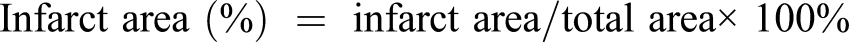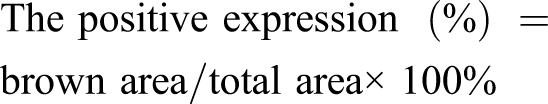Abstract
Objective
Shenfu injection (SFI) is commonly used for cardiac dysfunction in China. Adenosine receptors have been reported to exert anti-fibrosis effects. The intent of this study was to evaluate that SFI attenuates cardiac fibrosis through activating of adenosine A2a receptor (A2aR) in rats with myocardial ischemia-reperfusion (MI/R).
Methods
Sprague Dawley male rats were randomly divided into five groups, nine rats in each group. Injections in all rat groups were carried out prior to reperfusion, and in the sham and MI/R groups, only vehicle was injected. Injections in the remaining group were as follows: 5 mL/kg in the SFI group; 15 mg/kg nicorandil in the A2R agonist group; and 5 mL/kg SFI plus 5 mg/kg MSX-3 in the SFI + A2aR antagonist group. Changes in cyclic adenosine monophosphate (cAMP) and the development of myocardial infarction and cardiac fibrosis were documented among the groups. Additionally, the levels of A2aR, collagen Ⅰ, collagen Ⅲ, fibronectin, and matrix metalloproteinase-9 (MMP-9) were measured.
Results
Following injection with SFI or nicorandil, the cAMP concentration, infarct area, and cardiac fibrosis induced by MI/R injury were significantly decreased (p < 0.05). Additionally, the levels of collagen Ⅰ, collagen Ⅲ, fibronectin, and MMP-9 were clearly suppressed by SFI or nicorandil when compared with the MI/R group (p<0.01). However, the protective effects of SFI were counteracted by MSX-3. A negative correlation between A2aR and collagen I and collagen III was found (p = 0.00).
Conclusion
SFI activated the A2aR to reduce myocardial fibrosis caused by MI/R injury, which provided an underlying mechanism of action of SFI.
Introduction
Acute myocardial infarction (AMI) accounts for approximately 80% of patients in cardiogenic shock, which commonly manifests with blockage of coronary blood flow. 1 However, blood perfusion into ischemic myocardial tissues can cause additional tissue damage, referred to as myocardial ischemia-reperfusion (MI/R). 2 In clinical practice, MI/R can aggravate potentially reversible cardiac tissue damage, enlarging the area of necrotic myocardium, thereby worsening the patient’s condition and outcome.3,4 At present, there are no drugs that effectively reduce MI/R injury in patients with AMI.
After MI/R injury, due to the imbalance of myocardial extracellular matrix deposition and degradation, collagen fibers replace necrotic myocardial tissue, resulting in scar tissue accumulation and maturation, with the potential for adverse cardiac remodeling and cardiac dysfunction.5–7 Therefore, modulating the progression of cardiac fibrosis is necessary to reduce MI/R injury. Adenosine receptors, including adenosine 1 receptor (A1R), A2aR, A2bR, and A3R, are expressed in most organs.8,9 A pivotal finding was previously reported that extracellular cyclic adenosine monophosphate (cAMP) reduced cardiomyocyte hypertrophy through activating A1R, while delivering an anti-fibrotic signal to cardiac fibroblasts by A2R activation. 8 Moreover, da Silva et al. clarified that an A2aR agonist could prevent the development of myocardial infarction through counteracting the cardiac fibrosis and inflammatory processes. 10 These reports indicated that A2aR activation could be useful in preventing cardiac fibrosis and abnormal remodeling following the MI/R injury.
We previously determined that Shenfu injection (SFI) could increase the expression of myocardial adenosine receptors in rats with MI/R postconditioning. 11 SFI has been used in injury correlated disorders of the cardiac muscle for nearly 30 years in China, and its main bioactive components are ginsenosides, such as Re, Rc, Rb2, and Rb1. 12 As a traditional Chinese medicine used to treat patients with myocardial infarction treated by percutaneous coronary intervention, SFI reportedly can protect myocardium from MI/R injury, prevent myocardial fibrosis, and improve myocardial function. 13 Furthermore, cardiac fibrosis has been reduced by SFI through regulation of the transforming growth factor-beta (TGF-β)/Smads pathway in rats with heart failure. 14
Aim of the Work
Therefore, we hypothesized that SFI could prevent cardiac fibrosis induced by MI/R injury through activation of A2aR. In this study, we used nicorandil (A2aR agonist) and MSX-3 (A2aR antagonist) in conjunction with SFI to test our hypothesis. The SFI was purchased and its active components were extracted from the Radix Ginseng rubra and monkshood and prepared water soluble constituents for injection.
Material and Methods
Animals
Forty-five Sprague Dawley male rats, weighing 200–220g at 8 weeks, were purchased from Jinan Pengyue Experimental Animal Breeding Co., Ltd., license: SCXK (Lu) 20190003. The rats were raised at 24 ± 2°C, 50 ± 10% humidity, and 12 h light/dark cycle with food and water ad libitum. After 7 days, rats were used to perform experiments.
Animal Ethics
This study was approved by the Animal Care and Use committee of Yantaishan Hospital (Approval No. 202013), and all experiments followed the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (2011).
Animal model
As previously described, 15 the rats were anesthetized using 1.5% pentobarbital sodium (0.27 mL/100 g) and then ligated the left anterior descending coronary artery to induce myocardial infarction. Following 30 minutes of ischemia time, the various reagents were injected via the femoral vein (the reagents for each group are showed in the Animal groups section). Then, the rats underwent reperfusion for 24 h to assess cardiac function. In the sham operation group, the rats underwent the surgical procedures but without ligation of the left anterior descending coronary artery.
Animal Groups
The rats were randomly divided into five groups, 9 rats per group. In all groups, the specific infusions were administered after cardiac ischemia was completed. 1) In the sham group, the same volume of vehicle was infused through the femoral vein, but with added reagent. 2) In the SFI group, 5 mL/kg SFI (batch number, 1903120104, China Resources Sanjiu (Ya’an) Pharmaceutical Co., Ltd., China) was injected. 3) In the A2R agonist group, 15 mg/kg nicorandil (BP1103, Merck, Shanghai, China) was injected. 16 4) In the SFI + A2aR antagonist group, 5 mL/kg SFI and 5 mg/kg MSX-3 (M3568, Merck, Shanghai, China) were injected. 8 The nicorandil and MSX-3 were dissolved in 0.9% saline. The manufacturing process of SFI was in compliance with the guidelines of Good Manufacturing Practice and Good Laboratory Practice (WS3-B-3427-98, China), and the major components of SFI were confirmed by the Chinese government agency. The aconitine (C24H47NO11) in 1 mL of this product is no more than 0.1 mg, and the Rb1 (C54H92O23) in 1 mL of this product is more than 0.5 mg.
cAMP Levels in Platelet Suspension
After anesthetization using 1.5% pentobarbital sodium, blood was collected from the abdominal aorta of the rats. A concentrated platelet suspension was prepared using a rat peripheral blood platelet separation solution kit (Solarbio, P8570, Beijing, China). After washing with phosphate buffered saline (PBS), the platelet suspension was lysed using a high RIPA buffer (Solarbio, P0010, Beijing, China). The cAMP concentration was analyzed using a rat cAMP Elisa kit (ml002907, Jiangsu Enzymic Bio-technology Co., Ltd., China).
Enzyme Linked Immunosorbent Assay
The levels of interleukin (IL)-1β (MM-0922R2), IL-6 (MM-1011M2), and tumor necrosis factor (TNF)-α (MM-160002) in the serum were measured by ELISA kits (Jiangsu Enzymic Bio-technology Co., Ltd., China), according to the manufactures’ instructions.
Infarct Size Measurement
The cardiac infarct size in each group was measured after 24 h of reperfusion using 2,3,5-triphenyltetrazole chloride (TTC) staining. Briefly, the heart was transected horizontally into five sections and stained with 2% TTC solution (Solarbio, G3005, Beijing, China) for 15 min in darkness. The infarct tissue was unstained and appeared white.
Cardiac Fibrosis Measurement
Cardiac fibrosis was analyzed by Masson staining following 24 h of reperfusion. Briefly, the heart was filled with 4% paraformaldehyde solution for 48 h, and then paraffin was embedded. The paraffin-embedded tissues were then cut into 3 μm sections and stained with Masson trichrome staining reagent (solid green method, Solarbio, G1343, Beijing, China). Muscle fibers were dyed red and collagen fibers were dyed blue or green. The ratio of myocardial fibrosis was analyzed by Image J 5.0 software (National Institutes of Health, USA).
Immunohistochemistry
The paraffin-embedded sections (3 μm) were dewaxed and dehydrated and then treated with citrate buffer for 10 min at 95°C. After blocking with 5% bovine serum albumin (BSA) for 20 min, polyclonal primary antibodies A2aR (1:200, orb338920, Biorbyt, Shanghai, China), collagen I (1:500, orb312178, Biorbyt, Shanghai, China), and collagen III (1:200, orb213759, Biorbyt, Shanghai, China) were individually added overnight at 4°C. After washing with PBS, the secondary antibody rabbit IgG (H+L) antibody (1:1000, orb763497, Biorbyt, Shanghai, China) was added for 30 min at room temperature. Immunohistochemistry of the sections was prepared using the super sensitive polymer-HRP ISH detection system (DAB) substrate kit. The positive expression was quantified by Image J 5.0 software.
Western Blot
Total protein of myocardial tissues was extracted using a protein extraction kit (Solarbio, bc3640-50t, Beijing, China) and mixed with electrophoresis sample buffer. The proteins (40 μg) were separated by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE, Bio-Rad laboratories, Inc.) and transferred to a polyvinylidene difluoride membrane. The membranes were blocked with 5% non-fat milk for 1 h and then incubated at 4°C overnight with primary antibodies A2aR (1:800, orb338920, Biorbyt, Shanghai, China), collagen I (1:800, orb312178, Biorbyt, Shanghai, China), collagen III (1:800, orb213759, Biorbyt, Shanghai, China), fibronectin (1:800, orb10665, Biorbyt, Shanghai, China), matrix metalloproteinase-9 (MMP-9, 1:2000, orb36382, Biorbyt, Shanghai, China), and GAPDH (1:3000, orb166925, Biorbyt, Shanghai, China). After washing with Tris-buffered saline containing 0.1% Tween 20, the membranes were incubated with the secondary antibody rabbit IgG (H+L) antibody (1:1000, orb763497, Biorbyt, Shanghai, China) for 60 min. The signals were analyzed using an ECL chemiluminescence reagent (C05-07,004, Bioss, Beijing, China). The densitometric analysis of protein was quantified by Image J 5.0 software. The results of the proteins were expressed as relative optical density, compared with the GAPDH as the loading control.
Statistical Analysis
All data were expressed as mean ± standard deviation, and the statistical analysis was performed with SPSS 19.0 software (National Institutes of Health, USA). One-way analysis of variance was used to analyze the difference among groups, followed by Tukey’s test. The correlation analysis between the parameters of A2aR expression and the parameters of fibrosis was performed using Pearson correlation. p < 0.05 was considered statistically significant.
Results
SFI Inhibits MI/R-Induced Myocardial Injury via Activating A2aR
The study protocol is shown in Figure 1(A). In the present study, we found that nicorandil, an A2R agonist, had a similar role as SFI, significantly reducing the MI/R injury induced cAMP increases (Figure 1(B) and Supplementary Table 1S, shown in the Supplemental file, p < 0.01) and infarct area (Figure 1(C) and Supplementary Table 2S, shown in the Supplemental file, p < 0.01). Moreover, cardiac fibrosis was also decreased after nicorandil treatment when compared with the MI/R group (Figure 1(D) and Supplementary Table 3S, shown in the Supplemental file, p < 0.01). However, the protective effects of SFI were nullified by MSX-3, an A2aR antagonist (p < 0.01). SFI administration reduced MI/R induced infarct area, increases in cAMP, and cardiac fibrosis. Prior to reperfusion, rats injected with SFI (5 mL/kg), A2R agonist (nicorandil, 15 mg/kg), or A2aR antagonist (MSX-3, 5 mg/kg). (A) Protocol of this study; (B) cAMP concentrations were measured using ELISA kit; (C) infarct area was analyzed using triphenyltetrazole chloride staining; and (D) cardiac fibrosis was observed using Masson staining (scale bar = 200 μm). Compared with the sham group, **p < 0.01; compared with the MI/R group, ##p < 0.01; compared with the SFI group, &&p < 0.01. SFI, Shenfu injection.
SFI Increases the A2aR Expression After MI/R
In order to investigate whether SFI activated A2aR, the expression of A2aR in the myocardium of each group was determined using immunohistochemistry (Figure 2(A) and Supplementary Table 4S) and western blot (Figure 2(B) and Supplementary Table 4S). The results of Supplementary Table 4S are shown in the Supplemental file. Following MI/R injury, the expression of A2aR in the myocardium was clearly reduced (p < 0.01). The results indicated that SFI activated A2aR expression similar to nicorandil, compared to the MI/R group (p < 0.05). Additionally, MSX-3 injection counteracted activation of SFI when compared to the SFI group (p < 0.01). SFI activated A2aR expression following MI/R. (A) A2aR expression was analyzed using immunohistochemistry (scale bar = 100 μm) and (B) A2aR expression was analyzed using western blot and five animals were used in the creation of the graphs. Results analyzed by Image J 5.0. Compared with the sham group, *p < 0.05, **p < 0.01; compared with the MI/R group, #p < 0.05, ##p < 0.01; compared with the SFI group, &&p < 0.01. SFI, Shenfu injection.
SFI Decreases Collagen I and Collagen III Expression by Activating A2aR
To investigate expression of collagen I and collagen III with activation of A2aR, we used immunohistochemistry to analyze collagen I (Figure 3(A) and Supplementary Table 5S) and collagen III (Figure 3(B) and Supplementary Table 6S). The results of Supplementary Table 5S and Supplementary Table 6S are shown in the Supplemental file. Following MI/R injury, the levels of collagen I and collagen III notably increased in the cytoplasm of cardiomyocytes (p < 0.01). Injection of either SFI or nicorandil significantly suppressed the expression of collagen I and collagen III (p < 0.01), but MSX-3 administration clearly resisted the effects of SFI (p < 0.05). SFI decreased collagen I and collagen III expression induced by MI/R via activation of A2aR. Expression of collagen I (A) and collagen III (B) was analyzed using immunohistochemistry (scale bar = 100 μm). Results were analyzed using Image J 5.0. Compared with the sham group, **p < 0.01; compared with the MI/R group, #p < 0.05, ##p < 0.01; compared with the SFI group, &p < 0.05, &&p < 0.01. SFI, Shenfu injection.
SFI Decreases Fibronectin and MMP-9 Expression by Activating A2aR
The levels of collagen I, collagen III, fibronectin, and MMP-9 in the myocardium of each group were measured by western blot (Figure 4(A)). Consistent with the results of immunohistochemistry, the levels of collagen I and collagen III were significantly increased after MI/R injury compared with the sham control (p < 0.01, Figure 4(B) and (C) and Supplementary Table 5S and 6S). Similarly, the levels of fibronectin and MMP-9 were clearly elevated following MI/R injury (p < 0.01, Figure 4(D) and (E) and Supplementary Table 7S, shown in the Supplemental file). However, if either SFI or nicorandil was injected prior to reperfusion, the levels of above noted proteins were not elevated, indicating significant inhibition (p < 0.01). Administration of MSX-3 significantly counteracted the effects of SFI on the above proteins expression (p < 0.01). SFI decreased cardiac fibrosis associated protein expression induced by MI/R via activation of A2aR. (A) Collagen I, collagen III, fibronectin, and MMP-9 proteins were analyzed using western blot; relative expression of collagen I (B), collagen III (C), fibronectin (D), and MMP-9 (E) was analyzed by Image J 5.0. Five animals were used in the creation of the graphs. Compared with the sham group, **p < 0.01; compared with the MI/R group, #p < 0.05, ##p < 0.01; compared with the SFI group, &&p < 0.01. SFI, Shenfu injection.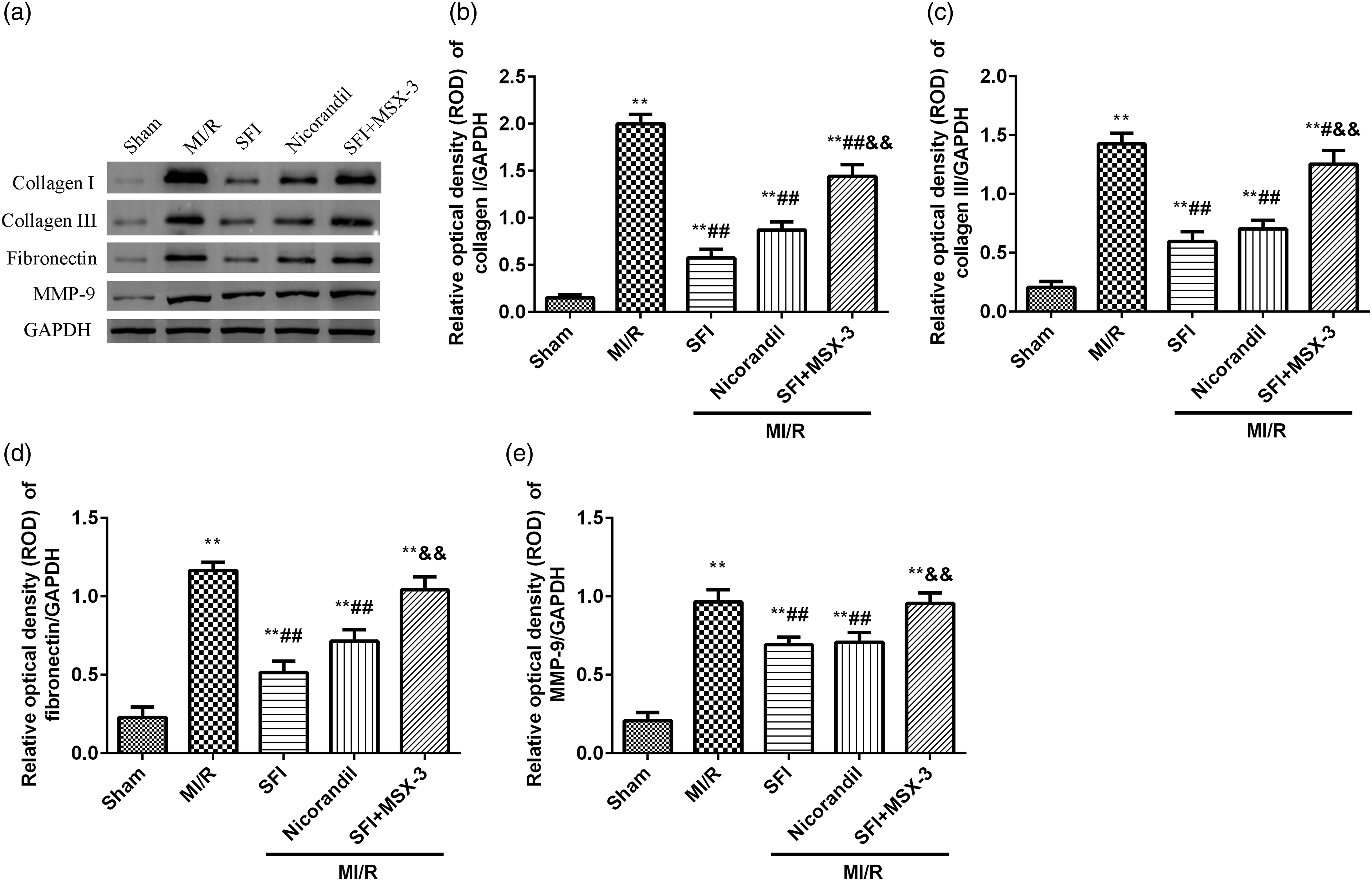
SFI Decreases Inflammatory Cytokine Expression by Activating A2aR
The levels of serum IL-1β, IL-6, and TNF-α were measured by ELISA (Figure 5 and Supplementary Table 8S). The results of Supplementary Table 8Sare shown in the Supplemental file. Following MI/R injury, these inflammatory cytokines were significantly increased compared with the sham group (p < 0.01). After injecting SFI, these inflammatory cytokines were clearly reduced compared to the MI/R group (p < 0.01). The effects of nicorandil were similar to SFI, and the effects of SFI on the inflammatory cytokines were nullified by MSX-3 (p < 0.05). SFI decreased inflammatory cytokines induced by MI/R via activation of A2aR. Levels of IL-1β (A), IL-6 (B), and tumor necrosis factor-α (C) in serum following MI/R were measured by ELISA kit. Compared with the sham group, **p < 0.01; compared with the MI/R group, #p < 0.05, ##p < 0.01; compared with the SFI group, &p < 0.05, &&p < 0.01. SFI, Shenfu injection.
The correlation between the parameters of A2aR expression and the parameters of fibrosis
The correlation analysis between the A2aR expression (immunohistochemistry) and the collagen I and collagen III (immunohistochemistry).

The correlation analysis between the A2aR expression (western blot) and the collagen I and collagen III (western blot).

Discussion
The results of our study confirmed our hypothesis that SFI prevented MI/R injury induced cardiac fibrosis through the activation of A2aR, which was the first time to demonstrate the mechanism of SFI. Cardiac injury is known to activate innate immunity, resulting in an inflammatory response, whereby neutrophils and monocytes/macrophages are recruited to the site of injury. 17 This immediate and active accumulation of high levels of neutrophils and monocytes in the infarcted area can evolve into adverse cardiac remodeling and result in a poor prognosis. 18 In this study, the inflammatory factors (IL-1β, IL-6, and TNF-α) in serum were clearly increased after MI/R, which suggested that an inflammatory response was trigged in the site of injury. In addition to phagocytosis, inflammatory cells also activate repair pathways, including the formation and deposition of scar tissue, which is principally composed of collagen and elastin/proelastin.17,18 In agreement with these descriptions, our results are also demonstrated that MI/R contributes to the formation of myocardial fibrosis, with increase in the expression of collagen I and collagen III. Cardiac fibrosis is characterized by exaggerated accumulation of extracellular matrix components in the myocardium. As extracellular matrix components in the myocardium, collagen, fibronectin, and MMP-9 play a pivotal role in cardiac fibrosis.19,20 We also found the expression of fibronectin and MMP-9 significantly increased after MI/R. For myocardial fibrosis induced by MI/R, injection of SFI prior to reperfusion could be greatly reduced by suppressing the expression of collagen I, collagen III, fibronectin, and MMP-9 proteins.
SFI, a traditional Chinese medicine, has typically been used in China for cardiac disorders such as heart failure21,22 and AMI.13,23 Multiple mechanisms of action accounting for improved myocardial function with SFI have been reported, including anti-oxidation,24,25 anti-inflammation,24,26 and anti-fibrosis. 14 In our results, SFI also showed the effects of anti-inflammation and anti-fibrosis to improve myocardial function. From our previous investigation, 11 SFI is associated with adenosine receptor activation in the treatment of MI/R. Furthermore, previous reports have indicated that A2aR activation could improve cardiac function and suppress cardiac fibrosis following cardiac injury.10,27,28 We analyzed the effects of SFI on A2aR in the treatment of MI/R. At the same time, the A2R agonist (nicorandil) and A2aR antagonist (MSX-3) were included in this study. SFI and nicorandil injection attenuated the cardiac remodeling, but MSX-3 significantly counteracted the effects of SFI. These findings contributed to our understanding of the therapeutic mechanism of SFI.
The mechanism of A2aR suppressing cardiac fibrosis may be associated with activation of extracellular signal-regulated kinases 1/2 (ERK1/2) through the G proteins (Gs)/protein kinase A (PKA) pathway 29 and activation of cAMP and Rap1 pathways. 30 In the heart, β-adrenergic receptors are mainly coupled with stimulating Gs, which activate adenylyl cyclase to generate the second messenger cAMP from ATP. The formation of intracellular cAMP is the strongest mechanism to enhance cardiac function, but the continuous activation of cAMP pathway can promote cardiac hypertrophy and fibrosis.8,31 In MI/R, the concentrations of cAMP were significantly increased, which was closely correlated with the canonical cAMP dependent PKA pathway.32,33 This explained the increase of cAMP after MI/R in our study. The activity of the cAMP-dependent PKA pathway was enhanced by MI/R, which further induced cardiomyocyte apoptosis and oxidative stress. Interestingly, the heart is equipped with the enzymes to metabolize cAMP to adenosine. 31 As has been well demonstrated, A2aR and A2bR signal through Gs to stimulate adenylyl cyclase and cAMP accumulation.34,35 In adult rat cardiac fibroblasts, a combination of cAMP-dependent signals generated via A2a and A2b receptors likely mediate adenosine’s effects, 36 and activation of A2R results in inhibition of collagen synthesis.8,37 Agreement on these findings, A2aR involves in the inhibition of cardiac fibrosis. Through our results, SFI suppressed the continuous activation of cAMP and activated A2aR expression, thereby reducing cardiac fibrosis induced by MI/R.
Nevertheless, with only a 20–25% downregulation of cAMP from injection of SFI or nicorandil in MI/R rats, even though SFI could improve MI/R, it could not fully control MI/R damage. SFI combined with other drugs or with postconditioning might offer an effective method to further control MI/R.
Conclusion
The results of the present study have provided insight into the underlying protective mechanism of SFI prior to perfusion following myocardial ischemia. SFI was shown to activate the A2aR that improved cardiac function and suppressed cardiac fibrosis, thereby preventing adverse cardiac remodeling.
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of Data and Material
All data generated or analyzed during this study are available from the corresponding author upon reasonable request.