Abstract
An efficient bioactive tracking separation strategy based on liquid-liquid extraction and high-speed counter-current chromatography (HSCCC) was developed and used to isolate bioactive natural products from the endophytic fungus Chaetomium globosum residing in Ginkgo biloba. Using HSCCC, the novel metabolite chaetoglobol acid (
Diabetes mellitus is a major chronic disease. 1 The incidence and prevalence of diabetes have risen sharply in recent years, and it is predicted that 642 million people might suffer from diabetes in 2040. 2,3 α-amylase catalyzes digestion of carbohydrates by hydrolyzing α-(1, 4)-glycosidic linkages and producing maltose and glucose from starches. α-glucosidase is a type of glycoside hydrolase, which is involved in the absorption of carbohydrates. Inhibiting the activity of these enzymes will delay the absorption of carbohydrates in the small intestine and decrease postprandial blood glucose levels. 4,5 Moreover, the inhibition of α-glucosidase also has positive effects of treating diseases such as virus disease, cancers, chronic heart failure, etc. 6,7 Nowadays, α-glucosidase inhibitors have been recognized as an efficient therapy in the treatment of type 2 diabetes. 8 The inhibition of membrane-bound α-glucosidases that reside in the cell lining of the small intestine decreases the digestion of starch and additional dietary sugars to the blood. This aims to prevent hyperglycemia and maintain normal blood sugar levels. 9 Therefore, the discovery of α-glucosidase inhibitors is significant in the development of treatments for type 2 diabetes.
Natural resources, especially endophytes, have proven to be an important source of structurally unique and bioactive secondary metabolites (eg, anticancer, antimicrobial, antifungal, allelopathic, anti-insect, and α-glucosidase inhibition activities). 10 -15 The Chaetomium fungi are the largest genus of saprophytic ascomycetes, with more than 350 species. Up till now, more than 200 metabolites with a wide range of bioactivities have been isolated from the genus Chaetomium. However, the number of identified species outweighs the number of isolated and characterized compounds, suggesting that the Chaetomium genus remains a rich source of potential bioactive secondary metabolites that are waiting to be discovered. 16
In the past, a number of separation techniques including liquid-solid and liquid-liquid have been developed for the isolation and purification of natural drug candidates. 17 More recently, high-speed counter-current chromatography (HSCCC) has emerged, as a unique liquid–liquid partition chromatography-based technique for resolving complex natural products. HSCCC achieves efficient separation without complications such as irreversible solute adsorption, contamination, reaction and deactivation from interactions with support matrix, all of which are common problems in typical chromatographic separation techniques. 18,19 In addition, CCC can support a larger stationary phase area compared to other liquid chromatography (LC) methods (eg. high-performance liquid chromatography; HPLC). A larger stationary phase allows for a more selective separation of some targeted compounds. 20
In our continuous screening for bioactive metabolites from Chaetomium sp,
12,13,21
-23
we found potential α-glucosidase inhibitory fractions from C. globosum crude samples. Thus, to obtain the α-glucosidase inhibitory constituents from C. globosum for further pharmacological investigation, using an efficient method combining liquid-liquid extraction with HSCCC, 1 new compound (
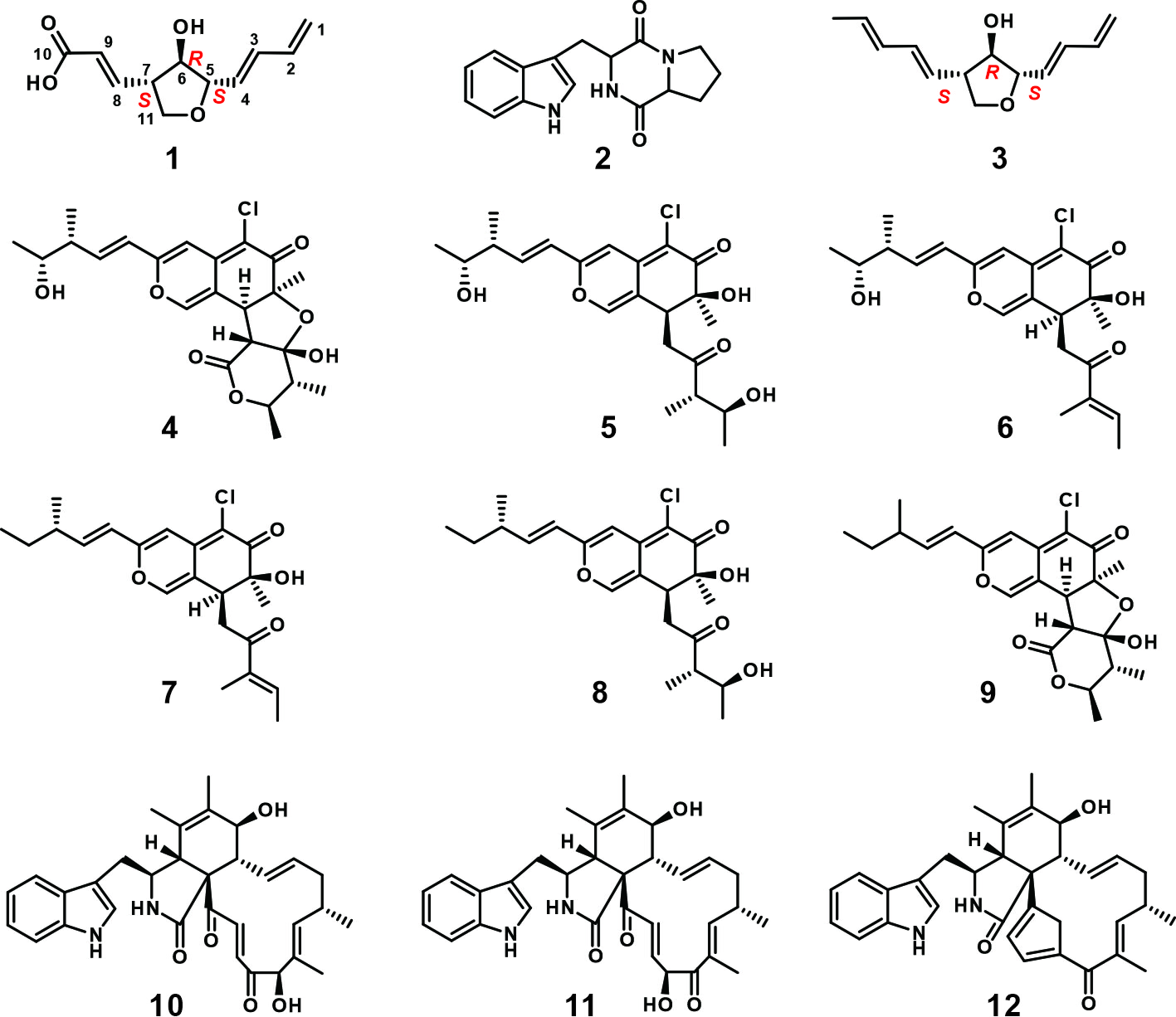
Chemical structures of the isolated compounds 1
Results and Discussion
α-Glucosidase and α-Amylase Inhibition Activity Tracking Liquid-Liquid Extraction and HSCCC Isolation
Two-phase solvent systems composed of 4 solvents were used in liquid-liquid extraction, and thus the components in the treated samples could be fractioned based on different polarities according to their K values.
24
A series of solvent systems based on hexane-ethyl acetate-methanol-water mixed at different volume ratios (9:1:1:9, 7:3:1:9, 5:5:1:9, 3:7:1:9, and 1:9:1:9) was selected for liquid-liquid extraction pretreatment of the crude samples of C. globosum. Fractions

HPLC chromatograms of fraction C (
K
a
of Compounds 1
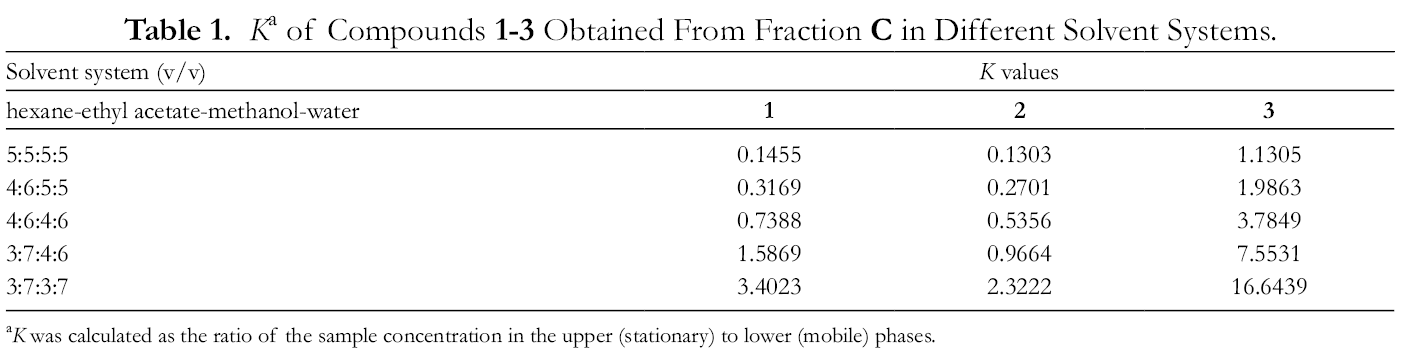
a K was calculated as the ratio of the sample concentration in the upper (stationary) to lower (mobile) phases.
Structure Elucidation
Twelve compounds were isolated and purified from C. globosum, including 1 new compound named chaetoglobol acid (
Chaetoglobol acid (
1H NMR (500 MHz) and 13C NMR (125 MHz) Spectroscopic Data for

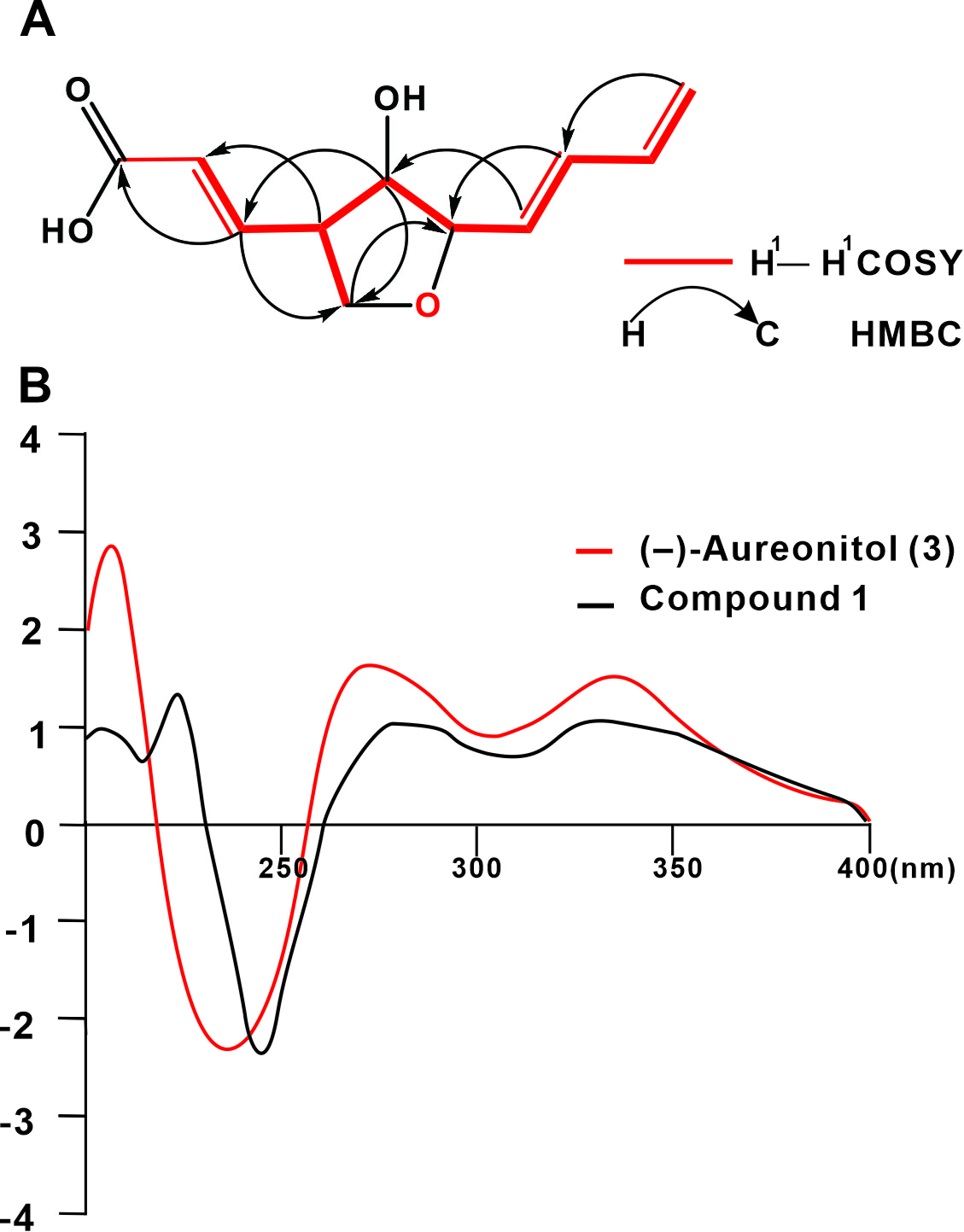
1H-1H Correlated Spectroscopy (COSY) and key Heteronuclear Multiple-Bond Correlation (HMBC) correlations of compound 1 (
(–)-aureonitol (
In Vitro α-Glucosidase and α-Amylase Inhibition
α-glucosidase and α-amylase inhibitory activities of compounds

Molecular docking of compound
α-Glucosidase and α-Amylase Inhibition.

In conclusion, in this study, 12 metabolites (
Materials and Methods
General
NMR spectra were obtained on Bruker Avance 400 MHz and Bruker Avance III 500 spectrometers with tetramethylsilane as an internal standard at room temperature. High resolution (HR) Electrospray Ionization Mass Spectroscopy (ESIMS) were recorded on a Thermo Fisher Scientific Q-TOF mass spectrometer. ESIMS were recorded on a Thermo Fisher LTQ Fleet instrument spectrometer. Electronic Circular dichroism (ECD) spectra were obtained on a Chirascan spectrometer. Silica gel (300, 400 mesh, Qingdao Marine Chemical Ltd., People’s Republic of China) and RP-18 gel (20, 45 µM, Fuji Silysia Chemical Ltd., Japan) were used for column chromatography (CC). Semi-preparative HPLC was performed on a Waters 1525EF liquid chromatography system equipped with Hypersil BDS C18 column (4.6 mm × 250 mm; 10.0 mm × 250 mm). The HSCCC experiment was conducted on a TBE-300B HSCCC system (Tauto Biotech, Shanghai, China), equipped with 3 polytetrafluoroethylene (PTFE) preparative coils (i.d. of the tubing = 1.6 mm, total volume = 300 mL) and a 20 mL sample loop. The revolution radius of the apparatus was 5 cm, and the β values of the multilayer coil ranged from 0.5 (at the internal terminal) to 0.8 (at the external terminal). For the apparatus, β = r/R, where r is the distance from the coil to the holder shaft, and R is the revolution radius or the distance between the holder axis amniocentesis of the centrifuge. Apparatus revolution speed was regulated with a speed controller within the range of 0 to 1200 rpm. The system was equipped with a model TBP5002 constant-flow pump (Tauto Biotech, Shanghai, China), a model UV-500 detector (XUYUKJ Instruments, Hangzhou, China) operating at 254 nm, and a model N2000 workstation (Zhejiang University, Hangzhou, China). A DC-0506 constant temperature-circulating implement (Shanghai Shunyu Hengping Instruments, Shanghai, China) was used to adjust the experiment temperature. Baker’s yeast α-glucosidase, hog pancreas α-amylase, nitrophenyl α-D-glucopyranoside (pNαGP), 2-chloro-4-nitrophenyl α-D-maltotrioside (G3-CNP), acarbose, and genistein were purchased from Sigma-Aldrich (St. Louis, MO).
Fungal Materials and Fermentation
The fungal strain C. globosum TY1, separated from the fresh bark of a medicinal plant Ginkgo biloba, was donated by Prof. J.C. Qin, College of Plant Science, Jilin University. 12,13 After growing on Potato Dextrose Agar (PDA) medium at 25 °C for 7 days, the mycelium of this strain TY1 was inoculated in PD liquid medium. The pH was adjusted to 6.0 prior to autoclaving. Fermentation was carried out in multiple 500-mL flasks containing 150 mL media on rotary shakers at 220 rpm/min, 25 °C for 14 days.
Measurement of Partition Coefficient (K)
The partition coefficients (K) for target compounds were determined by an HPLC analysis according to the previous study. 24 In brief, a small amount (1 mg) of the crude sample was dissolved into equal volume (0.8 mL) of the aqueous phase (lower phase) and organic phase (upper phase) of the thoroughly equilibrated 2-phase solvent system in a 2-mL test tube. After the equilibration was established, both the upper phase and lower phase were analyzed by HPLC and the peak areas of each compound in the upper phase and lower phase were recorded as A1 and A2, respectively. The partition coefficient (K) was then calculated by the following equation: K = A1/A2.
Extraction and Isolation
The culture broth (42 L) together with 5 L MeOH mycelium extraction was filtered by 8 layers of sterile gauze, and the filtrate was concentrated by a rotary evaporator under reduced pressure at 45 ℃ to give a crude extract (86.0 g). The crude extract was dissolved in the lower phase (500 mL) of hexane-ethyl acetate-methanol-water (9:1:1:9, v/v) in a separating funnel, after which an equal volume of the upper phase (500 mL) of the same solvent system was added and the mixture in the separating funnel was shaken. After equilibrium of the sample distribution between the two phases was achieved, the upper phase was separated. The lower phase was extracted twice by the upper phase. The upper phase was combined and evaporated to dryness as fraction
Chaetoglobol acid (
Enzyme Inhibition Assays 42,43
Commercially available α-glucosidase and α-amylase were selected as the target proteins, with pNαGP and G3-CNP as their substrates, respectively. Fractions
Statistical Analysis
All data are expressed as means ± the standard error of the mean (SEM). The statistical analysis was performed with GraphPad Prism 7.0 (GraphPad, La Jolla, CA, USA).
Molecular Docking
The molecular docking experiment of the ligand was performed using the crystal structure of α-glucosidase and maltose co-crystal (yeast isomaltase, PDB code: 3A4A), in the Discovery Studio 2017 R2. The CDOCKER package was used to prepare ligands and receptors. After the docking process, the resulting 3D complexes were shown by Discovery Studio (BIOVIA Software Inc, USA).
Supplemental Material
Supplementary Material 1 - Supplemental material for New Metabolite With Inhibitory Activity Against α-Glucosidase and α-Amylase From Endophytic Chaetomium globosum
Supplemental material, Supplementary Material 1, for New Metabolite With Inhibitory Activity Against α-Glucosidase and α-Amylase From Endophytic Chaetomium globosum by Jianzhao Qi, Dacheng Wang, Xia Yin, Qiang Zhang and Jin-Ming Gao in Natural Product Communications
Supplemental Material
Supplementary Material 2 - Supplemental material for New Metabolite With Inhibitory Activity Against α-Glucosidase and α-Amylase From Endophytic Chaetomium globosum
Supplemental material, Supplementary Material 2, for New Metabolite With Inhibitory Activity Against α-Glucosidase and α-Amylase From Endophytic Chaetomium globosum by Jianzhao Qi, Dacheng Wang, Xia Yin, Qiang Zhang and Jin-Ming Gao in Natural Product Communications
Footnotes
Acknowledgements
The authors sincerely thank Mr. Edward Neil Schmidt (Department of Chemistry, University of Alberta) for language editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the China Postdoctoral Science Foundation 2019M653760, and the National Science Foundation of China 31800031 and 81502938, as well as Natural Science Basic Research Plan in Shaanxi Province of China 2019JQ-046.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
