Abstract
Background
Breast cancer remains a significant global health burden despite advancements in treatment. While conventional therapies often induce adverse effects, there is growing interest in exploring natural alternatives. Acacia nilotica, a traditionally used medicinal plant, has shown promise in cancer management.
Objectives
This study investigated the therapeutic potential of A. nilotica extract and nanoparticles against 7,12-dimethylbenz[a]anthracene (DMBA)-induced breast cancer in albino rats.
Materials and Methods
The formation of A. nilotica crude solution and nanoparticles was done and characterized via dynamic light scattering (DLS) and Fourier-transform infrared spectroscopy (FTIR). A total of 60 female Wistar albino rats were divided into six groups (10 rats/group) and received care in compliance with the state authorities following the Saudi Arabian rules of animal protection. The animals in the first group were given distilled water, while those in the second group were administered DMBA (50 mg/kg). The third and fourth groups were treated with 10 mg/kg of the A. nilotica crude solution and nanoparticles, respectively. In the fifth and sixth groups, A. nilotica crude solution and nanoparticles were administered with 10 mg/kg after DMBA-induced breast cancer in rats, respectively. After sacrificing the rats, the blood and breast tissues were collected from each rat and processed for histological and apoptotic markers analyses.
Results
Our findings indicated that the average size of nanoparticles was 162.5 nm in diameter with 0.145 as a polydispersity index (PDI). Functional groups were confirmed via FTIR analysis for A. nilotica crude solution and nanoparticles. Histopathological analysis revealed a marked reduction in tumor size and cellular proliferation in nanoparticle-treated groups. Our data demonstrated that both formulations significantly inhibited tumor growth and induced apoptosis, as evidenced by altered Bcl-2 and BAX expression.
Conclusion
These findings suggest that A. nilotica nanoparticles warrant further investigation as a potential therapeutic strategy for breast cancer.
Introduction
Breast cancer topped the list of cancer in women in 157 countries out of 185 in 2022, with nearly 2.3 million new cases that led to approximately 670,000 death cases globally (WHO, 2024). Cancer research has uncovered valuable clues on targets for breast cancer therapy. This includes drugs that target and protect deoxyribonucleic acid (DNA), preventing cell cycle progression and division, or those that induce programmed cell death (apoptosis) and block pathways like estrogen receptor signaling that contribute to abnormal cell growth (Hamza et al., 2022). However, tumor cells have evolved various strategies to escape destruction by the body’s immune system or even the cell’s self-destruction mechanisms (Hanahan & Weinberg, 2000). One of which is by disturbing the apoptotic intrinsic pathway by aiding in overexpressing anti-apoptotic proteins such as B-cell leukemia/lymphoma 2 protein (Bcl-2), which prevents the release of cytochrome c to block intrinsic pathways. Conversely, they downregulate pro-apoptotic proteins, such as Bcl-2-associated X-protein (BAX), which, when activated in the cell cytoplasm, can form pores in the outer membrane of mitochondria, triggering an intrinsic programmed cell death pathway within the damaged cell (Jan & Chaudhry, 2019; Qian et al., 2022). Therefore, targeting the apoptotic pathways in cancer therapy presents a compelling field of investigation, regardless of the cancer type (Pfeffer & Singh, 2018). This approach aims to overcome the resistance mechanisms developed by cancer cells.
Current cancer therapies, like chemotherapy, often face limitations such as low success rates and resistance due to a lack of targeting specificity (Fauzi & Muchtaridi, 2020; Gueyo et al., 2021). This highlights the need for exploring alternative therapeutic approaches (El-Beltagy et al., 2021). In search of potential candidates for improved treatment with greater efficacy and reduced side effects of cancer therapy (Banik et al., 2017), natural products derived from plants are gaining interest due to their ability to produce various bioactive compounds to protect themselves against stress caused by environmental factors (Doan et al., 2021). Particularly in Asian countries like China, India, Japan, Iran, and Saudi Arabia (Sak, 2022), research is ongoing to explore their potential effects against inflammation-driven diseases and to find new powerful classes of anti-cancer agents (El-Beltagy et al., 2021). Notably, studies on Acacia nilotica (family: Fabaceae), a well-known medicinal plant rich in phenolic compounds, show promise for its anti-cancer properties (Abduljawad, 2020; Farzana et al., 2014).
To address the limitations of conventional therapies and explore the potential of natural products, researchers are increasingly turning to an emerging field known as nanomedicine that holds the potential to revolutionize healthcare by targeting diseases at the molecular level. These nanoscale particles can be designed to interact with biological systems in specific ways, enabling the development of novel diagnostic and therapeutic tools that target cancer cells with high selectivity and specificity, improving treatment efficacy and reducing side effects (Chauhan et al., 2020; Kim et al., 2010).
Building upon these facts and the growing interest in nanomedicine for cancer therapy, the current study aims to investigate further the therapeutic effects of A. nilotica—derived nanoparticles synthesized using an anti-solvent precipitation method on breast cancer induced by 7,12-dimethylbenz[a]anthracene (DMBA) in rats.
Materials and Methods
Plant Collection and Extract Preparation
Dried fruits composed of pods and seeds of A. nilotica were sourced from Wadi Hanifah, Riyadh, Saudi Arabia, and identified by a specialist at the herbarium (Department of Botany, College of Science, King Saud University, Riyadh, Saudi Arabia) as A. nilotica (L.) Willd. ex Delile. The collected fruits underwent a thorough cleaning process with tap water and subsequent rinsing with distilled water. Afterward, the fruits were air-dried at room temperature for 48 h. Once completely dry, the fruits were finely ground using a grinder, and the resulting powder was sieved to obtain a fine consistency (Sadiq et al., 2015). This powder was stored in a fridge (4°C) until further use. For the preparation of the crude solution, 10 mg of the finely powdered fruits were added to 1 mL of distilled water and mixed thoroughly to achieve a homogeneous solution.
Synthesis and Characterization of Plant Nanoparticles
A. nilotica nanoparticles were prepared via a sonication-assisted anti-solvent precipitation method, a bottom-up approach that utilizes the principle of nucleation. This principle involved solute molecules from the plant crude extract (homogeneously dispersed in methanol in this case) clustering together to form nanoparticles upon encountering an anti-solvent (water). Rapid and homogeneous mixing of the solvent and anti-solvent is crucial for achieving submicron-sized particles, according to the protocol of Kakran et al. (2012). Briefly, 250 mg of A. nilotica powder was dispersed in 10 mL of methanol using the sonication method. The mixture was then rapidly added to 25 mL of preheated (90–95°C) deionized (DI) water under constant stirring. This combined solution was sonicated using an ultrasonic water bath with a nominal operating frequency of 44 kHz (±6%) and a power output of 250 W for 5 h while maintaining the temperature at 37°C as possible. Following sonication, the mixture was stirred at 200–800 rpm for 1 h at room temperature. Then, the lyophilization process was done to increase both the long-term stability of nanoparticles and the shelf life of the drug product. The synthesized nanoparticles were characterized for size using dynamic light scattering (DLS), and functional groups using Fourier-transform infrared spectroscopy (FTIR) (Perkin-Elmer FTIR-spectrum BX, USA).
Experimental Animals
A total of 60 female Wistar albino rats aged 7 weeks old and weighing 180–200 g was obtained from the Department of Medical Research on Experimental Animals Administration in the Central Lab at King Saud University. The animals have been kept at standard housing facilities (25 ± 2°C, 45 ± 5% humidity, and 12 h light and dark cycles). They have been fed on a standard laboratory chow and water ad libitum. All animals have received human care in compliance with the state authorities following the Saudi Arabian rules of animal protection.
Breast Cancer Induction
Based on the previous study of Ahmed et al. (2016), a freshly prepared single dose of 50 mg/kg of chemical carcinogen DMBA (Sigma–Aldrich®, Darmstadt, Germany) diluted in distilled water was given using subcutaneously (SC) injection to induce breast cancer in experimental rats. The animals were palpated weekly starting from the 2nd week after DMBA administration to check for the tumor appearance.
Experimental Design
The animals were grouped into six groups of 10 animals each, and used for the experimental procedures and treated as follows:
Group 1 (Control): Untreated control. Group 2 (DMBA): Rats were given a single dose of DMBA dissolved in distilled water (50 mg/kg). Group 3: Rats received A. nilotica crude solution alone (no DMBA exposure) at a daily dose of 10 mg/kg body weight. Group 4: Rats received A. nilotica nanoparticles alone (no DMBA exposure) at a daily dose of 10 mg/kg body weight. Group 5: (DMBA and A. nilotica crude): Rats received a single dose of DMBA dissolved in distilled water (50 mg/kg) and a daily dose of 10 mg/kg body weight of A. nilotica crude for 6 weeks after tumor development. Group 6: (DMBA and A. nilotica nanoparticles): Rats received a single dose of DMBA dissolved in distilled water (50 mg/kg) and a daily dose of 10 mg/kg body weight of A. nilotica nanoparticles for 6 weeks after tumor development.
The dose of the A. nilotica crude solution and their nanoparticles, as well as the route of injection, were selected based on Sakthivel et al. (2012) and Alduraihem et al. (2023), respectively. Treatment was done for 6 weeks.
Sample Collection
After the completion of dosing, rats were sacrificed by CO2 asphyxiation (Akhouri et al., 2020). Blood samples were collected through the cardiac puncture of the experimental rats, the plasma was separated for biochemical tests, and tissues of the breast were fixed in 10% neutral buffered formalin (NBF) for the histopathological studies.
Qualitative Analysis of Tumor Characteristics
The excised breast tissues were fixed in 10% NBF for 24 h, washed in running water, and then processed in ethanol for dehydration. Then, they were cleared in xylene and impregnated in parablast for blocking. Subsequently, the excised tissues were cut into 5-µm-thick sections and divided into two groups, one stained with hematoxylin and eosin (H&E) according to (Feldman & Wolfe, 2014) protocol, and the other stained with Masson’s Trichrome according to (Chang and Kessler, 2008) protocol, for histopathological investigation under light microscope.
Quantitative Analysis of Tumor Characteristics
Cell count and tissue surface area were analyzed using computer software (ImageJ) on H&E-stained samples, while Masson’s Trichrome stained samples were used for the measurement of fibrosis and optical density percentages of the breast tissue using the same software (Baviskar, 2011).
Determination of Apoptotic Markers
Anti-apoptotic Bcl-2 and pro-apoptotic BAX protein levels were measured in blood plasma using an ELISA Kit (MyBioSource, Southern California, San Diego, USA). The procedure was performed according to the manufacturer’s instructions and levels were expressed as ng/mg of plasma protein.
Statistical Analysis
Data were analyzed using GraphPad Prism software, version 10.2. One-way ANOVA and post hoc Tukey tests. A p value less than 0.05 was considered statistically significant, while a p value less than 0.0001 was highly significant. Results were expressed as Mean ± Standard Error (SE).
Results
Characterization of A. nilotica Nanoparticles
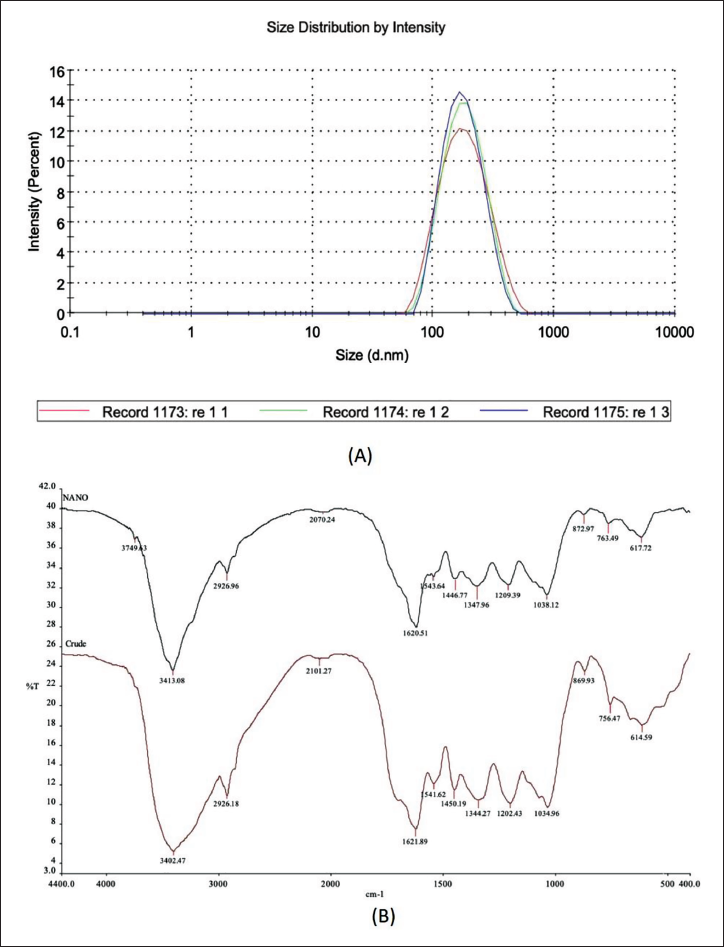
DLS analysis revealed nanoparticles with an average size of 162.5 nm (diameter) and a polydispersity index (PDI) of 0.145, indicating a relatively uniform size distribution (Figure 1A). FTIR analysis, on the other hand, confirmed the presence of characteristic functional groups in the crude extract of A. nilotica and the synthesized nanoparticles (Figure 1B). This suggests the successful incorporation of these biomolecules during nanoparticle formation.
(A) Dynamic Light Scattering (DLS) Average Size Result of Acacia nilotica Nanoparticles. (B) Fourier-transform Infrared Spectroscopy (FTIR) Spectrums for Nanoparticles of A. nilotica (A), and Crude Extract of A. nilotica (B).
Qualitative Analysis of Tumor Characteristics
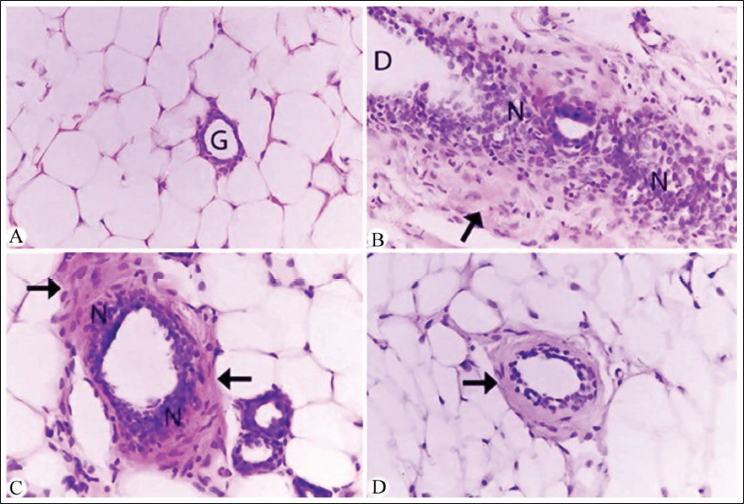
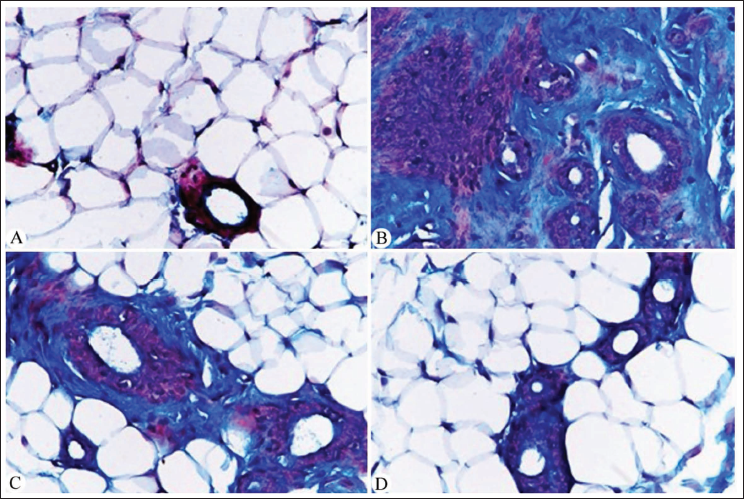
To assess the histopathological alterations in breast tissue, H&E staining was employed to evaluate tissue architecture and morphology (Figure 2). Additionally, Masson’s Trichrome staining was utilized to assess collagen deposition and fibrosis (Figure 3). Breast tissue from DMBA-treated rats exhibited characteristic features of breast cancer, including distorted glandular structures, dense collagenous stroma, and ductal hyperplasia (Figures 2B and 3B). In contrast, groups treated with A. nilotica extract or nanoparticles demonstrated improved tissue architecture with reduced cellular atypia and decreased collagen deposition (Figures 2C,D and 3C,D). Notably, the nanoparticle-treated group exhibited the most pronounced ameliorative effects on breast tissue morphology (Figures 2D and 3D).
Photomicrographs of Microscopic Section of Breast Cancer [Hematoxylin and Eosin (H&E)—400×]. (A) Normal Histological Structure of the Mammary Gland Surrounded by Adipose Tissue in the Control Untreated Group. (B) Albino Rat Breast Tissue After Tumor Induction by 7,12-Dimethylbenz[a]anthracene (DMBA) (50 mg/kg) Displaying Tumor Nest Cells (N), Dilated Glandular Duct (D), and Fibrosis Around the Mammary Glands (Black Arrow). (C) Mammary Gland in Albino Rats Treated with DMBA (50 mg/kg) and Acacia nilotica Crude Extract (10 mg/kg) Showed a Clear Decrease in the Glandular Duct Structure, Less Fibrosis Around the Gland (Black Arrows), and a Noticeable Reduction in the Number of Cancer Cells is Evident (N). (D) The Mammary Gland of Albino Rats Treated with DMBA (50 mg/kg) and A. nilotica Nanoparticles Solution (10 mg/kg) Showed an Improved Structure of the Gland and a Significant Decrescent in Fibrosis (Black Arrow).
Photomicrographs of Microscopic Sections of Breast Tissues (Masson’s Trichrome – 400×). (A) Normal Histological Structure of the Mammary Gland Surrounded by Adipose Tissue and No Fibrosis Around the Glands in the Control Untreated Group. (B) Albino Rat Breast Tissue After Tumor Induction by 7,12-Dimethylbenz[a]anthracene (DMBA) (50 mg/kg) Displaying Aggregations of Tumor Cells in Purple Color and Dense Collagenous Fibrosis Appears Stained in Blue Color Surrounding the Glands. (C) Post-Treated Albino Rats with DMBA (50 mg/kg) and Acacia nilotica Crude Extract (10 mg/kg) Displayed a Clear Improvement in the Collagenous Content Around the Glands. (D) Albino Rat Breast Tissue Showed a Healthy Structure and Significant Reduction in Collagenous Fibers Around the Mammary Glands After Treatment with DMBA (50 mg/kg) and A. nilotica Nanoparticles (10 mg/kg).
Quantitative Analysis of Tumor Characteristics
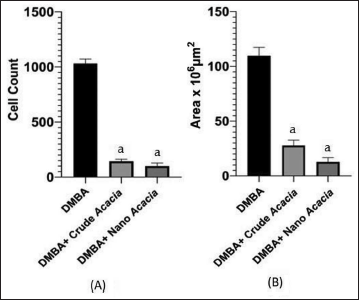
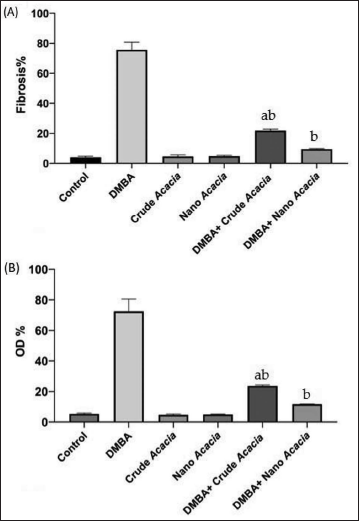
Further analysis was conducted using (ImageJ) software to evaluate tumor cell count and overall area using sections stained with H&E staining. Additionally, the tumor microenvironment was examined through quantification of fibrosis and optical density percentage (OD%) using sections stained with Masson’s Trichrome staining (Figures 4 and 5).
Quantitative Analysis of Tumor Cell Count (A) and Tissue Surface Area (B) Within Groups Treated with 7,12-Dimethylbenz[a]anthracene (DMBA), DMBA with Acacia nilotica Crude Extract and DMBA with Nanoparticles Solution. aSignificant Change at p < 0.05 with Respect to DMBA Group. Values are Expressed as the Mean ± Standard Error (SE).
Quantitative Analysis of Tumor Fibrosis (A), and Optical Density Percentages (B) Within Groups as Follows: Untreated Control and Treated with 7,12-Dimethylbenz[a]anthracene (DMBA), Groups Treated with Acacia nilotica Crude Extract and Nanoparticles Solution, and Groups Treated with DMBA and Either Crude Extract or Nanoparticles Solution of A. nilotica. aSignificant change at p < 0.05 with Respect to the Control Group. bSignificant Change at p < 0.05 with Respect to DMBA group. Values are Expressed as the Mean ± Standard Error (SE).
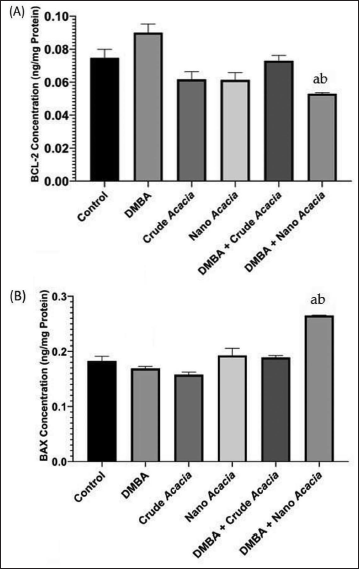
Effects of A. nilotica Crude and Nanoparticles on Apoptotic Markers
To elucidate the mechanisms underlying the anti-tumor effects of A. nilotica extract and nanoparticles, the levels of apoptotic markers, Bcl-2, and BAX proteins, were assessed in the plasma of experimental animals. Bcl-2 protein expression was significantly decreased in the A. nilotica nanoparticle group (0.05 ± 0.001) compared to the control (0.07 ± 0.005) and DMBA-only (0.09 ± 0.005) groups (p < 0.05) (Figure 6A). A significant increase in the expression of BAX, a pro-apoptotic protein, was observed in the nanoparticles group (0.25 ± 0.02) (at p < 0.05), compared to the control (0.17 ± 0.02) at (p < 0.05) and DMBA (0.18 ± 0.02) (at p < 0.0001) groups (Figure 6B). No significant alterations in Bcl-2 or BAX levels were detected in the group treated with DMBA, followed by A. nilotica crude extract.
Anti-Apoptotic (Bcl-2) and Pro-Apoptotic (BAX) Protein Level Markers in Control Untreated Albino Rat Breast Tissue, Treated Solely with 7,12-Dimethylbenz[a]anthracene (DMBA), Acacia nilotica Crude Extract or Nanoparticles Solution, DMBA Combined with Either Crude or Nanoparticles Solutions of A. nilotica. aSignificant Change at p < 0.05 with Respect to the Control Group. bHighly Significant Change at p < 0.0001 with Respect to the DMBA Group. Values are Expressed as the Mean ± Standard Error (SE).
Discussion
Previous studies have demonstrated the cytotoxic potential of A. nilotica extract and its nanoparticles against different types of human cancer cells, Alobaid et al. (2023) reported a dose-dependent cytotoxic effect of A. nilotica extract and nanoparticles fabricated using anti-solvent precipitation method (AN-NPs) on breast cancer cell line (MDA-MB-231), with AN-NPs exhibiting superior efficacy in suppressing cell viability and promoting programmed cell death in cancer cells compared to the crude extract. Similarly, another study demonstrated the anti-cancer activity of A. nilotica extract and A. nilotica silver nanoparticles (AgNPs) against SW620 and SW480 colon cancer cell lines, reporting a dose-dependent reduction in cell viability and increased cytotoxicity of AgNPs compared to the extract (Alduraihem et al., 2023). These findings collectively underscore the potential of A. nilotica-derived compounds, particularly nanoparticles, as a source of novel anti-cancer agents. To explore the therapeutic efficacy of A. nilotica nanoparticles in a more complex in vivo setting, this study investigated their effects on a DMBA-induced breast cancer rat model. Our findings indicate that both the crude extract and nanoparticles of A. nilotica exhibit promising anti-tumor properties, with improvement in nanoparticle-treated group results, suggesting their potential to induce cancer cell death and improve overall mammary gland health.
FTIR analysis confirmed the presence of functional groups within the A. nilotica nanoparticles, a key component contributing to their anti-cancer properties. Phenolic compounds of A. nilotica are well-known for their diverse bioactivities, including the induction of apoptosis (Huang et al., 2010) through multiple mechanisms, such as the intrinsic pathway involving mitochondrial dysfunction and the extrinsic pathway mediated by death receptors (Foyzun et al., 2022). Our findings support this notion, as we observed a significant decrease in Bcl-2 expression, an anti-apoptotic protein, in the group treated with DMBA and A. nilotica nanoparticles compared to the control and DMBA groups, and a concomitant increase in BAX expression, a pro-apoptotic protein, in the group treated with DMBA and A. nilotica nanoparticles compared to the control and DMBA groups. These alterations in apoptotic markers suggest that the phenolic components within the nanoparticles contribute to their anti-cancer effects by inducing apoptosis, which is consistent with Vieira et al. (2023) and Leena Panigrahi et al. (2024). However, further investigation is required to elucidate the specific mechanisms underlying the anti-apoptotic effects of these phenolic compounds, including their potential interactions with other cellular signaling pathways.
On the other hand, histopathological analysis confirmed the anti-tumor effects of both A. nilotica extract and nanoparticles in agreement with previous studies (Alduraihem et al., 2023; Alobaid et al., 2023; Revathi et al., 2017). Compared to the DMBA-only group, treatment groups exhibited significant reductions in tumor cell count (1,032 ± 41 vs. 144 ± 20 and 99 ± 29 cells), tissue surface area (110 ± 8 vs. 28 ± 50 and 13 ± 4 × 106 mm2), and fibrosis (75.67 ± 5.04% vs. 21.80 ± 0.98% and 9.43 ± 0.43%) (p < 0.05), which is a collagenous component associated with tumor progression and therapeutic resistance, that was markedly decreased in our treated animals. These findings align with previous studies linking reduced fibrosis to improved cancer outcomes (Rujchanarong et al., 2021; Sharma et al., 2022; Sheng et al., 2021).
Conclusion
Our findings demonstrated that nanoparticles exhibited superior anti-tumor properties. At the molecular level, nanoparticles induced apoptosis, as evidenced by the significant downregulation of Bcl-2 and upregulation of BAX. Moreover, histopathological analysis revealed significant reductions in tumor cell count, tissue surface area, and fibrosis in nanoparticle-treated groups. Further studies are required to elucidate the precise mechanisms of action and to evaluate the clinical translatability of these findings.
Footnotes
Abbreviations
None.
Acknowledgments
The authors want to thank Manal Awadh and Ibrahim Barakat for their help in the field.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This research was approved by the Research Ethics Committee (REC) at King Saud University (approval number KSU-SE-22-77).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Researchers Supporting Project (RSP2025R97), King Saud University, Riyadh, Saudi Arabia.
