Abstract
Background
Gentamicin (GM), a commonly used aminoglycoside anti-biotic, is effective against various bacterial infections. However, its clinical use is often limited due to its significant nephrotoxic side effects. Nephrotoxicity results from oxidative stress, inflammation, and apoptosis of renal cells. In previous research, Biochanin A (BCA), a natural isoflavone found in various plants, has demonstrated antioxidant, anti-inflammatory, and anti-apoptotic properties. This study explores the potential protective effects of BCA against GM-induced nephrotoxicity.
Objectives
To investigate the protective effects of BCA on GM-induced nephrotoxicity in male Wistar rats by assessing kidney function, oxidative stress markers, inflammatory response, and apoptotic regulation.
Materials and Methods
Male Wistar rats were divided into four groups, each group containing six animals: Group 1 as the control, Group 2 received BCA alone [50 mg/kg, intraperitoneal (IP)], Group 3 received GM alone (100 mg/kg, IP), and Group 4 received BCA (50 mg/kg, IP) along with GM (100 mg/kg, IP). At the end of the experiment day (on the 12th day), the animals were sacrificed, the organs were removed, and used for the following parameters.
Results
GM administration significantly increased the levels of creatinine, urea, uric acid, blood urea nitrogen (BUN), and lipid peroxidation levels and decreased the levels of anti-oxidant enzymes. Administration with BCA has significantly mitigated kidney function markers and oxidative stress while enhancing anti-oxidant activities. Furthermore, GM-induced rats exhibited significant upregulation in proinflammatory cytokines, proapoptotic proteins, and downregulation of anti-apoptotic proteins. Administration of BCA significantly downregulated proinflammatory cytokines and proapoptotic proteins and upregulated anti-apoptotic proteins in GM-induced rats. These results underscore the utility of necrotic and apoptotic markers in the early detection of tubular kidney damage.
Conclusion
This study conclusively demonstrates that BCA protects the kidneys from GM-induced oxidative stress, inflammation, and apoptosis of renal tubular cells, offering promise for mitigating GM-induced nephrotoxicity.
Introduction
Acute kidney injury (AKI), also known as acute renal failure, is a frequent and often severe clinical condition that can occur as a complication of various critical illnesses. It is characterized by a sudden and significant reduction in the glomerular filtration rate (GFR) and an increase in serum creatinine levels. These changes ultimately result in acute uremia, indicating the presence of toxic levels of waste products in the blood, as well as alterations in urine production (Kwiatkowska et al., 2021; Petejova et al., 2020). Various substances, including industrial chemicals, natural products, pesticides, and environmental contaminants, can harm the body’s organs to some extent (Kanna et al., 2015). Around 3.2% of adverse reactions caused by the drugs can lead to damage in the renal cells (Pierson-Marchandise et al., 2017).
Gentamicin (GM) belongs to aminoglycosides and is extensively used as an anti-biotic to treat contagions triggered by gram-negative bacteria (Brereton et al., 2018). Due to its risk of potential renal toxicity, GM is deterred despite high anti-bacterial efficiency (Mohamed et al., 2019). Up to 10–25% of individuals treated with GM have been reported to have AKI; this includes patients who just got one dose of the medication (Hayward et al., 2018; Sun et al., 2018). However, the exact mechanism of GM-induced renal cellular damage is yet unknown; the drug’s harmful effects on the kidney have been associated with the induction of hydroxyl radicals (·OH), superoxide anion (O2−), and hydrogen peroxide (H2O2) (Aurelien-Cabezas et al., 2023). GM-induced nephrotoxicity in preclinical research is characterized by histologic alterations such as interstitial fibrosis, tubular damage, and tubular atrophy (Moghadam et al., 2017; Otunctemur et al., 2013). Oxidative stress and nitrosative stress are significant contributors to the adverse effects induced by aminoglycosides such as GM in the kidneys (Balakumar et al., 2010). Despite conflicting evidence regarding their therapeutic effectiveness, the utilization of compounds possessing therapeutic properties has demonstrated success in mitigating nephrotoxicity caused by GM (Boroushaki et al., 2019; Saleem et al., 2019).
Biochanin A (BCA) (4′-methoxy-5,7-dihydroxy isoflavone) is a naturally occurring isoflavone isolated from the stem and leaves of red clover (Trifolium pratense L.) (Yan et al., 2021; Yu et al., 2019). Other plants that contain BCA include Carqueja (Genista tridentata L.), peanuts (Arachis hypogaea), and soy (Glycine max L.) (Pinto et al., 2020), chickpeas (Cicer arietinum L.), and alfalfa (Medicago sativa L.) (Deavours & Dixon, 2005; Li et al., 2015). In addition, BCA is present in red clover, cabbage, alfalfa, and many other herbal products (Yu et al., 2019). BCA is commonly recognized for a wide range of medicinal features, primarily to treat osteoporosis, asthma, and (Ko et al., 2011) arthritis (Felix et al., 2020; Liao et al., 2021) for its one of the most predominant therapeutic anti-inflammatory effects by inhibiting the initiation of tumor necrosis factor-alpha (TNF-α), nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), and other proinflammatory signaling and transcription factors (Puthli et al., 2013). In our study, we have assessed the outcome of the protective effect of BCA on GM-mediated nephrotoxicity in male Wistar rats.
Materials and Methods
Chemicals
BCA (purity – ≥96.5%) and GM were procured from Sigma–Aldrich, USA. Mouse enzyme-linked immunosorbent assay (ELISA) kits, including NF-κB, TNF-α, interleukin-6 (IL-6), and cyclooxygenase-2 (COX-2), were acquired from BOSTER in the USA. Primary anti-bodies such as Bax, Bcl-2, caspase-3, and β-actin were procured from Abcam in the USA. Secondary anti-bodies were obtained from Sigma–Aldrich in the USA. All chemicals used were of analytical grade and procured from Sigma–Aldrich in the USA.
Experimental Groups
In this study, 24 male Wistar rats, aged 32 days and weighing between 180 and 200 g, were used. The experimental period lasted 12 days, and the animals were randomly divided into four groups.
Group 1: Control animals.
Group 2: BCA control (50 mg/kg b.wt.) for 12 days.
Group 3: GM (100 mg/kg/day) intraperitoneal (IP) for 12 days.
Group 4: Both GM (100 mg/kg b.wt.) and BCA (50 mg/kg b.wt.), with BCA administered 1 h after GM injection, using saline as the vehicle for BCA.
Measuring Kidney Function Markers Level in Blood Serum
Standard diagnostic kits were purchased from IDEXX, Biological Products Trading Co., Ltd., China. Automatic Analyzer (IDEXX Catalyst One®) was used to determine the levels of creatine, urea, uric acid, and blood urea nitrogen (BUN) in blood serum. In urea, 1.0 mL of buffered enzyme solution, and 10 µL of sample were added, mixed thoroughly, and incubated at 37°C for 5 min. In parallel, 10 µL of the standard and 10 µL of distilled water (blank) were processed under the same conditions. To each tube, 1.0 mL of color-developing reagent was added and mixed well. After exactly 5 min of incubation at 37°C, 1.0 mL of distilled water was added to each tube, and the developed color was measured at 600 nm.
In uric acid, 1 mL of enzyme reagent and 25 µL of sample were added and mixed by gentle inversion. In parallel, 25 µL of standard and 25 µL of distilled water (blank) were processed under the same conditions. The tubes were incubated at 37°C for 5 min, and the developed color was measured at 510 nm.
In creatine, to a reagent mixture containing 0.5 mL of picric acid solution and 0.5 mL of sodium hydroxide, 0.1 mL of serum was added. The tubes were mixed thoroughly and incubated for 20 s. With the spectrophotometer set to zero absorbance using distilled water, the absorbance was measured at 510 nm after 20 s (A1) and again at exactly 45 s (A2). The change in absorbance (A2 – A1) was calculated for both the test and standard samples to determine the creatinine concentration in the test sample.
In BUN, 1.0 mL of sample and 1.0 mL of urease reagent were added to each tube and mixed thoroughly by gentle inversion or vortexing. Incubate all tubes at 37°C for 10–15 min to facilitate the enzymatic conversion of urea to ammonia. For color development, 1.0 mL of phenol solution and 1.0 mL of sodium hypochlorite solution were added to each tube, mixed well, and allowed the reaction to proceed at room temperature for 10 min to form a colored complex and read at 540 nm.
Enzymatic Antioxidant
Thiobarbituric Acid Reactive Substances (TBARS)
In a centrifuge tube, 0.5 mL of homogenate was mixed with 1 mL of TBARS (0.6%) and 3 mL of phosphoric acid (1%). The solution was subsequently heated for 45 min in a boiling water heating system. After cooling, the mixture was centrifuged for 20 min at 20,000 rpm, vortexed for 1 min, and 4 mL of n-butanol was added. After transferring to a new tube, the organic layer and its absorbance were read at 532 nm. The TBARS standard curve was created with concentrations ranging from 0 to 40 µM (Hosseinzadeh et al., 2005).
Superoxide Dismutase (SOD) Activity
A 0.5 mL of tissue homogenate was diluted with water to a total volume of 1.0 mL. To this, 2.5 mL of ethanol and 1.5 mL of chilled chloroform were added. The mixture was shaken for 90 s at 4°C and then centrifuged. The assay mixture, comprising 1.2 mL of sodium pyrophosphate buffer, 0.1 mL of phenazine methosulphate, and 0.3 mL of nitroblue tetrazolium, was combined with the enzyme extract. The reaction was initiated by adding 0.2 mL of nicotinamide adenine dinucleotide hydrogen (NADH) and incubated at 30°C for 90 s. The reaction was terminated by adding 1.0 mL of glacial acetic acid. The resulting mixture was shaken with 4.0 mL of n-butanol and left to stand for 10 min before centrifugation. The absorbance of the n-butanol layer was measured at 510 nm (Kakkar et al., 1984).
Catalase (CAT) Activity
To measure CAT activity, 0.1 mL of tissue homogenate was combined with 0.4 mL of H2O2. The reaction was stopped at time intervals of 15, 30, 45, and 60 s by adding 2.0 mL of a dichromate-acetic acid mixture. The samples were then boiled for 10 min, cooled, and the developed color was read at 620 nm (Sinha, 1972).
Glutathione Peroxidase (GPx) Activity
The assay began by mixing 0.2 mL of tris buffer with 0.2 mL of reduced glutathione (GSH) and 0.1 mL of H2O2. The reaction mixture was incubated at 37°C for 10 min. A control sample without homogenate was also prepared. The reaction was stopped by adding 0.5 mL of 10% trichloroacetic acid (TCA), followed by centrifugation (Beutler et al., 1963). The supernatant was then used to measure GSH content using Ellman’s method (1959).
Measurement of GSH
GSH was quantified using Ellman’s method (1959). A 0.5 mL sample of tissue homogenate was precipitated with 2.0 mL of 5% TCA. After centrifugation, 2.0 mL of the supernatant was mixed with 1.0 mL of Ellman’s reagent and 4.0 mL of 0.3 M disodium hydrogen phosphate solution. The yellow color formed was measured at 412 nm using a spectrophotometer.
ELISA Assay
ELISA kits are commonly used in research to measure specific proteins and biomarkers in biological samples. In this context, ELISA kits from BOSTER, USA, were utilized to assess various inflammatory markers such as NF-κB, TNF-α, COX-2, and IL-6 in serum (Sun et al., 2018). In the protocol, 50 µL of the sample and 50 µL of an anti-body cocktail were added to the experimental wells and incubated at 37°C for 1 h. The wells were then washed with 100 µL of 3,3′,5,5′-tetramethylbenzidine substrates and incubated for 10 min. Finally, 100 µL of stop solution was added, and the color development was read at optical density (OD) 450 nm using an ELISA reader. The levels of IL-6, TNF-α, NF-κB, and COX-2 in the serum were expressed as picograms per milligram of protein.
Western Blot
Lowry’s method was used to evaluate the protein content of kidney tissues that had been lysed using radioimmunoprecipitation assay (RIPA) buffer. After adding the loading buffer, the 50-µg protein sample was boiled for 5 min and then electrophoresed using sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). Following electrophoresis, the proteins were transferred to a nitrocellulose membrane (Bio-Rad). After blocking the membrane with 4% bovine serum albumin (BSA), the primary anti-body was allowed to incubate at 4°C for a whole night. In this study, the main anti-bodies used were Bax, Bcl2, caspase-3, and β-actin at a 1:1,000 ratio. After three washes with PBST (phosphate buffer saline with tween-20), the membrane was kept for 1 h at 4℃ with a horseradish peroxidase (HRP)-labeled secondary anti-body (1:7,500). Using enhancing chemiluminescence method (ECL) specific protein bands were detected (Chakrabarti et al., 2018).
Statistical Analysis
The results were presented as mean values with their corresponding standard deviations (SD). Statistical analysis was conducted using one-way analysis of variance (ANOVA), followed by Duncan’s Multiple Range Test, performed with the SPSS software package. A significance level of p ≤ 0.05 was set to identify statistically significant differences.
Results
Effect of BCA on GM-induced Kidney Function Markers in Blood Serum
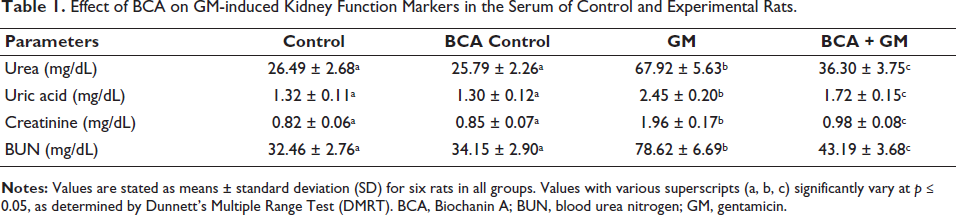
Administration of GM (100 mg/kg, IP) resulted in a notable upregulation in the amount of BUN, urea, uric acid, and creatinine levels as compared to the control group. In contrast, the treatment of BCA (50 mg/kg, IP) to GM (100 mg/kg, IP) group significantly reduces the levels of BUN, urea, creatinine, and uric acid compared to GM alone (Table 1). Additionally, there were no significant changes observed in the group administered with BCA (50 mg/kg, IP) alone.
Effect of BCA on GM-induced Kidney Function Markers in the Serum of Control and Experimental Rats.
Effect of BCA on GM-induced Anti-oxidant Activity in Kidney Tissue
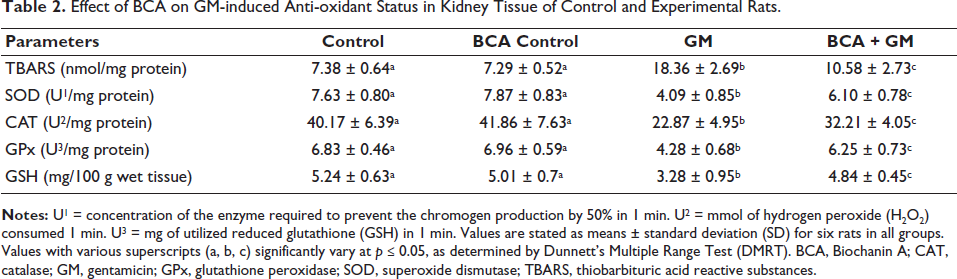
The effect of BCA on GM-induced AKI was associated with its anti-oxidant activity. We measured various anti-oxidation-related and oxidation markers, including TBARS, SOD, CAT, GPx, and GSH. As shown in Table 2, the GM (100 mg/kg, IP) group exhibited a significant increase in TBARS levels to 18.36 nmol/mg protein, while the activities of CAT, SOD, GPx, and GSH were significantly reduced to 7.63 U/mg protein, 4.09 U1/mg protein, 22.87 U2/mg protein, 4.28 U3/mg protein, and 3.28 mg/100 g wet tissue, respectively. Treatment with BCA to GM treated group has shown decreased TBARS levels and increased activities of CAT, SOD, GPx, and GSH as compared to the GM alone treated group. Additionally, BCA alone did not cause any significant changes in TBARS, SOD, CAT, GPx, and GSH activities compared to Group 1.
Effect of BCA on GM-induced Anti-oxidant Status in Kidney Tissue of Control and Experimental Rats.
Effect of BCA on GM-induced Inflammatory Markers in the Serum
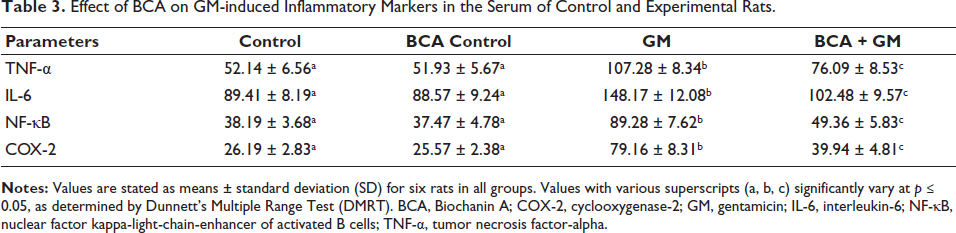
AKI is associated with an inflammatory response and oxidative tissue damage. As shown in Table 3, we examined the release levels of inflammatory cytokines in the serum of experimental animals by ELISA assay. Animals treated with GM (100 mg/kg, IP) alone showed a significant increase in NF-κB, COX-2, IL-6, and TNF-α levels compared to control rats. Conversely, treatment with BCA (50 mg/kg, IP) significantly reduced the GM-induced inflammatory cytokine response compared to the GM alone group. Notably, no significant changes in inflammatory cytokine levels were observed in the group treated with BCA (50 mg/kg, IP) alone.
Effect of BCA on GM-induced Inflammatory Markers in the Serum of Control and Experimental Rats.
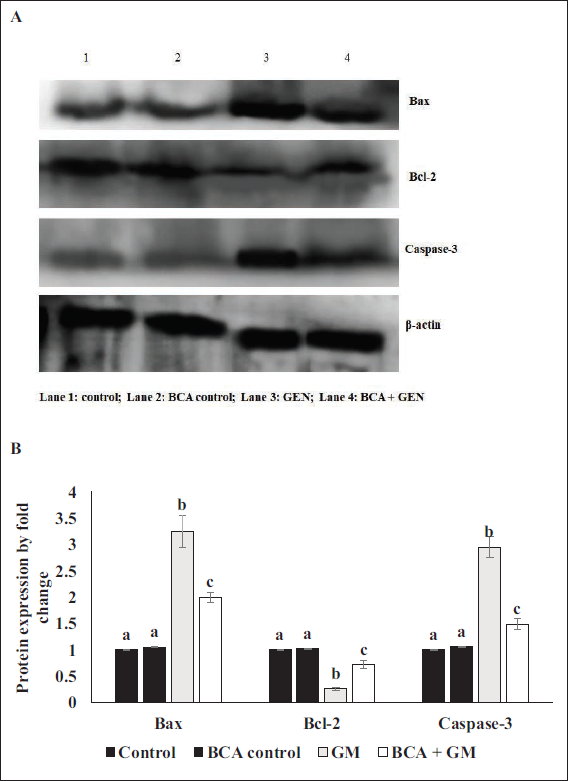
Effect of BCA on GM-induced Apoptotic Protein Expression in the Kidney Tissue
As shown in Figure 1, the GM (100 mg/kg, IP) treated group exhibited downregulated Bcl-2 protein expression and increased caspase-3 and Bax protein expression levels compared to the control group. However, in the group treated with BCA (50 mg/kg, IP) alongside GM (100 mg/kg, IP), the expression levels of caspase-3 and Bax were reduced, while Bcl-2 expression was significantly enhanced. There were no significant changes in the expression levels of apoptotic proteins in the group treated with BCA (50 mg/kg, IP) alone compared to the control group.
Discussion
Acute gram-negative bacterial contagions are often treated with aminoglycoside anti-biotics (GM). However, the main difficulty of GM administration is nephrotoxicity (Randjelovic et al., 2012). Reactive oxygen species (ROS) and inflammatory mediators are the main mechanisms behind GM-induced nephrotoxicity (Randjelovic et al., 2017). Research shows that GM is absorbed in the kidney’s proximal tubule. Additionally, its absorption in tubular cells reduces blood flow to the kidneys, lowering the glomerular purification rate and raising blood urea nitrogen and plasma creatinine levels (Emadi et al., 2021; Ghaznavi & Kadkhodaee, 2007). It, therefore, appears that using anti-inflammatory and anti-oxidant supplements can reduce the risk of this toxicity (Kurutas, 2016). Herbal medications are believed to offer numerous benefits due to the presence of various phytoconstituents with anti-inflammatory and anti-oxidant properties (Iqbal et al., 2022).
Earlier studies have exposed that BCA has potent anti-oxidant and anti-inflammatory effects (Alauddin et al., 2018; Aldhahri et al., 2022; Berköz et al., 2020). The protective benefits of BCA against GM-mediated acute nephrotoxicity in rats are probably being revealed for the first time in this study. Our findings showed that 12 days of BCA treatment could mitigate the damages developed by GM intoxication in the rat’s kidney. In this study, the amount of serum BUN, creatinine, urea, and uric acid was significantly upregulated, and it indicated renal alterations in GM-treated rats. Interestingly, the treatment of BCA significantly reduced the levels of blood biomarkers in our study. Similarly, other studies show a natural phenolic compound and flavonols such as curcumin and rosmarinic acid mitigate the serum biomarker alteration caused by the GM (Laorodphun et al., 2022; Tavafi & Ahmadvand, 2011).
GM administration to rats resulted in an increased formation of OH, O2−, and H2O2, specifically within the renal mitochondria. The oxidative damage caused by the ROS to the cellular structures and biomolecules, including proteins, deoxyribonucleic acid (DNA), and lipids is predominantly related to the reduced efficacy of anti-oxidant enzymes in the contribution of oxidative stress in nephrotoxicity. This impairment indicates a compromised characteristic of renal cells, such as the neutralizing ability and detoxification of ROS, thereby leading to oxidative stress-induced damage (Balakumar et al., 2010). In our study, we observed a significant increase in TBARS levels and significant decreases in GSH, CAT, SOD, and GPx in the kidney tissue of GM-treated rats (Balakumar et al., 2010). We found that treatment with BCA significantly increased in the activities of antioxidant enzymes in GM-induced group.. In another study, Kang et al. demonstrated Oxymatrine, a plant phytochemical, also retrieved the GM-mediated oxidative and nitrative stress, shown by the amplified GSH, CAT, and SOD activities and diminished TBARS (Kang et al., 2022). Similarly, the treatment with Glycyrrhiza glabra root extract in the GM-treated animal group has significantly increased the levels of Gpx and SOD parameters and decreased the TBARS levels (Nassan et al., 2021).
A toxic chemical that causes an inflammatory reaction in the renal tissue is likely to damage healthy kidney function, and the edema caused by the drug-devolved acute tubular necrosis probably contributed to the kidney tissue’s hypertrophy in the GM-treated animal group (Nafiu et al., 2019). The triggered oxidative stress is developed by the inflammatory process and is mediated by necrosis (Paquette et al., 2015). In our study, the inflammation shown in GM-treated animals has shown upregulation in proinflammatory cytokines such as TNF-α, IL-6, NF-κB, and COX-2. However, the treatment of BCA significantly downregulates the proinflammatory cytokines of the rat kidney, representing the anti-inflammatory activity of BCA. Apparently, in another study, Atsamo et al. (2021) demonstrated that the Cinnamomum zeylanicum extract has actively reduced the levels of the proinflammatory cytokine. Similarly, in another study, an ayurvedic medicinal compound, gymnemic acid diminished the count of TNF-α and IL-1β in rodents (Gumbar et al., 2023).
Apoptosis is essential for maintaining kidney physiological functions and plays a significant role in nephrotoxicity induced by various medications and kidney disorders (El Gamal et al., 2014). Extended use of GM medications may cause acute tubular apoptosis and kidney damage. It is commonly known that Bcl-2 functions as an anti-apoptotic protein during apoptosis, while Bax and caspase-3 work as proapoptotic proteins (Chen et al., 2017). In our study, GM treated group has shown upregulation in the expression of Bax, caspase-3, and diminished the Bcl-2 expression. Interestingly, the administration of BCA to rats treated with GM has shown significantly less apoptotic variation than GM-treated alone rats. Recently, Althunibat et al. (2022) have demonstrated that Formononetin, a natural plant flavonoid, has rescued against the GM-mediated apoptosis in the kidney tissues of rats and has shown diminished proapoptotic protein expression and upregulated anti-apoptotic protein expression. Likewise, apigenin, a natural plant flavonoid commonly present in many edible plants and known for its potent beneficial effects, has suppressed apoptosis by reducing the levels of proapoptotic protein as compared to the GM-administered animal group (Hussein et al., 2022).
Conclusion
The findings of this study provide compelling evidence that BCA can effectively protect against GM-induced nephrotoxicity in male Wistar rats. BCA improved markers of renal function markers, mitigated oxidative damage, and enhanced antioxidant enzyme activities. Moreover, BCA reduced the activation of proinflammatory cytokine production and proapoptotic proteins, alongside the upregulation of anti-apoptotic proteins. These results suggest that BCA holds significant potential as a therapeutic agent for preventing or minimizing GM-induced kidney damage by counteracting oxidative stress, inflammation, and apoptosis in renal cells. Future studies may explore BCA’s detailed molecular mechanisms and clinical applicability in mitigating nephrotoxic side effects in broader therapeutic contexts.
Footnotes
Abbreviations
AKI: Acute kidney injury; BCA: Biochanin A; CAT: Catalase; COX-2: Cyclooxygenase-2; ECL: Enhanced chemiluminescent; GM: Gentamicin; GPx: Glutathione peroxidase; GSH: Reduced glutathione; IL-6: Interleukin-6; NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B cells; SOD: Superoxide dismutase; TBARS: Thiobarbituric acid reactive substances; TNF-α: Tumor necrosis factor-alpha.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The Committee for the Care and Use of Laboratory Animals at Shaanxi Provincial People’s Hospital, Shaanxi Province (Xian, China) evaluated and approved the methodology of experiments.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
