Abstract
Background
Myocardial fibrosis following myocardial infarction (MI) is linked to decreased cardiac function and heart failure. Astragaloside IV (AST IV), a component of traditional Chinese medicine, has shown promise in animal studies for improving cardiac function.
Objectives
This study intends to investigate the role of AST IV in MI and its correlation with the sirtuin-3 (SIRT3)/β-catenin/peroxisome proliferator-activated receptor gamma (PPARγ) signaling pathway.
Materials and Methods
After the establishment of an animal model of MI, 60 rats were divided into a control group, model group (MI group), positive control group (sham group), low-dose AST IV group (0.8 mg/100 g), and high-dose AST IV group (3 mg/100 g). The animals were injected once a day for 4 consecutive weeks. To further investigate the mechanism of action of AST IV, another nine rats from the model group were randomly divided into the 3TYP group (5 mg/100 g), SKL2001 group (20 µmol/L, 10 mL/kg), and T0070907 group (0.15 mg/100 g), with three rats in each group. The rats in each group were intervened once a day by intraperitoneal injection and once a day by gavage for 4 consecutive weeks. Hemodynamics, cardiac function, ventricular weight, and infarct area were assessed at 1, 2, and 4 weeks post-surgery. Myocardial collagen content in the non-infarcted area was measured, the expression rate of transforming growth factor-beta and ED-positive cells was calculated, and reverse transcription polymerase chain reaction was used to detect SIRT3, β-catenin, and PPARγ mRNA expression.
Results
The left ventricular ejection fraction, short-axis shortening rate, and left ventricular mass in the model group were decreased obviously. AST IV treatment decreased systolic blood pressure, +dp/dtmax, –dp/dtmax, and left ventricular end-diastolic pressure, as well as reduced myocardial collagen deposition in rat hearts (p < 0.05). AST IV inhibited the SIRT3/β-catenin/PPARγ signaling pathway when alleviating myocardial fibrosis. Intervention with SIRT3 blocker 3-TYP, β-catenin agonist SKL2001, and PPARγ blocker T0070907 reversed the inhibitory effect of AST IV on the signaling pathway.
Conclusion
AST IV exhibits significant anti-fibrotic effects, effectively reducing collagen deposition in the MI model, improving cardiac function parameters, and decreasing markers such as creatine kinase-MB. It significantly regulates SIRT3, β-catenin, and PPARγ to exert cardioprotective effects. The key mechanism involves inhibiting the SIRT3/β-catenin/PPARγ, offering an innovative strategy for post-MI treatment.
Introduction
Cardiovascular diseases, particularly myocardial infarction (MI), are primarily caused by insufficient blood supply to the myocardium due to coronary artery obstruction (Femia et al., 2021), of which myocardial fibrosis is a major pathophysiological process (Hu et al., 2022). The abnormal increase in collagen fibers within myocardial tissue enhances myocardial stiffness, leading to reduced ventricular function and significantly impairing the heart’s pumping ability (González et al., 2019). Therefore, it is of great clinical significance to thoroughly investigate myocardial fibrosis’s pathological mechanisms post-MI and identify effective intervention strategies to improve cardiac function.
Traditional Chinese medicine is known to have certain advantages in the treatment of cardiovascular diseases (Wang et al., 2024). Various studies have shown that tanshinone IIA, extracted from Danshen, inhibits the proliferation and phenotypic transformation of rat cardiac fibroblasts, thereby reducing extracellular matrix production and delaying the progression of myocardial fibrosis (Ma et al., 2023). The Chinese patent medicine Tongxinluo improves myocardial circulation by promoting the regeneration of ischemic myocardial blood vessels, further inhibiting myocardial fibrosis. Tongxinluo also has low side effects and plays a crucial role in protecting the myocardium in patients with acute MI (Wei et al., 2024).
Astragaloside IV (AST IV) is an extract from the traditional Chinese medicine Astragalus membranaceus with anti-viral and anti-stress effects, as it also can protect cardiopulmonary function, enhance metabolism and blood circulation, and improve immunity and disease resistance (Zhang et al., 2020). Studies have found that AST IV can regulate signaling pathways such as mitogen-activated protein kinase (MAPK) and PI3K/Akt, influencing the activity of proteins such as P38, Erk, and JNK. This regulation impacts the proliferation and differentiation of cardiomyocytes, thereby improving symptoms of coronary heart disease (Wan et al., 2023). AST IV can also regulate nuclear factor kappa B (NF-κB), reducing the expression of CD11b/CD18 and ICAM-1, inhibiting the adhesion, migration, and infiltration of polymorphonuclear leukocytes (PMNs) and endothelial cells, which is significant for improving endothelial function and managing cardiovascular disease complications (Li et al., 2012). AST IV can increase the proliferation of T and B lymphocytes, participating in immune regulation and reducing inflammatory cytokines like interleukin 1 (IL-1), tumor necrosis factor-alpha (TNF-α), and TNF, exerting its anti-inflammatory effects (Wang et al., 2022). AST IV also inhibits the activity of caspase-3 and procaspase-9 and thereby hinders apoptosis when downregulating pro-apoptotic proteins and upregulating anti-apoptotic proteins. By activating Bad and mTOR, AST IV promotes cell survival and proliferation, as well as the differentiation of cardiomyocytes (Sun et al., 2021). However, the specific mechanisms by which AST IV affects myocardial fibrosis after MI remain incompletely understood and require further exploration.
Studies have shown that during the pathological process of MI, the expression of sirtuin-3 (SIRT3) significantly influences the progression of myocardial fibrosis (Su et al., 2023). The expression of β-catenin is closely related to myocardial fibrosis (Hu et al., 2021). Peroxisome proliferator-activated receptor gamma (PPARγ), a nuclear receptor transcription factor, plays a crucial role in improving myocardial function by regulating inflammation and fibrosis. It can be speculated that the effect of AST IV on myocardial fibrosis post-MI is related to its correlation with the SIRT3/β-catenin/PPARγ signaling pathway (Zhou et al., 2020). This study aims to analyze the mechanism of AST IV in myocardial fibrosis. By revealing AST IV’s mechanisms for treating cardiovascular diseases, the study might provide theoretical support for its clinical application, contributing to the modernization of traditional Chinese medicine as well as enhancing its influence in the field of international cardiovascular disease treatment.
Materials and Methods
Experimental Materials
AST IV injection was obtained from Dali Pharmaceutical Co., Ltd. (Z53021585), hydroxyproline kits from Shanghai ELISA Biotechnology Co., Ltd., monoclonal antibodies from Shanghai Beyotime Pharmaceutical Co., Ltd., rabbit anti-rat transforming growth factor-beta 1 (TGF-β1) antibody, S-P kits from Shanghai Biotechnology Co., Ltd. 3-TYP (SIRT3 inhibitor, Shanghai Yuanye Biotechnology Co., Ltd., 98% purity), β-catenin pathway agonist SKL2001 (HY-101085) and PPARγ inhibitor T0070907 were purchased from Med Chem Express, Inc. in the United States of America (HY-13202/CS-0462). And the instrument includes a R415 small animal ventilator (Rayward), a BL-420 biofunctional experimental system (Shanghai Shengjian Medical Instrument Equipment Development Co. Ltd.), Sartorius electronic balance (Sartorius, Germany), UV1901PC ultraviolet-visible spectrophotometer (Shanghai Aoxi Scientific Instrument Co., Ltd.), and HPIAS2000 image analysis software.
Animals
Sixty male Sprague Dawley (SD) rats weighing 200–250 g were purchased from Jiangsu Huachuang Xinnuo Pharmaceutical Technology Co., Ltd.
Methods
Establishment of MI Model
Under anesthesia with sodium pentobarbital (40 mg/kg), rats were then intubated and connected to a specialized animal ventilator. The chest cavity and pericardium were opened layer by layer at the 4th intercostal space to expose the heart, and the left anterior descending coronary artery was ligated with silk thread. In the control group, the wire was passed through the same part of the left ventricle but was not ligated. All other procedures were consistent with the experimental group.
Criteria for determining the success of MI modeling: significant elevation of the ST segment and lightening or whitening of the myocardium of the anterior wall of the left ventricle were observed in the standard limb lead II of the electrocardiogram.
Grouping and Treatment
Sixty rats were randomly positive in the control group (sham group), low-dose AST IV group, and high-dose AST IV group, control group, model group (MI group) (n = 12, each group). With the control group fed usually without any surgical procedure, the MI and sham groups received intraperitoneal injections of normal saline. In contrast, the AST IV groups received intraperitoneal injections of AST IV at a dose of 0.8 mg/100 g and 3 mg/100 g, respectively. To further investigate the mechanism of action of AST IV, another nine rats from the model group were randomly divided into the 3TYP group (SIRT3 blocker), SKL2001 group (β-catenin agonist), and T0070907 group (PPARγ blocker), with three rats in each group. Three rats in each group were given 5 mg/100 g of 3TYP by gavage in the 3TYP group, 10 mL/kg of 20 µmol/L SKL2001 in the SKL2001 group, 0.15 mg/100 g of SKL2001 in the T0070907 group, and an equal amount of saline intraperitoneally in each of the three groups every day. The positive control group (sham group), low-dose AST IV group, and high-dose AST IV group, as well as other model groups, were treated with equal amounts of saline by gavage. The rats in each group were treated by intraperitoneal injection and gavage once a day for 4 weeks. Rats were sacrificed at 1, 2, and 4 weeks post-surgery for testing various indicators.
Index Measurement
Hemodynamics and Cardiac Function
After anesthesia with sodium pentobarbital, the right common carotid artery of the rats was isolated and cannulated. The rats were stabilized for 10 min, and then the BL-420 biofunction experimental system was used to record systolic blood pressure (SBP), left ventricular end-diastolic pressure (LVEDP), +dp/dtmax, and –dp/dtmax.
Ventricular Weight and Infarct Area
Post-sacrifice, the weights of the whole heart and left ventricle were measured, and the infarct area was assessed. The ventricles were divided into two parts, and the apex portion was fixed. About 4 µm specimens were prepared after paraffin embedding for hematoxylin and eosin (HE) staining and Van Gieson (VG) staining. Image analysis software was used to measure the infarct area. VG-stained sections were also used to measure the collagen volume fraction (CVF) and perivascular collagen area (PVCA). To ensure accuracy, six non-endocardial and endocardial myocardial fields were randomly selected for measurement in each specimen, and the average value was taken.
Determination of Myocardial Collagen Content in Non-infarcted Areas
Approximately 100 mg of rat myocardial tissue was weighed to test hydroxyproline content, following the kit instructions strictly. Collagen content was calculated as: 7.46 × hydroxyproline content.
Immunohistochemistry
Three micrometer-thick paraffin sections were dewaxed and hydrated. The sections were incubated with methanol containing 3% H2O2 and then subjected to antigen retrieval via microwave technology. The sections were probed with primary antibodies (1:5,000), followed by secondary antibody (1:5,000) incubation. A DAB colorimetric agent was used for development, and hematoxylin was used for counterstaining. Sections were sealed with neutral resin. A blank control was included to ensure reliability, and the expression of ED-positive cells and TGF-β1 was detected. Image analysis software calculated the expression rates of TGF-β and ED-positive cells.
Determination of Myocardial Injury
Apical blood samples from each group of rats were collected into EDTA anti-coagulation tubes, centrifuged, and the supernatant was cryopreserved. The levels of creatine kinase (CK), creatine kinase-MB (CK-MB), and lactate dehydrogenase (LDH) were determined using an automatic biochemical analyzer.
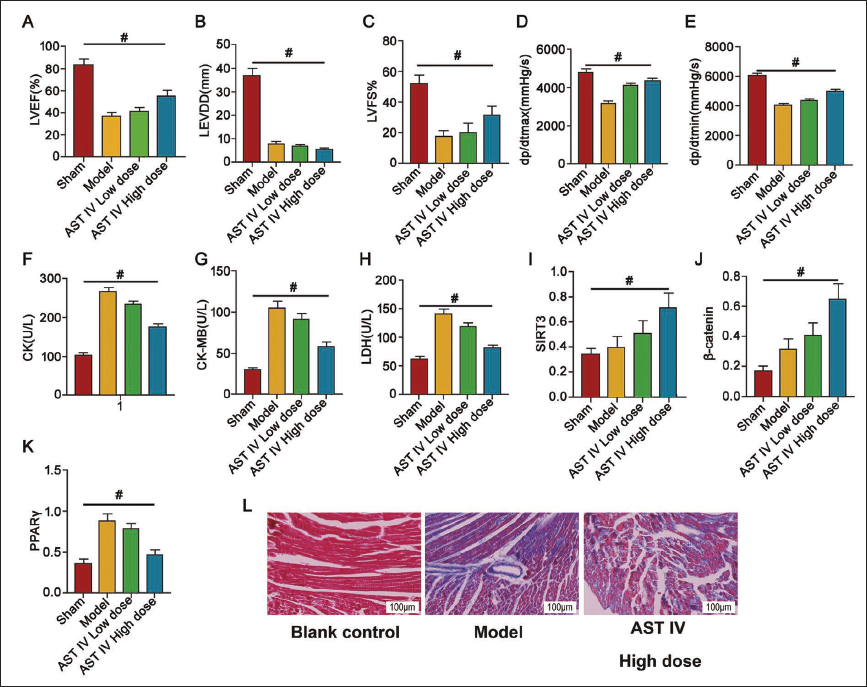
Quantitative Reverse Transcription Polymerase Chain Reaction (RT-qPCR)
Rat hearts were removed, and the left ventricle was isolated, washed, dried, and cleared of blood vessels. The tissue was divided into two parts: one part was fixed with 4% paraformaldehyde, dehydrated, made transparent, and embedded in paraffin; the other part was stored in EP tubes. Total RNA was extracted using TransZol, and mRNA was reverse-transcribed into cDNA and verified by PCR. The primers are listed in Table 1. With glyceraldehyde-3-phosphate dehydrogenase (GAPDH) as an internal control, the 2D method estimated the gene expression level.
Primers for Quantitative Reverse Transcription Polymerase Chain Reaction (RT-qPCR).
Western Blot Analysis
Protein samples were first lysed, followed by the addition of lysate, loading, electrophoresis, transfer, and blocking. The membranes were then incubated overnight with primary antibodies: SIRT3 (ab217319, 1:1,000), PPARγ (ab310320, 1:1,000), and β-catenin (ab32572, 1:1,000) and secondary antibody (goat anti-rabbit, 1:2,000) for 2 hours. Enhanced chemiluminescence reagents were used for detection.
Statistical Methods
GraphPad Prism 8.0 was used to analyze experimental data. Measurement data were presented as mean ± SD. Homogeneity of variance tests, pairwise LSD-t-tests, and ImageJ image processing were used for multiple group comparisons. A significance level of p < 0.05 was used for all tests.
Results
Establishment of an animal model of myocardial fibrosis post-MI and the beneficial effects of AST IV.
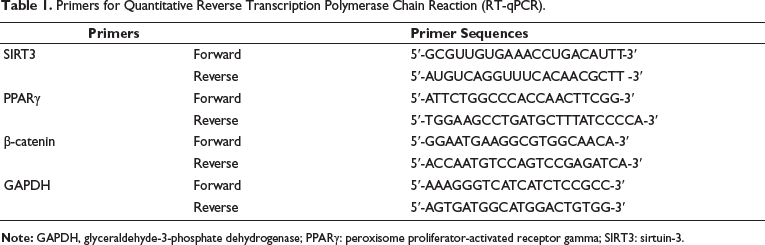
After successfully establishing a myocardial fibrosis model, we observed decreased left ventricular ejection fraction (LVEF) and short-axis shortening rate in the model group (Figure 1A–B) and sham group (p < 0.05). Following AST IV intervention, a dose-dependent improvement was noted, with values significantly higher than those in the model group (p < 0.05). With higher left ventricular mass index (LVMI) in the model group (Figure 1C) (p < 0.05), post-AST IV intervention, a dose-dependent reduction in LVMI was observed (p < 0.05).
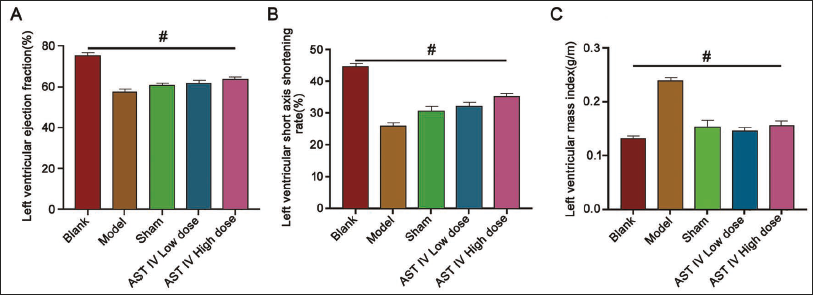
Interestingly, the SBP, +dp/dtmax, and –dp/dtmax of the sham group in week 1 to week 4 were greater than those in the MI group (Figure 2A, C–D), and LVEDP, infarct size, HW/BW, LVW/BW, CVF, and PVCA the MI group in week 1 to week 4 were significantly elevated (Figure 2B, E–J) (p < 0.05). Following AST IV intervention, hemodynamic indicators showed a positive correlation with dose (Figure 2A, C–D) and were both lower than in the sham group (p < 0.05). However, AST IV intervention showed a negative correlation with dose in other indicators (Figure 2B, E–I) (p < 0.05).
Microscopic examination of the model group revealed scattered, elongated rod-shaped cardiomyocytes with visible degeneration and necrosis (Figure 2J). Post-AST IV treatment improved tissue lesions, particularly in the high-dose group, where cardiomyocytes were neatly arranged, and lesions were reduced compared to the low-dose group. Therefore, the high-dose AST IV group was selected for subsequent experiments.
When detecting ED1-positive cells, a marker of myocardial fibrosis, it was found that in the non-infarcted myocardium at 2 and 4 weeks, both the model group (MI) and the MI + AST IV group exhibited significantly higher ED1-positive cell expression than the sham group (Figure 2K). This indicates that post-MI, myocardial collagen accumulation exceeds normal levels regardless of treatment, reflecting inevitable scarring and fibrosis stages in cardiac repair. Notably, post-treatment myocardial collagen content in the AST IV group was greatly reduced (p < 0.05), which suggests a positive effect of AST IV, potentially reducing myocardial fibrosis, maintaining myocardial structure and function, and mitigating adverse effects on cardiac structure.
AST IV inhibits the SIRT3/β-catenin/PPARγ signaling pathway and regulates myocardial fibrosis after MI.
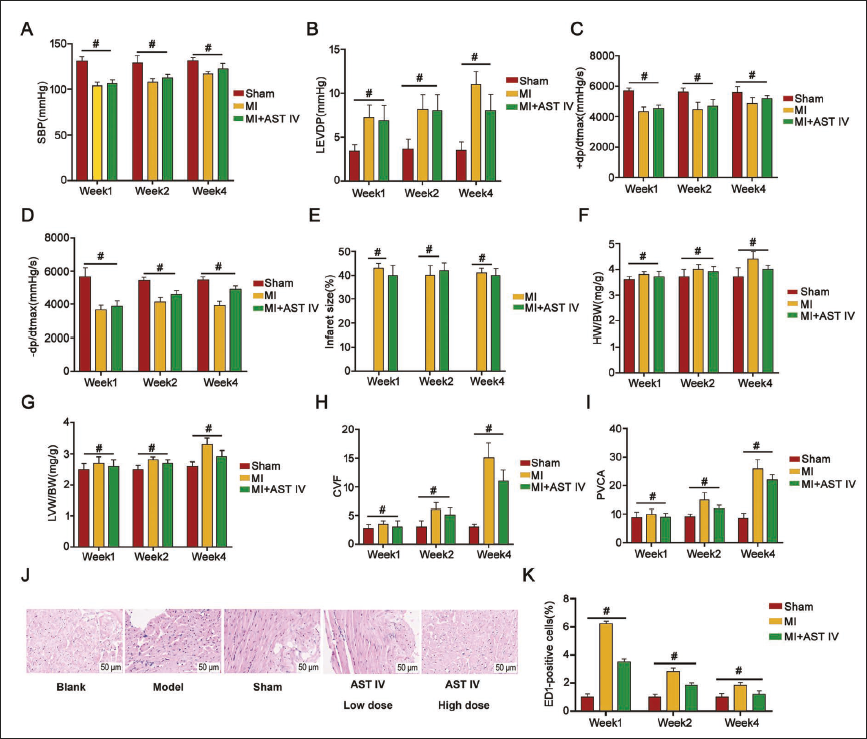
Key cardiac function parameters indicated significant changes in LVEF (%), left ventricular end-diastolic dimension (LVEDD) (mm), left ventricular fractional shortening (LVFS) (%), dp/dtmax (mmHg/s), and dp/dtmin (mmHg/s) (Figure 3A–E) at weeks 2 and 4 when comparing the control and sham groups with the MI and MI + AST IV groups (Figure 3A–E). This suggests cardiac function was impaired after MI, while AST IV treatment partially restored these parameters, indicating a protective effect on cardiac function. Additionally, the AST IV treatment attenuated myocardial damage in the model group, as demonstrated by a decline in the levels of CK, CK-MB, and LDH (Figure 3F–H).
At the molecular level, the expression of SIRT3, β-catenin, and PPARγ was significantly upregulated in the MI group. AST IV treatment markedly decreased the expression of these molecules (Figure 3I–K), suggesting that AST IV exerts its anti-fibrotic effect by regulating SIRT3, β-catenin, and PPARγ. This finding offers a novel insight into post-MI treatment. Notably, after AST IV intervention, a dose-dependent positive correlation was observed (Figure 3A, C–E, I–J), with values higher than those in the model group (p < 0.05). The model group HAD lower values than the sham group (Figure 3A–D, p < 0.05). After AST IV intervention, a dose-dependent negative correlation was seen (Figure 3B, F–H, K) (p < 0.05).
Further, Masson staining demonstrated a reduction in myocardial fibrosis in the AST IV treatment group (Figure 3L), confirming the positive role of AST IV in reducing myocardial fibrosis. This suggests AST IV inhibits the SIRT3/β-catenin/PPARγ signaling pathway, alleviating myocardial fibrosis after MI. This provides potential targets and strategies for MI treatment.
SIRT3 blocker/β-catenin agonist/PPARγ blocker reverses the inhibitory effect of AST IV
After various interventions, the protein expression in the T0070907 group was the lowest (Figure 4A–B). Further comparative analysis was conducted to evaluate the effects of AST IV and the traditional cardiovascular drug captopril on post-MI fibrosis. The captopril group showed the expected effects of lowering blood pressure and reducing angiotensin II production, thereby decreasing cardiac afterload and protecting myocardial cells from further damage. When comparing the effect of captopril and AST IV, we found that both treatments significantly reduced myocardial collagen deposition and alleviated cardiac fibrosis (Figure 5A–B). However, while captopril effectively regulated blood pressure, AST IV demonstrated a more significant inhibitory effect on the expression of α-SMA, an essential protein in fibrosis (Figure 5A). Unlike AST IV, captopril rarely exhibited a direct inhibitory effect on the SIRT3/β-catenin/PPARγ (Figure 5C–E); meanwhile, AST IV alone dramatically decreased the expression of SIRT3, β-catenin, and PPARγ (Figure 5C–E) and regulated TGF-β1 (Figure 5J), indicating that AST IV mitigates myocardial fibrosis by inhibiting this signaling pathway.
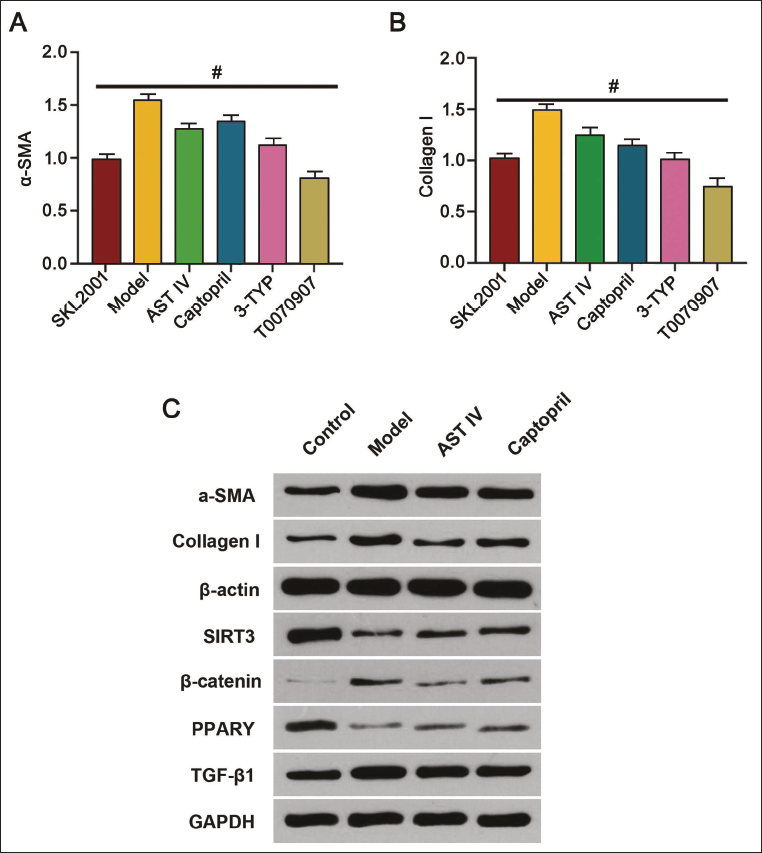
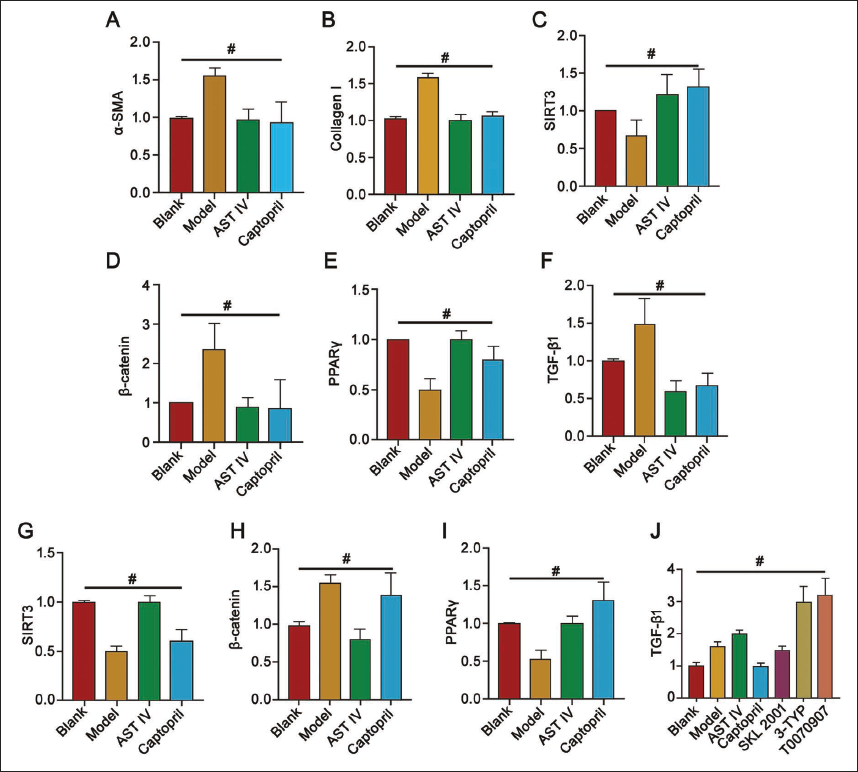
To further validate the underlying mechanism of AST IV in MI, 3-TYP (SIRT3 blocker), SKL2001 (β-catenin agonist), and T0070907 (PPARγ blocker) were applied to the rats. The levels of α-SMA and collagen I in the model group were elevated compared to the blank group but decreased in the AST IV group (Figure 4A–B, D, F, H) (p < 0.05). Additionally, SIRT3, β-catenin, PPARγ, and TGF-β1 levels were restored or upregulated after intervention (Figure 5C–I), effectively reversing the inhibitory effect of AST IV. Moreover, this reversal was manifested by increased expression of fibrosis markers α-SMA and collagen I (Figure 5A–J).
This finding underscores the pivotal role of the SIRT3/β-catenin/PPARγ signaling pathway in AST IV-mediated cardioprotection. The regulation of this pathway by AST IV suggests that targeting it could be a novel strategy for treating myocardial fibrosis. Furthermore, reversing AST IV’s effects through specific molecular interventions provides new insights into the regulatory networks involved in myocardial fibrosis.
Discussion
After MI, the damage and death of myocardial cells initiate a series of pathological processes, including cardiac remodeling and fibrosis, which significantly contribute to the development of heart failure (Jenča et al., 2021). Myocardial fibrosis is a critical factor in the progression of various heart diseases, MI, and hypertension (Liu, López de Juan Abad et al., 2021). This study’s successful construction of an MI model provided a reliable platform to demonstrate that AST IV can significantly mitigate fibrosis, thereby presenting a potential therapeutic strategy.
AST IV is recognized for its multiple pharmacological functions, including anti-inflammatory, antioxidant, and immune-regulating properties (Yang, Pan et al., 2023). Previous studies have shown that AST IV can reduce myocardial tissue damage in rat MI models and promote angiogenesis through the PKD1/HDAC5/VEGF pathway (Yang, Liu et al., 2023). Furthermore, AST IV protects cardiomyocytes from hypoxic injury when maintaining mitochondrial homeostasis (Zhang et al., 2022). In this study, we found that AST IV could reduce collagen deposition after MI, thereby reducing the extent of myocardial fibrosis. This anti-fibrotic effect is likely associated with AST IV’s anti-inflammatory and antioxidant properties (Yuan et al., 2023). Following AST IV treatment, the improvement in cardiac function indicators like LVEF and LVFS supports its cardioprotective effects. Additionally, the model group exhibited increased myocardial collagen accumulation, leading to scar formation and fibrosis. The administration of AST IV significantly reduced the excessive deposition of myocardial collagen, which helped in maintaining myocardial structure and function, thereby mitigating the adverse effects of cardiac remodeling. Similar findings have been reported in studies on dilated cardiomyopathy, where AST IV was shown to reduce LVEDD and type I collagen levels while increasing LVEF and PPARγ mRNA expression through the β-catenin/PPARγ pathway (Zhang et al., 2023).
Studies have confirmed that AST IV improves hemodynamic parameters, reduces aldosterone (ALD) and atrial natriuretic peptide (ANP), and alleviates heart failure in rats (Jing et al., 2021). In this study, AST IV treatment significantly improved LVEF and dp/dtmax and reduced CK-MB and LDH levels, key indicators of cardiac function, which correlate with reduced myocardial fibrosis. This supports the role of AST IV in effectively modulating myocardial fibrosis. AST IV achieves these effects by inhibiting myocardial cell death, reducing myocardial damage, and improving the condition of rats with MI under diabetes by activating the NRF2/1 pathway. On the other hand, it also reduces inflammatory reactions, decreases apoptosis in myocardial tissue cells, and mitigates scar tissue hyperplasia. By downregulating TLR4-like receptors, reducing DVL-1 and β-catenin proteins in myocardial tissue, and increasing LVEF while reducing left ventricular end-systolic diameter, AST IV helps improve ventricular remodeling through β-catenin regulation (Tang et al., 2018).
In a rat model of chronic heart failure, Shenfu injection reduced LVEF and LVFS and showed potential to improve myocardial fibrosis through the regulation of β-catenin protein, indicating that β-catenin plays a crucial role in myocardial fibrosis and could be an effective target for treatment (Liu, Zheng et al., 2021). Further studies have shown that in myocardial fibrosis in rats with chronic heart failure, dipeptidyl peptidase (DPP-4) increases the expression of p-GSK-3β and β-catenin proteins by reducing TGF-β1 and α-SMA proteins, improving myocardial fibrosis through the Wnt/β-catenin signaling pathway. This highlights the key role of the Wnt/β-catenin pathway in treating myocardial fibrosis. In hypertensive myocardial fibrosis, Tianma Gouteng Decoction upregulates the expression of β-catenin by increasing the levels of type I and III collagen and α-SMA proteins. This regulation of Wnt/β-catenin, along with the improvement of miR-150-3p expression, helps attenuate myocardial fibrosis, demonstrating the potential value of traditional Chinese medicine in this field.
This study found that AST IV significantly downregulated the mRNA expression of SIRT3, β-catenin, and PPARγ, indicating that AST IV exerts its effects by inhibiting this pathway. SIRT3 is known to regulate extracellular matrix production through deacetylase reactive oxygen species kinase in cardiac fibrosis, and AST IV’s inhibition is consistent with this mechanism. The regulation of β-catenin/TCF-β-catenin is also related to fibrosis, and AST IV’s inhibitory effect along with the reduction of TGF-β1 expression confirms its key role in regulating this pathway and inhibiting fibrosis.
It is noted that in experiments on myocardial fibrosis in neonatal mice, sFRP1 increased Dvl-1 and α-SMA proteins through the Wnt/β-catenin signaling pathway and decreased the OD value and the contents of type I and III collagen, which is beneficial for improving myocardial fibrosis. This underscores the important role of the Wnt/β-catenin signaling pathway in the development of myocardial fibrosis (Tao et al., 2021). Herein, we noted that compared with traditional drugs captopril, AST IV (an active ingredient extracted from A. membranaceus) showed a more significant inhibitory effect on α-SMA, a key protein in fibrosis, and improved the condition of MI. We further explored the specific mechanism underlying the role of AST IV in MI.
The captopril group did not show the same direct inhibition of the SIRT3/β-catenin/PPARγ signaling pathway as AST IV. This is different from traditional treatment strategies, which often focus on a single signaling pathway or target. In contrast, AST IV operates through multiple pathways, improving myocardial fibrosis and offering a basis for combination therapy.
The Wnt/β-catenin signaling pathway plays a crucial role in developing myocardial fibrosis (Zhang & Lu, 2024). sFRP1 can improve myocardial fibrosis through this pathway, particularly within the SIRT3/β-catenin/PPARγ signaling axis. Of note, administration-specific intervention agents such as 3-TYP, SKL2001, and T0070907 elevated the content of myocardial fibrosis markers (collagen I and signaling molecules). This indicates the reversibility of AST IV regulation and supports the central role of the SIRT3/β-catenin/PPARγ pathway in AST IV-mediated myocardial protection, emphasizing these pathways as therapeutic targets (Song et al., 2019). In the study of myocardial fibrosis in mice, AST IV showed its unique effect (Liu et al., 2019). It significantly reduced α-SMA and MMP-9 proteins, increased MMP-2 and TIMP-1, and improved myocardial fibrosis through the PPARγ/nuclear factor kappa B (NF-κB) signaling pathway. This enhanced left ventricular function, reduced myocardial sclerosis, and optimized post-MI outcomes. Additionally, in a rat model, AST IV upregulated PPARγ and LVSP expression and downregulated NF-κB expression, promoting myocardial protection via the PPARγ/NF-κB pathway, improving heart function and patients’ quality of life (Liu et al., 2020). In addition, some studies have focused on myocardial fibrosis in the context of type 1 diabetes in which AST IV regulates peroxidase activation to increase TGF-β1 and PPARγ proteins, reduce LVEF/A levels, and use the TGF-β1/ERK signaling pathway to inhibit diabetes-induced cardiac fibrosis and promote myocardial health (Gbr et al., 2021). In pressure overload experiments, AST IV reduced heart rate by regulating peroxidase activation, upregulating α-SMA and TGF-β, and significantly improved major symptoms caused by pressure overload through the PPARγ pathway. This led to reduced left ventricular damage and fibrosis in mice, improved myocardial fibrosis, and restored cardiac function. These findings further illustrate the promising potential of AST IV in various post-MI fibrosis models in which it improves cardiac function in MI.
Conclusion
In conclusion, this study indicates AST IV has a significant anti-fibrotic effect, effectively reducing collagen deposition in MI models and improving cardiac function. Mechanistically, AST IV regulates the expression of SIRT3, β-catenin, and PPARγ to mitigate myocardial fibrosis. In the MI model, the key role of AST IV lies in its impact on the SIRT3/β-catenin/PPARγ signaling pathway, providing innovative strategies and theoretical support for post-MI treatment. Future research can further explore the mechanisms of AST IV targeting multiple signaling pathways.
Footnotes
Abbreviations
AST IV: Astragaloside IV; CK-MB: Creatine kinase-MB; LVEDP: Left ventricular end-diastolic pressure; LVEF: Left ventricular ejection fraction; MI: Myocardial infarction; PPARγ: Peroxisome proliferator-activated receptor gamma; SBP: Systolic blood pressure; SIRT3: Sirtuin-3; TGF-β: Transforming growth factor-beta.
Acknowledgments
The authors gratefully acknowledge the Fourth Affiliated Hospital of Guangzhou Medical University for providing the necessary equipment for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethics Committee of the Fourth Affiliated Hospital of Guangzhou Medical University.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
