Abstract
Background
Maprotiline intervention is noted to alleviate the body’s inflammatory response effectively, but its specific molecular mechanism remains elusive.
Objectives
This study aimed to investigate the impact of maprotiline on the inflammatory response caused by isoflurane.
Materials and Methods
Microglia BV-2 cells were grouped into control group, isoflurane group, maprotiline group, and positive control group, and were respectively receiving treatment of isoflurane (10 µg/mL) alone, isoflurane and maprotiline (10 µg/mL), and isoflurane and oxymethylene ketone (10 µmol/L), with cells of control group cultured alone. After treatments, the enzyme-linked immunosorbent assay (ELISA) assay detected BV-2 cell activity, BV-2 cell inflammatory response expression (interleukin 1 beta (IL-1β), interleukin 6 (IL-6), tumor necrosis factor (TNF)-α), ionized calcium-binding adapter molecule 1 (Iba-1) protein expression, toll-like receptor 4 (TLR4) protein, p-inhibitor of nuclear factor kappa B (p-IκBα) expression, nuclear P65, and cytoplasmic P65 expression.
Results
The isoflurane group had the lowest BV-2 cell viability and the highest content of lactate dehydrogenase (LDH), followed by the maprotiline group and the positive control group (p < .05). As isoflurane induced the contents of IL-1β, IL-6, TNF-α, and Iba-1, the addition of maprotiline or oxymethylene ketone decreased the expressions of the inflammatory cytokines (p < .05) without significant difference between the maprotiline group and the positive control group (p > .05). The isoflurane group and the positive control group had the highest expression of TLR4 p-IκBα and nuclear P65 and the lowest expression of cytoplasmic P65, followed by the maprotiline group (p < .05). There was no significant difference between the isoflurane and positive control groups (p > .05).
Conclusion
Maprotiline may enhance microglia activation and alleviate and reduce the isoflurane-induced inflammation of microglia through inhibition of TLR4/nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) signaling pathway, P65 nuclear translocation, and IκBα phosphorylation.
Keywords
Introduction
Alzheimer’s disease is the most common disease, and the neuroinflammatory response mediated by microglia contributes to the progression of the pathology (Alhotan et al., 2021). Evidence has shown that such diseases are mainly responsible for the destruction of the homeostasis of the brain tissue and the activation of microglia, which stimulate the inflammatory cells in the brain tissue and further secrete inflammatory factors, thereby inducing nervous inflammatory reactions and triggering degenerative diseases (Anzai et al., 2021). In recent years, the incidence of Alzheimer’s disease has kept rising, and the disorder has seriously affected people’s quality of life (Biavasco et al., 2021). Despite advances in treatments, they only delay the progression of Alzheimer’s disease and still hardly cure it. The recurrence rate is relatively high. Therefore, it is essential to develop an actionable treatment to improve the outcome of Alzheimer’s disease.
Maprotiline, a tetracyclic compound, mainly inhibits the process of the central nerve presynaptic membrane to restrain norepinephrine reuptake, with anti-inflammatory and anti-oxidant effects (Chidambaram et al., 2021). It is noted that maprotiline could mediate multiple signaling pathways, such as toll-like receptor 4 (TLR4)/nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), to inhibit the activation of central microglia exert and interfere with the activity of related inflammatory cells, thereby reducing the release of inflammatory cytokines (De Biasi et al., 2021). In a study by Huang et al. (2021), the administration of maprotiline in isoflurane-induced microglia can significantly reduce the level of interleukin 6 (IL-6), but its mechanism is unknown. Maprotiline also inhibits the formation of fibrous plaques and further improves the cognitive ability of patients (Kimber et al., 2020). The above findings indicate that maprotiline exhibits efficacy in Alzheimer’s disease.
The surface of microglia is coated with plenty of TLR4 protein, which may drive the activation of the downstream NF-κB signaling pathway (Kumar et al., 2021; Lima et al., 2021). The inflammatory response process involves diverse signal transduction pathways, among which NF-κB is a more critical pathway, including the following factors such as p50, p52, p65, RelB, and C-Rel (Liu et al., 2021; Lopez-Sanz et al., 2021). Normally, the above-mentioned molecules can interact with each other and with IkappaB kinase (I-κB) in the cytoplasm (Ma et al., 2021). However, in the presence of related inflammatory factors and other factors, I-κB is easily phosphorylated and degraded, while NF-κB in the free state transfers from the cytoplasm to the nucleus, thereby regulating transcription and participating in the inflammatory response (McCarson & Fehrenbacher, 2021; Morisaki et al., 2021). It is reported that inhibiting the activity of the TLR4/NF-κB signaling pathway can significantly attenuate the neuroinflammatory response (Omage et al., 2021; Polese et al., 2021). Whether the effect of maprotiline on the inflammatory response of Alzheimer’s disease is related to the TLR4/NF-κB signaling pathway deserves investigation. Therefore, this study focused on the above-mentioned problems and detected the effect of maprotiline on the inflammatory response of microglia after obtaining microglia, to provide evidence for the treatment development for Alzheimer’s disease.
Materials and Methods
Reagents and Instruments
Microglia BV-2 cells were obtained from Sigma; FBS, Penicillin, and Streptomycin were purchased from Fermentas, Maprotiline from Beijing Novartis, and Gastrodin from Sichuan Cuiyirun. Other reagents used in this study included 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) (Invitrogen); enzyme-linked immunosorbent assay (ELISA) analyzer (Fermentas), lactate dehydrogenase (LDH) kit, 5% skimmed milk powder (Abcam), primary antibodies (TLR4, ionized calcium-binding adapter molecule 1 [Iba-1], 1:500; p-inhibitor of nuclear factor kappa B (p-IκBα), NF-κB-P65, 1:1,000; GAPDH, 1:1,000), and secondary antibodies (mouse anti-rabbit 1:200) (Shanghai Beyotime).
Cell Culture and Grouping
Microglia BV-2 were cultured in Dulbecco’s modified Eagle medium (DMEM) containing 10% FBS, penicillin (100 U/mL), and streptomycin (100 µg/mL) with the medium changed regularly. As the confluency reached 80%, the microglia were sub-cultured, and ones at log phase were collected for experiments. The cells were divided into four groups: control group, isoflurane group, maprotiline group, and positive control group. BV-2 cells were cultured alone to obtain a control group. The microglia in the isoflurane group, maprotiline group, and positive control group received isoflurane (10 µg/mL), isoflurane and maprotiline (10 µg/mL), and isoflurane and oxymethylene ketone (10 µmol/L), respectively. BV-2 cells were cultured in isoflurane and placed in an anesthesia box for 6 h, whose air intake was connected to isoflurane gas at the air inlet, and the air outlet was connected to the anesthetic gas monitor.
BV-2 Cell Viability Detection
MTT Assay
Cells were seeded onto plates and incubated with MTT solution (5 mg/mL). After culturing in 5% CO2, the mixture was centrifuged at 1,500 r/min for 10 min. The lower-layer solution was transferred to the plates and added to the dimethyl sulfoxide (DMSO) solution. 15 min later, the plate was placed on the enzyme-linked immunoassay analyzer for testing.
LDH
The supernatant of BV-2 cells was placed in an automatic biochemical analyzer. Turn on the machine, observe the pressure gauge, check the sample needle and reagent needle to confirm no contaminant, and use the reagent needle to take the BV-2 cell supernatant to the computer. After recording data, turn off the machine.
ELISA (Interleukin 1 Beta (IL-1β), IL-6, Tumor Necrosis Factor (TNF-α))
After cells were cultured for 24 h, the upper layer solution was taken and centrifuged at 4,000 rpm. The solution was incubated, washed, and mixed with a working solution and an enzyme-labeled antibody working solution. After washing, the sample was added to the color reagent (100 µL) and reacted for 15 min. 100 µL stop solution was then added, and ELISA was used to examine the optical density (OD) value.
Western Blot (TLR4, Iba-1, p-IκBα, NF-κB-P65)
With a bicinchoninic acid (BCA) method measuring protein concentration, total proteins were extracted from cells and boiled in a water bath for 10 min to get the sample protein. The protein was separated by electrophoresis and transferred to a membrane. After blocking with 5% skim milk powder, the membrane was co-cultured with primary antibodies (TLR4, Iba-1, 1:500; p-IκBα, NF-κB-P65, 1:1,000; GAPDH, 1:1,000) overnight at 4°C and secondary antibodies (mouse anti-rabbit 1:200) for 1 h. Upon washing with phosphate-buffered saline (PBS), the blot was developed and photographed.
Observation Indicators
Indicators detected in this study included BV-2 cell viability, contents of inflammatory response expression (IL-1β, IL-6, TNF-α), Iba-1 protein expression, TLR4 protein, p-IκBα expression, nuclear P65, and cytoplasmic P65 expression.
Statistical Analysis
Statistical Package for the Social Sciences (SPSS) 26.0 and GraphPad Prism software were used for data analysis. After the normality test, all measurement data were in line with normal distribution, which was expressed by
Results
Maprotiline Can Increase BV-2 Cell Viability
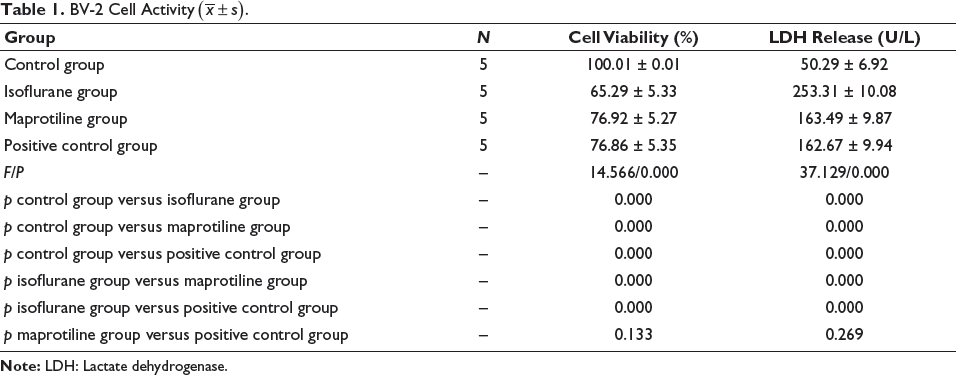
The isoflurane group had the lowest BV-2 cell viability and the highest LDH release. The addition of maprotiline and oxymethylene ketone similarly significantly restored cell viability and reduced the release of LDH (p < .05). However, the viability was still lower than that of the control group, and the LDH content was still higher. The difference between the maprotiline group and the positive control group was not significant (p > .05) (Table 1).
BV-2 Cell Activity
.
Maprotiline Improves the Inflammatory Response Induced by Isoflurane
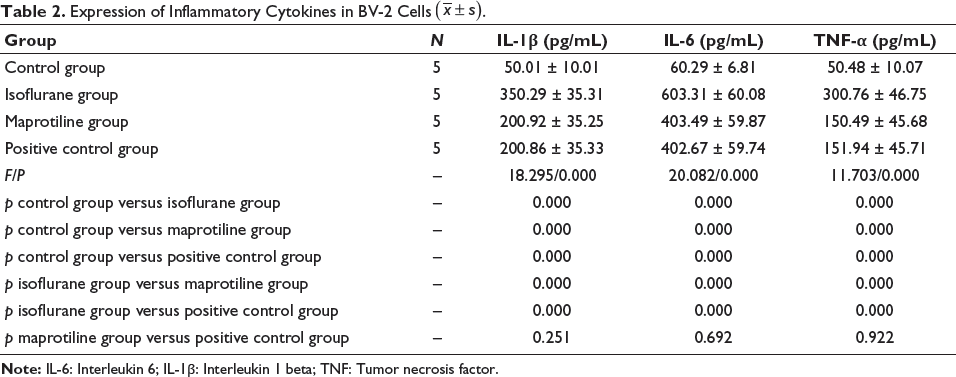
After treatment with isoflurane, the levels of IL-1β, IL-6, and TNF-α rose significantly, and they were significantly decreased by the treatment with maprotiline and oxymethylene ketone (p < .05), while the control group had the lowest level of inflammatory cytokines. The difference between the maprotiline group and the positive control group did not reach significance (p > .05) (Table 2).
Expression of Inflammatory Cytokines in BV-2 Cells
.
Maprotiline Inhibits BV-2 Cell Activation Induced by Isoflurane
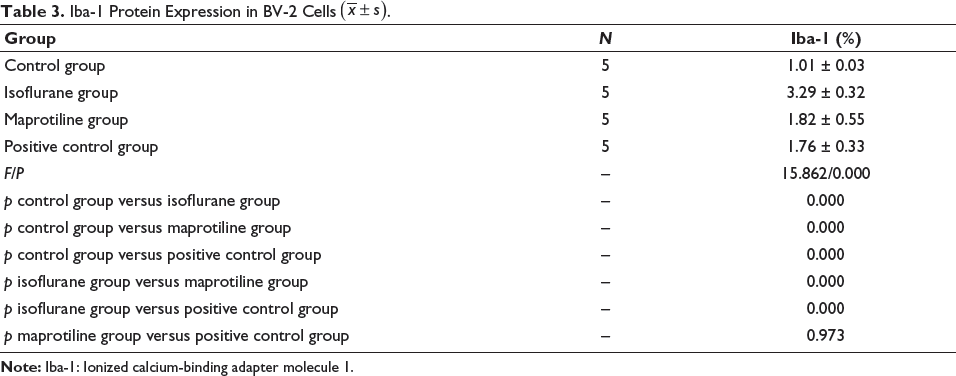
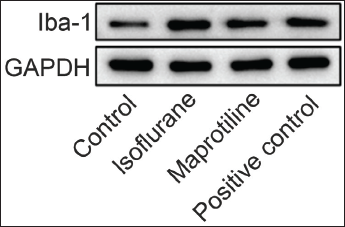
The Isoflurane group had the highest level of Iba-1 protein expression, significantly higher than the maprotiline group and the positive control group (p < .05), and there was no difference between the two groups (p > .05) (Table 3 and Figure 1).
Iba-1 Protein Expression in BV-2 Cells
.
Maprotiline Effect on Inflammation is Related to Inhibition of TLR4 Expression, Degradation of p-IκBα Expression, and Inhibition of NF-κB Nuclear Translocation
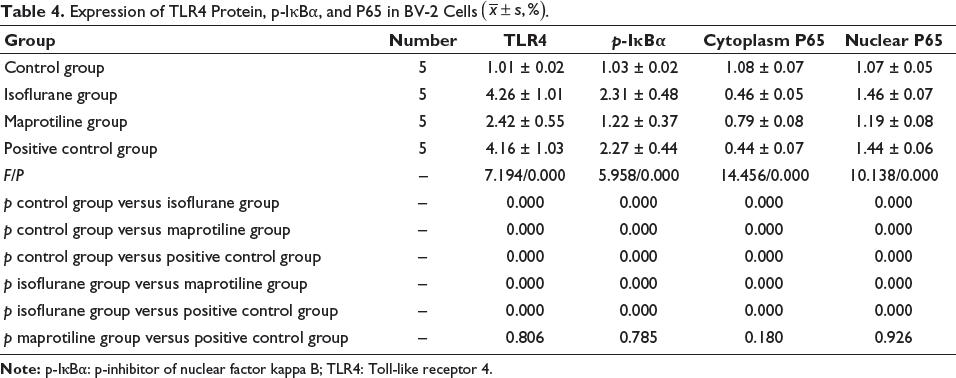
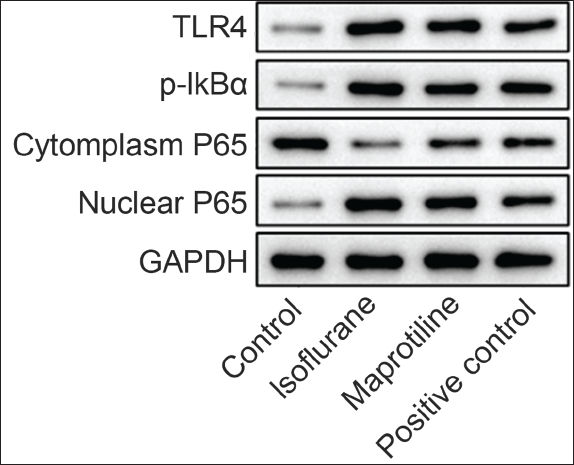
Compared with the control group, isoflurane treatment elevated the expression of TLR4 protein, p-IκBα, and nuclear P65, and, of note, the addition of oxymethylene ketone in the positive group rarely affected the expression (p > .05). Only maprotiline significantly decreased TLR4 protein, p-IκBα, and nuclear P65 expressions (p < .05) (Table 4 and Figure 2).
Expression of TLR4 Protein, p-IκBα, and P65 in BV-2 Cells
.
Discussion
This study focuses on the analysis of microglial inflammatory response induced by isoflurane and the impact of maprotiline on inflammation. After the microglia were administered isoflurane with an increased content of inflammatory cytokines, maprotiline was added to the microglia. The results show that the viability of BV-2 cells in the isoflurane group was decreased with increased production of LDH. There was no significant difference between the maprotiline group and the positive control group, and the cell viability of both groups was restored, and LDH production increased. These suggest that maprotiline and oxymetholone can increase the viability of microglia BV-2, but the mechanism underlying their impact on microglia still needs to be explored. The effect of maprotiline on the activity of microglia has been confirmed previously.
IL-1β, IL-6, and TNF-α are all direct products of the inflammatory reaction. When the activity of microglia is strengthened, it can secrete a large number of inflammatory factors and further promote the body’s neuroinflammatory response. The secreted inflammatory factors, in turn, stimulate microglia to trigger inflammation. Therefore, controlling the secretion of the above-mentioned inflammatory factors might alleviate the body’s neuroinflammatory response (So et al., 2021; Spronck et al., 2021). In this study, further observation of changes in the secretion of inflammatory factors in BV-2 cells found the highest level of IL-1β, IL-6, and TNF-α in the isoflurane group, whilst the levels in the maprotiline group and the positive control group were lower than those in the isoflurane group but still higher than those in the control group. These results indicate that maprotiline and oxymetholone may significantly inhibit the release of inflammatory factors (Sun et al., 2021). Our findings are supported by a study by Van et al. (2021), which indicates that maprotiline decreases the levels of inflammatory factors, thereby improving Alzheimer’s disease. However, the specific molecular mechanism remains to be studied.
Iba-1 is a common marker protein of microglia, and its expression could reflect the activity of microglia (Wang, Zhu et al., 2022). Herein, our results unraveled that the expression of Iba-1 protein was increased in the isoflurane group relative to the control group, indicating that isoflurane induces the activation of microglia BV-2. After treatment with maprotiline, the increased Iba-1 expression in microglia was decreased, suggesting that adding maprotiline significantly inhibits microglia activation and restrains the inflammatory response.
TLRs are common receptors on the cell surface and are related to the immune system (Wang, Trent et al., 2022). With increased activity of TLRs and their downstream pathways, they can stimulate cellular inflammatory factors and, in turn, cause inflammatory responses (Wang et al., 2021). TLR4 is highly expressed in microglia and is involved in the body’s inflammatory response through neurotoxicity mediated by microglia (Yan et al., 2021). In the present study, the results showed that compared with the control group, the TLR4 protein level in the isoflurane group was higher, and its level was reduced by the addition of maprotiline, suggesting that TLR4 activity was activated by isoflurane, but the isoflurane induction effect was relieved by maprotiline. Furthermore, the highest expression of p-IκBα and nuclear P65 was observed in the isoflurane and positive control groups, with the lowest level of cytoplasmic P65 expression. Maprotiline treatment still significantly diminished the expression of p-IκBα and nuclear P65. This shows that the activity of the TLR4/NF-κB signaling pathway in microglia increases upon isoflurane treatment, and it can be suppressed by only maprotiline rather than oxymetholone, which mediates inflammation. Mechanistically, maprotiline exerts an inhibitory effect on the activation of microglia BV-2, inhibiting TLR4 protein expression, degrading the phosphorylation level of IκBα, inhibiting P65 nuclear translocation, and regulating the TLR4/NF-κB signaling pathway. Maprotiline hence inhibits the level of inflammatory factors and ultimately improves the inflammatory response of microglia induced by isoflurane. Previous studies have consistently proved that microglia inflammation can also be affected by the TLR4-TRIF signaling pathway (Yu et al., 2021), but it has rarely been elucidated whether maprotiline may simultaneously mediate multiple pathways (Zheng, Chen et al., 2021; Zheng, Li et al., 2021).
Conclusion
Collectively, maprotiline increases BV-2 cell viability and significantly reduces the isoflurane-induced inflammation of microglia through inhibiting the activation of microglia BV-2 and inhibition of TLR4/NF-κB signaling pathway and IκBα phosphorylation level and P65 nuclear translocation, thereby improving the condition of Alzheimer’s disease. However, this study also has some limitations. As for the effect on the inflammatory response of microglia, whether the maprotiline’s impact is concentration dependent, and the optimal concentration, still needs further analysis. In addition, this study is still limited to basic experiments and has not been analyzed for patient safety. The accuracy of the conclusion still needs to be confirmed by many experimental studies.
Footnotes
Abbreviations
ELISA: Enzyme-linked immunosorbent assay; LDH: Lactate dehydrogenase; NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B cells; TLR4: Toll-like receptor 4; TNF-α: Tumor necrosis factor-α.
Acknowledgments
The authors gratefully acknowledge Huashan Hospital Laboratory for providing the necessary equipment for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the ethics committee of the Huashan Hospital.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
