 3.92).
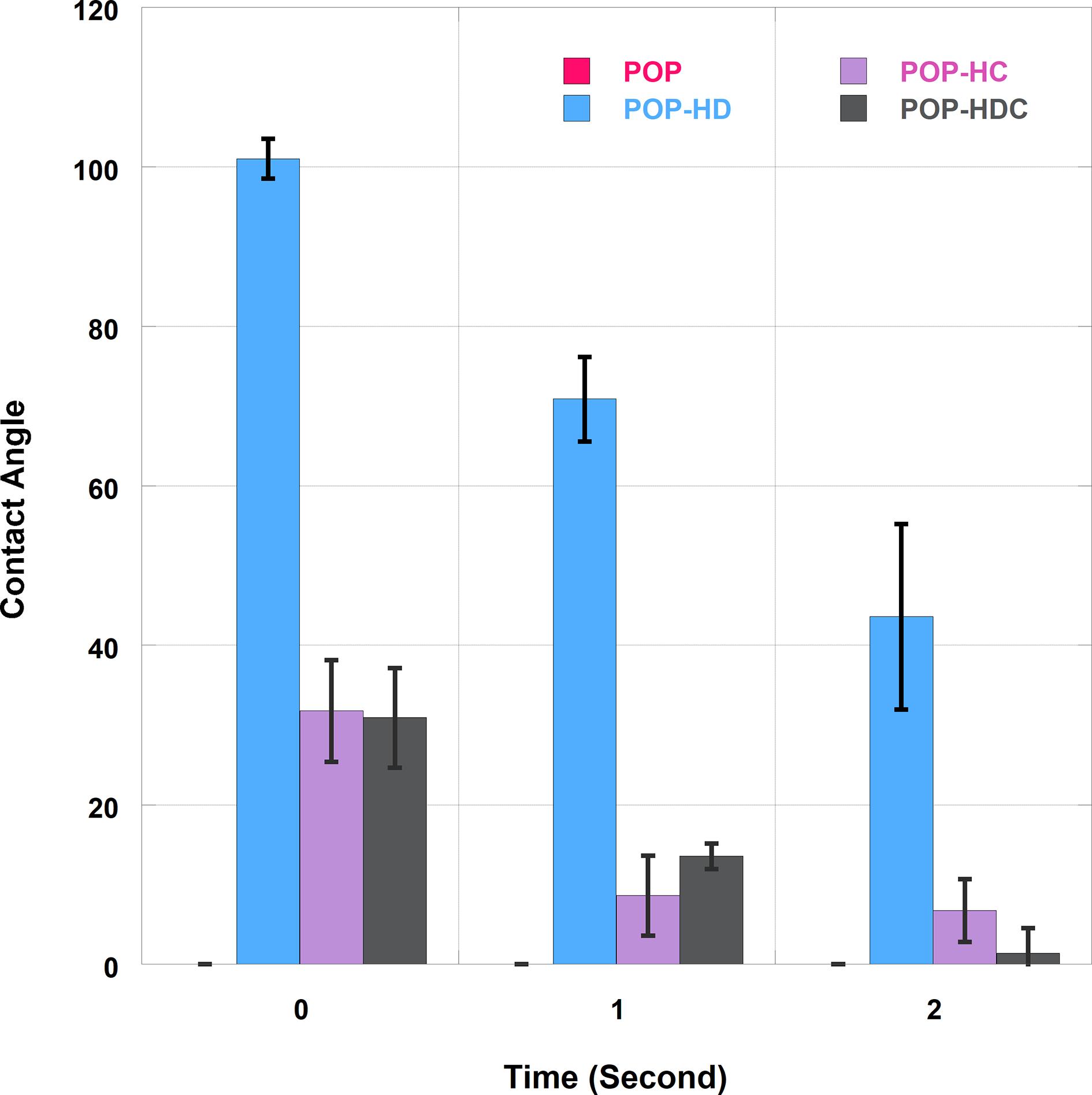
Contact angle of the specimens of POP-HD, POP-HC, and POP-HDC. For the POP, the contact angle was 0° for the whole duration. DHT, dehydrothermal treatment; HC, hydrogel loaded with cinnamaldehyde; HD, hydrogel crosslinked with DHT; HDC, hydrogel loaded with cinnamaldehyde and crosslinked with DHT; POP, Plaster of Paris.

The result of subcutaneous implantation of POP, POP-HD, POP-HC, and POP-HDC at 4 × 13 magnification with hematoxylin and eosin staining. All specimens result in good tissue reaction. DHT, dehydrothermal treatment; HC, hydrogel loaded with cinnamaldehyde; HD, hydrogel crosslinked with DHT; HDC, hydrogel loaded with cinnamaldehyde and crosslinked with DHT; POP, Plaster of Paris.
Contact Angle of the Specimens.

Abbreviations: DHT, dehydrothermal treatment; HC, hydrogel loaded with cinnamaldehyde; HD, hydrogel crosslinked with DHT; HDC, hydrogel loaded with cinnamaldehyde and crosslinked with DHT; POP, Plaster of Paris.
Surface Roughness, Diametral Tensile Strength, and Hardness
Material surface roughness or topography is an important factor that influences the cellular adhesion process. Hallab and co-wokers 29 confirmed that cellular adhesion is associated with the biomaterial surface roughness and surface energy. Materials with lower surface energy (ie, polymers) show an increase in cellular adhesion strength associated with increased surface roughness. POP was shown to have the smoothest surface (0.9 ± 0.20), compared with POP-HD (4.37 ± 0.30) and POP-HC (4.36 ± 0.15). The surface roughness of the specimens crosslinked with the combination of DHT and CA (POP-HDC) decreased to 2.97 ± 0.25.
The diametral tensile strength of POP (13.16 ± 1.56 MPa) was the highest among those tested. POP-HD (6.57 ± 0.28 MPa) had a similar diametral tensile strength as POP-HC (6.68 ± 0.05 MPa). Table 2 shows that POP-HDC decreased until 5.28 ± 0.11 MPa. The phenomena corroborate our previous study 4 in which addition of gelatin hydrogel to POP (with GA crosslinking) decreased diametral tensile strength (DTS) by almost a half, from 13.57 ± 1.56 to 7.14 ± 0.78 MPa. This proves that flexibility of the hydrogel plays an important role in decreasing the mechanical strength of the composite. 4,7 Hoare et al 30 stated that due to the unique physical properties of a hydrogel, which is highly porous, it tends to have a low tensile strength. Furthermore, CA can act as a plasticizer, which reduces the intermolecular forces of the polymer chains. The combination of polyethylene-co-vinyl acetate with CA has improved flexibility and extensibility. 31
Surface Roughness and DTS (MPa) of the Specimens.

Abbreviations: DHT, dehydrothermal treatment; DTS, diametral tensile strength; HC, hydrogel loaded with cinnamaldehyde; HD, hydrogel crosslinked with DHT; HDC, hydrogel loaded with cinnamaldehyde and crosslinked with DHT; POP, Plaster of Paris; VHN, Vickers Hardness Number.
The results for hardness measurement are listed in Table 2. The hardness (in Vickers Hardness Number) of POP (27.43 ± 6.36) was the highest of those tested, followed by POP-HDC (19.07 ± 2.11), POP-HD (12.03 ± 2.51), and POP-HC (16.63 ± 0.93). It is interesting that the hardness numbers of POP-HD and POP-HC are almost the same as that of POP-HDC. Hardness is the resistance of a solid material against various permanent shape changes when a compressive force is applied. 32 Hardness measurement can be used to estimate the crosslinking density. 33 In our study, the higher hardness number found for POP-HDC compared with both POP-HD and POP-HC is a proof of the relationship between hardness and crosslink density. The combination effect of CA and DHT treatment can influence the hardness number of POP-HDC. In humans, the mean value of bone hardness ranges from 33.300 Vickers Hardness Number (VHN) (head) to 34.157 VHN (the proximal metaphysis), 35.246 VHN (the distal metaphysis), and 43.820 VHN (the diaphysis). 34
Gross Assessment in In Vivo Studies
None of the animals died during the study and, clinically, there were no cases of infection. Post-implantation histological observation at 14 days indicated the data as shown in Table 3. Subcutaneous implantation represents a common model for examination of biomaterial characteristics. 35 -37 After 2 weeks’ implantation, the tissue reaction showed a good response to all specimens. Figure 3 shows the histological pictures post subcutaneous implantation. Clinically, there was visible mild swelling, which reduced day by day in 1 week. The result of the histological grading after 2 weeks implantation is also depicted in Table 3. Inflammatory cells (Figure 4), like macrophages, fibroblasts, neutrophils, collagen fibers, and new blood vessels, were observed.

Giant inflammatory cells surrounding the specimens at 40 × 13 magnification with hematoxylin and eosin staining.
Observation of the Tissue Reaction Surrounding the Implant.

Abbreviations: DHT, dehydrothermal treatment; HC, hydrogel loaded with cinnamaldehyde; HD, hydrogel crosslinked with DHT; HDC, hydrogel loaded with cinnamaldehyde and crosslinked with DHT; POP, Plaster of Paris.
P < .05 (significance).
The reaction zone semiquantitatively is shown by varying degrees of thickness (score 3-4). The thickness of cell layers varies in number due to the presence of cells in the fibrous tissue matrix. 9 The reaction zone qualitatively showed that there was fibrous tissue, mature, nondense, resembling connective or fat tissue in the noninjured region (score 4) for all groups. In this zone, adipose tissue was frequently noticed around the implant. This may be a criterion of implant compatibility. 35
The interface qualitatively showed that there were scattered foci of macrophages and foreign body cells surrounding the implant for all groups (score 3). Macrophages were the dominant cell type at the implant surface, playing a major role in cellular responses and tissue reaction. The tissue in the interstitial space showed that there were several blood vessels, young fibroblasts invading the spaces, and visible macrophages for all groups (score 3). The tissue that grows into these porous implant materials supports connective tissue ingrowth. Wide, variable stages were depicted: areas that are only composed of inflammatory cells, inflammatory cells with some connective tissue, young immature connective tissue with only a few inflammatory cells, and mature connective tissue or fat tissue. 36
Implant degradation for POP showed at score 2, and the other groups between 2 and 3. This indicated that POP is more rapidly resorbed in vivo than the others. CaCO3 hydrogel with CA retarded implant degradation. These findings were the same as in the previous study. 7
Discussion
POP or gypsum is a self-setting, biocompatible, and osteoconductive biomaterial. Gypsum has been successfully used in the treatment of a variety of bone defects, but it has a poor resorption rate in vivo. 4 Successful bone regeneration depends on the resorption rate. In a previous study, Dewi and co-workers showed that incorporation of CaCO3 hydrogel with POP improved the properties of the POP composite by decreasing the degradation rate of POP. It was also shown that addition of CaCO3 hydrogel to POP increased bone formation, angiogenesis, and collagen density. 7
Hydrogels are crosslinked hydrophilic polymer structures that can imbibe large amount of water or biological fluids. 38 They are 3D networks of water-soluble polymers, 30 but have limitations related to their use in load-bearing applications, such as low tensile strength. High water content and large pore sizes often result in relatively rapid degradation over a few hours to a few days. 30
In this study we used type B gelatin (Nitta Gelatin, Osaka, Japan) to combine POP with a hydrogel material. Gelatin, as a protein, has a variety of functional groups such as –NH2, -OH, and –COOH. 24 Gelatin is produced by a denaturation process from triple helix collagen to a single strain molecule. 24 However, the hydrogel has some drawbacks in terms of its mechanical properties in the physiological condition, although they have physicochemical similarity to the native extracellular matrix. 24 To improve their mechanical properties, several crosslinking methods can be used. The carboxyl (-COOH) and amine groups (-NH2) of the gelatin are functional groups that are targets of a crosslinking agent to create a peptide bond (Figure 5). The bonding will change the gelatin networks to be interconnected with each other. 24 It is also known that the crosslinking process defines several biochemical and structure modifications such as decreasing antigenicity, increasing mechanical properties, reducing solubility, and reducing the rate of biodegradability. 24,38,39

Schematic reaction in (A) hydrogel loaded with cinnamaldehyde, (B) hydrogel loaded with cinnamaldehyde and crosslinked with DHT, and (C) hydrogel crosslinked with DHT. DHT, dehydrothermal treatment.
Crosslinking methods can be divided into 2 main groups: chemical, for example by GA or carbodiimides, and biophysical, which includes the use of ultraviolet light and DHT crosslinking. The DHT process is a common technique for stabilizing collagen and collagen composite materials. It is a physical treatment that involves subjecting collagen to increased temperature (>98 °C) while under vacuum. 40 This removes water from the collagen molecules, resulting in the formation of intermolecular crosslinks through condensation reactions. An advantage of DHT crosslinking is that it does not involve the use of cytotoxic reagents.
Crosslinking of collagenous samples with GA involves the reaction of free amino groups of lysine or hydroxylysine residues of polypeptide chains with the aldehyde groups of GA. 41 GA is the most widely used crosslinking molecule due to its low cost and excellent efficiency for the stabilization of collagenous materials, 42,43 but GA is cytotoxic. 40
Eldin et al 44 succeeded in combining chitosan with trans-CA by Schiff base reaction. The presence of amine groups in the polymeric chain leads to the preparation of Schiff bases (-RC = N-) by reaction with aldehyde and ketones. On the other hand, CA is also known to have antimicrobial properties. 17,44,45 The addition of CA at different concentrations (4%, 6%, 8%, and 10%) inside the composite resulted in a decrease in the swelling ratio and degradation rate. 45 Swelling behavior was influenced by crosslink density, while swelling capacity can be modified by chemical or physical crosslink methods. 46
The SEM results of hydrogel with CA showed a smaller pore size than the other ones without CA. Pore size and porosity of the scaffold are important to determine the cells that grow into the scaffold and extracellular matrix formation. 28 This study reports how CA influenced the surface topography of the CaCO3 hydrogel compared with those that used crosslinking agent such as GA and DHT.
Babu and co-workers 23 used cinnamaldehyde as a crosslinking agent to enhance the delivery of baicalein by chitosan nanoparticles. The α, β-unsaturated aldehyde can act in the same manner as GA that was used as a crosslinking agent for gelatin. 47 Although GA is known to be cytotoxic, it was used as a crosslinking agent in the making of a porous hydroxyapatite–gelatin composite scaffold for bone tissue engineering. 48
The POP-HD and POP-HC composites have almost similar surface roughness (4.37 ± 0.30 and 4.36 ± 0.15) compared with that of POP (0.19 ± 0.20). This indicates that gelatin hydrogel plays a role in increasing surface roughness. The combination of DHT and CA (POP-HDC) decreased the surface roughness of the composite to 2.97 ± 0.25. It can be assumed that the CA and DHT crosslinking caused the hydrogel molecule to be more tight than the others. In this case, the crosslinking process is proven to decrease the surface roughness of the composite.
The contact angle of the hydrogel with CA was 31.77° ± 6.40° (POP-HC) and 30.92° ± 6.24° (POP-HDC), while the contact angle of the composite without CA was 101.02° ± 2.48° (POP-HD). When the contact angle is less than 90°, it is categorized as a hydrophilic material. The polymer is hydrophilic since its macromolecular chains (amide and carboxyl groups) are hydrolyzed quickly in the presence of water. 49 This result also showed that the CaCO3 hydrogel addition to POP enhanced its water barrier properties. This can be shown by its contact angle, which increased from 0° ± 0° (POP) to 101.02° ± 2.48° (POP-HD). However, CA addition decreased the contact angle by almost one third (more hydrophilic). This is different from the study of Ojagh 50 in which he found that addition of cinnamon decreased the hydrophilicity of the chitosan films; this might be due to the loss of free amino and hydroxyl groups resulting in a decreased moisture content of the edible film. 51 This indicated that cinnamon acts as a crosslinking agent when combined with chitosan. In our study it was shown that the addition of hydrogel with CA into POP (POP-HC and POP-HDC) increased hydrophobicity, but not higher than 90° compared with the one without CA.
Bonding or adherence of 2 materials depends on wetting, which is determined by the hydrophilicity or polarity. Wetting depends on the energies or surface tension of the interfaces between 2 materials. Hydrophilicity of the material influences the adsorption of blood protein that will promote cellular attachment onto the material surface. 51 Hydrophilicity refers to the ability of the molecule to bond transciently with water through hydrogen bonding, while hydrophobic substances interact within themselves and with other substances through Van der Waals forces and have either low or no capacity to form hydrogen bonds. Wetting is often characterized by measuring the contact angle formed between a liquid drop and a solid surface based on interfacial interaction concepts. The conventional method to evaluate wettability and surface energy is by drop shape analysis. 52 -55
Our results showed that the combination of POP-HD and POP-HC had almost similar DTS (6.57 ± 0.28 and 6.68 ± 0.05 MPa) compared to POP (13.16 ± 1.56 MPa). This indicated that the gelatin hydrogel decreased DTS by increasing the composite flexibility. Meanwhile, the combination of DHT and CA (POP-HDC) decreased DTS to 5.28 ± 0.11 MPa. In view of this, it is proven that CA is a plasticizer, which enables reduction of the intermolecular force of the polymer chain.
Biocompatible medical devices are needed to solve the problem in cases of body tissue damage but, to some extent, body reaction is far from perfect. Complications, such as thrombosis, infection, excessive inflammation and fibrosis, and impaired function, are unfortunate for the patient. The application of biomaterials to humans often fails because they act as a foreign body. The tissues responds to them with an inflammation reaction, wound healing, and scar formation. 56
The results of this study showed that 14 days post implantation, the tissue reaction had a good response to all specimens. Capsule thickness rating showed that only POP had a variable thickness with the score ranging from 3 (5, 9 layers of fibroblasts) to 4 (1, 4 layers of fibroblasts). The other groups had thickness ratings with a score of 3. The capsule is an inflammatory and healing reaction. It depends on implant geometrical properties such as shape, size, and surface topography. Large granules cause a thick encapsulation and the small particles evoke a more localized inflammatory reaction. There is a correlation between the rate of capsulation and the rate of resorption. 9 The POP implants were degraded faster than their combination with hydrogel. 7 Among the groups, POP is the most hydrophilic, and so was degraded fastest. The surface roughness of the POP was the lowest, which means that it has a smooth surface area. We assume that the capsule thickness of the POP related to hydrophilicity, surface roughness, and degradation rate.
The semiqualitative reaction zone showed score 4 for all specimens. The capsule was fibrous, mature, not dense, resembling either connective tissue or fat tissue in the noninjured regions. Besides, adipose tissue was found around the implant. This indicated implant compatibility. 37 The interface zone for all specimens showed a score of 3. After 14 days of implantation, there was still remaining material in all groups (score 2-3). It is possibly the reason why scattered foci of macrophages and foreign body cells are present. The interstice zone had a score of 3 for all groups. The material was degraded (score 2-3). In conjunction with this, new blood vessels were developed to provide a nutrition for the wound-healing process. Young fibroblasts were found, besides several macrophages as a result of the degradation of the implant material.
A number of inflammatory cells (neutrophils, monocytes, macrophages, and lymphocytes) express biochemical mediators affecting fibroblastic and angiogenic responses. 10 Neutrophils invade the wound site to scavenge debris, exclude microorganism and damaged tissue, and initiate the release of pro-inflammatory cytokines such as tumor necrosis factor α (TNF-α) and IL-1β. 57,58 The macrophages continue the process of phagocytosis releasing platelet derived growth factor (PDGF) and transforming growth factor β. Monocytes or macrophages play a critical role in managing innate and adaptive immunity, including inflammatory molecules such as TNF-α and nitric oxide (NO). 59
The overall results of the histological observation indicated that although POP has less capsule layers, the tissues around the capsules, in the interface, and within the interstice zone show the same score for all groups. It can be concluded that POP, POP-HD, POP-HC and POP-HDC have almost similar biocompatibility.
Conclusion
The SEM results of the hydrogel with CA showed smaller pore size than the one without CA. This indicates that cinnamaldehyde can act as a crosslinking agent besides GA or DHT methods. It was also found that gelatin hydrogel plays a role in increasing surface roughness. Also, the combination of DHT and CA (POP-HDC) decreases surface roughness of the composites due to crosslinking that tightens the molecules of the hydrogel. Furthermore, it was proven from this study that the crosslinking process decreased surface roughness, thus smoothing the surface of the composites. It was also shown that gelatin hydrogel decreased DTS by increasing composite flexibility. The combination of DHT and CA (POP-HDC) showed less decreased DTS because CA can act as a plasticizer, which reduces the intermolecular force of the polymer chain. We also found that combining POP to hydrogel with CA (POP-HC and POP-HDC) increased hydrophobicity, but not higher than 90° compared with the one without CA. In term of biocompatibility, POP has less capsule layers, but the tissue around the capsules, in the interface and within the interstice zone, showed the same score as the other groups. This can be an indicator that POP, POP-HD, POP-HC, and POP-HDC have almost similar biocompatibility, thus showing the biocompatibility of CA for the development of biomedical composites.
Experimental
Specimen Preparation
Gypsum or POP and calcium carbonate (CaCO3) were obtained from Wako Pure Chemical Industries Ltd. (Osaka, Japan). Type B gelatin was provided by Nitta Gelatin Inc., Osaka, Japan and CA by Merck, Germany. All other chemicals were of pure analytical grade. The CaCO3 hydrogel with 4% CA was made by mixing 10 mL Tween 80 solution (Sigma Aldrich, Germany) with 4 mL CA (stirred for 30 minutes), and then 40 mL H2O was added to 2.5 g CaCO3 (stirred for 1 hour). Subsequently, the hydrogel was prepared by mixing 5 g gelatin and 46 mL H2O until it was swollen and ready to mix with the CA–CaCO3 solution in a 37 °C waterbath; stirring was continued for 2 hours. Then, the pH was adjusted to 7.4, the solution placed in a refrigerator for 1 day at −20 °C, followed by freeze drying for 3 days. After that, the specimens were crosslinked by the DHT method in a vacuum oven for 72 hours, at 140 °C. CaCO3 hydrogel–CA beads were made by grinding the freeze-dried blocks. The beads were sieved through a 150-µm mesh and then added to CaSO4.1/2 H2O powder. The powders were used to prepare cylindrical specimens for the various assays using a water/powder ratio of 1/2. The cylinders were put in an incubator at 37 °C for 24 hours to allow them to set completely. The list of the specimens is given in Table 4.
Sample Distribution.

Abbreviations: DHT: Dehydrothermal crosslink; HC, hydrogel loaded with cinnamaldehyde; HD, hydrogel crosslinked with DHT; HDC, hydrogel loaded with cinnamaldehyde and crosslinked with DHT.
Surface Topography by SEM
The surface microstructure of the dried specimens was observed by SEM using a JEOL-JSM-T300 (Tokyo, Japan) at 20 kV linked to an energy-dispersive X-ray spectrometer. The specimens were dried and sputter-coated with a gold layer prior to measurement. The accelerating voltage was 20 kV, the specimen temperature was approximately 20 °C, and the column vacuum was 7 × 10–4 Pa. Magnification from 500 up to 3500× was used to visualize the surface morphology.
Surface Roughness
The surface roughness was investigated using surface roughness tester Starret SR 300, series 014036, Japan. Samples 1 mm2 were set on a range of roughness at 200 µm (n = 3).
Mechanical DTS Testing
DTS of the specimens was studied using a universal testing machine (Zwick/z 0.5, Ulm Germany) at a constant crosshead speed of 1 mm/min. Specimens with a diameter of 6 mm and a thickness of 3 mm were used in the assay (n = 3). The DTS was recorded in MPa.
Hardness Measurements
The dried samples were prepared for hardness measurement and the readouts were taken at 3 different positions on each specimen. Measurements were made on specimens of approximately 1 mm thickness. The Vickers Microhardness Tester (Shimadzu, Japan) generates small indentations at 100 × g force. The dimensions of the indentations, in both horizontal and vertical diameters, would then be measured to generate a hardness value or VHN.
Contact Angle
The contact angles of the samples were measured by the sessile drop method. A small drop of PBS was deposited on the composite disk. Interaction between liquid–composite disk surfaces was captured by a customized home-made device connected with a digital camera. The drop profile images were edited and optimized with Image-J analysis software. The contact angle is defined as the angle between the surface of the specimen and tangent line at the point of contact of the PBS droplet with the surface. 42,53 Figure 6 shows a schematic diagram of the contact angle measurement as referred to in the previous study. 60

Sessile drop technique to measure contact angle as referred to Yulianto and Rinastiti. 60
In Vivo Experimental
Surgical procedures
The protocol for the research involving the animal subjects for this study was approved by Health and Medical Research Ethics of the Faculty of Dentistry UGM No. 001465/KKEP/FKG-UGM/ 2018 with annual renewal until the end of the study. Animal experiments were carried out using healthy, adult male Wistar rats (The Human Society of the United States, 2006). Animals were individually housed in rooms at 22 °C with 12 hours light/dark cycles and handled regularly for 7 days to become acclimatized to their new housing and diet in the Integrated Research and Testing Laboratory (LPPT), Universitas Gadjah Mada. Ten Wistar rats, age 120 days, weight 250‐300 g, were used for the experiment (n = 5 in each group).
The animals were treated under general anesthesia by intramuscular injection of ketamine (11, 22 mg/kg body weight) in combination with xylasin (0.55, 1.1 mg/kg body weight). The surgical area was shaved and scrubbed with sterile gauze soaked with betadine.
Subcutaneous implantation
Four cylindrical implants per animal were placed into a subcutaneous pouch through a 1-cm-long dorsal midline incision on both sides of the vertebral column. The skin was closed with resorbable 2.0 vicryl suture material (Ethicon, Johnson and Johnson Indonesia, Jakarta, Indonesia). Immediately following implantation, to reduce the risk of perioperative infection, each rat was injected with antibiotics, Interflox- 100 (Interchemix, Horsterweg 26, Maastricht, the Netherlands) at 10 mL/20‐40 kg intramuscularly during 5 days after surgery and analgesic intraoral. All animals were fed mouse pellets, and water was given ad libitum.
Two weeks after that, the rats were sacrificed by an overdose of ketamine and xylazine. Thereafter, each implant and its surrounding tissue were excised immediately for further histological processing. The sections were removed and immersed in 10% phosphate-buffered formalin for 24 hours. Processing of tissue included dehydration in graded ethanol from 50% to 100%, infiltration with paraffin, and routine histological processing and staining with hematoxylin–eosin and Mallory’s trichrome.
Histological observations
A semiquantitative and qualitative histological analysis of the retrieved specimens was carried out using a light microscope (Nikon Eclipse E-600) with Optilab, Micronos, Indonesia. The histological grading scale evaluations were performed (Table 5) to investigate the soft tissue response to the implants, and the images were projected with a 520× magnification.
Histological Grading Scale for Soft-Tissue Implant (Referred to Bodde et al., 2007).

Footnotes
Acknowledgments
Authors thank the Ministry of Research, Technology, and Higher Education of the Republic of Indonesia for supporting the study. The authors have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study received funding from Ministry of Research, Technology, and Higher Education of the Republic of Indonesia.
