Abstract
Introduction
Vancomycin (VCM) has an antibacterial effect against aerobic and anaerobic gram-positive bacteria, which is a kind of glycopeptide antibiotic. It has been used to treat methicillin-resistant Staphylococcus aureus infections. 1 VCM is mainly excreted via kidney. Trials showed that VCM induced nephrotoxicity. The VCM-induced nephrotoxicity has been reported to occur in 5–25% of patients who were administered. 2 The mechanisms of VCM-induced nephrotoxicity remain unclear yet. A current study suggested that oxidative stress might underlie in the pathogenesis of VCM-induced renal injury. 3 The cause might be indirectly generated reactive oxygen species (ROS) associated with inflammatory events. ROS may produce cellular injury via several mechanisms, which include peroxidation of membrane lipids, protein denaturation and DNA damage. 4 Dose and duration of administration are limited owing to the fact that VCM induces renal toxicity. 5
Thymoquinone (TQ), the main constituent of the volatile oil from Nigella sativa seeds, is reported to possess a strong antioxidant property. 6 It neutralizes oxygen radical anions acting as a cleaner. TQ has been shown to increase the level of antioxidant enzymes such as glutathione peroxidase (GSH-Px), glutathione reductase and catalase. 7 TQ acts as a powerful superoxide radical scavenger. Similarly, it is effective against the superoxidized radicals. 7 –9 TQ protects organs against oxidative damage that was induced by a variety of free radical-generating agents. These agents include the cisplatin-induced nephrotoxicity, 10 doxorubicin-induced cardiotoxicity 11 and carbon tetrachloride-evoked hepatotoxicity. 12 Its effects against nephrotoxicity are induced by various chemicals. 10 ,13 Effects of TQ on the VCM-induced nephrotoxicity have not been investigated. The aim of this study was to investigate the potential protective effect of TQ on VCM toxicity in renal tissue of rats.
Materials and methods
Animals
Experimental procedures were approved by Faculty of Veterinary, Ethics Committee, Mustafa Kemal University (Antakya, Turkey), for the use and care of laboratory animals. A total of 28 male Wistar albino rats, weighing 200–250 g, were purchased from the Laboratory Animal Production Unit of Saki Emirli (Ankara, Turkey). All rats were kept in an environment with controlled temperature (23 ± 2°C) and humidity (55–60%), and they were acclimated for 1 week before the start of the experiment. They were housed in a facility with a 12-h light–dark cycle. All animals were fed balanced rat chow (Ankara Yem Sanayi, Ankara, Turkey) and were provided tap water to drink ad libitum.
Experimental protocol
The rats were randomly divided into four groups consisting of seven rats per group: (a) control (C) group: normal saline was injected intraperitoneally at a dose of 0.3 ml and continued at 24-h intervals for 8 days in the control group rats; (b) VCM group: VCM (Vancocin-CP flacon 0.5 g, Lilly, Istanbul, Turkey) was injected intraperitoneally at a dose of 200 mg/kg, which is the dosage scheme reported to cause marked nephrotoxicity in rats. 14 VCM treatments were started 1 day after the first administrations of TQ and continued at 12-h intervals for 7 days; (c) VCM plus TQ group: VCM was injected intraperitoneally at a dose of 200 mg/kg and continued at 12-h intervals for 7 days; TQ (TQ dissolved in saline and heated at 50°C; obtained from Sigma Chemical Co. St. Louis, Missouri, USA) was injected intraperitoneally at a dose of 10 mg/kg and continued at 24-h intervals for 8 days; (d) TQ group: TQ was injected intraperitoneally at a dose of 10 mg/kg and continued at 24-h intervals for 8 days.
Specimen collection
Rats were anesthetized with intramuscular ketamine hydrochloride (Ketalar; 50 mg/kg; Eczacibasi, Istanbul, Turkey) and killed 24 h after the last injection. Blood samples were taken from portal vein into tubes. After centrifugation at 4000 r/min for 5 min at 4°C, the serum was removed for several biochemical estimations. After killing the animal, both the kidneys were excised from the rats; the left kidney was washed with prechilled physical saline and frozen promptly in a deep freezer for biochemical analysis, until further use. All samples were protected under −80°C until analysis. Both kidneys were removed, weighed, decapsulated and divided equally into two longitudinal pieces. One-half of the left kidney was placed in formaldehyde solution for routine histopathological examination by light microscopy. The entire right kidney and the other half of the left kidney (1 g) were washed with physiological saline for analysis of malondialdehyde (MDA), superoxide dismutase (SOD) and GSH-Px.
Biochemical analysis
Renal impairment was determined by blood urea nitrogen (BUN) and serum creatinine (Cr). The levels of BUN and Cr were assayed with an autoanalyzer (Architect c8000, Abbott) using the commercial Abbott diagnostic kits.
Before we started to analyze, the tissues were homogenized with prechilled physical saline in tissue homogenizer, Teflon homogenizer (Ultra Turrax IKA T25 Basic, Germany), after cutting the kidneys into small pieces with a scissors (for 2 min at 5000 rpm). Analyses of level of MDA protein were carried out at this stage. Levels of lipid peroxidation (as MDA) in renal homogenate were stated with the thiobarbituric acid reaction by the method of Esterbauer and Cheeseman. 15 The values of MDA were expressed as nanomoles per gram protein. The homogenate was then centrifuged at 3000 r/min for 10 min to remove debris. Clear supernatant fluid was taken, and analyses of GSH-Px and SOD activities and measurements of protein concentrations were carried out in this stage. Activity of total SOD was detected according to the method of Sun et al. 16 The principle of this method is resided inhibition of nitro blue tetrazolium (NBT) reduction by the xanthine–xanthine oxidase system as a superoxide generator. Activity was inspected in the ethanol phase of the lysate after 1.0 ml of ethanol chloroform mixture (5:3, v/v) was added to the same volume of sample and centrifuged. One unit of SOD was described as the amount of enzyme causing 50% inhibition in the NBT reduction rate. The activity of SOD was expressed as units per milligram protein. GSH-Px (EC 1.6.4.2) activity was determined by the method of Paglia and Valentine. 17 The protein content in the renal tissue was assayed in homogenate, supernatant and extracted samples according to the method of Lowry et al. 18
Histological evaluation
The kidneys were fixed in 10% formaldehyde, sampled and embedded in paraffin for light microscopic evaluation. Tissues were sectioned into 3–4 mm in thickness and stained with hematoxylin and eosin (H&E). Six slides from each group were examined by a pathologist who was unaware of the treatment regimens used. Kidney sections were evaluated semiquantitatively.
Tubular epithelial alterations (dilatation, desquamation, vacuolization, necrosis, atrophy and casts), interstitial inflammatory cell infiltration, edema and glomerular alterations were examined. All histopathological parameters were graded as follows: (−): showing no changes; mild (+): single cell necrosis, slight degenerative changes, few foci of dilatation, casts, inflammatory infiltration and edeme; moderate (++): for all changes at different foci throughout the kidney; severe (+++): extensive and marked changes. 19
Statistical analysis
Statistical evaluations were performed using the “SPSS 13.0 for Windows” packet program. In general, any significant differences between these groups were evaluated using the Kruskal-Wallis test. The Mann-Whitney U test was used to compare the groups with each other. Results are presented as mean ± SD; p < 0.05 was regarded as statistically significant, p < 0.01 was regarded as highly statistically significant.
Results
Levels of BUN and serum Cr
Levels of serum BUN and Cr were summarized in Table 1. Levels of serum BUN and Cr in the VCM group were found to be significantly higher than that of the control groups (p < 0.01 BUN, p < 0.01 Cr). Levels of BUN and Cr in the VCM + TQ group were detected to be significant regressed when it was compared with VCM group (p < 0.01 BUN, p < 0.01 Cr).
Levels of BUN and Cr in the serum of the four groups of rats a

VCM: vancomycin; TQ: thymoquinone; BUN: blood urea nitrogen; Cr: creatinine.
aResults are presented as mean ± SD.
b p < 0.01 compared with control group.
c p < 0.01 compared with VCM group.
Levels of renal MDA and activities of renal SOD and GSH-Px
Table 2 summarizes levels of MDA and the activities of renal SOD and GSH-Px enzymes in all groups. Levels of MDA in the VCM groups were increased compared with control (p < 0.01). On the other hand, treatment with TQ reversed the enhanced levels of MDA in the VCM + TQ group, compared with VCM groups (p < 0.01). In the VCM group, when the activities of SOD were compared with the control group, there was no significant difference for statictical analysis (p > 0.05). Treatment with TQ significantly increased the activities of SOD in the VCM + TQ group when compared with control group (p < 0.05). The activities of GSH-Px were significantly decreased in the VCM group in comparison with the control group (p < 0.01). Activities of GSH-Px were significantly increased in the VCM + TQ group in comparison with the VCM group (p < 0.001).
MDA, SOD and GSH-Px values in the kidney of the four groups of rats (n = 7 for each group) a

MDA: malondialdehyde; SOD: superoxide dismutase; GSH-Px: glutathione peroxidase; VCM: vancomycin; TQ: thymoquinone.
aResults are presented as mean ± SD.
b p < 0.01 compared with control group.
c p < 0.01 compared with VCM group.
d p < 0.05 compared with control group.
e p < 0.001 compared with VCM group.
Kidney histopathology
Table 3 summarizes kidney histopathology of all groups. In histologic examination, control samples of kidneys showed normal kidney morphology (Figure 1). VCM caused significant changes in tubular epithelium like vacuolization; desquamation, atrophy and necrosis; interstitial edema and inflamation in general architecture (Figure 2). But, administration of TQ provided a great improvement in the renal morphology (Figure 3). Tubular and glomerular structures were seen close to their normal structures in the VCM + TQ group.

Control rat showing normal tubular architecture, tubules, and glomerules that appear normal (H&E ×200).

The epithelial cell vacuolization, desquamation, interstitial inflammation (i), luminal casts (tc) and dilatation (d) of renal tubules are clearly observed in the kidney of vancomycin-treated rat (H&E ×200).
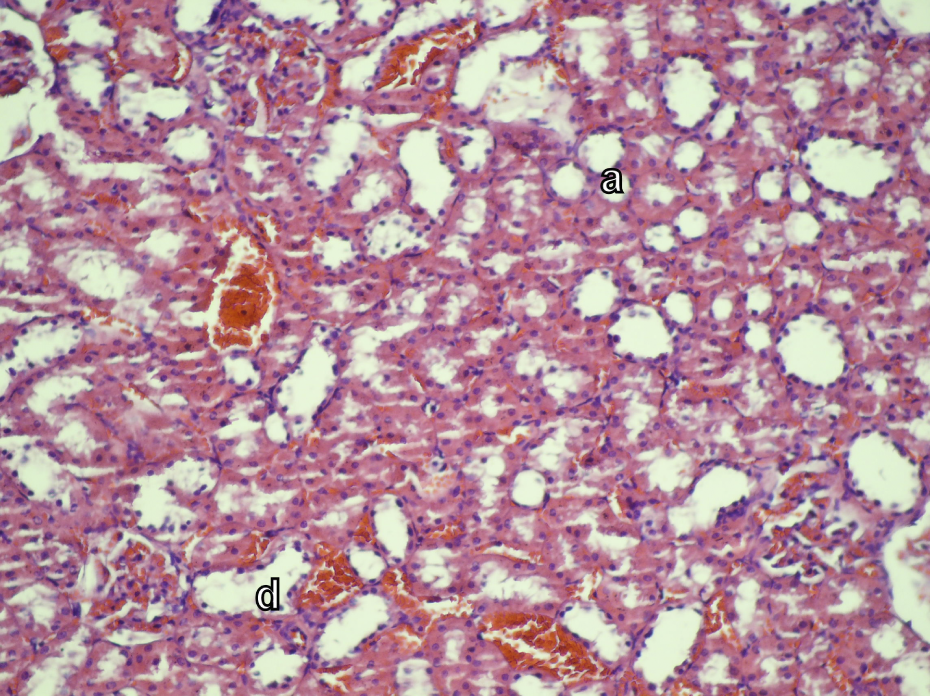
Almost all morphology is preserved in timokinon- plus vancomycin-treated rat. Mild degeneration and tubular dilatation (d) and atrophy (a) are observed in some tubules (H&E ×200).
Quantification scores of the data showing histopathological changes affected by VCM and VCM + TQ groups in kidney tissue a

VCM: vancomycin; TQ: thymoquinone.
aHistopathologic assesment of the experimental parameters was scored as follows: score (−): no meaningful histopathologic change; score (+): mild degree; score (++): moderate degree; score (+++): severe degree; n = number of observations.
Discussion
In this study, the protective effect of TQ on VCM-induced nephrotoxicity was investigated. Hereby, oxidant and antioxidant parameters (levels of MDA as well as activities of SOD and GPX) in kidney tissue and levels of BUN and Cr in serum were studied. Histopathological changes in renal tissue were also evaluated. When a literature review is performed, we saw that our study was the first trial in which the protective effect of TQ on VCM-induced nephrotoxicity was investigated on the basis of biochemical and histological data.
VCM usage has led to increased infections in β-lactam-resistant gram-positive microorganisms in recent years. 20 Administration and dosage of VCM are limited due to renal injury induced by VCM. 5 The molecular mechanism of renal damage of VCM has not been known yet. 2,5 But, recent studies have proposed that one of the underlying mechanisms of the pathogenesis of renal injury may be oxidative stress. 3,19,21 On the other hand, there are different studies which demonstrate that oxidative damage has vital role in nephrotoxicity models, which were induced by VCM. 22,23
The kidney is a highly sensitive to toxic damage. 24 In recent studies, significant increase in the levels of serum BUN and Cr was detected following VCM treatment. 14,21,25 Levels of serum BUN are significant for the detection of late renal tissue damage, while levels of serum Cr are important for the detection of early renal failure. 26 In our study, levels of serum BUN and Cr in the VCM group were found to be significantly higher than that of the control group. These results pointed out that VCM administration causes serious nephrotoxicity in rat model. Levels of BUN and Cr in the VCM + TQ group were found to be less than that of VCM group.
Aerobic metabolism is of utmost importance in generating reactive oxygen products. 27 King and Smith emphasized that VCM played a stimulant role for free radical production and oxidative phosphorylation by increasing oxygen consumption and cellular adenosine triphosphate concentration. 28 Free radicals can cause cellular injury, DNA damage, peroxidation of membrane lipids and protein denaturation via various mechanisms. 4 Lipid peroxidation leads to damage in the structure and function of membrane. This damage results in the generation of various end products such as MDA. 29 Thus, there may be a direct proportion between MDA and lipid peroxidation. Accordingly, increase in the level of MDA is accepted as an indicator of increase in lipid peroxidation, and a decrease in the level of MDA is accepted as an indicator of decrease in lipid peroxidation. 30
Nishino et al., 3 Oktem et al. 19 and Cetin et al. 31 reported a significant increase in the levels of renal MDA in nephrotoxicity models, induced by VCM given intraperitoneally for 7 days at a dose range of 200–400 mg/kg. They emphasized that lipid peroxidation was evoked by oxidative damage. This also played an important role in the pathogenesis of nephrotoxicity of VCM. 3,19,31 Similarly, when levels of MDA in VCM group were compared with the control group, a significant increase was observed in VCM group. In the VCM + TQ group, the levels of MDA were found to be significantly less than those of the VCM group. These results indicated that lipid peroxidation was involved in the pathogenesis of nephrotoxicity induced by VCM. On the other hand, we considered that TQ was able to create a retrospective effect by decreasing lipid peroxidation.
Cells have protective enzymes and antioxidant molecules such as natural SOD and GSH-Px against destructive effects of free radicals. Overrelease of free radicals may surpass the antioxidative capacity of biological systems and lead to serious cellular damage. 32 SOD is the most important protective enzyme against oxidative stress in renal tubulus. 24 In the recent studies, a significant decrease in the activities of SOD was found in renal injury which was induced by VCM. 3,19 In our study, a similar result was observed in the activity of SOD but the decrease was not statistically significant. But, a significant decrease was detected in the activity of GSH-Px. In the VCM + TQ group, a statistically meaningful increase was observed in the activity of SOD when compared with the control group. Activity of GSH-Px in the same group was found to be significantly higher compared with the VCM group. El-Abhar et al. have showed that a high-dose TQ (50–100 mg/kg) raised the activities of SOD in the injury model which was induced by ischemic–reperfusion. 33 Al-Majed et al. have carried out a study by inducing neuronal injury on rat hippocampus via ischemia. They found an increase in the levels of MDA and a decrease in the levels of SOD. They also showed a decrease in MDA, and SOD level returned to normal following TQ administration (5 mg/kg per day oral). 34 Fouda et al. have induced a renal injury on rats with mercuric chloride and found a decrease in the activity of renal GSH-Px. They also found improvements in these enzyme levels following TQ administration (10 mg/kg per day). 35 Sayed-Ahmed and Nagi carried out a nephrotoxicity model, which was induced by gentamycin and found a decrease in the levels of GSH-Px. But they detected an increase in these enzyme activities following TQ treatment. 36 Compared to the literature, our study also showed similar results. Accordingly, TQ raised the levels of SOD and GSH-Px and proved antioxidant-protective effect in renal injury, which was induced by VCM.
Recent studies emphasized that antioxidant substances provided renal protective effect via diminishing lipid peroxidation. 19,21,28,37 TQ has been the focal point in the pharmacological studies in recent years due to its strong antioxidant property. TQ protects organs against oxidative damage that was induced by a variety of free radical-generating agents. Additionally, it protects organs against cisplatin-induced nephrotoxicity, 10 doxorubicin-induced cardiotoxicity 11 and carbon tetrachloride-evoked hepatotoxicity. 12 When 10 mg/kg TQ was administered for ameliorating doxorubicin-induced cardiotoxicity and nephrotoxicity, we saw that TQ protected rat tissues. 11,38 In our study, increase in the activities of SOD and GSH-Px and decrease in MDA were detected after administering TQ against renal injury which was induced by VCM. Furthermore, histopathological evaluations supported these findings. All results indicate that TQ has an antioxidant effect on renal damage which was induced by VCM.
Pathogenesis of medicine-originated nephrotoxicity may be linked with the accumulation of medicines within cells. Various substances that contain VCM are reabsorbed through the epithelium of renal proximal tubulus. Reports showed that VCM toxicity occurs in the proximal tubulus and its vicinity. 39 Cetin et al. have stressed that nephrotoxicity by VCM arose from acute tubulointerstitial damage. 31 Oktem et al. have showed that 7-day VCM protocol (200 mg/kg per day) caused heavy degeneration in cortical tubular cells. 19 In our study, various parenchymal damages such as interstitial edema, tubular dilatation, tubular cell desquamation and vacuolization were observed in histopathological examination of rat kidney, which was extracted following VCM injection. These injuries were significantly improved by giving TQ. These results demonstrated that VCM-induced renal failure could be ameliorated by TQ.
These results indicated that TQ was a retrospective agent that lessens both toxic damage of VCM and lipid peroxidation of superoxide radicals. TQ may show this effect by decreasing the lipid peroxidation in the renal tubular cells.
In our study, we proved that VCM induced kidney damage by raising the levels of serum BUN, Cr and MDA. Also, we histologically indicated that TQ protected kidney damage by diminishing the levels of serum BUN, Cr and MDA and increasing the levels of SOD and GSH-Px. TQ had a marked positive effect on VCM-induced nephrotoxicity. TQ ameliorated oxidative status, biochemical damage and histopathological changes.
As a result, renal tubular damage with the increased levels of MDA can support the role of oxidative stress in VCM toxicity. TQ can interact with ROS as a general radical scavenger during VCM toxicity. We think that TQ may have a useful role as a novel retrospective agent for preventing the nephrotoxic damage of VCM.
