Abstract
Background
Epithelial-mesenchymal transition (EMT) is crucial in liver cancer progression. Developing novel medicines to disrupt the progression of EMT might be a novel approach in managing liver cancer.
Objectives
This study investigates the protective role of Astragalus polysaccharide (APS), a bioactive compound derived from Astragalus membranaceus, in modulating EMT in HepG2 liver cancer cells.
Materials and Methods
The effects of APS on EMT progression were evaluated by assessing cell viability, proliferation, invasion, migration, cell cycle, and apoptosis in HepG2 cells. Furthermore, the involvement of the NOD-like receptor protein 3 (NLRP3) inflammasome in this process was explored.
Results
APS treatment significantly inhibited the viability, proliferation, invasion, and migration of HepG2 cells while promoting cell cycle arrest and apoptosis. Notably, APS reduced the expression of NLRP3 inflammasome components, including NLRP3, apoptosis-associated speck-like protein, and caspase-1, while decreasing the levels of inflammatory cytokines. In addition, the activator Nigericin dampened the effect of APS by partially rescuing the activity of NLRP3. Specifically, Nigericin promoted the development of EMT inhibited by APS and exacerbated the malignant transformation of HepG2 cells improved by APS.
Conclusion
Our findings demonstrated the function of APS in liver cancer and the process by which APS inhibits NLRP3 inflammasome activation to protect the EMT of HepG2 cells. Our results showed that by targeting NLRP3, the application of APS appears to be a viable tactic for preventing the progression of liver cancer.
Introduction
Among the five most lethal tumors, liver cancer is the only one whose incidence rate is rising annually with prevalent malignancies. It has been determined that liver cancer, particularly in China, accounts for half of the worldwide burden (Song et al., 2022). The early manifestations of liver cancer are not typical, and most patients are diagnosed in the late stage, which poses a serious threat to their poor prognosis and quality of life (Anwanwan et al., 2020). Therefore, investigating efficient targets and treatment modalities for liver cancer is critical. Epithelial-mesenchymal transition (EMT) is a fundamental event in tumor metastasis, which is typified by alterations in the cellular architecture of epithelial cells, a decrease in polarized and specialized cell–cell interaction, and the presentation of mesenchymal-like cell shape and functional traits. During EMT, epithelial cells undergo profound changes in their structure and function, which enable them to acquire more aggressive behaviors, such as increased motility and invasiveness, allowing cancer cells to spread to distant organs and tissues (Dongre & Weinberg, 2019). EMT is vital in the initiation and metastasis of liver cancer (Sharma & Kanneganti 2021), and how to prevent EMT in liver cancer cells is vital in cancer management.
The activation of a polymeric cytoplasmic protein complex known as the NOD-like receptor protein 3 (NLRP3) inflammasome causes inflammation response mediated by caspase 1/interleukin-1 (IL-1), which contributes to inflammatory disorders and tumor malignancies. NLRP3 inflammasome activation is regarded to have controversial roles, and its impact on tumor progression remains explored. NLRP3 activation mitigates the formation of tumors, but there is also evidence that it may protect against some types of cancer (Tezcan et al., 2020). For instance, activating NLRP3 inflammasome inhibits hepatocellular carcinoma (HCC) proliferation, induces cell cycle arrest, and suppresses migration, suggesting its potential as a therapeutic target (Dai et al., 2023). However, it is also suggested that NLRP3 inflammasome promotes HCC progression by activating inflammatory pathways and enhancing tumor-promoting cytokine release while it inhibits cancer development by inducing pyroptosis, which enhances the susceptibility of HCC cells to immune-mediated killing (Tang et al., 2023). Therefore, further investigation of the role of NLRP3 in HCC is needed.
Astragalus polysaccharide (APS), also known as Astragalus membrane pod or Astragalus Mongolia, is a water-soluble heteropolysaccharide that is isolated, concentrated, and refined from the dried root of the legume plant. When it comes to Astragalus medication, it is the most prevalent and immunoactive ingredient (Zhang et al., 2024). Astragalus has immunomodulation, anti-inflammation, anti-tumor, anti-oxidation, and other effects in traditional Chinese medical theory (Fu et al., 2014). In particular, its potential to combat tumors has recently drawn a lot of attention from researchers (Li et al., 2020). So far, the effects of astragalus in treating tumors have been confirmed in various carcinoma, including gastric cancer (Chen et al., 2022), ovarian cancer (Guo et al., 2020), and breast cancer (Li et al., 2019). However, the specific effects of APS on liver cancer require further explorations. APS has been shown in studies to dramatically increase the death of liver cancer cells and to protect against liver damage (He et al., 2024; Yang et al., 2024). However, its potential mechanism still requires clarification. Remarkably, research has shown that APS can reduce protracted inflammatory reactions by inhibiting NLRP3 inflammasome activation (Xu et al., 2021). Furthermore, APS has been suggested to block NLRP3 inflammasome activation, thereby reducing inflammation (Tian et al., 2017). These facts prompt that NLRP3 inflammasome suppression may be a factor in the way that APS affects liver cancer.
Against this background, it is clear that NLRP3 inflammasome is a key element leading to EMT in cancer cells. Given APS’s regulatory impact on NLRP3, this study aimed to explore its effects on NLRP3 inflammasomes in liver cancer cells and to examine whether APS can improve liver cancer outcomes by inhibiting the EMT process through NLRP3 modulation.
Materials and methods
Cell Culture
In this study, HepG2 cells (Procell, China), a human liver cancer cell line, were used to investigate the effects of APS. Cells were cultured in minimum essential medium (MEM) (Pricella, China) with 20% fetal bovine serum (Excell Bio, China), penicillin (100 units/mL), and streptomycin (50 units/mL) at 37°C in a 5% CO2 atmosphere. Cells were digested with 0.25% trypsin (Gibco, China), and after detachment, 5 mL of complete culture medium was added to terminate digestion. In the CON group, HepG2 cells were treated with the vehicle without any interference. For the APS group, HepG2 cells were treated with 400 mg/L APSs (Jonln, China) for 48 h. For the APS + Nigericin group, cells were pretreated with 10 nM Nigericin (MedChemExpress, USA) for 1 h before being treated with 400 mg/L APS for 48 h.
Cell Counting Kit-8 Assay
To perform the cell counting kit-8 (CCK-8) assay, cells were seeded into 96-well plates at a density of 2 × 103 cells per well, followed by a 2-h incubation with CCK-8 solution. Absorbance at 450 nm was recorded using an RT-6000 microplate reader (Rayto, China). The proliferation rate was determined as: cell survival rate (%) = [(OD of sample − OD of PBS)/(OD of control − OD of PBS)] × 100.
Colony Formation Experiment
Cells were seeded in six-well plates at 1,000 cells per well and cultured for 14 days. Colonies were fixed with 4% paraformaldehyde (Beyotime, China) for 20 min and stained with 0.2% crystal violet (Sigma–Aldrich, USA) for 5 min. After washing and air drying, images of the colonies were captured. The colony formation rate was calculated as: clone formation rate (%) = (number of colonies/number of seeded cells) × 100.
Transwell Assay
HepG2 cells (1–5 × 10⁵ cells/mL) were seeded into transwell chambers coated with matrix gel. The lower chamber was filled with 600 µL of culture medium containing 10% FBS. After phosphate-buffered saline (PBS) washing, cells were fixed with 4% paraformaldehyde for 10 min. The upper chamber was removed, and the fixation solution in the lower chamber was discarded. Crystal violet staining solution (Sigma–Aldrich, USA) was applied. The chamber was immersed in the staining solution for 10 min, rinsed thoroughly with water, and allowed to dry. Stained cells were observed and imaged under a microscope.
Wound Healing Assay
HepG2 cells were seeded in six-well plates at 1 × 106 cells per well and incubated for 4 h. Once adherent, cells were treated with 0, 50, 100, 200, or 400 mg/L of APS, or with 400 mg/L APS + Nigericin. When cell confluence exceeded 90%, scratches were made using a pipette tip along the marked lines on the plate. The scratch area was recorded under a microscope at 0 h and again at 24, 48, and 72 h.
The Detection of Cell Apoptosis
Apoptosis in HepG2 cells was measured using an Annexin V-FITC kit (Beyotime, China). Cells were digested, centrifuged at 1,000 rpm for 5 min, and resuspended in 1X Annexin V binding solution (1 × 106 cells/mL). A 100 µL suspension was mixed with 5 µL Annexin V-FITC and 5 µL PI solution, incubated at room temperature in the dark for 15 min, and diluted with 400 µL Annexin V binding solution. Fluorescence was detected within 1 h using an Attune NxT flow cytometer (Invitrogen, USA), and data were analyzed with FlowJo software.
The Detection of Cell Cycle
The cells were digested with 0.1% trypsin, washed three times with 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) containing 0.2% Bovine serum albumin (BSA), and adjusted to 2 × 106 cells/mL. Propidium iodide (0.5 mL) was added for staining, followed by incubation in the dark for 30 min. After centrifugation and washing, the suspension was readjusted to 2 × 106 cells/mL. Red fluorescence (488 nm) and light scattering were analyzed using flow cytometry, and the results were processed using the FlowJo software.
Western Blot
Total protein was extracted using radioimmunoprecipitation assay (RIPA) lysis buffer (Beyotime, China) and quantified with a BCA kit (NCM Biotech, China). Proteins were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinylidene fluoride (PVDF) membranes (Millipore, USA). Membranes were blocked with 5% skim milk (BD, USA) and incubated overnight at 4°C with primary antibodies against target proteins (Abcam, UK; Proteintech, China; Thermo Fisher, USA). After incubation with HRP-conjugated secondary antibodies (Bioss, China) for 1 h at room temperature, signals were visualized using a chemiluminescence imaging system (Tanon, China) and analyzed with ImageJ software.
Enzyme-linked Immunosorbent Assay (ELISA)
Levels of inflammatory cytokines IL-1β and IL-18 were quantified using ELISA kits (Hybio, China). Supernatants were collected, and standard curves were generated following the manufacturer’s protocol. Cytokine levels were determined using an RT-6000 microplate reader (Rayto, China).
Real-time Polymerase Chain Reaction (PCR) Analysis
Total ribonucleic acid (RNA) was extracted using the Cell/Tissue Total RNA Isolation Kit V2 (Vazyme, China). RNA purity and concentration were assessed using Nano 600 (Jiapeng, China). cDNA was synthesized with the HiScript III 1st Strand cDNA Synthesis Kit (Vazyme, China). Real-time PCR was performed with the Taq Pro Universal SYBR qPCR Master Mix (Vazyme, China) on a CFX96 Touch system (Bio-Rad, USA). Primer sequences were: E-cadherin (F: TACCCTGGTGGTTCAAGCTG; R: CAAAATCCAAGCCCGTGGTG), N-cadherin (F: GGCGTTATGTGTGTATCTTCACT; R: GCAGGCTCACTGCTCTCATA), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (F: TCCAAAATCAAGTGGGGCGA; R: AAATGAGCCCCAGCCTTCTC). Relative messenger RNA (mRNA) levels were calculated using the 2–∆∆Ct method.
Immunofluorescence Staining
Cells were seeded in six-well plates at a density of 1 × 106 cells/well at 1 × 106 cells per well and treated as per experimental groups. After fixation with 4% paraformaldehyde (Beyotime, China) for 20 min and permeabilization with 0.25% Triton X-100 (Beyotime, China) for 20 min, cells were blocked with goat serum (Bioss, China) for 30 min. Primary antibodies against E-cadherin and N-cadherin (1:200, Abcam, UK) were applied, and the samples were incubated overnight at 4°C. Secondary antibodies (1:200, Abcam, UK) were added and incubated at room temperature in the dark for 1 h. Fluorescence images were captured using an EVOS M5000 fluorescence microscope (Thermo Fisher, USA), and colocalization and fluorescence intensity were analyzed with ImageJ software.
Statistical Analysis
Data are expressed as Mean ± Standard Error of the mean (SEM). Statistical analyses were conducted using GraphPad Prism 8 software. One-way analysis of variance (ANOVA) was used to compare multiple groups, and differences between two groups were assessed using the unpaired t-test. A p value of less than 0.05 was considered statistically significant.
Results
APS Inhibited the Malignant Transformation of HepG2 Liver Cancer Cell Phenotype
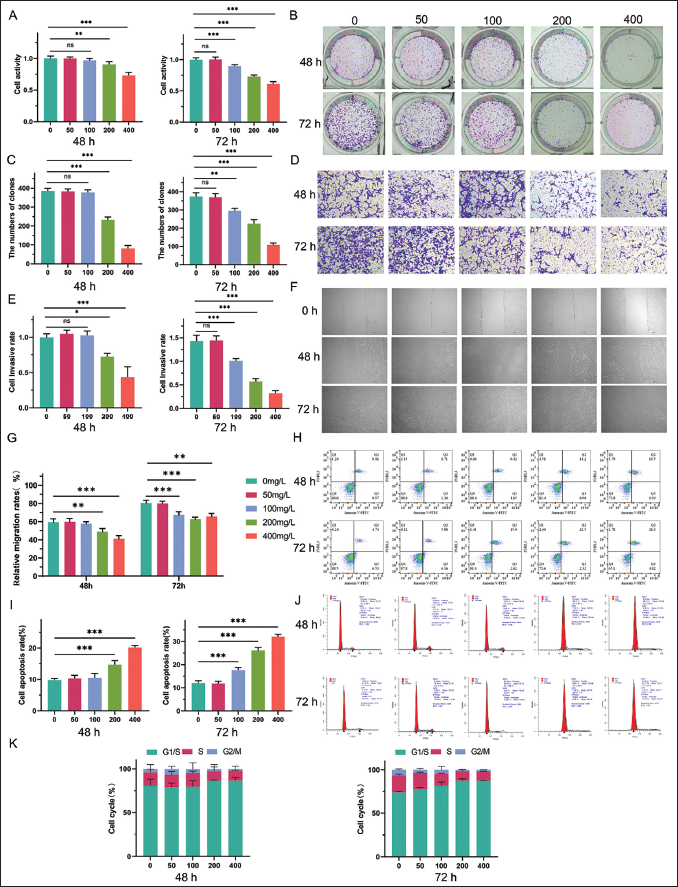
To assess the impact of APS on liver cancer cells, we treated HepG2 cells with APS at concentrations of 50, 100, 200, and 400 mg/L for 48 and 72 h, respectively, to evaluate its effects on liver cancer progression. First, to analyze the effect of APS on liver cancer cells, we selected APS at concentrations of 50, 100, 200, and 400 mg/L to treat HepG2 cells for 48 and 72 h, respectively, to evaluate its role in liver cancer. It was shown that the activity of HepG2 cells was significantly reduced following a 48-h APS therapy at 200 and 400 mg/L. For 72 h of administration, APS had a considerable impact on HepG2 cell viability at concentrations of 100, 200, and 400 mg/L (Figure 1A). Additionally, similar results were noted for the proliferation, invasion, and migration abilities of HepG2 cells (Figure 1B–G). Forty-eight hours after 200 mg/L APS and 72-h after 100 mg/L APS administrations, the apoptosis rate of HepG2 cells was notably upregulated (Figure 1H and I). Furthermore, the percentage of cells in the G1/S phase increased, whereas cells in the S phase reduced when incubating 200 mg/L APS for 48 h and 100 mg/L APS for 72 h. These observations suggest that APS treatment leads to a G1-phase arrest in the HepG2 cells, preventing progression into the S phase and thus inhibiting cell proliferation (Figure 1J and K).
The Effect of Astragalus Polysaccharides (APSs) on the Functional Phenotype of HepG2 Liver Cancer Cells. HepG2 Cells Were Divided Into 0, 50, 100, 200, and 400 mg/L of APS-treatment Groups. (A) Cell Counting Kit-8 (CCK8) Assay for the Changes in Cell Proliferation Ability After APS Treatment. (B) Colony Formation Experiment to Detect the Changes in Cell Growth. (C) The Quantitative Analysis of Colony Formation Ability. (D) Transwell Assay for the Changes in Cell Invasion Ability After APS Treatment. (E) The Quantitative Analysis of Cell Invasion Ability. Scale Bar, 50 µm. (F) Wound Healing Assay for the Migration Ability of Cells. Scale Bar, 50 µm. (G) The Quantitative Analysis of Cell Migration Ability. (H) Flow Cytometry to Detect the Apoptosis Rate of Cells. (I) The Quantitative Analysis of the Apoptosis Rate. (J) Flow Cytometry to Detect the Cell Cycle. (K) The Quantitative Analysis of Cell Cycle. The Above Experiments Tested the Results at 48 and 72 h, respectively. *p < 0.05, **p < 0.01, and ***p < 0.001 Between Groups.
APS Prevented the Activation of NLRP3 Inflammasomes in HepG2 Cells
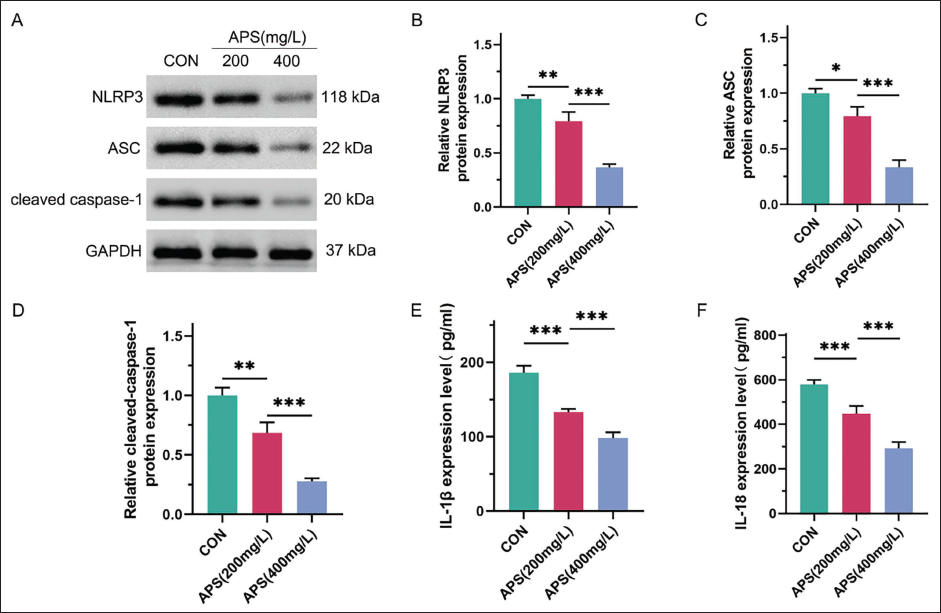
We then explored the function of APS on NLRP3 inflammasomes in HepG2 cells. We found that NLRP3 inflammasomes were activated in HepG2 cells, as indicated by markedly increasing levels of NLRP3, apoptosis-associated speck-like protein (ASC), and cleaved caspase-1, key components of the inflammasome. After treatment with APS at concentrations of 200 and 400 mg/L, the expression levels of various components of inflammasomes were dramatically downregulated (Figure 2A). Furthermore, the inhibitory impact was more pronounced in the APS treatment group at 400 mg/L (Figure 2B–D). We also examined the content of relevant inflammatory factors. ELISA experiments showed that APS at 200 and 400 mg/L notably reduced the elevated expressions of IL-1β and IL-18 (Figure 2E and F). Our data indicated that APS repressed the NLRP3 inflammasomes in cultured HepG2 cells.
The Effect of Astragalus Polysaccharides (APSs) on the Expression Level of NOD-like Receptor Protein 3 (NLRP3) Inflammasome in HepG2 Liver Cancer Cells. HepG2 Cells Were Divided into Control, 200, and 400 mg/L of APS-treatment Groups. (A) Western Blot Assay for the Protein Expression Levels of NLRP3, Apoptosis-associated Speck-like Protein (ASC), and Cleaved Caspase-1. Glyceraldehyde 3-phosphate Dehydrogenase (GAPDH) was Set as a Reference. (B–D) Quantitative Analysis of the Protein Expression Levels of NLRP3, ASC, and Cleaved Caspase-1. (E and F) Enzyme-linked Immunosorbent Assay (ELISA) Assay for the Content of Inflammatory Factors in Each Group of Cells, Including IL-1β and IL-18. *p < 0.05, **p < 0.01, and ***p < 0.001 Between Groups.
APS Arrested EMT of Liver Cancer Cells by Inhibiting NLRP3 Activation
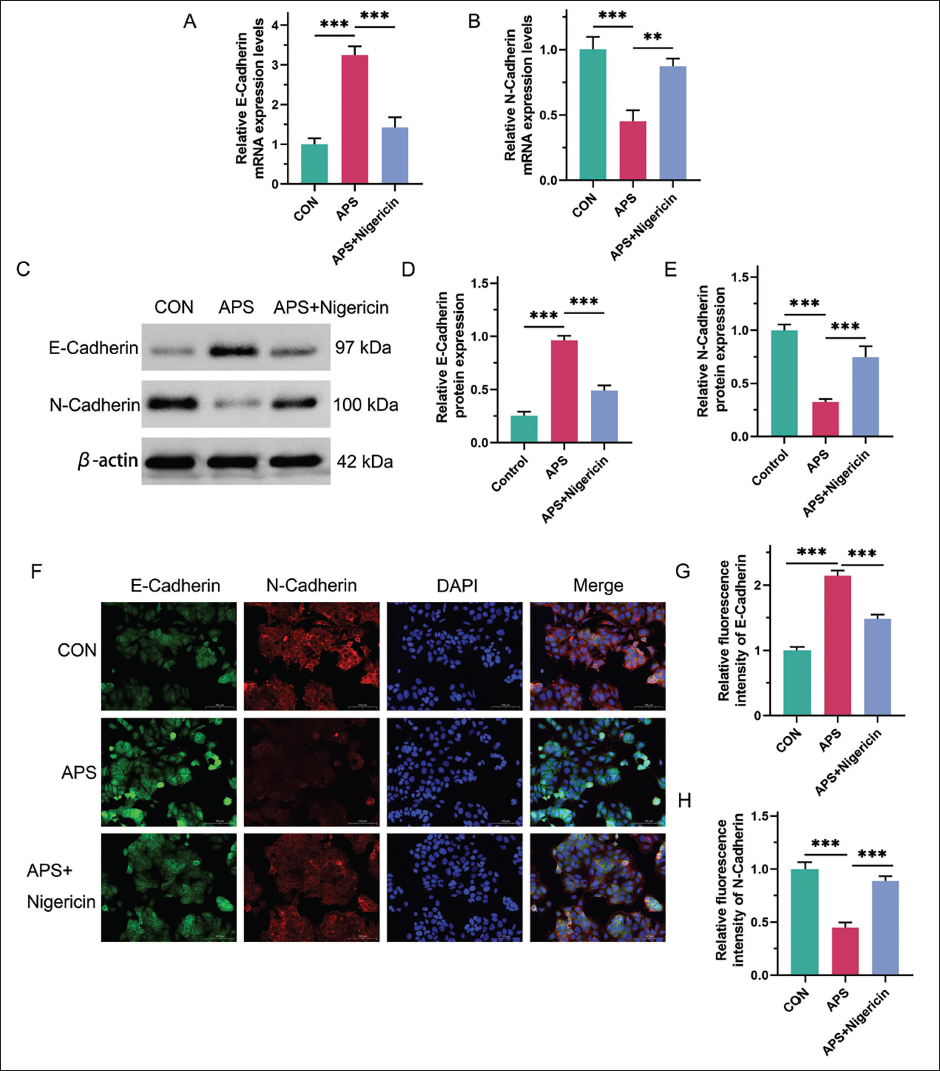
Building on the previous findings, we selected 400 mg/L APS for the subsequent experiments. Considering the pivotal role of NLRP3 inflammasomes in regulating EMT, we further explored the effect of APS on EMT in HCC. Our focus was on two key molecules involved in the EMT process, E-cadherin and N-cadherin, which are known markers of epithelial and mesenchymal states, respectively. We found that APS treatment significantly reduced N-cadherin expression, alongside with a notable increase in E-cadherin expression, suggesting that APS could inhibit the EMT process (Figure 3A and B). However, when cells were treated with the NLRP3 inhibitor Nigericin, E-cadherin transcription was downregulated, and N-cadherin transcription was upregulated, suggesting that NLRP3 activation reversed the effects of APS on the expression of these markers (Figure 3A and B). Also, we observed E-cadherin expression was inhibited, and N-cadherin expression was promoted after APS treatments (Figure 3C–E). Additionally, immunofluorescence staining revealed that Nigericin treatment diminished the enhanced fluorescence intensity of E-cadherin induced by APS, while it strengthened the fluorescence of N-cadherin, further supporting the reversal of APS effects by NLRP3 activation (Figure 3F–H). These results collectively suggest that APS effectively inhibits EMT in HepG2 cells, and this protective effect is reversed when NLRP3 inflammasomes are activated.
The Effect of Activating NLRP3 Inflammasome on Astragalus Polysaccharides (APSs) Preventing Epithelial Mesenchymal Transition in HepG2 Liver Cancer Cells. HepG2 Cells Were Divided into Control, 400 mg/L APS, 400 mg/L APS + NOD-like Receptor Protein 3 (NLRP3) Activator (10 nM Nigericin) Groups. (A and B) Real-time Polymerase Chain Reaction (PCR) for the messenger ribonucleic acid (mRNA) Expression Levels of E-cadherin and N-cadherin in Different Groups. (C) Western Blot Assay for the Protein Expression Levels of E-cadherin and N-cadherin. β-actin Was Set as a Reference. (D and E) Quantitative Analysis of the Protein Expression Levels of E-cadherin and N-cadherin. (F) Immunofluorescence Staining to Observe the Expression of E-cadherin and N-cadherin. Green Indicates E-cadherin Staining, Red Indicates N-cadherin Staining, Blue Indicates 4′,6-diamidino-2-phenylindole (DAPI) Staining of the Cell Nucleus, and Merge is a Synthetic Image. Scale Bar, 100 µm. (G and H) Quantitative Analysis of Fluorescence Intensity of E-cadherin and N-cadherin. **p < 0.01 and ***p < 0.001 Between Groups.
APS Alleviated Malignant Transformation of HepG2 Liver Cancer Cell Phenotype by Inhibiting NLRP3 Activation
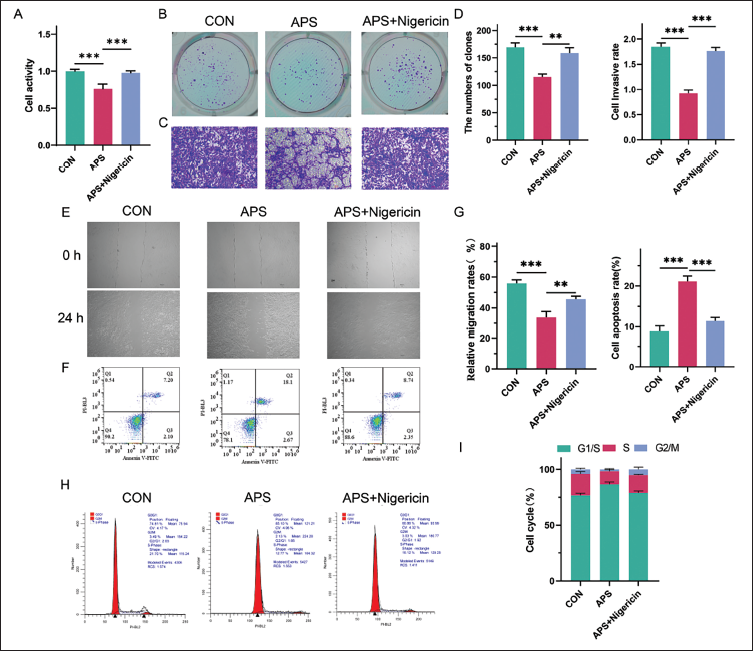
We then looked at how NLRP3 activation affected the function of HepG2 cells treated with APS to evaluate the role of NLRP3 inflammasome-mediated EMT in APS-induced inhibition of liver cancer. We found that Nigericin enhanced the vitality, proliferation, invasion, and migration ability of HepG2 cells treated with APS. Besides, treatment with Nigericin reduced the apoptosis that APS induced in HepG2 cells (Figure 4A–G). Additionally, Nigericin also improved cycle arrest in HepG2 cells given APS treatment (Figure 4H–I). Overall, these findings validated that APS ameliorated liver cancer via blocking NLRP3 inflammasomes and, subsequently, EMT.
The Effect of Activating NOD-like Receptor Protein 3 (NLRP3) on Astragalus Polysaccharides (APSs) Preventing Malignant Phenotypic Transformation of HepG2 Liver Cancer Cells. HepG2 Cells Were Divided into Control, 400 mg/L APS, 400 mg/L APS + NLRP3 Activator (10 nM Nigericin) Groups. (A) Cell Counting Kit-8 (CCK8) Assay for the Changes in Cell Proliferation Ability. (B) Colony Formation Experiment to Detect the Changes in Cell Growth. (C) Transwell Assay for the Changes in Cell Invasion Ability. Scale Bar, 50 µm. (D) The Quantitative Analysis of Colony Formation Ability and Cell Invasion Ability. (E) Wound Healing Assay for the Migration Ability of Cells at 24 h. Scale Bar, 50 µm. (F) Flow Cytometry to Detect the Apoptosis Rate of Cells. (G) The Quantitative Analysis of Cell Migration Ability and the Apoptosis Rate. (H) Flow Cytometry to Detect the Cell Cycle. (I) The Quantitative Analysis of Cell Cycle. **p < 0.01 and ***p < 0.001 Between Groups.
Discussion
HCC is one of the leading causes of cancer-related mortality worldwide. Despite advancements in surgical techniques, such as liver transplantation and resection, and therapies like sorafenib for advanced-stage disease, the prognosis for HCC patients remains unsatisfying. The complexity of HCC’s molecular mechanisms, including its ability to resist conventional therapies, metastasize, and undergo EMT, presents a significant challenge for effective treatment (De Gaetano et al., 2024; Yang et al., 2024). Recently, TCM has been emerging as a potential complementary treatment for various cancers, including HCC. Many components derived from TCM have demonstrated anti-cancer properties, including the ability to inhibit tumor growth, prevent metastasis, and induce apoptosis (Wang et al., 2020; Xiang et al., 2019). Among these, APSs, a bioactive compound isolated from Astragalus membranaceus, have shown promising effects in modulating immune responses, suppressing inflammation, and directly inhibiting tumor cell growth. However, the precise mechanisms of APS have not been fully elucidated. Here, we confirmed that APS exhibits significant anti-tumor effects on liver cancer, particularly by impeding the process of EMT, a key driver of cancer metastasis. Our findings indicate that APS inhibits the activation of the NLRP3 inflammasome, a crucial regulator of inflammation, which in turn blocks EMT in HepG2 liver cancer cells. This is the first time that APS has been shown to prevent liver cancer progression through the modulation of NLRP3 inflammasome-mediated EMT. These results not only provide new insights into the molecular mechanisms underlying the anti-cancer effects of APS but also suggest a novel therapeutic strategy for HCC, particularly for overcoming EMT-related drug resistance.
APS, the major ingredient in A. membranaceus, has attracted increasing attention in cancer treatments for its immune regulatory effects (Li et al., 2022). APS is proven to ameliorate liver injury (Huang et al., 2021) and fibrosis (Sun et al., 2023), and may promote Doxorubicin-induced HCC apoptosis (Li et al., 2023). Moreover, recent studies reported that APS attenuated PD-L1-mediated immunosuppression to exert an anti-tumor effect (He et al., 2022). Its anti-tumor mechanisms include immune system enhancement through activation of macrophages, dendritic cells, T lymphocytes, and NK cells, promoting cytokine production and immune cell maturation. APS also induces apoptosis and inhibits cancer proliferation by interfering with the cell cycle and signaling pathways like PI3K/Akt, ERK/MAPK, and NF-κB (Li et al., 2022). Additionally, it reshapes the tumor microenvironment, improving the efficacy of immunotherapies and traditional treatments while mitigating side effects. Future applications of APS hold promise, particularly as an adjuvant therapy in cancer immunotherapy, given its potential to enhance efficacy and reduce toxicity in clinical settings (Kong et al., 2021). In our investigation, we prepared APS at varying concentrations to stimulate HepG2 cells, revealing a dose- and time-dependent inhibitory effect on malignancy and survival. These observations are consistent with prior studies demonstrating the anti-tumor potential of APS in various cancers through the modulation of key cellular processes such as proliferation, invasion, and migration (Kong et al., 2021; Li et al., 2022). Notably, APS effectively induced cell cycle arrest, a crucial mechanism for halting uncontrolled cancer cell proliferation. We found that as the concentration and stimulation time of APS increased, the malignancy and survival of HepG2 cells were inhibited, which are markers of cancer progression. HepG2 cell cycle arrest was also successfully brought on by APS at the same time. Our results provided preliminary evidence for the therapeutic benefit of APS against liver cancer.
NLRP3 is a proinflammatory inflammasome that typically participates in regulating the tumor microenvironment, regulating the malignancy and apoptosis of tumor cells. In addition, NLRP3 inflammasome can enhance anti-tumor immune response and affect the effectiveness of tumor treatment (Tengesdal et al., 2023). Therefore, NLRP3 might be a highly promising target in cancer treatment. NLRP3 inflammasome is composed of the promoter molecule NLRP3, responsible for receiving danger signals; caspase-1, responsible for inducing the secretion of inflammatory factors; and ASC, serving as a bridge between NLRP3 and caspase-1 (Fu & Wu, 2023). The inhibition of NLRP3, ASC, and caspase-1 by APS in our study suggests its ability to attenuate inflammation-driven EMT. This mechanism is particularly relevant, as EMT is important in enhancing invasion, metastasis, and chemoresistance in liver cancer. Furthermore, our observation that Nigericin—a known activator of the NLRP3 inflammasome—partially rescues these effects highlights the specificity of APS in targeting inflammasome-mediated pathways. This meant that APS could inhibit NLRP3 inflammasome activation in liver cancer. Thus, we are interested in investigating the mechanism of NLRP3 in APS against liver cancer.
As demonstrated above, we have confirmed once more how the NLRP3 inflammasome inhibited the growth of liver cancer cells when it comes to APS. The NLRP3 activator is still added to HepG2 cells treated with APS. According to our research, the capacity of HepG2 cells for invasion, migration, and survival was greatly increased when NLRP3 inflammasomes were activated, which weakened the function of APS. This further demonstrated the crucial role of NLRP3 inflammasome in APS treatment of liver cancer. However, while its activation appears to induce certain anti-tumor immune responses under specific conditions, it may also promote tumor cell survival and metastasis when dysregulated. This paradoxical effect highlights the complexity of targeting inflammasomes in cancer therapy and emphasizes the need for a more nuanced approach. In the context of liver cancer, where inflammation is vital in both tumor progression and immune evasion, understanding the exact mechanisms by which NLRP3 inflammasomes influence cancer cell behavior is critical. It is possible that the activation of NLRP3 in combination with APS may trigger a compensatory inflammatory response, promoting the survival and metastasis of liver cancer cells. Therefore, future research might focus on optimizing the therapeutic window of APS by exploring strategies that selectively modulate NLRP3 inflammasome activity, minimizing its potential protumorigenic effects while enhancing its anti-tumor efficacy.
APS’s ability to inhibit EMT progression in liver cancer is particularly noteworthy, as EMT is a well-established driver of liver cancer malignancy. Normal epithelial cells have a regular shape, strong cell connections, and barrier and secretory activities. However, after undergoing EMT transition, cells lose their natural polarity and develop higher migratory and invasion capabilities in addition to increased resistance to drug resistance and apoptosis (Davis et al., 2014; Sabouni et al., 2023). It is reported that the activation of NLRP3 inflammasomes promotes the progression of colorectal cancer and is associated with EMT (Marandi et al., 2021). By producing proinflammatory cytokines, the NLRP3 inflammasome may aid in the formation of EMT (Peng et al., 2020). Thus, we assessed whether APS inhibited NLRP3 to influence the EMT process. Meanwhile, it was discovered that E-cadherin expression reduced and N-cadherin expression rose by the addition of NLRP3 activator based on APS treatment. That is to say, the NLRP3 activator reversed the inhibitory effect of APS on EMT. Through this mechanism, APS may prevent the epithelial phenotype loss and mesenchymal marker gain that contribute to tumor cell invasiveness and metastasis. The modulation of EMT markers, coupled with the observed reduction in proinflammatory cytokine levels, underscores APS’s multifaceted role in impairing the tumor microenvironment.
In summary, our studies provide evidence that APS, by inhibiting NLRP3 inflammasome, prevents the process of EMT in liver cancer. According to our research, there is a strong correlation between the high expression of NLRP3 and the development of EMT in HepG2 cells. The ability of APS to target multiple molecular pathways, including immune modulation and inflammasome regulation, underscores its potential as a promising adjunct therapy for liver cancer. Future studies are needed to validate these findings in vivo and explore the clinical applicability of APS in combination with existing therapies, especially for patients with advanced HCC or those resistant to current treatments. As an active ingredient of natural drugs, further investigation is necessary to elucidate the metabolic processes behind the absorption, distribution, metabolism, and excretion of APS. Additionally, while our findings at the cellular level are promising, they remain limited to in vitro experiments and require further validation in animal models. Future studies should prioritize in vivo experiments to confirm the therapeutic potential of APS in preventing EMT in liver cancer. Specifically, these studies should aim to evaluate the absorption, distribution, metabolism, and excretion properties of APS, as these factors are crucial for understanding its bioavailability and pharmacokinetics. In addition, it is essential to assess APS’s efficacy in appropriate animal models of liver cancer, which will provide more comprehensive data on its anti-tumor effects and therapeutic relevance. Detailed investigations into the molecular mechanisms by which APS inhibits the NLRP3 inflammasome and prevents EMT are also needed to fully elucidate its mode of action. Furthermore, long-term studies are necessary to assess both the safety and potential side effects of APS treatment, including its impact on liver function, immune response, and overall health. These in vivo validations will be crucial in establishing APS as a viable therapeutic strategy for liver cancer and could potentially pave the way for clinical trials in human patients.
Footnotes
Abbreviations
APS: Astragalus polysaccharide; ASC: Apoptosis-associated speck-like protein; BCA: Bicinchoninic acid; BSA: Bovine serum albumin; ELISA: Enzyme-linked immunosorbent assay; EMT: Epithelial-mesenchymal transition; FITC: Fluorescein isothiocyanate; GAPDH: Glyceraldehyde 3-phosphate dehydrogenase; HEPES: 4-(2-Hydroxyethyl)-1-piperazineethanesulfonic acid; HCC: Hepatocellular carcinoma; MEM: Minimum essential medium; NEAA: Non-essential amino acids; NLRP3: NOD-like receptor protein 3; PBS: Phosphate-buffered saline; PCR: Polymerase chain reaction; PI: Propidium iodide; PVDF: Polyvinylidene fluoride; RIPA: Radioimmunoprecipitation assay; SDS-PAGE: Sodium dodecyl sulfate-polyacrylamide gel electrophoresis; IL-1: Interleukin-1; IL-18: Interleukin-18; CCK8: Cell counting kit-8.
Acknowledgments
None.
Author Contributions
Hongtao Sui and Caihua Zhang designed the study and performed the experiments. Hai Yang collected the data. Zongyuan Huang analyzed the data. Hongtao Sui and Caihua Zhang prepared the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
