Abstract
Background
One of the leading causes of serious liver disease worldwide is drug-induced liver injury, which is brought on by either the direct hepatotoxic effects of a medication or one of its reactive metabolites. One often-used medication over the counter is acetaminophen (APAP). Although APAP is safe in small amounts, an overdose can result in rapid liver damage and even death from acute liver failure. It is common knowledge that the pharmaceutical business has been using more and more plant products in recent years. Traditional Chinese medicine has utilized patchouli, scientifically known as Pogostemon cablin (P. cablin) Benth, a member of the Lamiaceae family, since the Eastern Han period.
Objectives
The current study aims to assess pogostone’s (PO) hepatoprotective effects on rats’ APAP-induced toxicity.
Materials and Methods
APAP was used to produce toxicity after two different dosages of PO were given. Liver function markers and biochemical parameters, bilirubin, and protein profile were used to determine the extent of hepatoprotection. The indicators of oxidative stress in the liver and kidney tissue were examined, including superoxide dismutase, catalase, glutathione, and malondialdehyde along with the histopathological analysis.
Results
PO has been found to return hepatic and renal antioxidant levels, as well as liver function markers, to normal, compared to the aberrant levels detected in the APAP-treated group. After being observed, it was discovered that the liver weight and liver index were comparable to the positive control. A liver histological examination was also performed, and the results indicated that co-administration with PO significantly reduced the histopathological lesions in the liver caused by APAP.
Conclusion
As a result, the current study demonstrates that PO has hepatoprotective effects against liver damage brought on by APAP.
Introduction
The liver, the most intricate organ in the body, performs a variety of vital tasks that contribute to the preservation of the interior environment. It is essential to the metabolism of proteins, fats, and carbohydrates. Through its xenobiotic metabolism, it also contributes to the excretion and detoxifying process of numerous endogenous and foreign substances (Ilavenil et al., 2016). About half of all abdominal organ injuries occur in the liver, making it the most frequently injured abdominal organ. A common target organ for much chemically induced damage is the liver. Numerous oxidative processes result in reactive metabolites, which have the potential to cause oxidative stress and, as a result, harm or damage to the liver. The kind of toxic chemical, the degree of intoxication, and the exposure type or chronic all influence the many forms of liver damage (Soliman et al., 2020). Several metabolic processes are distorted in cases of hepatic damage. Reactive oxygen species (ROS) and reactive nitrogen species (NOS) have a well-established involvement in the development and course of liver-related illnesses, including non-alcoholic steatosis, hepatocellular carcinoma, and alcoholic and viral hepatitis. For the treatment of liver illnesses, steroids, vaccinations, and anti-viral medications have been used; however, prolonged administration of these medications can have unfavorable side effects.
A significant health issue arises when its function is compromised. India has lost almost 18,000 lives annually because of impaired liver function (Ilavenil et al., 2016). Diseases affecting the liver must be treated, and they need to be treated carefully and thoroughly. A small number of conventional medications have been shown to be hepatotoxic at specific doses, despite their ability to protect the liver and promote liver function (Abirami et al., 2015). In clinical practice, drug-induced liver damage (DILI) is a common adverse effect because the liver microsomes metabolize a wide range of substances, including herbal remedies and complementary medicines. Before developing jaundice, individuals with fulminant liver failure who have never had liver disease before were present with hepatic encephalopathy, and coagulopathy is the most harmful clinical manifestation.
Rats’ toxicity to acetaminophen (APAP) (N-acetyl-p-aminophenol, paracetamol) is a widely accepted experimental model for assessing the hepatoprotective potential of plant extracts. APAP, also referred to as paracetamol in Europe, is an aminophenol that functions primarily as an anti-pyretic and an analgesic. While therapeutic amounts of APAP are thought to be safe, an overabundance of the drug might cause fatal liver damage (Liao et al., 2023). The most well-known type of APAP-induced hepatotoxicity is acute, severe hepatocellular damage brought on by an unintentional or intentional overdose. Excessive doses of APAP have a direct, toxic effect that is the cause of the harm. The liver is the primary organ responsible for metabolizing and excreting APAP; overuse of the medicine can harm the liver and cause toxicity linked to both the drug and its metabolite, N-acetyl-p-benzoquinone imine (NAPQI). It is well known that NAPQI causes oxidative stress, which in turn generates the release of free radicals such as ROS and NOS, as well as the depletion of cellular glutathione (GSH), a natural antioxidant level. Hepatotoxicity ultimately comes from an imbalance in the cellular antioxidant defense mechanism in liver hepatocyte cells. APAP is frequently employed as a model liver toxin for the experimental validation of hepatoprotective medications due to its well-known mechanism of hepatotoxicity (Bharali et al., 2014). Serum alanine transaminase (ALT) and aspartate transaminase (AST) levels often rise significantly 24–72 hours after consumption, signaling the onset of hepatic injury. Clinical signs, such as jaundice, disorientation, liver failure, and in rare cases, death, appear 48–96 hours later. Renal insufficiency is also frequently demonstrated. The majority of APAP’s conversion to innocuous glucuronate or sulfate conjugates occurs in the urine (Igami et al., 2015). Since the mid-1980s, there have been reported occurrences of APAP-induced hepatotoxicity in the United States, and all indications point to an increasing incidence going forward. According to reports, this is one of the pharmaceutical products that causes drug-induced liver injury the most frequently. An estimate of the mortality rate for overdose patients is 0.4%, which translates to 300 deaths per year in the United States. The drug’s dose is the primary determinant that determines the onset and severity of APAP hepatotoxicity; however, some contend that the interval between APAP intake and N-acetylcysteine (NAC) therapy is just as significant, if not more so. The occurrence and degree of liver damage following APAP overconsumption are influenced by numerous additional factors (Yoon et al., 2016).
Naturally occurring bioactive phytochemicals found in plants include antioxidants, which can delay or prevent the oxidation of lipids and other molecules by preventing the start or spread of oxidative chain reactions (Azim et al., 2017). Numerous studies have shown that because of specific interactions and synergisms, natural compounds with antioxidant activity are useful in preventing liver diseases associated with oxidative stress. The goal of the current medicinal research is to identify antioxidants that are found naturally, especially in plants. There are numerous plant species that have been discovered to have biomolecules that might be used to make hepatoprotective medications, and research is still being done to identify the best one. Therefore, the purpose of the current study was to determine whether using pogostone (PO) could lessen the hepatic damage caused by APAP.
PO (C12H16O4) is the major constituent and one of the secondary metabolites of Pogostemonis herba. The dried aerial portion of Pogostemon cablin (Blanco) Benth is known as P. herba (Yi et al., 2013). P. cablin is a plant native to the Philippines that is also found in tropical regions of Asia. It is currently grown commercially in West Africa, India, China, Indonesia, Malaysia, Singapore, and Vietnam (Swamy & Sinniah, 2015). Numerous pharmacological actions, such as anti-bacterial, anti-fungal, anti-influenza virus, anti-inflammatory, and antioxidative effects, have been shown to exist in it (Li et al., 2014). P. cablin is well known for its oil, which is utilized in the perfume and cosmetic industries as well as a food additive (Zhao et al., 2022). It is thus a significant medical herb with enormous business potential in the perfume sector. Numerous bioactivities, including antioxidant, anti-inflammatory, and immunosuppressive properties, have been identified for PO. Previous studies demonstrated an obvious protective effect of PO on endotoxin-induced acute lung injury (ALI) in mice (Yang et al., 2018), ALI, and lung cell injury (Zhang et al., 2021). According to research on its pharmacokinetic characteristics, PO taken orally has a high bioavailability and is easily absorbed (Rezazadeh et al., 2022).
This study’s main objective is to cause APAP-induced liver damage in rats and assess if PO can heal the resulting damage. Utilizing serum biochemical markers and parameters, the functionality of the liver was assessed. The antioxidant capacity of PO in kidney and liver tissues was also investigated. To detect obvious changes, the histological changes were also assessed. These results may serve as a foundation for comprehending PO’s function in the management of liver damage.
Materials and Methods
Materials
Prior to the start of the study, all chemicals, reagents, kits, and equipment were purchased commercially and made available.
Experimental Animals
The applicable national regulations and institutional policies for animal use and care were followed, and the rats were acquired from the Approved Animal Canter with the appropriate ethical approval for animal experimentation studies. For the investigation, male Wistar rats were obtained. Animals were kept in 12-hour light–dark cycles in polycarbonate cages and regulated ambient temperature between 23°C and 25°C with 55%–60% relative humidity. They had free access to drinking water and normal rat food pellets.
The animals were split up into five groups at random; each group had six rats, each of which weighed between 150 and 160 g. The first group functioned as a normal control by receiving only water for 8 days, the second group was induced with APAP at 750 mg/kg bwt/8 days, the third and the fourth group as an experimental group where APAP stimulated groups were administered with 15 mg/kg bwt and 30 mg/kg bwt of PO, respectively, and the fifth group served as a positive control was the APAP exposed rats were treated with 100 mg/kg bwt of Silymarin (SIL).
Effects of PO on Liver Function Markers of APAP Intoxicated Rats in Serum
Blood samples were centrifuged to extract serum, and an automated analyzer measured the levels of AST, ALT, and alkaline phosphatase (ALP) present in the serum. Serum from the experimental animals was tested for liver function biochemical indicators like ALT, AST, and ALP using standard kits that were supplied by the provider (Abirami et al., 2015).
Effects of PO on Liver Biochemical Parameters of APAP Intoxicated Rats in Serum
Blood samples were taken via direct heart puncture under light ether anesthesia, following an APAP dosing period. The serum was separated using centrifugation at 2,500 rpm for 15 minutes, and standard kits were used to analyze the total bilirubin and total protein.
Effects of PO on Liver Weight and Liver Index
The carbon dioxide method was employed to sacrifice the animals, and the liver was taken out and promptly weighed following the collection of blood samples. The formula used to determine the liver index was: rat liver weight/rat weight × 100%.
Identification of Important Indicators of Oxidative Stress in Liver and Kidney Tissues
After being cleaned and homogenized, a portion of the experimental animals’ liver tissue that was sacrificed was stored at a pH of 7.4. After centrifuging the contents for 20 minutes at 4°C at 10,000 × g, the supernatant was examined for the presence of GSH, catalase (CAT), and superoxide dismutase (SOD). Using commercial assay kits, the activities of SOD and GSH and the level of CAT were measured in accordance with the manufacturer’s instructions. The colorimetric approach was utilized to quantify the malondialdehyde (MDA) level, which serves as an indicator of lipid peroxidation (Soliman et al., 2020).
Effect of PO on Histopathology in Liver Tissues
Immediately following animal sacrifice, the removed livers were cleaned with buffer, sliced into small pieces, preserved in a 10% formalin solution, and finally embedded in paraffin wax. Hematoxylin-eosin was used to stain the tissues that had been fixed. Under a microscope, the histological alterations were inspected. A digital image recorder was used to take photographs of the sections (Ilavenil et al., 2016).
Statistical Analysis
The data were reported as mean ± standard deviation (SD) (n=6) and were obtained from different tests. The results were statistically analyzed using the GraphPad Prism program. The SPSS 17.0 Windows statistical program and one-way analysis of variance (ANOVA) were also used for the statistical analysis. A statistically significant difference between the groups was denoted by p < 0.05.
Results
Effects of PO on Liver Function Markers of APAP Intoxicated Rats in Serum
Animal experiments were conducted to look into the impact of PO and SIL treatment on APAP-induced liver injury. The increased serum hepatic enzymes (ALT, AST, and ALP) in the rats given an APAP overdose indicated severe liver damage and cell necrosis. In the meantime, the elevated activities of blood hepatic marker enzymes were considerably reduced by oral treatment of PO (15 mg/kg), PO (30 mg/kg), and SIL with APAP in contrast to the normal control group (Figure 1). The PO treatment’s suppression of the AST, ALT, and ALP levels was similar to that brought about by treatment with SIL, which has a well-known, strong hepatoprotective agent.
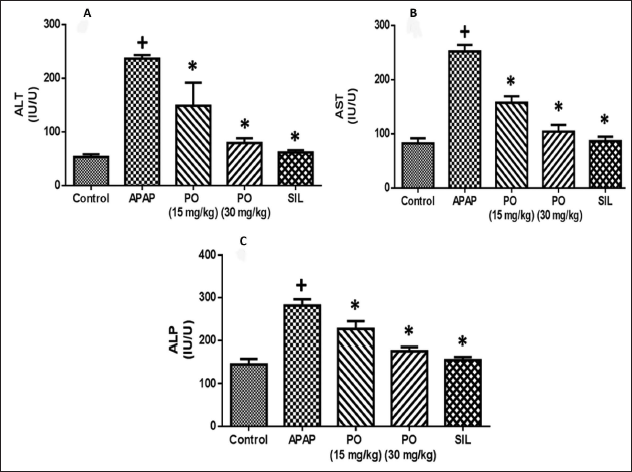
Effects of PO on Liver Biochemical Parameters of APAP Intoxicated Rats in Serum
Figure 2 shows how PO affected the amounts of total protein and serum total bilirubin. The control group treated with APAP alone had a substantially higher level of total bilirubin in its serum than the normal group. When compared to the animals treated with APAP alone, the levels of total bilirubin were significantly lower in the PO (15 and 30 mg/kg) administered animals, respectively. In animals provoked with APAP, administration of conventional pharmaceuticals, SIL, also considerably lowered the total bilirubin level when compared with the PO-treated group. Comparing the APAP-treated control group to the normal control, there was a significant decrease in total protein levels. When PO (30 mg/kg) was administered, the total protein level increased dramatically to a maximum while SIL (positive control) likewise boosted the amount to a high. This predicted the hepatic impairment caused by PO.
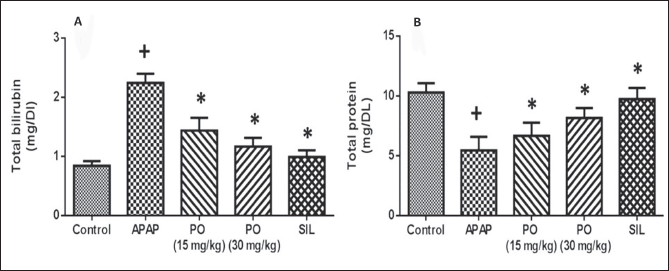
Effects of PO on Liver Weight and Liver Index
When compared to a hepatotoxic control, the PO effect on liver weight was dramatically lessened; SIL, a standard medication, had a similar effect. Moreover, the animals treated with APAP had a more pronounced increase in liver index than the ones treated with PO and SIL (Figure 3).
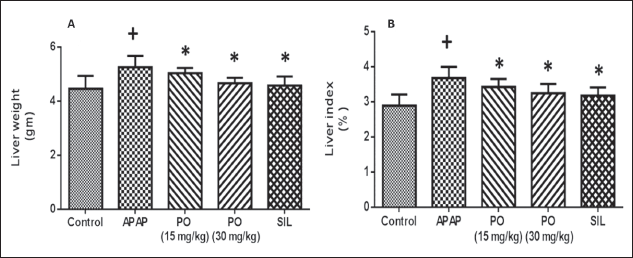
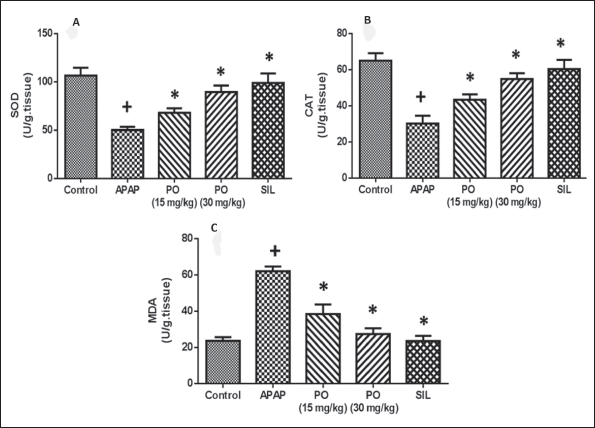
Determination of Key Oxidative Stress Markers in Liver Tissues
Reduced levels of SOD, CAT, and GSH in Figure 4 indicated that APAP intoxication led to an oxidative stress state in the liver. Comparatively to the negative control group, the experimental animals’ livers produced more MDA due to increased lipid peroxidation after receiving APAP. PO administration was successful in restoring the liver’s GSH reserves and significantly increased CAT and SOD activity. Conversely, the increased MDA levels were significantly reduced during PO therapy. At a maximal dose of 30 mg/kg of PO, the levels and activities were comparable to those of the positive control SIL, which is comparable to the normal control rats.
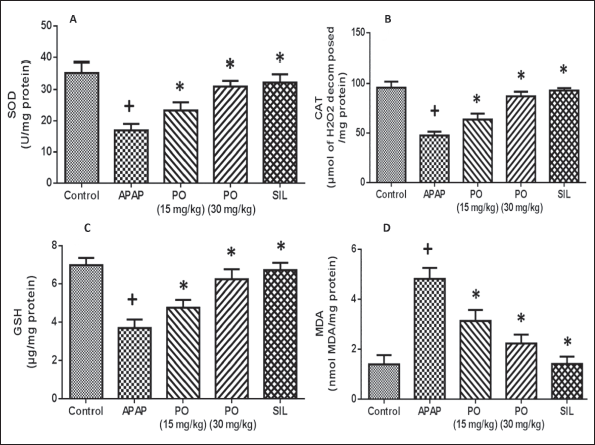
Antioxidant Activity in Kidney Tissue
Figure 5 shows that when APAP was administered, there was a large increase in MDA levels and a significant drop in SOD and CAT levels when compared to control values. These biochemical markers in the kidney tissues of the PO-treated groups were nearly back to normal.
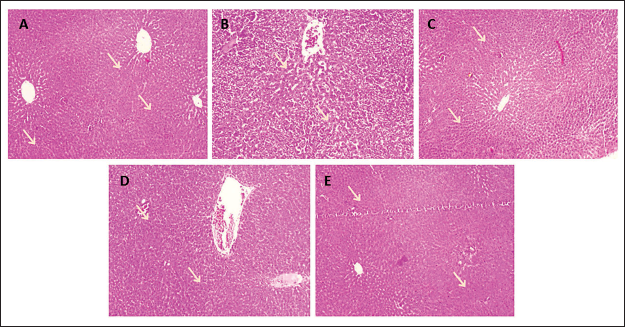
Effect of PO on Histopathology in Liver Tissues
Figure 6 displays the rats’ histological liver sections from each of the five experimental groups. Understanding the organization of tissues at every level of structure, including that of molecules and macromolecules, whole cells, intercellular materials, tissues, and organs is the main goal. The control rat’s normal liver segment displayed no histopathological changes, and the surrounding hepatocytes gave the impression that the central vein’s histological structure was normal. The tissue called the periductal sac encircling the dilated cystic bile ducts in Group 2 of rats treated with APAP displayed edema and severe congestion and dilatation in the portal vein, which was accompanied by the infiltration of inflammatory cells. Rats treated with 15 mg/kg of PO for APAP-induced congestion in the portal vein and a little infiltrate of inflammatory cells around the bile ducts were seen. Positive control SIL has normal hepatic architecture, whereas a maximal dose of 30 mg/kg of PO showed little indication of histological changes.
Histopathological Changes in the Liver after Being Treated with Acetaminophen (APAP) and/or Pogostone (PO). (A) Normal Liver Section from Control Rat Shows No Histopathological Alteration and the Normal Histological Structure. (B) APAP Treated Rats Showed Severe Congestion and Dilatation in the Portal Vein Associated with Inflammatory Cells Infiltration and Edema in the Periductal Tissue Surrounding the Cystic Dilated Bile Ducts. (C) APAP + PO 15 mg/kg Treated Rats Showed Congestion in the Portal Vein Associated with Few Inflammatory Cell Infiltrations Surrounding the Bile Ducts. (D) APAP + PO 30 mg/kg Treated Rats, There was No Histopathological Alteration Recorded. (E) SIL + APAP Received Positive Control (100 mg/kg bwt) (Hematoxylin and Eosin (H&E)).
Discussion
In general, the action of chemicals or medications’ intermediate products can potentially cause harm to the liver. Guidelines for acceptable levels in both pharmacological and toxicological settings may be provided by a better quantitative comprehension of the connection between the detoxifying process of xenobiotics and liver damage. Specifically, pharmaceutical drug development can be streamlined by accurately predicting the toxicity profile of lead candidates. An opportunity to recognize individualized medication based on the patient’s surroundings, active biomarkers, and genetics arises from a deeper comprehension of how liver damage first manifests (Mardinoglu et al., 2018). One important target organ for the metabolism of medications and xenobiotics is the liver. The buildup of hazardous intermediate metabolites, such as NAPQI, is linked to APAP toxicity (Gopinathan, 2016). This study confirmed the hepatic damage caused by APAP intoxication in rats and assessed the potential of PO as a safe natural medication alternative.
AST, ALT and ALP activities can be estimated from serum liver enzyme levels, and these results can be used as quantitative markers to determine the kind and degree of hepatocellular damage caused by APAP (Ramachandran & Jaeschke, 2019). Hepatocytes’ transport function gets disrupted during liver damage, which results in interruption to the plasma membrane and elevated activity of these enzymes, which in turn causes cellular leakage and loss of integrity (Hewitt et al., 2007). Rats given paracetamol suffer significant liver damage when exposed to high concentrations of ALP. The heightened production of ALP by bile canaliculi lining cells in response to cholestasis and elevated biliary pressure could be one reason for the elevated blood ALP level (Fernández-Murga et al., 2018). PO is well-known to have a wide range of therapeutic uses in conventional medicine, and significant research has shown that the majority of these claims are supported by the drug. Based on the serum levels of ALT, AST, and ALP in the treated groups, PO therapy significantly and dose-dependently decreased the release of these three liver enzymes. Its hepatoprotective properties were comparable to those of SIL, the positive control. Based on the aforementioned results, our study demonstrated the hepatoprotective action of PO by showing that it may considerably reduce the elevated activity of serum liver enzymes following the administration of PO at varying doses to the APAP group.
The majority of serum proteins are mostly derived from the liver. Due to faulty liver protein production, a hepatotoxic scenario will result in a decrease in total protein levels (Ramachandran et al., 2018). As a byproduct of heme in the reticuloendothelial system, bilirubin’s elevation in the bloodstream can be attributed to many factors such as excessive synthesis, heightened hemolysis, reduced conjugation, or compromised bilirubin transport (Santos & Christian, 2022). These outcomes were in line with those found in our investigation, which showed that an overdose of APAP led to a marked rise in blood total bilirubin and a decrease in total protein. In our investigation, PO administration resulted in a return of the level of bilirubin and total protein to the normal range, demonstrating its hepatoprotective effect.
Oxidative stress is the adverse outcome linked to the generation of free radicals in vivo; it is defined as elevated intracellular ROS that causes damage to lipids, proteins, and DNA (Junren et al., 2021). SOD and CAT are two examples of antioxidant enzymes that are crucial for shielding organisms against ROS. SOD is the main defensive mechanism against the effects of oxidative stress. CAT is one of the most important intracellular enzymes in the detoxification of the oxidant hydrogen peroxide (Ighodaro & Akinloye, 2018). MDA, or the amount of lipid peroxide, is produced when polyunsaturated fatty acids in biological membranes peroxidize. The assessment of MDA levels serves as a gauge for structural changes and damage to cellular membranes (Samsonov et al., 2017). Next, reduced GSH detoxifies these compounds. The nucleophilic targets of macromolecules in cells can be covalently attached to via this intermediate when GSH levels are low, which ultimately results in cell death (Potęga, 2022).
When PO was administered, the liver tissues showed decreased MDA levels and elevated GSH, SOD, and CAT activity levels. According to earlier research, PO demonstrates a strong potential for antioxidant defense by promoting the removal of hydroxyl and superoxide anion free radicals, as well as by inhibiting lipid peroxidation (Junren et al., 2021). All these data indicate the beneficial role of PO in restoring equilibrium to the APAP-treated liver’s unbalanced antioxidant system. The kidneys play a crucial part in maintaining the body’s fluid balance, balancing the electrolyte balance, and eliminating waste products such as urea and creatinine (Merrill & Chambliss, 2020). As a result, using PO was associated with higher levels of lowered CAT and SOD levels as well as lower levels of MDA. The current study’s findings clearly show that PO therapy was successful in reducing renal toxicity.
Histopathological observations of APAP-induced tissue damage in the liver revealed substantial congestion and dilatation of the portal vein, as well as infiltration of inflammatory cells and swelling in the periductal tissue encircling the cystic enlarged bile ducts (Nouioura et al., 2023). In rat models, PO treatment reduces histopathological lesions. The outcomes of biochemical tests and liver sample histopathological analysis concurred, suggesting that PO can prevent APAP-induced hepatotoxicity. It is still unclear, therefore, exactly which biochemical pathway PO uses to carry out its hepatoprotective effect. It will take further research to determine the molecular mechanism behind its therapeutic activity.
Conclusion
APAP ingestion and the ensuing hepatotoxicity are a serious issue that continues to affect people worldwide. In APAP-induced experimental rats, PO returns all hepatic functions and oxidative stress indicators to the appropriate ranges, according to all profiles of hepatoprotective study, suggesting that they can function as hepatoprotectants. Additionally, it lessened the liver’s histological lesions. As a result, to determine its therapeutic potential as a hepatoprotective drug and to clarify the precise molecular and biochemical mechanisms involved, more research utilizing more models of experimental liver injury is necessary. PO is a promising candidate for more investigation in the trials of new drugs.
Footnotes
Abbreviations
ALP: Alkaline phosphatase; ALT: Alanine transaminase; AST: Aspartate transaminase; ANOVA: Analysis of variance; APAP: Acetaminophen; CAT: Catalase; DILI: Drug-induced liver injury; GSH: Glutathione; H&E: Hematoxylin and eosin; MDA: Malondialdehyde; NAPQI: N-Acetyl-p-benzoquinone imine; NOS: Reactive nitrogen species; PO: Pogostone; ROS: Reactive oxygen species; SD: Standard deviation; SIL: Silymarin; SOD: Superoxide dismutase.
Acknowledgments
Nil.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the Ethical Committee of The First Affiliated Hospital of Shandong First Medical University & Shandong Provincial Qianfoshan Hospital (Approval No. 2024081501).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
