Abstract
Background
Paclitaxel is commonly used as a cancer chemotherapy drug that frequently causes peripheral neuropathic pain. Inflammasome is a multiprotein complex consisting of Nod-like receptor proteins (NLRPs), apoptosis-associated speck-like protein, and caspase-1, which functions to switch on the inflammatory process and the release of interleukin-1β. Growing evidences have supported that peripheral interleukin-1β is critical in enhancing paclitaxel-induced neuropathic pain. However, whether activation of NLRP3 inflammasome in peripheral nerve contributes to paclitaxel-induced neuropathic pain is still unclear.
Results
Paclitaxel induced mechanical allodynia of rats from day 3 and worsened gradually till 3 weeks after injection. Paclitaxel resulted in expression of NLRP3 and activated fragments of caspase-1 and interleukin-1β in L4-6 dorsal root ganglia and sciatic nerve three weeks after injection, indicating activation of NLRP3 inflammasome. The expression of NLRP3 was located in CD68-labeled macrophages infiltrating in L4-6 dorsal root ganglia and sciatic nerve, and paclitaxel increased the expression of NLRP3 in macrophage. Moreover, the paclitaxel elicited mitochondria damage, which became swollen and enlarged in macrophages and axons of sciatic nerve three weeks after injection. In vitro, paclitaxel increased the number of damaged mitochondria and mitochondrial reactive oxygen species production in the rat alveolar macrophage cell line NR8383. The administration of a non-specific reactive oxygen species scavenger, phenyl-N-tert-butylnitrone, markedly alleviated mechanical allodynia and inhibited the activation of NLRP3 inflammasome in L4-6 dorsal root ganglia and sciatic nerve of the paclitaxel-induced neuropathic pain model.
Conclusions
Paclitaxel induced mechanical allodynia and activation of NLRP3 inflammasome in infiltrated macrophages of L4-6 dorsal root ganglia and sciatic nerve. Paclitaxel elicited mitochondria damage and reactive oxygen species production may result in activation of NLRP3 inflammasome in peripheral nerve, which contributes to paclitaxel-induced neuropathic pain.
Keywords
Introduction
Chemotherapy-induced neuropathic pain is the most common and serious adverse effect of effective chemotherapeutic agents. Paclitaxel is a commonly used cancer chemotherapy drug that frequently causes peripheral neuropathic pain.1,2 Since the mechanisms underlying this dose-limiting side effect are poorly understood, there is no reagent that has been proven effective against symptoms of neuropathic pain. 3
The discovery of inflammasome provides a new opportunity for the study of the neuroimmune mechanism of chemotherapy-induced neuropathic pain. The inflammasomes, recently identified innate immune complexes, are now known to be responsible for triggering inflammation in response to several molecular patterns. 4
The inflammasome acts as an activating scaffold for proinflammatory caspase-1, cleaves and activates prointerleukin (IL)-1β. 5 IL-1β, one of the first cytokines discovered in the 1980s, is a potent mediator of fever, pain, and inflammation. 6 A growing body of evidence has supported that proinflammatory cytokines, such as peripheral IL-1β, is critical in enhancing pain in paclitaxel-induced neuropathic pain.7,8 In recent years, the relationship between inflammasome and pain has been gradually demonstrated. Dysregulation of inflammasome is the cause of pain-related diseases such as complex regional pain syndrome, 9 gouty arthritis. 10 However, whether the inflammasome is involved in paclitaxel-evoked neuropathic pain is still unknown.
Reactive oxygen species (ROS) play a causal role in the development and maintenance of paclitaxel-induced pain. It was found that pharmacological inhibition of ROS significantly reversed established paclitaxel-induced pain and prevented the development of paclitaxel-induced pain. 11 Paclitaxel also patented cold hyperalgesia in diabetes by increasing mitochondrial ROS production and poor radical scavenging. 12 Previous studies suggested that Nod-like receptor protein 3 (NLRP3) inflammasome activity is positively regulated by ROS derived from mitochondrion.13,14 We hypothesized that paclitaxel induces the processing and release of IL-1β from ROS-induced activation of the NLRP3 inflammasome.
Here, using a rat neuropathic pain model induced by repeated intraperitoneal paclitaxel injections, we examined whether activation of NLRP3 inflammasome in peripheral nerve contributes to paclitaxel-induced neuropathic pain, and whether paclitaxel-induced mechanical allodynia and activation of NLRP3 inflammasome can be attenuated by ROS inhibitor phenyl-N-tert-butylnitrone (PBN).
Methods
Ethics statement
All animal experimental procedures carried out in this study were approved by the Laboratory Animal of the Ethics Committee of Huazhong University of Science and Technology and were in compliance with the guidelines for animal care set forth by this Committee.
Animal models
Experiments were carried out on male adult Sprague Dawley rats (200–250 g) purchased from Experimental Animal Center of Tongji Medical College of Huazhong University of Science and Technology. The following number of rats were used: 48 rats for paclitaxel treatment experimental research (including the behavioral tests, western blotting, and real-time polymerase chain reaction (PCR) quantitation); 24 rats for paclitaxel plus PBN treatment study (including the behavioral tests and western blotting); and 32 rats for paclitaxel and PBN treatment research (behavioral tests only). The rats were individually housed in cages with a 12-h light/dark cycle and had free access to food and water. Rats were injected intraperitoneally (i.p.) with 2 mg/kg paclitaxel (days 1, 3, 5, and 7) as previously described.15,16 Paclitaxel of 2 mg/ml was prepared with 0.9% sterile saline for injection from the clinical formulation of 5 ml/30 mg paclitaxel (TAXOL, Bristol Myers Squibb SRL, Italy). Separate rats received the same volume of saline on four alternate days and were used as vehicle control.
Behavioral tests
The rats were habituated to the testing environment for 30 min. Mechanical allodynia was assessed by placing rats on an elevated mesh floor, and the mechanical threshold was measured by the “up-down” method. 17 After an acclimation period of 30 min, a series of calibrated von Frey filaments (Stoelting, Wood Dale, USA) were applied perpendicularly to the plantar surface of both hindpaws with sufficient force to bend the filament for 6 s. Brief withdrawal or paw flinching was considered as a positive response. The test was repeated two to three times in each rat, and the mean value was calculated. The investigators involved in behavioral tests and biochemical assays were blinded to the drug injection throughout the study.
Western blotting
The L4-6 segments of dorsal root ganglia (DRGs) and the sciatic nerves were removed on day 21 after the animals were anaesthetized with an overdose of sodium pentobarbitone (120 mg/kg, i.p.) and decapitated. Total protein was isolated from the tissue minced with scissors and homogenized in radioimmunoprecipitation assay lysis buffer with 40 mg/ml (Beyotime Biotechnology, Nanjing, China) and 2 mM phenylmethylsulfonyl fluoride, which was then centrifuged at 12,000 g for 15 min at 4℃. The pellet was discarded and protein concentrations from the supernatant were determined using the Enhanced BCA Protein Assay Kit (Beyotime Biotechnology, China). A 60-mg protein of each tissue was denatured with sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) loading buffer at 95℃ for 5 min and separated on an 8% to 10% glycine-SDS-PAGE gel. The proteins were transferred onto a polyvinylidene fluoride membrane, blocked for 1 h in 5% non-fat dry milk in Tris-buffered saline (TBS) with 0.1% Tween-20.
The membrane was probed with primary antibodies of rabbit anti-NLRP3(1:800, Santa Cruz, CA, USA) or mouse anti-caspase-1 (1:500, Santa Cruz, CA, USA) or rabbit anti-IL-1β (1:500, Santa Cruz, CA, USA) or mouse anti-β-actin antibody (1:5000; Santa Cruz, CA, USA) at 4℃ overnight. After three washes in 0.1% TBS-Tween-20 (pH 7.4), the membrane was then incubated by horseradish peroxidase-labeled IgG from Santa Cruz Biotechnology: goat anti-rabbit secondary antibody (1:20,000) or goat anti-mouse secondary antibody (1:20,000) for 1 h at room temperature and washed 3 times with TBS with 0.1% Tween-20. The enhanced chemiluminescence method (ECL Plus western blotting detection reagents, Pierce, Rockford, IL, USA) was used to reveal the protein bands according to the manufacturer’s protocol. The optical density of each band was then performed using the Image J software (NIH, Bethesda, MD, USA) and normalized with the housekeeping gene β-actin.
Real-time PCR quantitation
List of primers used for real-time PCR.
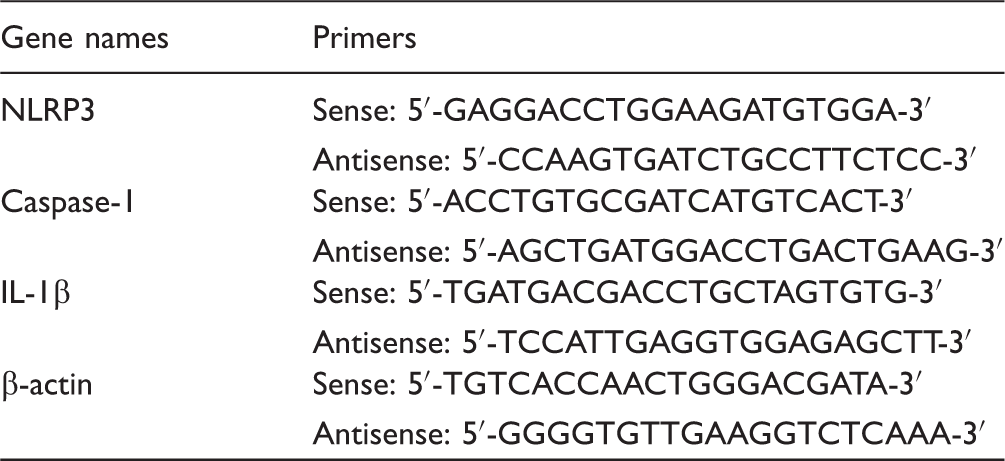
PCR: polymerase chain reaction; NLRP: Nod-like receptor protein; IL-1β: interleukin-1β.
Double-immunofluorescence labeling
Rats were deeply anesthetized with an overdose of sodium pentobarbitone (120 mg/kg, i.p.) and were transcardially perfused with sterile saline (37℃) and 4% paraformaldehyde (4℃) in 0.1 M phosphate buffer at pH 7.4. The L4-6 DRGs and sciatic nerve tissues were harvested after perfusion immediately. Tissues were postfixed at 4℃ in the perfusion fixative for 4 h, cryoprotected in 30% sucrose overnight, and sectioned at 20 µm on a cryostat, in a plane parallel to the long axis of the L4-6 DRGs and sciatic nerve. The sections were mounted onto gelatin-coated slides, air-dried overnight. The slides were washed in 0.01 M PBS with 0.05% Tween-20 and blocked for 1 h with 5% donkey serum and 0.2% Tween-20 in 0.01 M PBS and then incubated with rabbit anti-NLRP3 (Santa Cruz CA, USA, 1: 200), and mouse anti-CD68 (1:200; Serotec, Oxford, UK) for identification of macrophages at 4℃ overnight with the primary antibody diluted in PBS containing 5% donkey serum albumin. The anti-CD68 antibody specifically labels macrophages in the tissues. 18 The sections were washed 4 times with 0.05% Tween-20 in PBS for 5 min and incubated with a mixture of secondary antibodies: donkey anti-mouse IgG conjugated with Dynight 488 (1:400; Jackson ImmunoResearch, West Grove, PA, USA) and donkey anti-rabbit IgG conjugated with Dynight 594 (1:500; Jackson ImmunoResearch, West Grove, PA, USA). Sections were washed 4 times with 0.05% Tween-20 in PBS for 5 min and treated with the fluorescence-mounting medium to avoid quenching of fluorescence before being mounted on cover glass. Slides of negative controls were incubated with TBS instead of primary antibodies, which resulted in no positive labeling in the tissues.
Flow cytometric analyses
NR8383 cells (Procell, Wuhan, China) were cultured at 37℃, 100% humidity, and 5% CO2 in Kaighn’s F-12K Nurtient Mixture (Gibco, NY, USA), 20% fetal bovine serum (Biological Industries, Israel), 2 mM l-glutamine, 100 µg/ml penicillin, and 100 µg/ml streptomycin. Approximately 1.2 × 106 cells were seeded in 2.5 ml medium each. NR8383 cells were treated with paclitaxel (100 nmol/L) stimulation overnight. 19 Mitochondria were measured by fluorescence levels upon staining with MitoTracker green (Life Technology, City of New York, New York, USA) at 100 nM to mark all mitochondria, MitoTracker deep red (Life Technology, City of New York, New York, USA) at 100 nM to mark active mitochondria, and MitoSOX to detect ROS in the mitochondria for 30 min at 37℃. Cells were then washed with PBS solution and re-suspended in 300 µl cold PBS solution for fluorescence-activated cell sorting analysis. The results were analyzed with FlowJo7.6.2 software.
Drug treatment
In order to evaluate the protective effect of PBN, rats were randomly divided into vehicle control (vehicle of paclitaxel), paclitaxel + PBN treatment, paclitaxel + vehicle (vehicle of PBN) groups, and PBN alone groups. Rats in paclitaxel + PBN group were received intraperitoneal injection of PBN (100 mg/kg, 20 mg/ml in sterile saline, Sigma, St. Louis, MO) at the same time with the paclitaxel injection (on the first, third, fifth, seventh day). Rats in the paclitaxel + vehicle group were treated with the same volume of sterile saline. Rats in PBN alone group were received intraperitoneal injection of PBN on the first, third, fifth, seventh day.
In order to evaluate whether PBN can inhibit the existed allodynia induced by paclitaxel, rats were randomly divided into vehicle control (vehicle of paclitaxel), paclitaxel + PBN treatment, and paclitaxel + vehicle (vehicle of PBN) groups. Rats in paclitaxel + PBN group received intraperitoneal injection of PBN (100 mg/kg), twice daily at 12 h interval for 3 days beginning on the 17th day after paclitaxel treatment.
Statistical analysis
SPSS 17.0 (IBM Corporation, Armonk, NY, USA) was used to evaluate significance, and GraphPad Prism 6 (GraphPad Software Inc., La Jolla, CA) was used to make figures. Data are presented as means ± SEM. Significance between two groups was evaluated using non-paired Student’s t-test. Two-way analysis of variance (ANOVA) with Bonferroni’s post hoc test was used in the withdrawal thresholds between different groups and time points. One-way ANOVA with Bonferroni’s post hoc test was used for comparison between three or more groups or time points. A p value of less than 0.05 was defined as significant differences.
Results
Paclitaxel induced mechanical allodynia and activation of NLRP3 inflammasome in DRGs and sciatic nerve
Rats showed a slight decrease in mechanical withdrawal threshold one day after paclitaxel injection. The decrease became more pronounced over time, achieving a significant difference from control group four days after treatment (Figure 1). In contrast, withdrawal threshold of rats treated with vehicle remained stable throughout the experiment (Figure 1). The expression of NLRP3 was significantly increased in the L4-6 DRGs and sciatic nerve at one, two, and three weeks after paclitaxel treatment (Figure 2(a) to (d), p < 0.05). Paclitaxel also upregulated expression of activated fragments of caspase-1 and IL-1β in L4-6 DRGs and sciatic nerve, indicating activation of NLRP3 inflammasome (Figure 2(a) to (d), p < 0.05). Paclitaxel also significantly increased the mRNA levels of NLRP3, caspase-1, and IL-1β in L4-6 DRGs and sciatic nerve at one, two, and three weeks after paclitaxel treatment (Figure 2(e) and (f), p < 0.05). Paclitaxel also increased the mRNA and protein expression of NLRP3, caspase-1, and IL-1β protein in tibial nerve but not in spinal cord (Supplementary Figure 2). These data suggested that paclitaxel-induced mechanical allodynia was parallel with the activation of NLRP3 inflammasome.
Paclitaxel-induced mechanical allodynia in rats. The behavior test shows mechanical withdrawal threshold (in grams) for vehicle control (CON) and paclitaxel-treated (PAC) groups. Data are expressed as means ± SEM (n = 8 in each group). *p < 0.05, compared with the control group (non-paired Student’s t-test). Paclitaxel increased the mRNA and protein expression of NLRP3, caspase-1, and IL-1β in L4-6 DRGs and sciatic nerve at one, two, and three weeks. The representative western blot images shown in (a) and (b) illustrate the expression of NLRP3, activated fragments of caspase-1, and IL-1β in L4-6 DRGs (a) and sciatic nerve (c). Images (b) and (d) represent relative level of percentage of protein of control group (CON) corresponding to (a) and (c). The relative mRNA levels of NLRP3, caspase-1, and IL-1β were quantified in L4-6 DRGs (e) and sciatic nerve (f). Data are expressed as means ± SEM (n = 8 in each group). *p < 0.05, compared with the control group (non-paired Student’s t-test).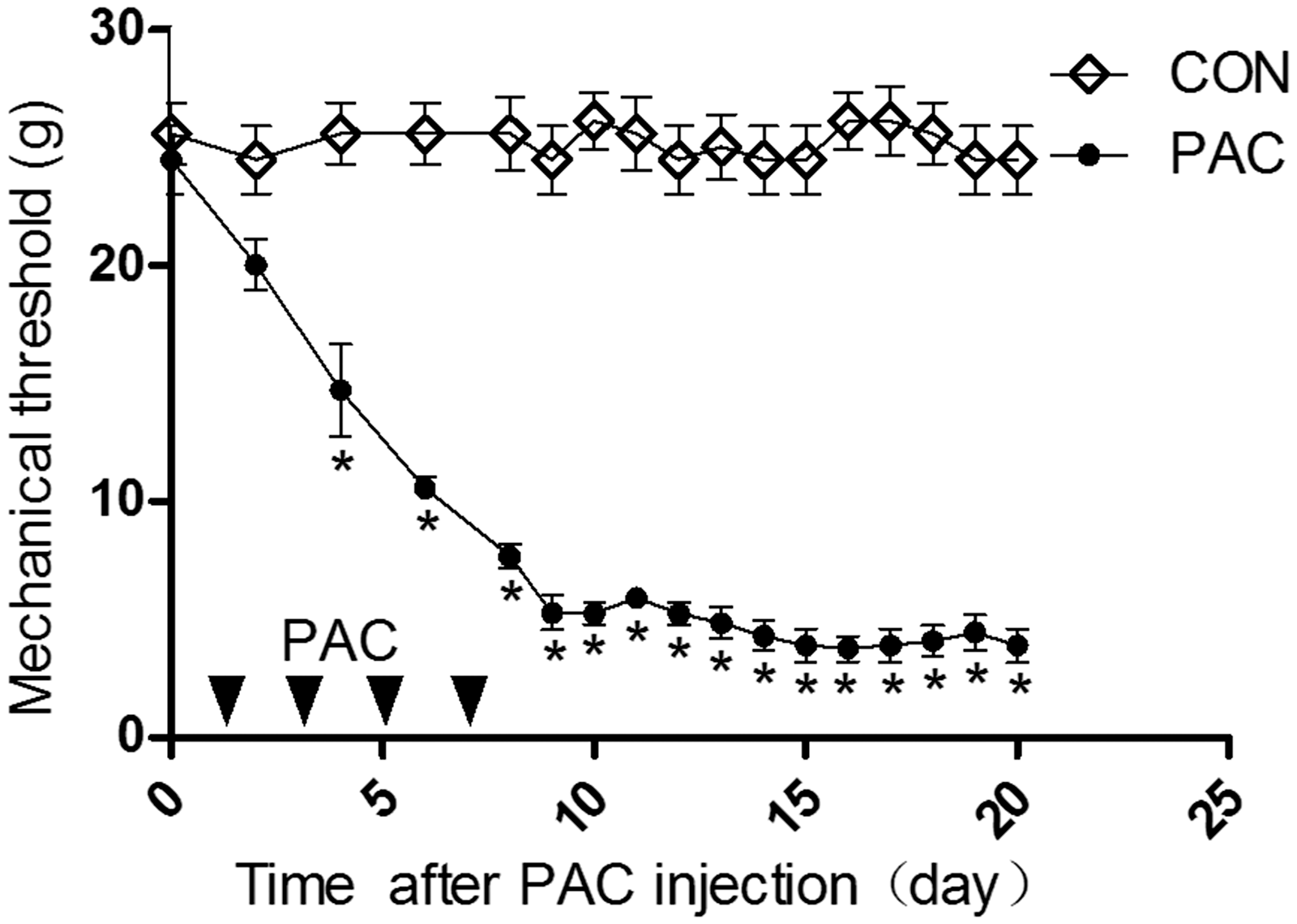

Paclitaxel increased the expression of NLRP3 in macrophages of L4-6 DRGs and sciatic nerve
Immunohistochemistry was used to define the cellular localization of NLRP3 in L4-6 DRGs and sciatic nerve. As shown in Figure 3, NLRP3 was coexisted with CD68-positive infiltrated macrophages in L4-6 DRGs and sciatic nerve in both vehicle control and paclitaxel group three weeks after paclitaxel treatment (Figure 3(a) and (c)). Paclitaxel increased the number of macrophages expressing NLRP3 in L4-6 DRGs (p < 0.05, Figure 3(b)) and sciatic nerve (p < 0.05, Figure 3(d)). These data suggested that paclitaxel significantly increased the expression of NLRP3 in macrophages.
The coexistence of NLRP3 and infiltrated macrophages in L4-6 DRGs and sciatic nerve after paclitaxel injection. Immunohistochemistry images of L4-6 DRGs and sciatic nerve sections in (a) and (c) show the CD68-positive macrophage (green) and NLRP3 immunoreactive cells (red) in vehicle control (CON) and paclitaxel-treated (PAC) groups at three weeks after the first injection of paclitaxel. The bottom panels in (a) and (c) are overlay images showing double-labeled NLRP3 and macrophage (Merge, yellow). Summary data in (b) and (d) show the percentage of the area of double-stained cells% in the area of total macrophage. Data are expressed as means ± SEM (n = 8 in each group). *p < 0.05, compared with the control group(non-paired Student’s t-test).
Paclitaxel induced ultrastructural damage of mitochondria in macrophage-like cell of sciatic nerve
Electron microscopic examination of the sciatic nerve sections revealed the characteristic appearance of ultrastructural damage of sciatic nerve three weeks after paclitaxel treatment. As shown in Figure 4, evident damage to myelinated fibers in the sciatic nerve, including axonal swelling, altered myelination, and the appearance of loose and breakdown of the myelin sheath, compared with a more compact wall of the myelin sheath in vehicle group (Figure 4(a) and (b)). Paclitaxel also elicited swollen and enlarged mitochondria in macrophage-like cell (Figure 4(c), (d), and (e)) and axon (Figure 4(c), (d), and (f)) in sciatic nerve.
Paclitaxel-induced ultrastructural changes of the sciatic nerve. (a), vehicle control; (b, c, d, e, and f), paclitaxel injection. The ultrastructural images of the sciatic nerve are control and paclitaxel-treated groups at three weeks after the first injection of paclitaxel. Image (a) shows several normal myelinated (star) and normal mitochondria (arrow, 1700 × , Scale bar, 2 µm). Image (b) shows paclitaxel-induced evident damage to myelinated fibers in the sciatic nerve (star) and the enlarged mitochondria (arrows 1700 ×, Scale bar, 2 µm). Image (c) shows mitochondria in macrophage-like cell (arrow head) and axon (arrow) in sciatic nerve following paclitaxel chemotherapy (1700 ×, Scale bar, 2 µm). Image (d) is enlargement of solid line square area of Image (c) (5000 ×, Scale bar, 1 µm). Image (e) is enlargement of arrow head pointed area of Image (d) (11,500 ×, Scale bar, 500 nm). Arrow heads show markedly enlarged mitochondria in macrophage-like cell. Image (f) is enlargement of arrow pointed area of Image (d). Arrows show markedly enlarged mitochondria in axon (11,500 ×, Scale bar, 500 nm).
Paclitaxel increased the number of damaged mitochondria and mitochondrial ROS production in macrophages of NR8383 cell line
Mitochondria were found to be damaged in macrophage-like cells in sciatic nerve after paclitaxel treatment, so we choosed the NR8383 cell line from rat to do in vitro experiment. We used flow cytometry to detect damaged mitochondria and mitochondrial ROS production induced by paclitaxel in NR8383 cells (Figure 5). This was determined by using three types of mitochondria-specific labels that distinguished intact (MitoTracker deep red), total (MitoTracker green), and ROS-generating mitochondria (MitoSOX). As shown in Figure 5(a), P2 area was infected with only MitoTracker green and did not catch MitoTracker deep red, namely the damaged mitochondria. Paclitaxel significantly increased the damaged mitochondria (Figure 5(b), p < 0.05). As shown in Figure 5(c), the fluorescence intensity of MitoSOX can detect ROS-generating mitochondria of cell. The data showed that paclitaxel significantly increased the number of ROS-generating mitochondria. It suggested that paclitaxel might increase mitochondrial injuries and mitochondrial ROS production in macrophages.
Paclitaxel-induced mitochondria damage and mitochondrial ROS production by flow cytometry in macrophages of NR8383 cell line. Image (a) shows flow cytometry of NR8383 cells treated with paclitaxel (PAC) or vehicle control (CON) overnight. (b), summary data show the ratio of cells in P2 zone out of total NR8383 cells of (a). Image (c) summary data show the relative of fluorescence intensity of mitoSOX. Data are expressed as means ± SEM (n = 3 in each group). *p < 0.05, compared with the control group (non-paired Student’s t-test).
PBN alleviated the mechanical allodynia and inhibited activation of NLRP3 inflammasome in L4-6 DRGs and sciatic nerve after paclitaxel treatment
ROS generation promotes the NLRP3 inflammasome activation.
20
The contribution of ROS to paclitaxel-induced neuropathic pain was assessed by evaluating the effects of intraperitoneally administered PBN on mechanical allodynia and NLRP3 inflammasome activation (Figure 6).
Effect of systemic multiple PBN administration on mechanical allodynia and activation of NLRP3 inflammasome in L4-6 DRGs and sciatic nerve after paclitaxel treatment. (a), time course of mechanical withdraw threshold (in grams) in vehicle control (CON), paclitaxel + vehicle of PBN (PAC+VEH), and paclitaxel + PBN (PAC+PBN) groups. PBN or vehicle was injected intraperitoneally twice daily at 12 h interval for 3 days beginning on day 17, as indicated by arrows. The representative western blot images shown in (b), (c), and (d) illustrate the expression of NLRP3, activated fragments of caspase-1, and IL-1β. Data are expressed as means ± SEM. *p < 0.05, compared with the control group. #p < 0.05, compared with the PAC+VEH group (two-way ANOVA with Bonferroni’s post hoc test was used in (a). One-way ANOVA with Bonferroni’s post hoc test was used for comparison between three groups in (b), (c), and (d).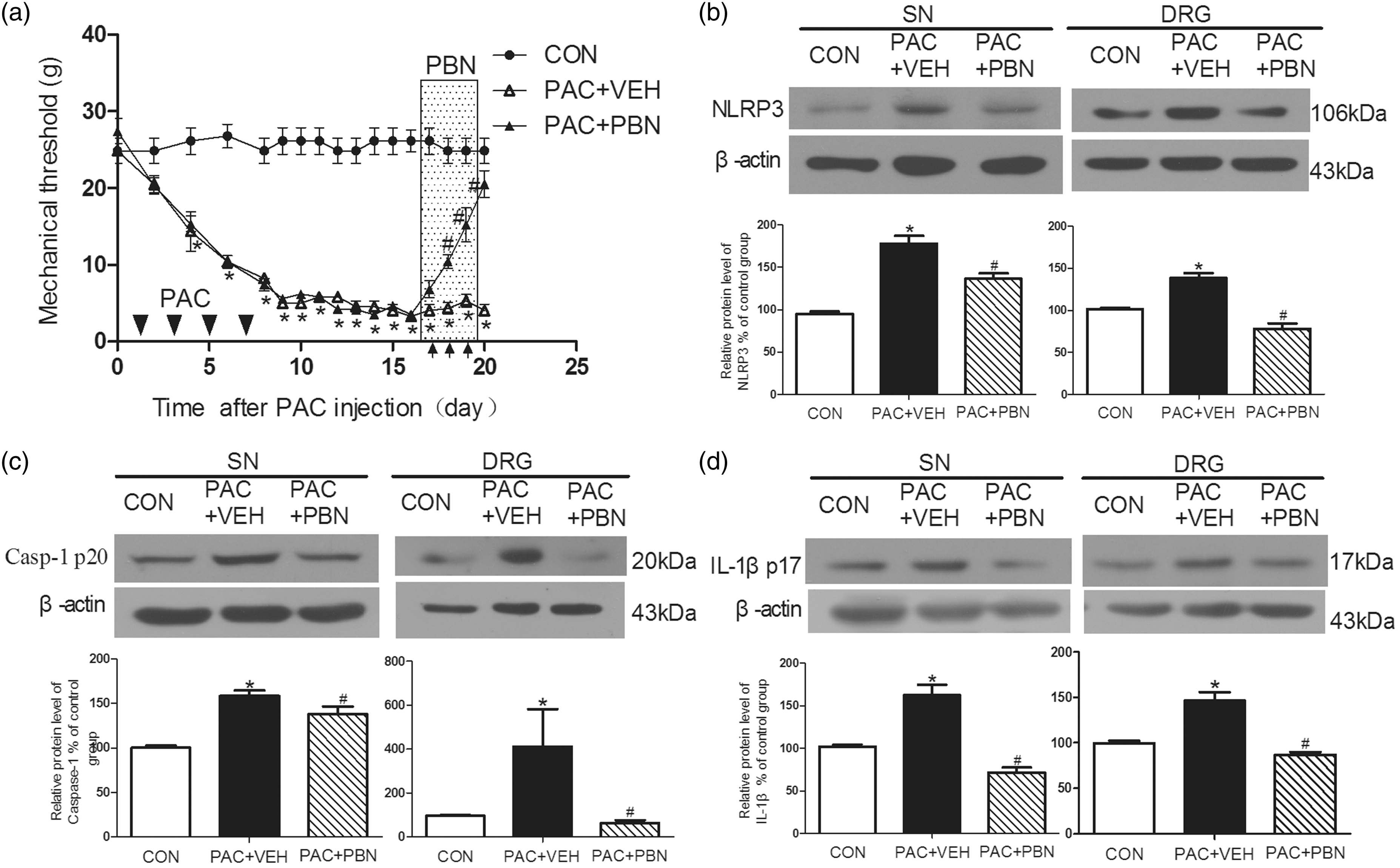
The paclitaxel + vehicle group showed significant decrease in mechanical threshold (Figure 6(a), Supplementary Figure 1). PBN alone did not affect mechanical threshold comparing with vehicle control group (Supplementary Figure 1). PBN treatment (100 mg/kg) on the third, fifth, and seventh day of PBN and paclitaxel injection significantly increased mechanical thresholds compared with those in the paclitaxel + vehicle group. The anti-nociceptive effect of PBN was not detected after the seventh day of PBN and paclitaxel injection (Supplementary Figure 1). It is consistent with previous study that early treatment of PBN has no preventive effects on the development of paclitaxel-induced neuropathic pain in rats. 21
In contrast, PBN did inhibit the existed allodynia induced by paclitaxel. From 17 to 19 days after paclitaxel treatment, the PBN was injected intraperitoneally twice daily at 12 h interval, which significantly increased the withdrawal threshold comparing with paclitaxel + vehicle group from 18 to 20 days after paclitaxel treatment. After three weeks of paclitaxel treatment, the expression of NLRP3 and activation fragments of caspase-1 and IL-1β in L4-6 DRGs and sciatic nerve were significantly higher in paclitaxel + vehicle group comparing with vehicle control group (Figure 6(b) to (d)). PBN treatment significantly reduced the expression of NLRP3 and activation fragments of caspase-1 and IL-1β in L4-6 DRGs and sciatic nerve in comparison to rats in paclitaxel + vehicle group (Figure 6(b) to (d)).
Discussion
In the present study, we demonstrated that the paclitaxel induced mechanical allodynia and activation of NLRP3 inflammasome in DRGs and sciatic nerve. The expression of NLRP3 was located in CD68-labeled infiltrated peripheral macrophage, and paclitaxel increased the expression of NLRP3 in macrophages. Moreover, paclitaxel elicited mitochondria damage in macrophages and axons of sciatic nerve. In vitro, paclitaxel increased the number of damaged mitochondria and mitochondrial ROS production in the rat alveolar macrophage cell line NR8383. The administration of a non-specific ROS scavenger, PBN, markedly alleviated mechanical allodynia and inhibited the activation of NLRP3 inflammasome in DRGs and sciatic nerve of the paclitaxel-induced neuropathic pain model.
Chemotherapy-induced pain is the most common treatment-limiting complication encountered by cancer patients receiving taxane-, vinca alkaloid-, or platin-based chemotherapy.22,23 Chemotherapy-induced pain improves or completely resolves in 51% of patients but becomes chronic in the remainder, affecting the quality of life in cancer survivors. 24 Paclitaxel is a widely used chemotherapeutic agent that has a dose-limiting side effect of painful peripheral neuropathy. 25 Currently, there is no effective therapy for the prevention or treatment for chemotherapy-induced painful peripheral neuropathies. Because there are large variabilities in the degree of pain symptoms in the rats with cancer, paclitaxel applied on normal rats has been an internationally accepted animal model of neuropathic pain.15,16
Inflammasome is a multiprotein complex consisting of NLRPs, apoptosis-associated speck-like protein, and caspase-1, which functions to switch on the inflammatory process and the release of IL-1β. 26 Since IL-1β appears to be a potential mediator of pain,27,28 the role of the inflammasome in paclitaxel-evoked peripheral neuropathy has aroused significant interest. In our study, we found that paclitaxel increased expression of NLRP3 and activated fragments of caspase-1 and IL-1β in L4-5 DRGs and sciatic nerve. Previous studies had reported that in peripheral mechanisms of complex regional pain syndrome, the NLRP1 inflammasome participated in the process of peripheral sensitization. 29 Other studies have shown chronic constriction injury of the sciatic nerve (CCI)-induced NLRP1 activation in the astrocytes of the spinal dorsal horn. 30 However, whether NLRP3 inflammasome is activated in neuropathic pain in rat has not been previously described.
Our study found that paclitaxel-induced neuropathic pain is parallel with activation of NLRP3 inflammasomes for the first time. Since paclitaxel promotes the processing of pro-IL-1β to mature IL-1β, 31 and proinflammatory cytokine IL-1β may induce peripheral sensitization of sensory neurons and mechanical allodynia.32,33 Paclitaxel treatment also induced mRNA expression of IL-1β and intrathecal injection of IL-1ra reversed paclitaxel-induced allodynia, which suggested that blockage the effect of IL-1β could alleviate the paclitaxel-induced neuropathic pain. 34 Thus, IL-1β produced by NLRP3 inflammasome activation may contribute to the development of the paclitaxel-induced neuropathic pain. Moreover, the expression of NLRP3 is increased in infiltrated macrophages. Other studies have proved that paclitaxel significantly increased density of macrophages in DRGs and the sciatic nerve of paclitaxel-treated rats.35,36 Consistently, our study found that paclitaxel upregulated the expression of NLRP3, the key components of the NLRP3 inflammasome, in macrophages of L4-6 DRGs and sciatic nerve. Previous study showed that chemotherapeutic drugs activated the NLRP3 inflammasome and induced secretion of IL-1β from primary mouse macrophages. 37 It suggested that activation of the NLRP3-dependent inflammasome might be derived from the macrophages.35,38
The dysfunction of mitochondrial has a critical role in development of various neurological disorders of the central and peripheral nervous system including neuropathic pain. 39 Paclitaxel treatment has been associated with an accumulation of typical mitochondria in the sensory nerves of rats.14,23 In the present study, paclitaxel induced the damage of mitochondria and ROS production in macrophages in vivo and in vitro. In addition, ROS over-production has been proven to be critical for NLRP3 inflammasome activation in response to a number of stimuli. 31 In environment-induced dry eye disease, the ROS-NLRP3-IL-1β signaling pathway might play a priming role. 40 In the present study, paclitaxel treatment may result in an accumulation of atypical mitochondria and ROS-NLRP3 activation in the sciatic nerve. In addition, paclitaxel also induces damage of mitochondria in axons of sciatic nerve, which is in line with the classical point that paclitaxel has a toxic effect on the mitochondria in axons of the peripheral nerve. This effect is manifested by a structural change—an abnormal incidence of axonal mitochondria that are swollen and vacuolated.41,42
Furthermore, we found that the intraperitoneal administration of a non-specific ROS scavenger, PBN, markedly alleviated paclitaxel induced mechanical allodynia and activation of NLRP3 inflammasomes in DRGs and sciatic nerve of the paclitaxel-induced neuropathic pain in rats’ model. This result is consistent with previous studies that PBN inhibits mechanical hypersensitivity evoked by spinal nerve ligation, 43 visceral inflammation, 44 and paclitaxel-induced inflammation, 11 which demonstrate that mitochondrial ROS-producing profiles are increased in the spinal cord following peripheral nerve injury or inflammatory stimulus. 45 Therefore, our data suggest a model whereby paclitaxel result in NLRP3 inflammasome activation because of ROS-induced oxidation. Thus, our study suggests that mitochondrial dysfunction and ROS over-production may be critical for paclitaxel-induced NLRP3 inflammasome activation. ROS scavenger PBN may attenuate the activation of NLRP3 inflammasomes and thus inhibit paclitaxel-induced neuropathic pain.
Conclusion
In summary, we found that paclitaxel induced mechanical allodynia and activation of NLRP3 inflammasome in rat DRGs and sciatic nerve. PBN markedly inhibited paclitaxel-induced mechanical allodynia and activation of peripheral NLRP3 inflammasome activation. The activated NLRP3 inflammasomes existed in infiltrated peripheral macrophages, and paclitaxel elicited mitochondria damage and ROS production may result in activation of NLRP3 inflammasome. Our study provides new information that the activation of NLRP3 inflammasome in peripheral nerve contributes to paclitaxel-induced neuropathic pain.
Footnotes
Author Contributions
ML and MJ conceived and designed the experiments. MJ did most of the experiments and analyzed the data. CW and FG helped with the western blotting experiment. HX and NS helped with rat model and behavior test experiments. PP, JL, XY, and HL helped with the data collection. XM, BT, and JS helped with the cell culture and flow cytometric analyses. MJ, HX, and ML wrote the manuscript. All authors reviewed the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the National Natural Science Foundation of China (81473768) and the major project of National Natural Science Foundation of Hubei province (No. 2015CFA094).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
