Abstract
Background
In Mexico, the fruit of the Manilkara zapota (L.) P. Royen (Chicozapote) tree is widely consumed; however, numerous studies have identified bioactive compounds within its leaves.
Objectives
To evaluate the bioactive properties of M. zapota leaf extracts, focusing on their antioxidant, anti-diabetic, and anti-hypertensive potentials, and assessing their efficacy in mitigating inflammation.
Materials and Methods
This study evaluated the antioxidant properties of aqueous (AE), ethanolic (EE), and hydroalcoholic (HE) extracts, utilizing 2,2-diphenyl-1-picrylhydrazyl (DPPH) and 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) assays. Anti-diabetic activity was assessed by inhibiting the enzymes α-amylase and α-glucosidase, while anti-hypertensive efficacy was measured via angiotensin-converting enzyme (ACE) inhibition. The anti-inflammatory potential was also examined in vivo using a murine model of 12-O-tetradecanoylphorbol-13-acetate-induced ear edema.
Results
The DPPH assay revealed that all extracts exhibited equivalent antioxidant capacities at the same concentration, with no significant statistical difference (p < 0.05). Conversely, the ABTS assay demonstrated that AE possessed superior activity (4.6−28.5%) across varying concentrations. EE exhibited the most potent inhibition of α-amylase, α-glucosidase, and ACE, with 81.8%, 58.9%, and 87.8%, respectively. In anti-inflammatory assays, EE at a 5 mg dosage significantly outperformed other extracts.
Conclusion
These findings suggest that M. zapota leaf extracts, mainly through EE, could offer a viable natural alternative for reducing inflammation and potentially managing diabetes and hypertension.
Introduction
Inflammatory conditions pose a significant global health challenge, affecting 80% of the worldwide population. This widespread prevalence underscores an urgent need for innovative treatment strategies that combine efficacy with minimal adverse effects. The therapeutic arsenal currently includes non-selective non-steroidal anti-inflammatory drugs (NSAIDs) such as indomethacin, phenylbutazone, and naproxen, alongside cyclooxygenase-2 (COX-2) selective inhibitors like nimesulide and celecoxib. However, the clinical utility of these agents is constrained by potential side effects, including gastrointestinal toxicity, fluid retention, hypertension, and renal impairment, necessitating cautious use (Okin & Medzhitov, 2012; Rodríguez et al., 2020).
The problem at hand is the dire need for therapeutic alternatives that not only effectively manage inflammation but also mitigate the risk of adverse effects associated with conventional drugs. This issue became particularly pronounced during the COVID-19 pandemic, which highlighted the vulnerabilities of individuals with inflammatory conditions further exacerbated by underlying health issues such as diabetes mellitus (DM) and arterial hypertension (AHT). The pandemic’s toll emphasized the need for immune-boosting interventions. Medicinal plants, long revered for their therapeutic benefits without the burden of significant adverse reactions, emerged as a viable option for enhancing immune defenses. Their accessibility and empirical validation over centuries make them an attractive alternative to synthetic pharmaceuticals. In response to this challenge, the investigation explores the efficacy of Manilkara zapota leaves, historically utilized in traditional medicine, as a scientifically validated treatment for inflammatory diseases (Manjili et al., 2020; Rodríguez et al., 2020).
Amidst growing interest in traditional medicine, governments and academic institutions have intensified efforts to validate the empirical knowledge of medicinal plants through rigorous scientific investigation. The significance of M. zapota cannot be overstated. This plant, native to tropical regions of the Americas, has been utilized in traditional medicine for various ailments, yet its potential for modern therapeutic applications remains largely untapped (Galeotti & Bayry, 2020; Magaña et al., 2010; Regalado et al., 2015).
In this context, our study aimed to systematically evaluate the bioactive properties of M. zapota leaf extracts, focusing on their antioxidant, anti-diabetic, and anti-hypertensive potentials, and assessing their efficacy in mitigating inflammation. This was achieved through comprehensive in vitro analyses and in vivo assessments using a mouse model of atrial edema induced by 12-O-tetradecanoylphorbol acetate (TPA).
Materials and Methods
Sample
Leaf samples were collected from M. zapota trees from April 2021 to March 2022. The specimens were cataloged in the herbarium of the Academic Division of Biological Sciences, where they were assigned voucher number 037112. Post-collection, the leaves were desiccated at ambient temperature in a shaded, ventilated room, utilizing a portable dehumidifier (h2otek model rdp-56l/d-424, Mexico). Subsequently, the dried leaves were pulverized (40 mm mesh) to standardize particle size.
Phytochemical Extracts
Phytochemical extracts were prepared at a concentration of 5% (w/v) following the protocol described by Dutta et al. (2010), specifically: distilled water for the aqueous extract (AE), 99.9% ethanol for the ethanolic extract (EE), and a 70:30 (v/v) ethanol:water mixture for the hydroalcoholic extract (HE).
Extracts were subjected to lyophilization (Virtus Freezemobile Sentry 2.0 freeze dryer, at –40°C and 0.045 mbar). Then, dry extracts were preserved in a desiccator at ambient temperature.
Total Polyphenol Content (TPC)
The TPC was quantified using the Kim et al.’s (2013) protocol. Initially, 0.125 mL of the extracted sample was combined with 0.625 mL of Folin–Ciocalteu (1:10 dilution) and vortexed for 10 s. The absorbance was measured at a wavelength of 760 nm (Thermo Scientific Evolution 220, UV-vis spectrophotometer, USA). TPC results were expressed in micrograms of gallic acid equivalents (GAE) per milligram of dry extract.
In vitro Biological Screening
2,2-Diphenyl-1-picrylhydrazyl (DPPH) Assay
Adopting Shimada et al. (1992), the free radical scavenging capacity of the extracts was assessed using the DPPH radical. Absorbance was measured at 517 nm with a Thermo Fisher Scientific Evolution 220 UV-vis spectrophotometer. Ascorbic acid (0.1 mg/mL) served as a positive control. Extract analyses were conducted in duplicate, expressing antioxidant activity as a percentage of free radical scavenging.
2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) Assay
Following Pukalskas et al. (2002), the ABTS radical scavenging activity was quantified. An ABTS stock solution was prepared and mixed with the extracts (10 µL) in microtubes; absorbance was read at 734 nm after 6 min. Ascorbic acid (0.1 mg/mL) was the positive control, with results expressed as percentage radical scavenging.
In vitro α-Amylase Inhibition Assay
Utilizing Dineshkumar et al. (2010), α-amylase inhibitory activity was evaluated by mixing starch, dimethyl sulfoxide (DMSO), the extract, pancreatic α-amylase, Tris–HCl buffer, and 3,5-dinitro-2-hydroxybenzoic acid (DNS). After heating and cooling, absorbance was measured. Acarbose (150 µg/mL) acted as a positive control.
In vitro α-Glucosidase Inhibition Assay
Per Dineshkumar et al. (2010), α-glucosidase activity was inhibited by incubating the enzyme with extracts and p-nitrophenyl-glucopyranoside, stopping the reaction with sodium carbonate, and measuring absorbance at 405 nm.
In vitro Angiotensin-converting Enzyme (ACE) Inhibition Assay
According to Hayakari et al. (1978), ACE inhibitory activity was determined by monitoring the hydrolysis of hippuryl-
In vivo Anti-inflammatory Effect
Ethical Considerations
All the procedures were conducted in accordance with the Guide for the Care and Use of Laboratory Animals.
Anti-inflammatory Evaluation by TPA-induced Ear Edema Model in Mice
The mice weighing 28–40 g were divided into experimental groups of five individuals each. Groups were randomly assigned and maintained under controlled environmental conditions—temperature at 21 ± 1°C, relative humidity at 55%, and a 12-h light/dark cycle—with ad libitum access to water and standard rodent feed (Prolab 2500 Rodent Diet, Circulo ADN SA de CV), meeting their nutritional needs.
The anti-inflammatory efficacy of M. zapota extracts was assessed through the progression of ear edema post-TPA application; this involved measuring the thickness increase in the treated ear over 4 h. The ear receiving only TPA (2.5 µg/ear) served as the negative control, showing consistent thickness increases due to the absence of anti-inflammatory treatment. Conversely, the untreated left ear acted as a baseline control, exhibiting no thickness changes. Indomethacin (500 µg/ear), a widely recognized anti-inflammatory drug, was employed as the positive control.
Extract doses (1 and 5 mg/ear) were dissolved in DMSO and topically applied to the right ear, with a total volume of 20 µL per ear. Four hours post-treatment, euthanasia was carried out via cervical dislocation, and a 7 mm diameter punch biopsy was taken from each pinna. The difference in weight between the treated and untreated ears was recorded to calculate the percentage of inflammation reduction using the following equation:
Experimental Design
In vitro Evaluation
The study employed a completely randomized design to assess the in vitro effects of M. zapota extracts. The independent variables were the types of extracts analyzed (aqueous, ethanolic, and hydroalcoholic), while the dependent variables were the observed activities (antioxidant, anti-diabetic, and anti-hypertensive).
In vivo Evaluation
A bifactorial design was implemented for the in vivo phase, incorporating two factors: types of extracts (three levels) and treatment dose (two levels). The primary response variable was the reduction of inflammation in the inner ear, quantified through changes in ear thickness and weight.
Statistical Analyses
Statistical in vitro and in vivo analyses were conducted using an analysis of variance (ANOVA) and Tukey’s post hoc test for mean comparisons among different treatments. These analyses aimed to identify significant differences between the treatment groups. All statistical procedures were performed utilizing GraphPad Prism 5 software (GraphPad, San Diego, CA, USA).
Results
Quantification of TPC in M. zapota Extracts
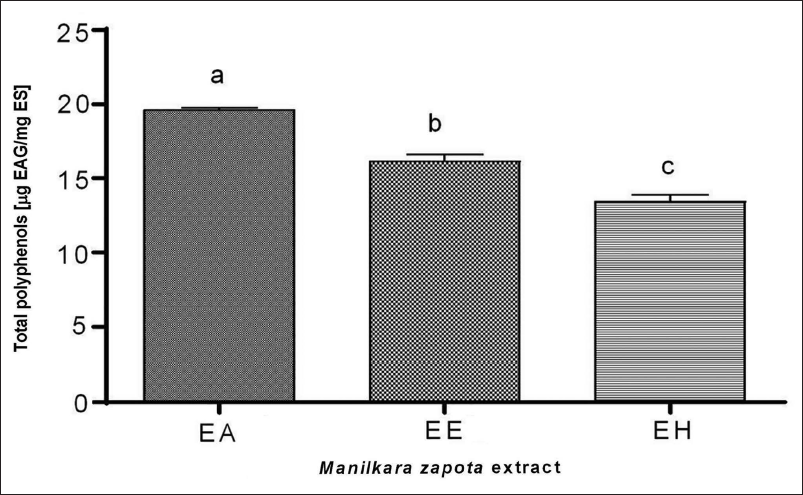
We quantified TPC across different M. zapota extracts (Figure 1). Among the extracts analyzed, the AE exhibited the highest TPC at 19.6 µg GAE/mg extract, surpassing the EE and the HE, which presented concentrations of 16.2 and 13.5 µg GAE/mg extract, respectively. Statistical analysis revealed significant disparities in TPC among the extracts (p < 0.05).
Total Polyphenols in Aqueous (AE), Ethanolic (EE), and Hydroalcoholic (HE) Extracts of M. zapota Leaves. a–c Letters Indicate Significant Statistical Differences Between Each Extract (p ˂ 0.05).
In vitro Biological Evaluation of M. zapota Leaf Extracts: Antioxidant and Enzyme Inhibition Assays
DPPH and ABTS Assays
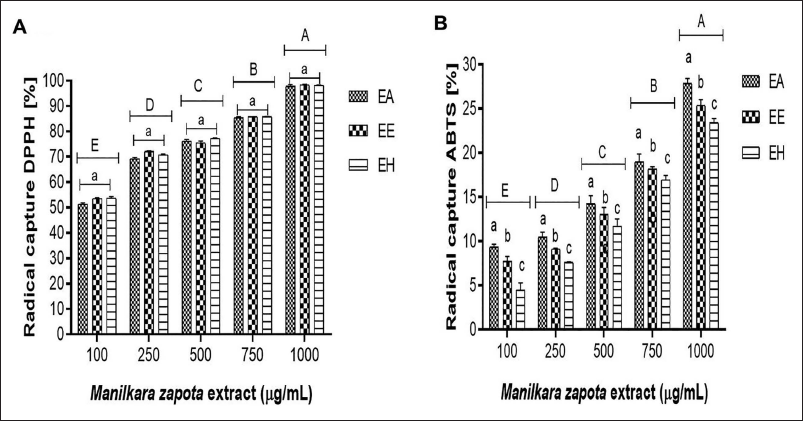
M. zapota leaf extracts, at concentrations ranging from 100 to 1,000 µg/mL, demonstrated a DPPH free radical scavenging activity between 50.6% and 93.5%, with significant statistical variations observed across the concentrations tested (p < 0.05). A direct correlation between the extracts’ concentration and their antioxidant capacity was observed (Figure 2A), albeit without significant differences across the extracts at equivalent concentrations. Conversely, ABTS radical scavenging activity varied from 4.6% to 28.5%, displaying significant statistical differences (p < 0.05), with the AE outperforming others in terms of radical scavenging efficiency (Figure 2B).
Free Radical Scavenging in Aqueous (AE), Ethanolic (EE), and Hydroalcoholic (HE) Extracts of M. zapota Leaves. A–E Uppercase Letters Indicate Statistical Differences Between Extract Concentrations (p ˂ 0.05).
Enzyme Inhibition: α-Amylase and α-Glucosidase
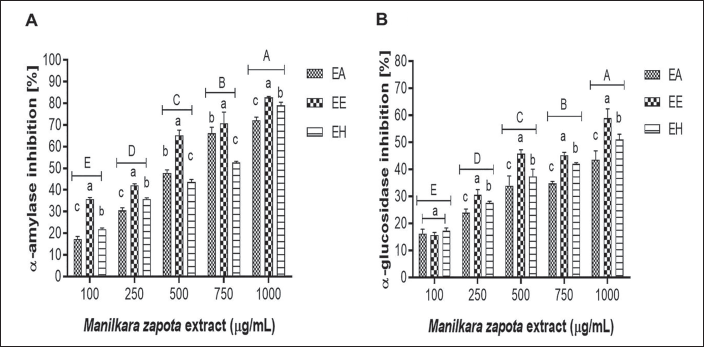
Inhibition rates of α-amylase ranged from 22.4% to 81.8% across the tested concentrations (100–1,000 µg/mL). Similarly, α-glucosidase inhibition spanned from 13.2% to 58.9%, with ethanol extracts showing the most substantial inhibition rates, albeit with minimal significant statistical differences among the extracts (p < 0.05) (Figure 3).
Percentage Inhibition of α-Amylase and α-Glucosidase in Aqueous (AE), Ethanolic (EE), and Hydroalcoholic (HE) Extracts of M. zapota Leaves. A–E Uppercase Letters Indicate Statistical Differences Between Extract Concentrations (p ˂ 0.05).
ACE Inhibition
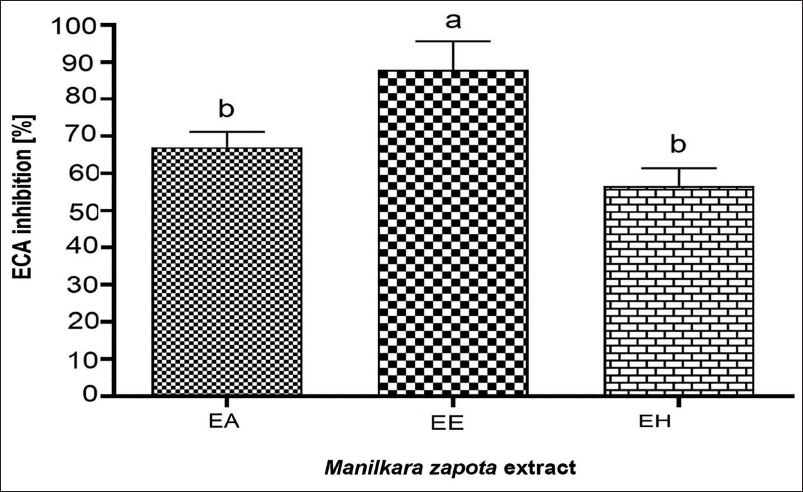
In this work, ethanol leaf extracts demonstrated the highest ACE inhibition rate (87.8%) with significant statistical differences noted (p < 0.05), outperforming aqueous and hydroalcoholic extracts, which showed inhibition rates of 67% and 56.7%, respectively (Figure 4).
Angiotensin-converting Enzyme (ACE) Inhibition Percentage in Aqueous (AE), Ethanolic (EE), and Hydroalcoholic (HE) Extracts of M. zapota Leaves at 1,000 µg mL–1. a–b Lowercase Letters Indicate Significant Statistical Differences Between Each Extract (p ˂ 0.05).
Evaluation of Anti-inflammatory Activity Using a TPA-induced Ear Edema Model in Mice
Severe and progressive swelling induced by TPA leads to an increase of 148.2% in swelling magnitude (Figure 5A). Indomethacin, in contrast, showcased a potent anti-inflammatory effect, significantly reducing ear edema swelling to a mere 2.1%. Upon application of M. zapota extracts at 1 and 5 mg/ear dosages, a noticeable interruption in the typical inflammation pathway was observed, markedly distinct from the negative control group treated solely with TPA. The EE and HE were particularly effective, maintaining inflammation levels beneath the 50% threshold.
Time Course of Total Polyphenol Content (TPA)-induced Atrial Inflammation and Anti-inflammatory Effect of Aqueous (AE), Ethanolic (EE), and Hydroalcoholic (HE) Extracts of M. zapota Leaves (1 and 5 mg).
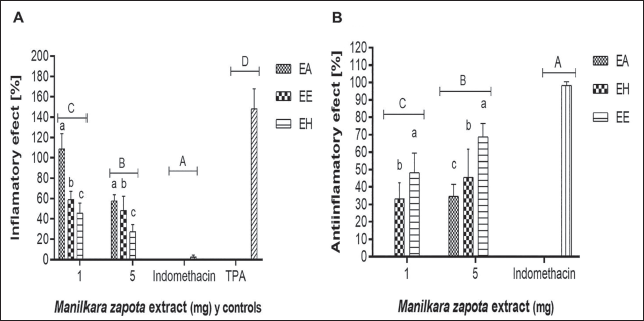
The dose-dependent evaluation of M. zapota extracts’ anti-inflammatory effects revealed a pronounced decrease in edema with escalating extract dosages. At a 5 mg/ear concentration, the EE displayed the most significant anti-inflammatory activity (68.9%), outperforming the HE and AE, which showed effects of 45.6% and 34.7%, respectively, at identical dosages. These outcomes demonstrated significant statistical differences among the extracts (Figure 5B), emphasizing a dose-responsive mitigation of inflammation.
Interestingly, the EE demonstrated superior anti-inflammatory effects at a dosage of 5 mg/ear compared to the AE, which exhibited negligible anti-inflammatory activity at 1 mg/ear.
Discussion
Notably, AE demonstrated the superior extraction efficiency for polyphenols (Figure 1). This efficacy likely stems from the high polarity of deionized water, which effectively mobilizes phenolic compounds, including tannins of high polarity. Given the Sapotaceae family’s composition, which includes alkaloids, tannins, phenols, flavonoids, steroids, terpenes, glycosides, and saponins, this result aligns with expectations (Mondal et al., 2022). Additionally, M. zapota leaves are known to harbor a plethora of secondary metabolites such as myricetin-3-O-α-
This investigation’s findings position the TPC in AE of M. zapota leaves above the levels previously reported by Harris et al. (2016), who identified a TPC of 5.5 mg GAE/g in aqueous plant extracts. However, Rodríguez-García et al. (2019) observed TPCs like our findings, with 17.4 mg EC/g in aqueous leaf extracts of M. zapota. While other research focused on different plant components yielded varying TPCs, such as 12.9 mg GAE/g in ethanolic stem extracts (Hilma, 2018), the results underline the influence of factors like soil type, climatic conditions, and agronomic practices on TPC (Carrera-Lanestosa et al., 2020). The decision to analyze leaf extracts was informed by documented higher concentrations of phytochemicals like phenolics in this vegetative organ (Villarreal-Ibarra et al., 2015). Polyphenols contribute by eliminating free radicals, forming chelates with catalytic metals, activating antioxidant enzymes, regenerating α-tocopherol radicals, and inhibiting oxidases, in addition to reducing oxidative stress, which enhances their positive effects on health, thanks to their antioxidant action (Adefegha, 2018). Oxidative stress, resulting from the production of free radicals (reactive oxygen species (ROS)), is associated with the development of various chronic and degenerative diseases, such as DM, hypertension, inflammation, stroke, cancer, and neurodegenerative disorders. Plant-based antioxidants can play a key role in preventing and treating these conditions linked to oxidative stress (Adefegha, 2018; Zhang et al., 2023).
An augmentation in antioxidant activity corresponds to the escalating concentrations of M. zapota extracts (Figure 2A), yet without significant statistical variance (p > 0.05) across extract types at identical concentrations. Islam et al. (2020) documented an 86.3% scavenging efficiency in the ethanolic extract of M. zapota leaves at a 100 µg/mL concentration, approximately 30% higher than the results observed in this investigation under similar conditions (Islam et al., 2020). Contrarily, Mohd Tamsir et al. (2020) identified remarkably high antioxidant activities, 93.6%, and 92.9%, in aqueous and ethanolic leaf extracts, respectively, at a 250 µg/mL concentration (Mohd Tamsir et al., 2020). These findings align closely with those reported by Kshanmugapriya et al. (2011), who noted antioxidant activities of 64.5%, 50.5%, and 52.8% in ethanolic, aqueous, and acetonic seed extracts of M. zapota at a 500 µg/mL concentration, respectively (Kshanmugapriya et al., 2011).
The variability in results could be attributed to the differential phytochemical composition present across various vegetative organs of M. zapota; yet, both seed and leaf extracts have been characterized by their content of steroids, triterpenoids, phenols, glycosides, and carbonyls, with a relatively lower presence of coumarins and tannins (Mohanapriya et al., 2019). In assessing ABTS radical scavenging efficiency at uniform concentrations, AE demonstrated superior antioxidant activity. However, these outcomes are lesser than those reported by Islam et al. (2020), who found a 56.2% antioxidant activity in ethanolic leaf extracts at 100 µg/mL. Meanwhile, Kshanmugapriya et al. (2011) reported antioxidant activities of 60.6% and 51.8% in seed extracts at a 500 µg/mL concentration, and Kannan et al. (2013) observed 63.2% and 70.4% in ethanolic extracts of the pulp and skin, respectively, at a 1,000 µg/mL concentration (Kannan et al., 2013). The distinguished antioxidant potential of M. zapota leaf extracts could be inherently linked to their phytochemical profile (Karle et al., 2022).
The α-glucosidase and α-amylase inhibition results (Figure 3) exceed those reported by Rodríguez-García et al. (2019) and align with Mondal et al. (2022), who highlight the potential of M. zapota extracts in the management of postprandial hyperglycemia and type 2 DM by interfering with carbohydrate absorption (Ibrahim et al., 2019). M. zapota may help treat postprandial hyperglycemia in individuals with type 2 diabetes by inhibiting the enzymatic activity of α-glucosidase and α-amylase (Bangar et al., 2022).
DM is associated with excessive production of ROS, which contributes to the development of diabetic complications such as insulin resistance and β-cell dysfunction. These cells are particularly vulnerable to oxidative damage, leading to an imbalance in insulin secretion (Adefegha, 2018). Phenolic compounds present in plant-based foods have been shown to reduce blood hyperglycemia levels, thanks to their ability to inhibit powerful enzymes such as α-amylase and α-glucosidase (Abusufyan et al., 2018). The inhibitory action of polyphenols is largely due to the hydroxyl groups in their structures. In addition, it has been observed that they contribute significantly to these enzymatic inhibitions due to: (a) unsaturated 2,3 bonds in conjugation with the 4-carbonyl group, (b) catechins with catechol-type structures, and (c) ellagitannins with β-galloyl groups in the C-1 position of glucose (Adefegha, 2018).
Hypertension and DM are closely related, increasing the risk of developing various cardiovascular diseases. One of the main underlying causes of these chronic conditions is the excessive production of ROS (Ademiluyi et al., 2016; Zhang et al., 2023). The ACE transforms angiotensin I into angiotensin II, a potent vasoconstrictor, and inhibits the action of bradykinin. Research has shown that phenolic compounds have a structure–function relationship in inhibiting ACE, either by chelating the zinc ion at its active site or by facilitating the formation of hydrogen bonds between phenols and amino acid residues at that site (Ademiluyi et al., 2016).
The potent ACE inhibitory activity shown in this study (Figure 4) could be attributed to the effectiveness of ethanol in extracting intermediate polarity substances, thereby affecting enzyme–substrate interactions. These results, although promising, are somewhat inferior to those reported by Das and De (2013) and Shree et al. (2021), marking this study as potentially the first to explore ACE inhibition in M. zapota, highlighting its relevance in the management of hypertension.
On the other hand, the results of the anti-inflammatory effect reported in the present study are modest compared to the anti-inflammatory effects of ethyl acetate and methanolic extracts of M. zapota leaves reported at 78% and 49% at 400 µg/mL, respectively, on carrageenan-induced hind paw edema for 5 h (Konuku et al., 2017); however, they highlight the therapeutic potential of M. zapota extracts. Similarly, Hossain et al. (2012) evaluated the methanolic extract of M. zapota root on carrageenan-induced hind paw edema for 4 h, observing anti-inflammatory effects of 59.7% at 400 mg/kg of body weight (Hossain et al., 2012).
The bioactive compounds such as flavonoids and polyphenols, known for their in vitro and in vivo anti-inflammatory properties, and tannins, which inhibit cyclooxygenase-1 and exhibit anti-phlogistic activity, are optimally extracted with solvents of intermediate polarity (Das & De, 2013). Thus, the anti-inflammatory mechanism of M. zapota could be attributed to the anti-phlogistic action of tannins, suggesting that M. zapota extracts could serve as a complementary natural anti-inflammatory agent, in line with Ayurvedic medicinal practices endorsed by the Pharmaceutical Association (Konuku et al., 2017). Other studies have shown that M. zapota leaf extracts exhibit significant anti-inflammatory activity, acting as a potential prostaglandin E2 (PGE2)-reducing agent (Hossain et al., 2012), inactivating pro-inflammatory enzymes such as cyclooxygenase (COX) and lysyl oxidase (LOX) and reducing nitric oxide (NO) production in mouse cells (Rivas-Gastelum et al., 2023).
Conclusion
This study has demonstrated that among the various extracts of M. zapota, the AE exhibited the highest antioxidant activity. The ABTS assay revealed statistically significant differences between the tested extracts, as this method allows the reaction with a wide range of antioxidants, including hydrophilic and lipophilic compounds. For this reason, the ABTS method was able to identify more differences compared to the DPPH method.
The EE was identified as the most potent in terms of anti-diabetic and anti-hypertensive activities, displaying the most significant enzymatic inhibition. Moreover, at a concentration of 5 mg/ear, EE also manifested superior anti-inflammatory effects. These observations suggest that AE likely harbors more phytochemicals with pronounced antioxidant capabilities. Meanwhile, EE, by extracting bioactive compounds of intermediate polarity, appears to enhance anti-diabetic, anti-hypertensive, and anti-inflammatory actions in comparison to AE and HE. Given these promising results, it is recommended that future research focus on isolating and characterizing novel anti-inflammatory molecules within M. zapota leaf extracts. Through such endeavors, M. zapota could emerge as a viable natural remedy for managing and potentially preventing conditions associated with DM and AHT, alongside mitigating inflammation.
Footnotes
Abbreviations
ABTS: 2,2′-Azino-bis (3-ethylbenzothiazoline-6-sulfonic acid); ACE: Angiotensin-converting enzyme; AE: Aqueous extract; AHT: Arterial hypertension; ANOVA: Analysis of variance; COX: Cyclooxygenase; COX-2: Cyclooxygenase-2; DM: Diabetes mellitus; DMSO: Dimethyl sulfoxide; DNS: 3,5-Dinitro-2-hydroxybenzoic acid; DPPH: 2,2-Diphenyl-1-picrylhydrazyl; EE: Ethanolic extract; GAE: Gallic acid equivalents; HE: Hydroalcoholic extract; LOX: Lysyl oxidase; NO: Nitric oxide; NSAIDs: Non-selective non-steroidal anti-inflammatory drugs; PGE2: Prostaglandin E2; ROS: Reactive oxygen species; TPA: 12-O-Tetradecanoylphorbol-13-acetate; TPC: Total polyphenol content.
Acknowledgments
None.
Authors’ Contributions
Conceptualization: A.C-L.; methodology, formal analysis, and investigation: A.T-M., J.J.A-F, N.dC.J-P., M.A.E-B., P.G-A., J.A.A-L.; software: A.T-M., A.C-L. supervision: A.C-L.; visualization: J.A.A-L., A.C-L., N.dC.J-P., F.A.G-A., P.G-A.; resources: A.C-L.; writing – original draft: A.C-L., A.T-M.; review and editing: J.A.A-L. All authors have read and agreed to publish this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The experimental protocol adhered strictly to the Mexican Official Standard NOM-062-ZOO-1999. All methods used in this study were conducted in accordance with relevant guidelines and regulations and approved by the Institutional Animal Care and Use Committee of “Comité para el Cuidado y Uso de Animales de Laboratorio (CCUAL-FM-UAEM)” with the project “Estandarización de modelos experimentales con enfermedades asociadas al síndrome metabólico: obesidad, diabetes e hipertensión” [Ethical approval no. (005/2018)]. This ensured the ethical treatment and welfare of the laboratory animals throughout the study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Consejo de Ciencia y Tecnología del Estado de Tabasco (CCyTET), through the “Programa para el Desarrollo por la Ciencia, la Tecnología y la Innovación del Estado” (PRODECTI-2020-01/018), with the project: “Evaluation of anti-inflammatory and immunomodulatory activity in vitro and in vivo of traditional medicinal plants cultivated in Tabasco, Mexico.”

