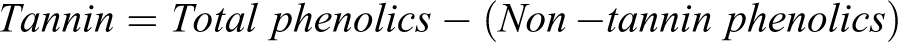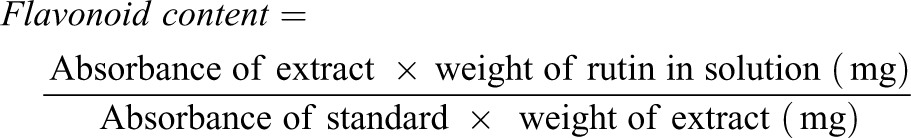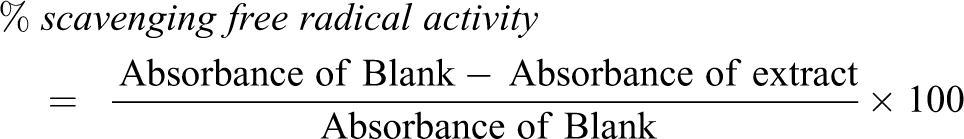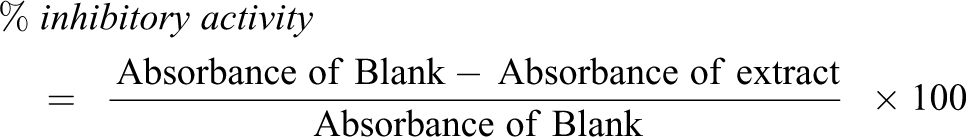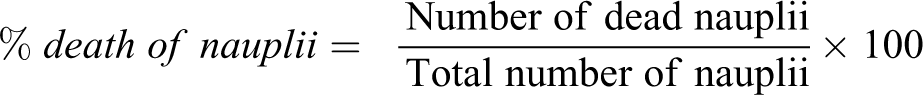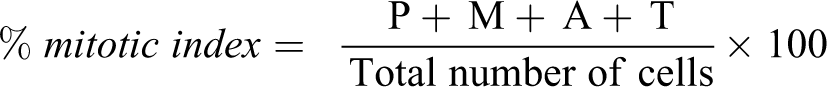Abstract
Health forecasters predict that cases of diabetes will double in 2030; hence proactive action is required to salvage this problem. Thus, this study was undertaken to evaluate the toxicological and anti-diabetic potential of n-hexane extract of T. bangwensis leaves on α-amylase and α-glucosidase activity. The phytochemical screening, antioxidant activity as well as the inhibitory effect of the plant extract was determined by UV-spectrophotometry method while brine shrimp and Allium cepa methods were used for the toxicity study. Preliminary phytochemical screening detected the presence of flavonoid, phenol, tannin, alkaloid and cardiac glycoside whereas phlobatanin, steroid, terpenoid and saponin were absent. The result also showed that flavonoid concentration was the highest compared to others. The 2,2-diphenyl-1-picrylhydrazine (DPPH) and nitric oxide (NO) results showed that the plant extract exhibited significant antioxidant activity particularly at the highest concentration (100 µg/ml). Brine shrimp lethality result showed that the highest mortality rate of nauplii and median inhibition concentration (IC50) are 97% and 7.46 ± 0.33 µg/ml respectively. Furthermore, the results also revealed that mitotic index, root growth length and mitotic division (cytotoxicity indicators) decreased as concentration increases. Finally, the results showed that the plant extract exhibited significant inhibitory effect on α-amylase and α-glucosidase activities at 100 µg/ml; nevertheless, the effect was higher on α-amylase than α-glucosidase activity. In summary, the significant antioxidant and inhibitory effects may be attributed to the presence of the phytochemicals mentioned above. It can therefore be concluded that T. bangwensis leaves may demonstrate potent anti-diabetic effect.
Introduction
The international diabetes federation (IDF) predicts that cases of diabetes mellitus which is the third killer disease threatening global population beside cancer and cardiovascular disease will double in 2030. 1,2 Diabetes mellitus is a chronic metabolic disorder or an endocrine systemic problem caused by either low insulin secretion and release, insulin resistance/insensitivity of pancreatic β-cells or insulin action to down-regulate blood glucose level resulting in hyperglycaemic condition thereby triggering several diabetic complications/abnormalities such as hyperglycaemia, hyperlipidaemia, ophthalmopathy, neuropathy, atherosclerosis, renal failure, ulceration, reproductive disorder to mention but a few. 3 –5 Furthermore, diabetes mellitus also occur due to imbalance between glycaemic index and glucose intolerance which increases the risk of individual’s progression to Type 2 diabetes (T2D) (also called non-insulin dependent diabetes mellitus (NIDDM) or maturity onset diabetes mellitus) and contributes approximately 90–95% of all cases of diabetes mellitus. 2,6
Since elevated blood glucose level is the major implicated factor of diabetes mellitus therefore inhibition of carbohydrate metabolizing enzymes such as alpha amylase and alpha glucosidase reduces the rate of glucose influx into the circulatory bloodstream thereby preventing postprandial rise/increase in blood glucose and invariably reducing drastically the effects associated with diabetes mellitus complications. 7,8 Several reports have established that oral hypoglycaemic drugs are effective in the treatment and management of cases of diabetes mellitus, however, they show undesirable adverse side effects such as gastrointestinal discomfort, oedema, renal failure, overweight and so forth. 9 –11 It is based on these toxicity outcomes that encouraged researchers to inquire about natural products with comparative advantage and little or no adverse side effect.
Some natural products have been isolated and screened which afterwards were found to be effective in the treatment and management of diabetes mellitus. 12 –14 Nevertheless, there is a need for further discoveries. Interestingly, studies showed that T. bangwensis leaf is ethno-medicinally beneficial as anti-inflammatory, hypolipidemic, antimicrobial, anticancer, hypotensive agent and so on. Mistletoe is a woody shoot parasite that belongs to the Loranthaceae family and consists of the genera namely: Phragmanthera, Oncocalyx, Tapinanthus, Plicosepathus, Globumetula, Agelanthus, Helixanthera, Berhauria, and Engderina. 15,16 Previous studies documented that Tapinanthus species such as aqueous-ethanol extract of Tapinanthus globiferus leaves and aqueous leaf extract of Tapinanthus butungii possess hypoglycaemic property. 17,18 However, information on the anti-diabetic property of T. bangwensis is scanty, therefore, the present study was undertaken to screen for the toxicological and anti-diabetic potential of n-hexane extract of T. bangwensis leaves so as to establish the safety status and therapeutic effect of the plant in the treatment of diabetes mellitus. African mistletoe (also known as T. bangwensis or Loranthus bangwensis) (Engl & K. Krause) Danser which is a citrus-dependent plant of African origin is called by the Hausas, Igbos and Yorubas as Kauci (Kanchi), Awurusie (or Apari) and Afomo onisana while the English acronyms are ‘all heal tree’ ‘bird lime’ or ‘tree of life’. 19
Materials and methods
Chemical reagents
Methanol, ethylacetate, n-hexane, sodium carbonate, (Na2CO3), Iron chloride (FeCl3), ammonium hydroxide, sodium hydroxide (NaOH) and hydrochloric acid (HCl) were bought from Chimex chemical Limited, Lagos, Nigeria. Ascorbic acid, Folin-ciocalteu reagent, gallic acid, rutin, α-amylase, α-glucosidase, 2,2-diphenyl-1-picrylhydrazine (DPPH), Dinitrosalicylic acid (DNSA), 4-nitrophenyl-d-glucopyroinoside (pNPG) acetic acid, orcein stain, picric acid, glacial acetic acid and polyvinyl polypyrrolidone (PVPP) were purchased from Sigma-Aldrich company, St Louis, USA while starch soluble was obtained from J. T. Baker Inc., Phillipsburg, USA
Authentication of plant material
Fresh leaves of T. bangwensis bought from Mushin market in Lagos were authenticated by Mr Adeleke of the Department of Pharmacognosy, University of Lagos. The unique characteristic that distinguished it from other Tapinanthus species is the presence of match-like flower with red/purple corolla tip. The plant leaves were then deposited in the University’s herbarium for documentation and a voucher number (LUH 3856) was assigned.
Extraction of plant material
The leaves of the plant were washed with distilled water and air-dried for 5days at 25–28°C. They were blended using electric blender and 1.5 kg powdered mass of T. bangwensis leaves was obtained. Approximately, 500 g of the powdered leaves of T. bangwensis was then extracted with 1.5 L of 100% methanol solvent. The content was left to stand and after 2days, it was filtered using Whatman filter paper and the filtrate concentrated by evaporation at room temperature. The final weight obtained was 40.85 g. Furthermore, approximately 25.0 g of methanol extract of T. bangwensis was extracted by solvent partitioned method using separating funnel in a resulting mixture of ethylacetate-water. The upper portion which was ethylacetate extract was collected, air-dried by evaporation and the mass yield was 10.2 g. Subsequently, 7.0 g of ethylacetate extract of T. bangwensis was further subjected to column chromatography extraction using 100% n-hexane as the eluting solvent. A total of five fractions in 100 ml bottle were collected and concentrated by evaporation at room temperature. The mass yield of solid n-hexane extract obtained was 1.58 g which was then used for this study (Figure 1).
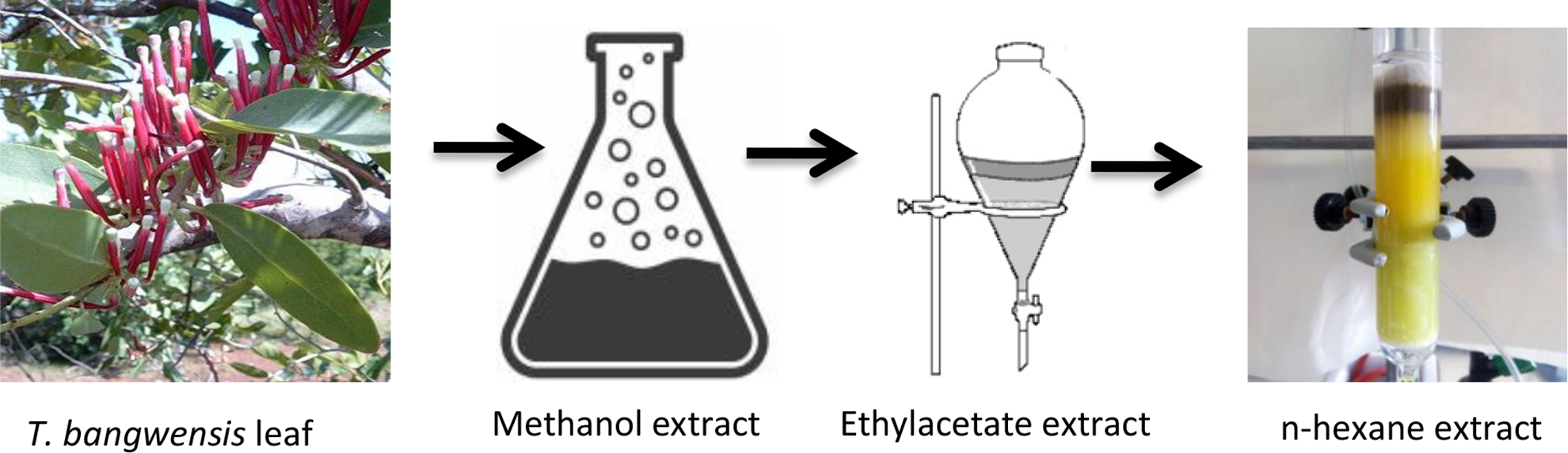
Schematic diagram of extraction of T. bangwensis.
Phytochemical screening of plant extract
Qualitative phytochemical screening
Preliminary phytochemical screening of alkaloid, terpenoid, saponin, tannin, flavonoid, steroid, phlobatanin phenol and cardiac glycoside were determined systematically using the method described by Geetha and Geetha. 20
Quantitative phytochemical screening of plant extract
Quantification of phenolic content
Total phenolic content was quantified using Folin-Ciocalteu reagent (FCR) described by Siddhuraju and Becker. 21 100 µl of 1 mg/ml of the extract dissolved in methanol was mixed with 750 µl of FCR (diluted in 10-fold) and allowed to stand for 5 mins at 25°C. 750 µl of 0.57 M Na2CO3 solution was then added to the mixture and after 60 mins, the absorbance was measured at 725 nm. The result obtained was expressed as gallic acid equivalent (mgGAE/100 g).
Quantification of tannin content
The tannin content was estimated by the method described by Siddhuraju and Becker. 21 Briefly, 1.0 ml of distilled water was added to 100 mg of polyvinyl polypyrrolidone (PVPP) followed by addition of 100 µl of 1 mg/ml of extract dissolved in methanol. The reaction mixture was vortexed for 15 mins at 4°C. After 10 mins, it was then centrifuged at 3000 rpm at 25°C. The absorbance of the supernatant which contains the simple phenolic was then measured at 725 nm. The result was expressed as non-tannin phenolic and the tannin content was calculated as follows:
Quantification of flavonoid content
Flavonoid content was determined according to the method described by Kumaran and Karunakaran. 22 100 µl of the extract in methanol (1 mg/ml) was mixed with 100 µl of 20% AlCl3 in methanol, followed by addition of a drop of acetic acid and then made up to 5 ml with methanol. After 40 mins, the absorbance was then taken at 415 nm. Blank sample contains 100 µl of extract, a drop of acetic acid and then made up to 5 ml with methanol. The absorption of standard rutin solution (0.5 mg/ml) in methanol was measured under the same condition. The amount of flavonoid in the extract in rutin equivalent was calculated as:
Quantification of alkaloid content
The alkaloid content in the plant extract was estimated by the method described by Herin et al. 23 Approximately, 0.2 g of the extract was weighed into a conical flask and 100 ml of 10% acetic acid in ethanol was added and allowed to stand for 4hours. The solution was then filtered and the filtrate obtained was concentrated on a water bath at 40°C. 1.0 ml 0.5 M NH4OH was added dropwise to the solution until a precipitate was formed. The precipitate was then collected, washed with dilute ammonium hydroxide and the content filtered. The residual sample which contains the alkaloid was dried and weighed.
Quantification of cardiac glycoside
The concentration of cardiac glycoside was quantified by the method of Yassa et al. 24 10% of extract was mixed with 10 ml freshly prepared Baljet’s reagent (95 ml of 1% picric acid and 5 ml of 10% NaOH solution). After 1hour, the mixture was diluted with 20 ml of distilled water and the absorbance was read at 495 nm. The standard curve was plotted by preparing 10 ml of different concentrations (12.5–100 mg/ml) of securidaside. Total cardiac glycoside was expressed as mg of securidaside per g of extract.
Antioxidant activity of the plant extract
2,2-Diphenyl-1-picrylhydrazine (DPPH) assay
The antioxidant property of n-hexane extract of T. bangwensis was evaluated using the DPPH free radical scavenging assay described by Nithiananthan et al. 25 and Zuraini et al. 26 Different concentrations of the extract were prepared (25, 50, 75 and 100 µg/ml) and 5 ml of 0.004% (w/v) solution of DPPH (or 4 mg of DPPH in 100 ml of methanol) was added. The solution obtained was vortexed and incubated at 25°C for 30 mins in a relatively dark place and afterwards the absorbance was measured at 517 nm. The blank contains 80% (v/v) methanol only. The absorbance was measured in triplicates for each concentration. Ascorbic acid was used for comparison as positive standard. DPPH scavenging free radical activity was calculated as:
Nitric oxide antioxidant assay
The antioxidant activity of the plant extract using nitric oxide assay was determined by the method described by Alisi and Onyeze. 27 Briefly, 5.0 ml of the reaction mixture which contains 5 mM sodium nitroprusside (SNP) in phosphate buffered saline (pH = 7.3) with or without the extract at different concentrations (25, 50, 75 and 100 µg/ml) was incubated at 25°C for 180 mins under a polychromatic light source. The nitric oxide free radicals generated interact with oxygen to produce the nitrite ion which was assayed at 30 min intervals by mixing 1.0 ml of incubation mixture with an equal amount of Griess reagent (1% sulphanilamide in 5% phosphoric acid and 0.1% naphthylethylene diaminedihydrochloride). The absorbance of the chromophore formed (purple azo dye) was measured at 546 nm in triplicates for each concentration and the percentage inhibition calculated as:
Toxicological study of the plant extract
Brine shrimp Lethality assay
The brine shrimp lethality assay used in this study was that described by Geethaa et al. 28 Artemia salina eggs were obtained from the Aquatic animal research centre, University of Lagos while salt water was collected from the Bar beach in Victoria Island, Lagos. 300 eggs were placed into 500 ml of salt water in a 1000 ml volumetric flask. After 36 hrs, the larvae (nauplii) hatched at 25°C in an aerated condition and continuous illumination. The different concentrations of the plant extract (10, 100 and 1000 µg/ml), and control (salt water) were prepared in test tubes and 10 nauplii were added into each of the test tube. After 24 hrs, the number of dead nauplii was counted under an intensive illumination. The percentage death was calculated as below while inhibition concentration (IC50) was determined.
Cytotoxicity study using Allium cepa assay
The cytotoxic effect of n-hexane extract of T. bangwensis leaves using Allium cepa was determined by the method of Fiskesjo. 29 Thirty pieces of onion bulbs (Allium cepa Linn) were used. The outer scales were removed carefully before initiating the test to avoid destroying the root primordia. The onion bulbs were then placed in tap water and after 24 hours the five best growing bulbs were exposed to the test solutions (0.01, 0.03, 0.06 and 0.1 mg/ml) for 4days in the dark at 25°C. The root growth length were then measured 24hourly using a ruler. For chromosomal aberration evaluation 5–7 mm of the onion root tips were excised and fixed in ethanol-glacial acetic acid solution (3:1 v/v). After 60 mins, the root tips were then hydrolysed in 1 N hydrochloric acid at 60°C and subsequently rinsed with distilled water. The root tip was then squashed in a glass slide and stained with 2% aceto-orcein for 10 mins. It was covered carefully with a slide cover to avoid air bubbles and sealed with fingernail polish. Ten slides were prepared for each concentration and the control and were then viewed under a microscope mounted on a camera. The mitotic index was then calculated as:
where P, M, A and T represents the number of dividing cells in prophase, metaphase, anaphase and telophase respectively.
Inhibitory effect of the plant extract
Inhibitory effect on α-amylase activity
The inhibitory effect of n-hexane extract of T. bangwensis on α-amylase was determined by the method of Kazeem et al. 30 A 250 µl of the extract/acarbose at different concentrations (25, 50, 75 and 100 µg/ml) were incubated with 500 µl of 2U/ml α-amylase in 100 mM phosphate buffer (pH = 6.8) for 20 mins at 37°C. 250 µl of 1% starch dissolved in 100 mM phosphate buffer was then added to the reaction mixture and incubated for 1hour at 37°C. 500 µl of dinitrosalicylic acid (DNSA) (colour forming agent) was then added and boiled for 10 mins. The mixture was cooled and diluted with 5 ml of distilled water. The control was prepared in the same manner as the test sample with distilled water replacing the extract. The absorbance was measured at 540 nm and the inhibitory activity calculated as:
Inhibitory effect on α-glucosidase activity
The inhibitory effect of n-hexane extract of T. bangwensis was determined by the method of Kazeem et al. 30 A 250 µl solution of the extract/acarbose at different concentrations (25, 50, 75 and 100 µg/ml) were incubated with 500 µl of 1.0U/ml α-glucosidase solution in 100 mM phosphate buffer (pH = 6.8) at 37°C for 15 mins. 5000 µl of 3 mM of 4-nitrophenyl-d-glucopyronoside (pNPG) solution in 100 mM phosphate buffer was added and the mixture was further incubated at 37°C for 25 mins and 5% w/v Na2CO3 was added. The mixture was cooled at 25°C and 5 ml of distilled water added and vortexed for 10 mins. The control contained all the component of the test sample except the extract. The absorbance of the released p-nitrophenol was measured at 405 nm and the percentage inhibitory activity calculated as:
Statistical analysis
All data obtained from this study were statistically expressed as Mean ± SD. The significant difference was compared with ANOVA and Tukey’s post hoc test while the confidence limit was considered at p < 0.05.
Results
Preliminary phytochemical screening of n-hexane extract of T. bangwensis revealed the presence of flavonoid, phenol, tannin, alkaloid and cardiac glycoside while phlobatanin, steroid, terpenoid and saponin were not detected (Table 1). Furthermore, the result also showed that the concentration of these phytochemical compounds were remarkable, nonetheless, flavonoid concentration was the highest compared to other phytochemicals that were quantified. (Figure 2)
Qualitative Phytochemical Screening of n-hexane extract of T. bangwensis.

Abbreviation: (+) indicates detected while (–) indicates not detected.
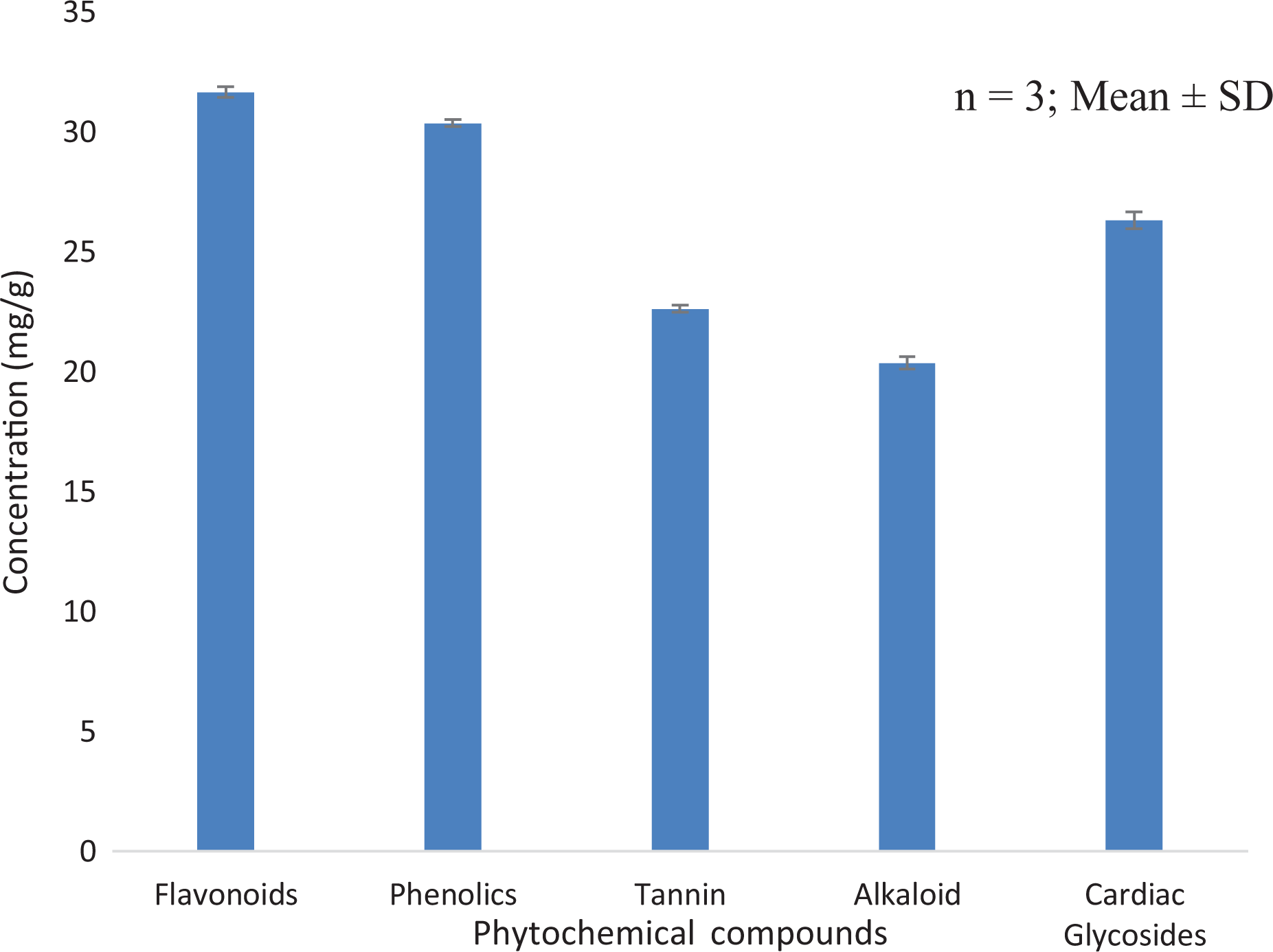
Concentration of phytochemical compounds in n-hexane extract of T. bangwensis.
The results of the antioxidant activity of the plant extract using 2,2-diphenyl-1-picrylhydrazine (DPPH) and nitric oxide assays showed that the plant extract exhibits significant antioxidant activity with the highest inhibition of DPPH free radicals observed at the highest concentration (100 µg/ml) (Figure 3 and 4). However, the antioxidant activity of ascorbic acid was higher compared to that of the plant extract in both assays.
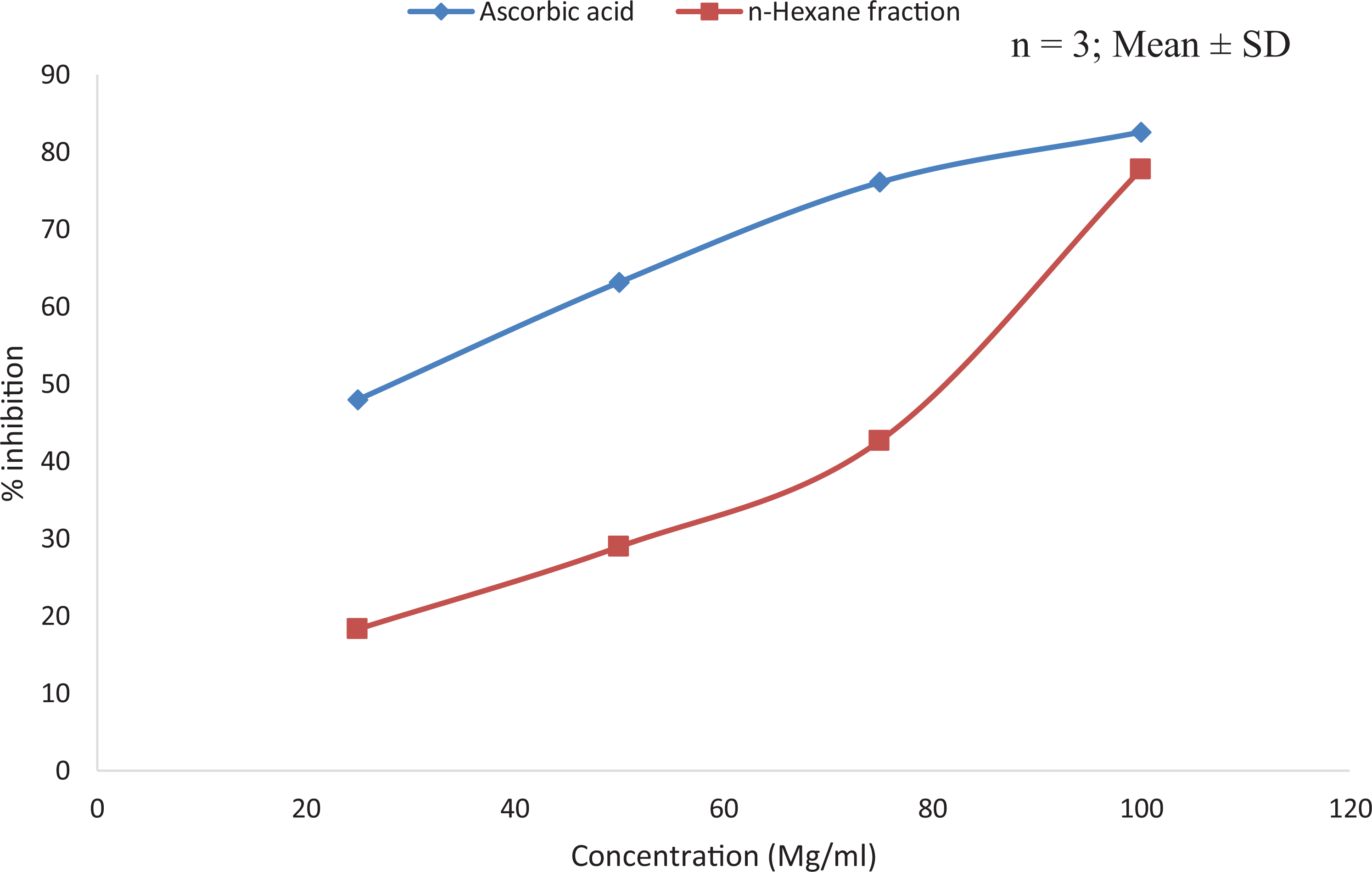
Antioxidant activity of n-hexane extract of T. bangwensis using 2,2-diphenyl-1-picrylhydrazine antioxidant model (DPPH).
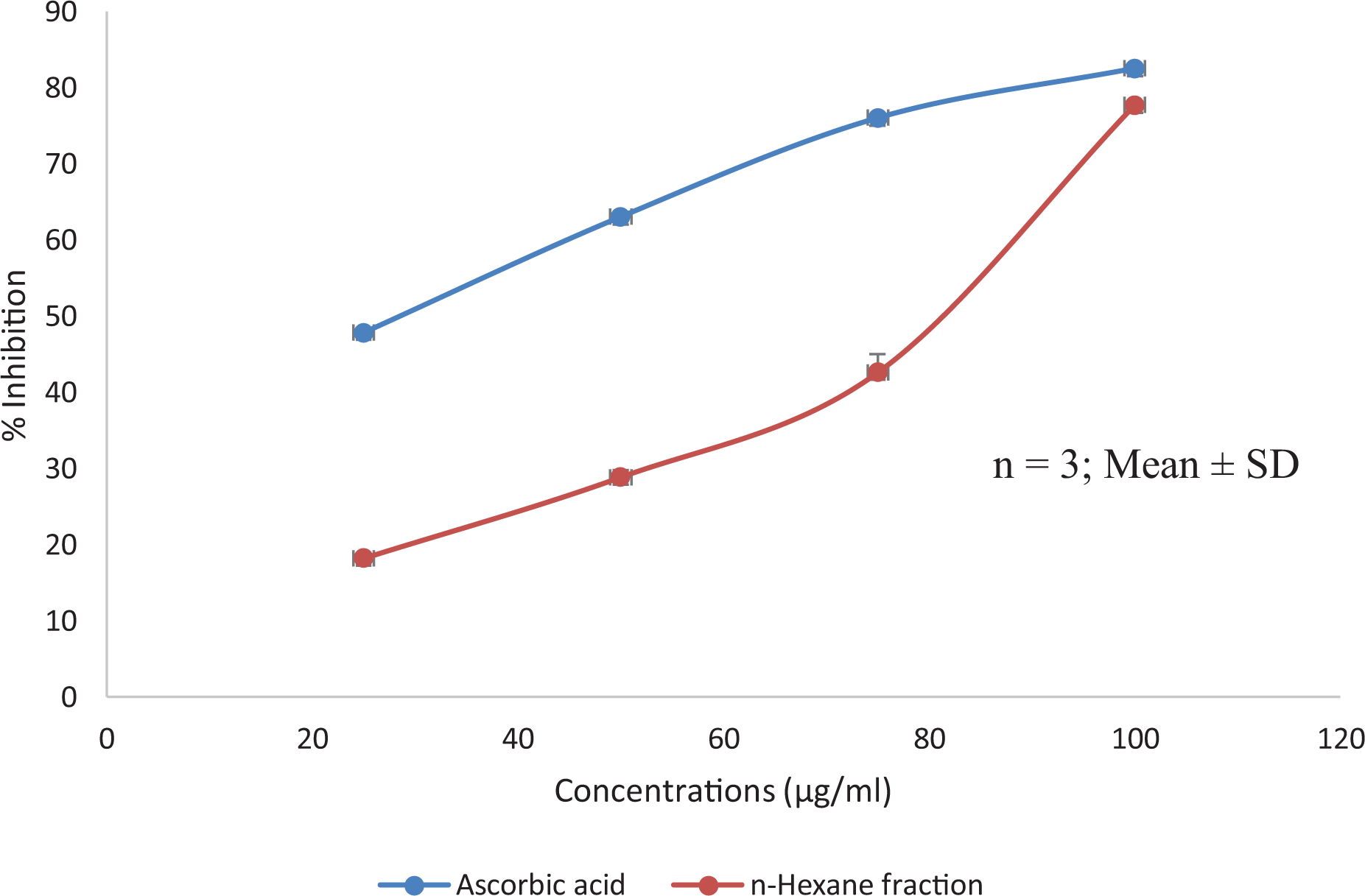
Antioxidant activity of n-hexane extract of T. bangwensis using nitric oxide antioxidant model.
Table 2 and 3 shows the results of the toxicological effect of the plant extract of T. bangwensis using Brine shrimp lethality assay and Allium cepa test. The brine shrimp lethality result showed toxic effect characterized by increase in nauplii mortality rate as concentration increases with the highest mortality rate (97%) observed at 1000 µg/ml. The median inhibition concentration (IC50) was found to be 7.46 ± 0.33 µg/ml (Table 2). There was significant difference between the normal control (saltwater) and the plant extract at different concentrations at p < 0.05. Also, the result of the Allium cepa test showed evidence of cytotoxicity such as decrease in root growth length (Figure 5) as well as decrease in mitotic index and mitotic division (Table 3) as concentration increases compared to the control (distilled water). From Table 3, it was observed that there was significant difference between the control (distilled water) and the plant extract at different concentrations at p < 0.05. The predominant chromosomal aberrations observed from Table 3 were bridged and sticky chromosomes compared to other chromosomal aberrations while laggard and multipolar chromosomes were absent. The various chromosomal aberrations are shown in Figure 6.
Toxicological effect of n-hexane extract of T. bangwensis using Brine shrimp model.
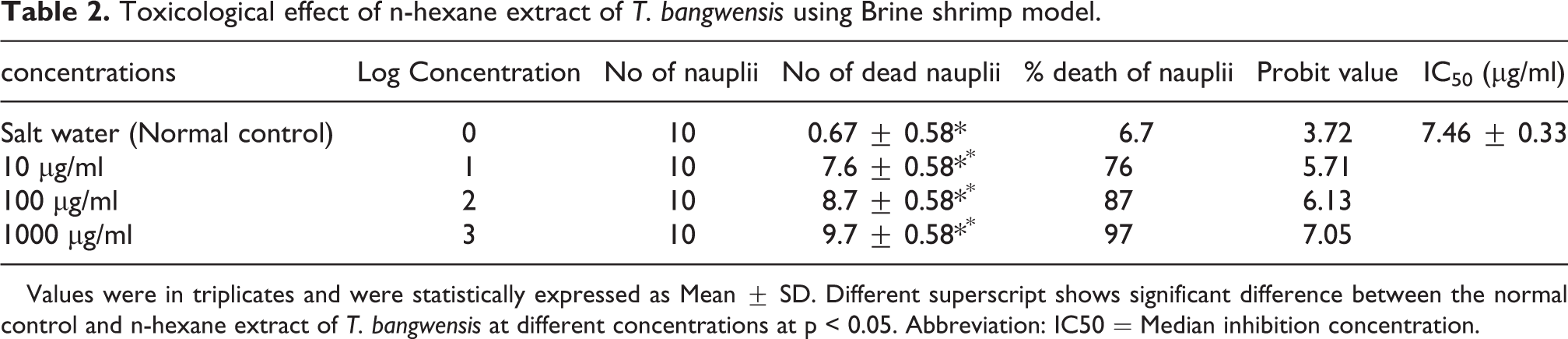
Values were in triplicates and were statistically expressed as Mean ± SD. Different superscript shows significant difference between the normal control and n-hexane extract of T. bangwensis at different concentrations at p < 0.05. Abbreviation: IC50 = Median inhibition concentration.
Cytotoxic effect of n-hexane extract of T. bangwensis using Allium cepa test.
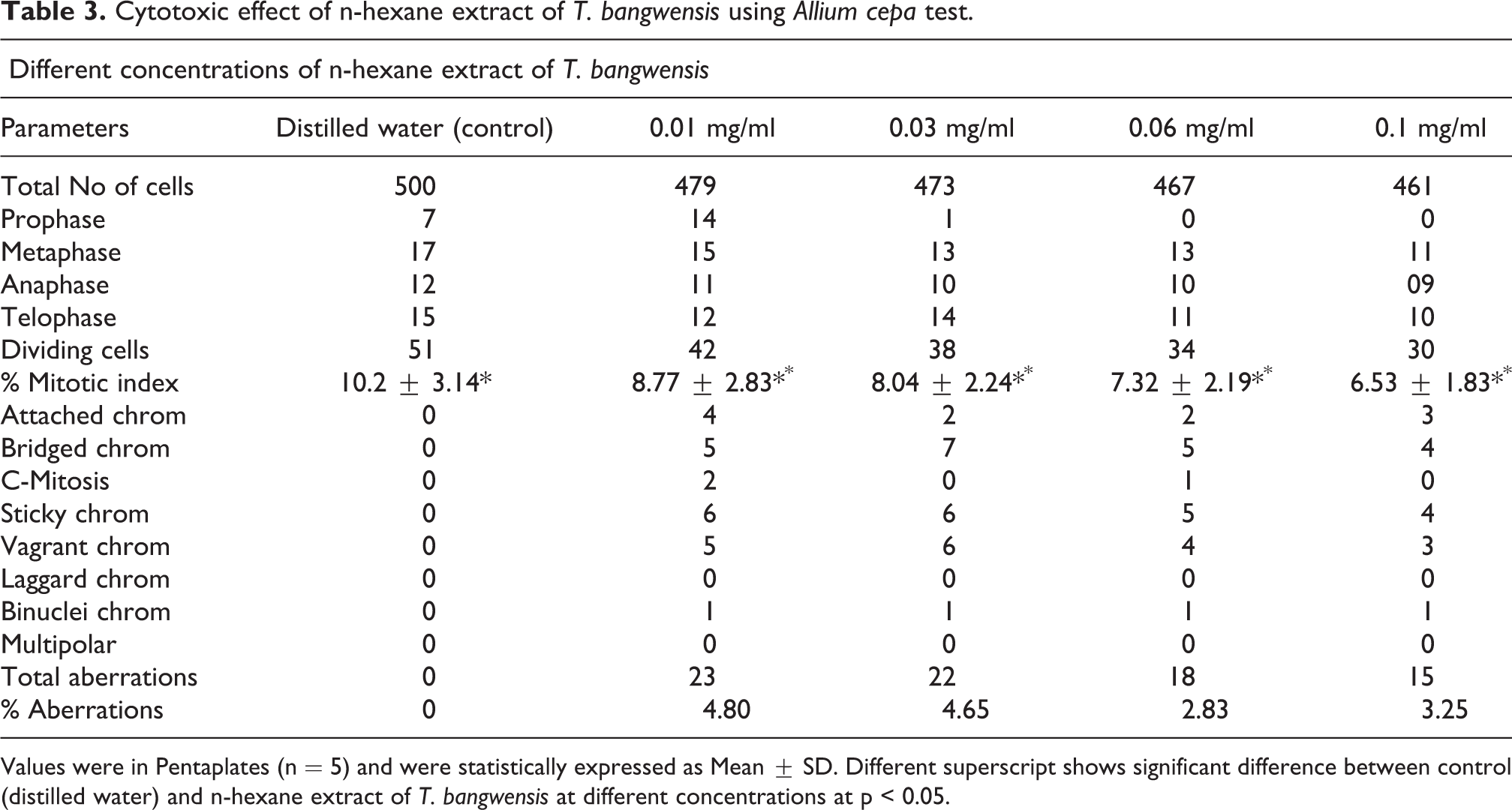
Values were in Pentaplates (n = 5) and were statistically expressed as Mean ± SD. Different superscript shows significant difference between control (distilled water) and n-hexane extract of T. bangwensis at different concentrations at p < 0.05.
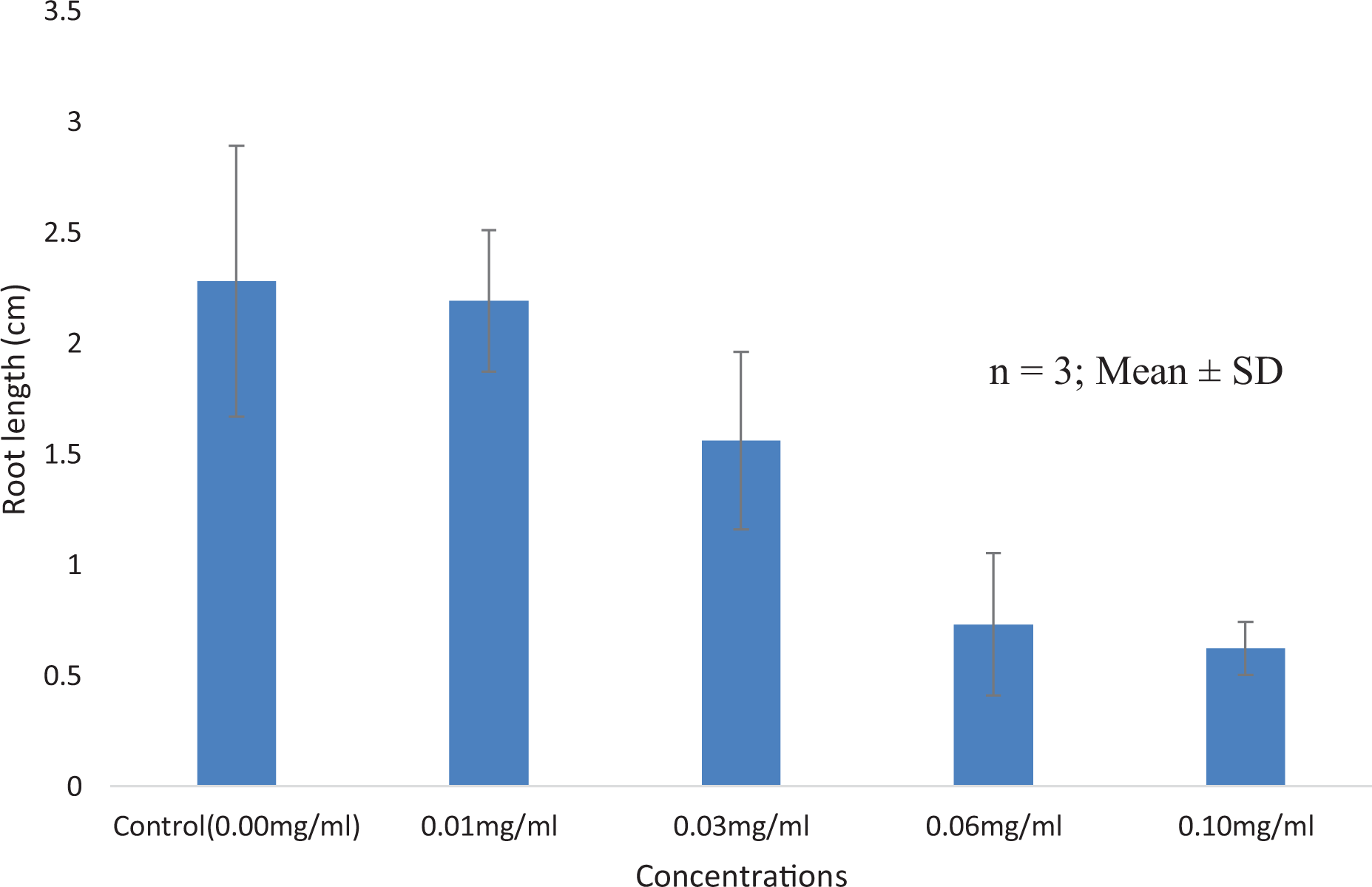
Effect of n-hexane extract of T. bangwensis on root growth length on Allium cepa.
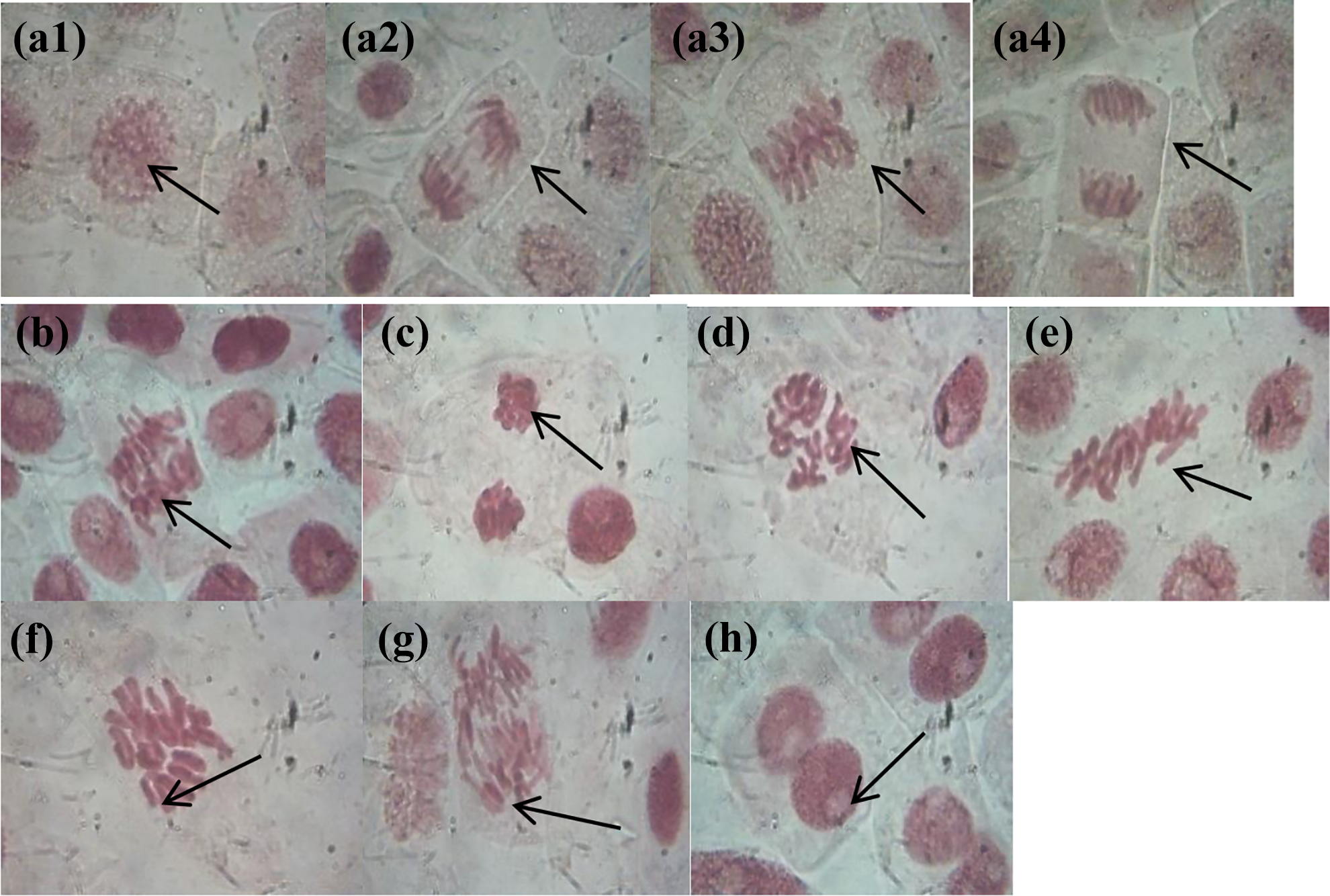
Photomicrograph of different chromosomal aberrations showing the effect of n-hexane extract of T. bangwensis leaves. Abbreviations: a1 = Prophase a2 Anaphase, a3 Metaphase, a4 Telophase, b = Bridged Anaphase, c = Sticky Telophase, d = C-Mitosis, e = Attached chromosome, f = Vagrant chromosome, g = Bridged Telophase and h = Binuclear chromosome.
The results of the inhibitory effect on α-amylase and α-glucosidase activities demonstrate that the plant exhibited significant inhibitory effects on both α-amylase and α-glucosidase activities particularly at the highest concentration (100 µg/ml). On the other hand, the inhibitory effect was higher on α-amylase activity compared to α-glucosidase activity. The median inhibition concentration (IC50) determined shows that the inhibitory effect of the plant extract on α-amylase activity was 59.15 ± 0.21 µg/ml while that of α-glucosidase was 73.29 ± 0.52 µg/ml. Meanwhile, the inhibition concentration at 90% (IC90) on α-amylase activity was 122.30 ± 0.45 µg/ml whereas that of α-glucosidase activity was found to be 139.52 ± 1.45 µg/ml. Nonetheless, the inhibitory activity of acarbose (standard drug) was higher compared to the plant extract. The IC50 of acarbose on α-amylase and α-glucosidase activities are 35.42 ± 0.22 µg/ml and 40.51 ± 0.11 µg/ml while the IC90 for α-amylase and α-glucosidase activities are 87.88 ± 0.20 µg/ml and 89.90 ± 0.34 µg/ml respectively. The results also showed significant difference between the control (acarbose) and n-hexane extract of T. bangwensis at p < 0.05 (Tables 4 and 5).
Inhibitory effect of n-hexane extract of T. bangwensis on α-amylase activity.
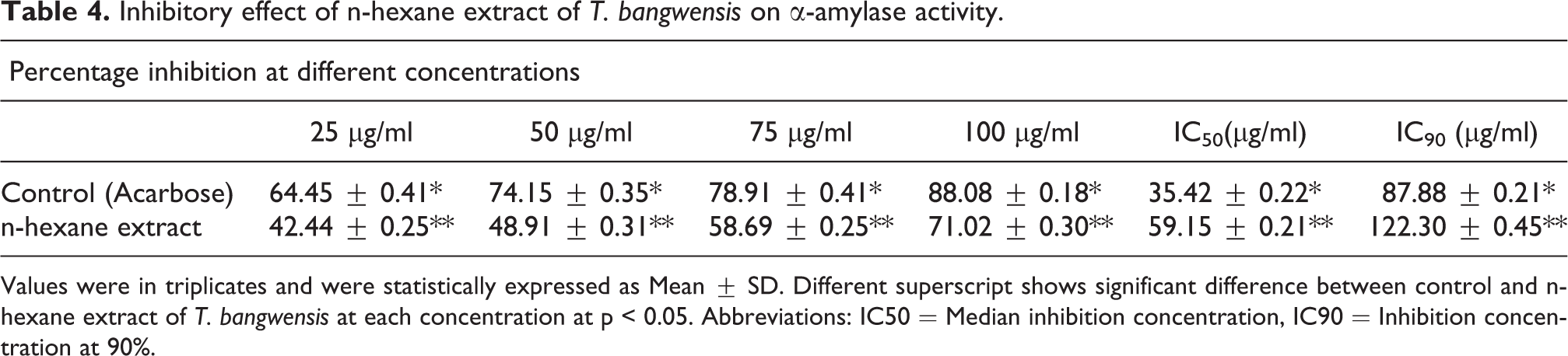
Values were in triplicates and were statistically expressed as Mean ± SD. Different superscript shows significant difference between control and n-hexane extract of T. bangwensis at each concentration at p < 0.05. Abbreviations: IC50 = Median inhibition concentration, IC90 = Inhibition concentration at 90%.
Inhibitory effect of n-hexane extract of T. bangwensis on α-glucosidase activity.
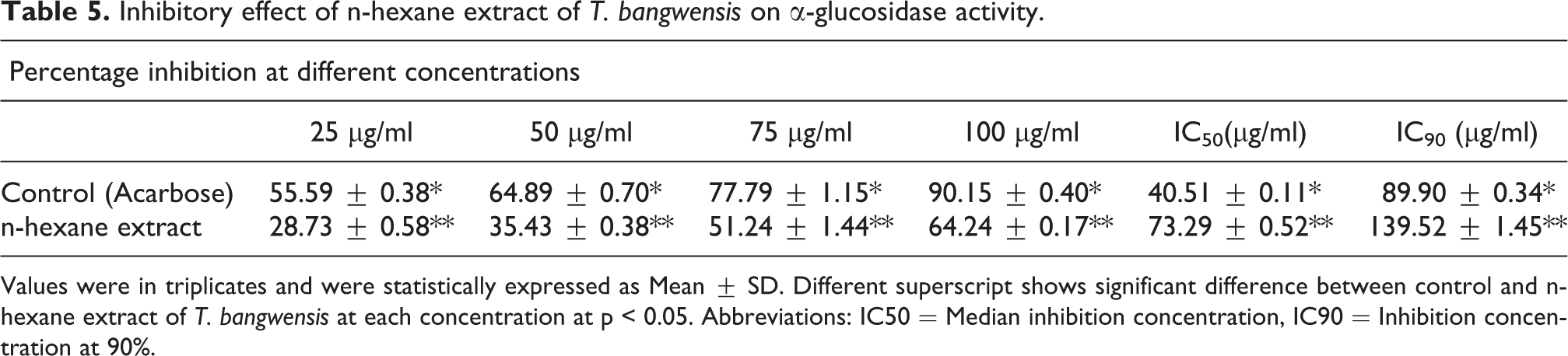
Values were in triplicates and were statistically expressed as Mean ± SD. Different superscript shows significant difference between control and n-hexane extract of T. bangwensis at each concentration at p < 0.05. Abbreviations: IC50 = Median inhibition concentration, IC90 = Inhibition concentration at 90%.
Discussion
The present study was undertaken to evaluate the toxicological and anti-diabetic potential of n-hexane extract of T. bangwensis leaves as a possible alternative remedy for the treatment and management of hyperglycaemic conditions. Phytochemical screening revealed the presence of flavonoid, phenol, tannin, alkaloid and cardiac glycoside. Studies have shown that the mechanistic action of phytochemical compounds as antioxidant agent is basically either by inhibiting/reducing oxidant species (free radicals) generation or stabilizing their effect by donating electron for them to attain a stable configuration. 31,32 For instance, flavonoid and phenolic compounds scavenge free radicals by transferring hydrogen to the electron deficient molecule (free radical) thereby transforming it into a stable molecule. 33 The remarkable antioxidant activity exhibited by the plant extract may be attributed to the high amount of flavonoid and phenol content. Therefore, the positive correlation obtained between antioxidant activity and high flavonoid and phenol content in this study is in agreement with previous research finding. 34
An investigation into toxicity study using Ames/salmonella test, cellular response test, micronucleus test, chromosomal aberration assay, cytotoxicity/genotoxicity assay, chronic and sub-chronic assay as well as brine shrimp lethality and Allium cepa test reveals that many plants used as food or in traditional medicine contain mutagenic, cytogenic, genotoxic and carcinogenic agents. 35,36 –39 The brine shrimp lethality assay which is a useful preliminary bioassay (or first-line toxicity tool) used for evaluating the toxicological effect of fungal toxins, plant extracts, heavy metals, pesticides, effluents was developed by Michael et al and one advantage of this assay is its rapidity, inexpensiveness and simplicity. 40 –42 It is noteworthy that the results obtained from this assay is generally acceptable and correlates with cytotoxic and anti-tumour properties. 43 Furthermore, the Allium cepa model studies the cytotoxic, inhibitory/mitodepressive and chromosomal aberration (abnormalities/changes in the DNA structure) effects which probably could be due to either chromatin dysfunction, disturbance in the cell cycle induced by the interaction of alkaloid with the DNA the inhibition of DNA synthesis or blocking of the G2 phase of the cell cycle from entering mitosis or chromatin dysfunction (DNA interaction). 44 –46 For instance, chromosome fragments indicate chromosome breaks and could be a consequence of anaphase/telophase bridges. 47 Subsequently, sticky anaphase/telophase chromosome observed in this study may be attributed to the influence of mutagens on the physical chemical properties of DNA, protein or formation of complexes with phosphate groups in DNA which leads to loss in the normal chromosomal appearance thereby resulting in ‘stick-surface’ causing chromosome agglomeration. 48 –52 This suggest that the plant extract may possess mutagenic potential which may possibly outweigh its therapeutic potential. The results of the brine shrimp lethality and Allium cepa test demonstrate that n-hexane extract of T. bangwensis leaves shows cytotoxic effect characterized by high mortality rate and decrease in root growth length, mitotic index (%MI) and mitotic division respectively.
Several strategic approaches have been explored in the treatment and management of diabetes mellitus such as stimulation of adenosine monophosphate dependent-protein kinase (AMPK), blockage of ATP gated potassium ion channels in β cells, stimulation of peroxisome proliferator-activated receptors activities (PPAR γ), glucagon-like peptide-1 (GLP-1) modulation among others. 11,53,54 These mechanisms of actions are directed at either enhancing insulin secretion/release, insulin sensitivity or reducing glycolysis by the liver. Currently, a new approach has been developed in the management of postprandial hyperglycaemia (PPH) by inhibiting α-amylase and α-glucosidase activity. The reduction in postprandial hyperglycaemia is important because it reduces advanced glycation and products formation which has been identified as a major risk factor for cardiovascular complications of diabetes mellitus. 55 Previous studies reported that the inhibition of these carbohydrate digesting enzymes may be beneficial to diabetic patients with impaired insulin tropic response particularly when used in combination with other hypoglycaemic agents. 56 Interestingly, the present study produced a significant inhibitory effect against the activities of α-amylase and α-glucosidase particularly at the highest concentration (100 µg/ml) and the results obtained is in agreement with other findings. 57,58 The inhibitory effect observed may be due to high flavonoid, phenol and alkaloid content which agrees with previous findings. Furthermore, the inhibitory action may probably be either due to the protective role of antioxidant phytochemicals against attack by free radicals on pancreatic/intestinal membrane thereby preventing elevated enzyme activity or increase in the concentration of the plant extract resulting in increase in the inhibition of the enzymes activities. 59,60,56
Conclusion
The present research findings showed that n-hexane extract of T. bangwensis leaves exhibited significant antioxidant activity and inhibitory effect on α-amylase and α-glucosidase activity particularly at 100 µg/ml. Furthermore, the antioxidant and inhibitory effects observed in this study may be attributed to high concentration of the phytochemical compounds mentioned above. It can, therefore, be concluded that T. bangwensis leaves may demonstrate potent anti-diabetic property. Within the scope of the toxicological models used, the results suggest that the plant extract may be cytotoxic.
Footnotes
Acknowledgements
We acknowledge the technical expertise of Mr Obu Francis and Mr Sunday Adenekan, Department of cell Biology and genetics and Department of Biochemistry respectively. All of the University of Lagos. Appreciation also goes to Ndam Shaifa, Department of Biochemistry and Forensic Science, Nigeria Police Academy, Wudil., Kano.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The ethical approval for the use of Artemia salina eggs in this research work was obtained from the ethics committee for the care and use of laboratory animals of Nigeria Police Academy, Wudil, Kano, Nigeria.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.