Abstract
Background and Objectives
An overdose of acetaminophen (APAP) usually leads to acute liver injury, and oxidative stress is one of the fundamental mechanisms used to characterize it. Procyanidins (PCs) can reduce the oxidative stress in the liver of mice. This study aimed to investigate the potential protective role of PCs against APAP-induced acute liver injury.
Materials and Methods
Experiments were performed on male Kunming mice in six groups: phosphate-buffered saline, PCs, APAP, and PCs pretreated with 10, 50, and 100 mg/kg. The mice were peritoneally injected with PCs 30 min before the administration of APAP. First, survival rates of mice were scored every 12 hr for three days in succession. Furthermore, serum levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), total bilirubin (T-Bil), total cholesterol (TC), triglyceride (TG), tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and interleukin-1 (IL-6) were determined. Additionally, histological analysis and hepatic oxidative stress including the levels of superoxide dismutase (SOD), malondialdehyde (MDA), and glutathione (GSH) were assessed. Finally, the protein expression of nuclear factor erythroid 2-related factor 2 (Nrf2) and SOD-1 was detected by Western blotting.
Results
The data indicated that PCs improved survival rates of APAP-induced liver injury in mice models. Moreover, PCs could reduce the elevated serum levels of ALT, AST, T-Bil, TC, TG, TNF-α, IL-1β, and IL-6 due to APAP exposure with a dose-dependent manner. Besides, PCs pretreatment attenuated hepatic histopathological damage and oxidative stress which manifested the increases of SOD and GSH, whereas the decrease of MDA. Furthermore, PCs enhanced the protein expression of Nrf2 and SOD-1 in the PCs pretreatment groups compared with the APAP group.
Conclusion
PCs ameliorated APAP-induced acute liver injury, and Nrf2 signaling pathway modulating antioxidative stress might be involved in it.
Introduction
Acute liver injury (ALI) is a worldwide disease that poses a serious threat to human health. Many factors are involved in the pathogenesis, including viral infections, drug and ethanol exposure, radiation damage, and accidental food poisoning.[1–3] In recent years, drug-induced liver injury (DILI), consisting of more than 50% of ALI, has markedly increased due to drug abuse[4]. It is a governing cause of high mortality.[5] Meanwhile, DILI has become an essential obstacle to clinical therapies, leading to a major concern for the withdrawal of drug candidates.[6]
Acetaminophen (APAP) was initially introduced to the market as an extensively used analgesic and antipyretic agent by Von Mering in 1893,[7] which is also the most commonly known drug to induce liver injury when it is overdosed. It has become one of the main reasons for acute liver failure (ALF) in the United States, leading to about 30,000 cases of hospitalization and around 500 deaths each year.[8] Mechanistically, accidental or intentional intake of APAP overdose often results in mitochondrial dysfunction and reactive oxygen species (ROS) production, which impairs cellular macromolecules and finally leads to acute liver necrosis.[9–11] At present, there is no specific drug to treat APAP intoxication besides antidote N-acetylcysteine (NAC),[12] which often have many inevitable side effects such as nausea, vomiting, urticaria, and delirium, and its clinical application was limited.[13] Hence, it is exigent to develop a newer, more effective, and safer medicine to prevent and treat liver injury induced by APAP.
The significant toxicity of APAP is caused by N-acetyl-p-benzoquinone imine (NAPQI), consisting of cytochrome P450-mediated hepatotoxic adducts.[14] NAPQI could form covalent bonds with other intracellular proteins, leading to dysfunction of mitochondria, oxidant stress, and lipid peroxidation in liver cells.[15,16] Simultaneously, increased NAPQI products override the detoxification of glutathione (GSH) S-transferase (GST), used out of GSH, and affect the synthesis of GSH, which leads to the decreased activity of GST,[17] and mitochondrial proteins by a mechanism related to oxidative stress.[18] Eventually, it leads to increased ROS, impaired cellular macromolecules, and hepatocellular necrosis.[19,20] Thus, the current research mainly focused on finding a new drug against APAP.[21–23]
Procyanidins (PCs), identified as condensed tannins, have drawn much attention for their protective effect in the liver, existing widely in fruits, vegetables, nuts, seeds, and flowers.[24] Besides, they have beneficial biological effects in humans, such as antioxidative effects.[25] Researchers have conducted studies on PCs for more than 50 years, especially since the 1980s, leading to much more profound knowledge and research findings concerning PCs. In addition, investigators have found that PCs can reduce the level of malondialdehyde (MDA) and ROS in the liver cells of mice, while they can increase the expression of superoxide dismutase (SOD).[26] Furthermore, PCs can inhibit lipid peroxidation, directly promote the antioxidant ability in vivo, and reduce liver cell injury, providing liver protection. A transcription factor named the nuclear factor erythroid 2-related factor 2 (Nrf2) plays a special function for protecting the lever from oxidative stresses.[27] Under normal circumstances, Nrf2 is combined with its inhibitor Kelch-like ECH-associated protein 1 (Keap-1) in the cytoplasm. Ubiquitylation and proteasomal degradation of Keap-1 regulate the expression of Nrf2. Nevertheless, intracellular ROS accumulation results in critical cysteine oxidation of Keap-1. Then, released Nrf2 activates antioxidative enzymes.[28] A previous study has indicated that activating the Nrf2 signaling pathway plays an important role in APAP-induced ALI.[29] Surprisingly, whether PCs can reduce ALI induced by APAP induced via the Nrf2/SOD-1 signal pathway is still unknown. We hypothesized that the antioxidant properties of PCs would ameliorate the oxidative stress caused by APAP-induced free radicals hence prevent liver injury via the Nrf2/SOD-1 signal pathway. Therefore, the purpose of this study is to explore the potent protective effect of PCs on APAP-induced ALI in vivo.
Materials and Methods
Study Area
The study was carried out at the Department of Pharmacy, Chongqing Medical University, from February 2016 to December 2017.
Materials
Standard substance of PCs, product code B2615-100 mg, was purchased from Shanghai Yuanye Bio-Technology Co. Ltd (Shanghai, China), the purity of which was more than 95.2%. Alanine aminotransferase (ALT), aspartate aminotransferase (AST), tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and interleukin-1 (IL-6) were measured by a multiscan spectrum, obtained from Nanjing Jiancheng Bioengineering Institute, Jiangsu, China. Serum levels of T-Bil, TC, and TG were detected by commercial kits from Human Diagnostics Worldwide, Germany, and what’s more MDA, GSH, and SOD were measured by detection kits from Shanghai Beyotime Biotechnology Co. Ltd, Shanghai, China.
Animals
In this study, the experimental protocol strictly followed the rules of animal ethics procedures and regulations of the People’s Republic of China. The Animal Ethical and Experimental Committee approved all animal experiments in this research of Chongqing Medical University. All surgeries were carried out under sodium pentobarbital anesthesia to minimize suffering. Male Kunming mice of 6–8 weeks and weight of 18–22 g, purchased from the Experimental Animal Center of Chongqing Medical University under specific pathogen-free conditions, were placed in a full automatic temperature-controlled room at 22 °C ± 2 °C with 50% ± 5% humidity. All mice were acclimatized for 7 days before experiments and exposed to 12 hr dark and light cycle with free access of food and water. The Animal Ethical and Experimental Committee approved all animal experiments in this research of Chongqing Medical University. All surgeries were carried out under sodium pentobarbital anesthesia to minimize suffering.
Experimental Design
The experiments consist of two parts. First, a total of 60 mice were divided into six groups with each group comprising 10 mice: (a) phosphate-buffered saline (PBS) control, (b) PCs (100 mg/kg), (c) APAP model (400 mg/kg), (d) PCs (10 mg/kg) + APAP (400 mg/kg), (e) PCs (50 mg/kg) + APAP (400 mg/kg), and (f) PCs (100 mg/kg) + APAP (400 mg/kg). PCs were administered once by injection into the peritoneal cavity of mice 30 min before the injection of APAP. Second, the PBS control and APAP groups were intraperitoneally infused in mice with an equal volume of sterile PBS and 400 mg/kg APAP. Eight hours after APAP treatment, mice were euthanized by cervical dislocation, and then their blood and liver specimens were collected to evaluate liver injury. First, the liver specimen was fixed in the concentration of 10% neutral buffered formalin for morphological observation. Then, the rest of liver tissue was cleaned with PBS solution reserved at −80 °C in the refrigerator. On Day 1, after the final immunization, mice’s survival rates were carefully assessed every 12 hr for 7 days in succession to explore the posttreatment impact with PCs on the mortality of APAP (400 mg/kg)-treated mice. What’s more, the cumulative survival curve was depicted using the Kaplan–Meier method.
Assessment of Liver Functions
Blood samples were obtained from orbital venous sinuses in mice. Furthermore, serum was isolated by centrifugation at 12,000 g and 4 °C from the whole blood. AST, ALT, T-Bil, TC, and TG were applied to indicate hepatocyte functions, which were measured by the multiscan spectrum according to instructions of assay kits.
Measurement of Serum Inflammatory Cytokines
Serum levels of inflammatory cytokines, including TNF-α, IL-1β, and IL-6, were tested by the enzyme-linked immunosorbent assay (ELISA) following the commercial kits’ instructions. The optical density was conducted on an automatic ELISA reader at 450 nm, with concentration expressed as pg/mL (Dakewe Biotech Co., Shenzhen, China).
Histological Analysis
Liver tissues from the ALI model mice were removed under sterile conditions and fixed in 10% formalin for consecutive 24 hr before regular histological processing. Then, these liver tissues were embedded in paraffin, deparaffinized, dehydrated, sliced into 5 µm sections, and stained with hematoxylin and eosin (H&E). Technicians were blinded to the specific group of each section and conducted conventional morphological evaluations using a light microscope (Olympus, Tokyo, Japan). Besides, liver slices were then observed at ×100 magnifications by a single pathologist. What’s more, six photomicrographs were taken for each group to evaluate APAP-induced hepatic necrosis. Furthermore, the percentage of the necrotic area was measured by the Image J software (Bethesda, USA). Moreover, scores were recorded from 0 to 4(no abnormality to most severity), according to standards based on exuding, edema, hemorrhage, and inflammatory infiltration.[26]
Determination of Hepatic Oxidative Stress
Frozen liver tissues were homogenized in the 4 PBS, and the supernatants were collected via centrifugation at 3,000× g and 4 °C for 15 min. Next, the supernatants were applied for the biochemical analysis of oxidative stress, including SOD, MDA, and GSH. And the procedures were complied with their manufacturers’ instructions. With normalizing the total protein concentration of the same sample, the MDA content was detected by the absorbance measured at 540 nm, SOD activity was measured at 450 nm, and the GSH level was measured at 340 nm.
Western Blotting
Liver tissues were lysed in protease lysis buffer and phosphate inhibitors for 30 min. Bicinchoninic acid protein measurement (Beyotime, China) was applied to measure protein concentrations. And 20 µg proteins were separated in 12% sodium dodecyl sulphate-polyacrylamide electrophoresis and electrophoretically transferred on the polyvinylidene difluoride (PVDF) membrane. The PVDF membrane was blocked with 5% defatted milk for 1 hr and incubated at 4 °C with a primary antibody of murine-originated monoclonal antibody (Nrf2 and SOD-1), and β-actin served as a protein controller. The next day, the PVDF membrane was incubated at 37 °C with horseradish peroxidase-conjugated secondary antimouse immunoglobulin G for 1 hr, which was diluted at the ratio of 1:1000 after washing with phosphate-buffered saline solution three times.
Statistical Analysis
All numerical data were presented as mean ± standard deviation (SD). Statistical significance was confirmed by one-way analysis of variance (ANOVA), which was followed by a post hoc least significant difference test for multiple comparisons. Scoring experiments were marked blindly. Survival statistics were compared with Kaplan–Meier curves. Student’s t-test and Mann–Whitney U-test were used for parametric and nonparametric analyses to calculate P values. Besides, one-way ANOVA with Bonferroni correction was used relying on the sample distribution and variation in figure legends (SPSS statistics, version 18.0 and GraphPad Prism, version 6.0). P < .05 was considered a significant difference, and such differences were indicated in the figures by asterisks (Figures 2–6).
Results
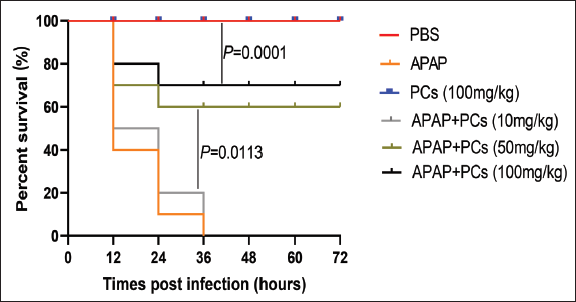
PCs Improved the Survival Rates in Mice
In the in vivo experiment, the survival rate of mice pretreatment with PCs was apparently improved in the control group. The groups did not exhibit any mice died within 72 hr after the administration of PBS, PCs respectively. Mice pretreated with APAP with a dose of 400 mg/kg died within 36 hr; however, those pretreated with PCs of 100 mg/kg had the highest survival rate. Besides, mice pretreated with PCs with a dose of 50 mg/kg and 100 mg/kg exhibited much higher survival rates than those of the APAP model group and the PCs group with a low dose of 10 mg/kg (P =.0113). Furthermore, the survival rates in the medium- and high-dose of the PCs group varied between 60% and 70%, and this indicator tended to be higher during the ending observation (Figure 1).
Protective effect of PCs in the APAP-induced model group. Kunming mice (n = 10) were immunized with PBS, APAP (400 mg/kg), PCs (100 mg/kg), APAP + PCs (10 mg/kg), APAP + PCs (50 mg/kg), and APAP + PCs (100 mg/kg). The survival rate of mice was monitored for three consecutive days in every 12 hr. P values were calculated by the Mantel–Cox log-rank test.
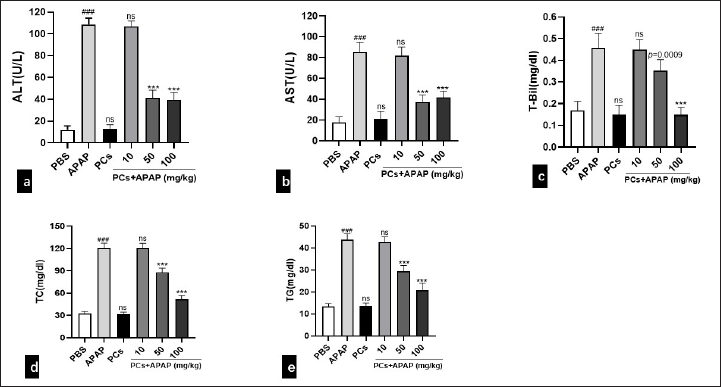
Pretreatment of PCs Improved the Damaged Liver Functions Induced by APAP in Mice
In order to assess the liver functions of mice, we measured serum biochemical levels of ALT, AST, and T-Bil. Serum levels of ALT, AST, T-Bil, TC, and TG increased significantly in the APAP group, suggesting that the model of ALI induced by APAP was successfully established. Compared with the PBS group, the serum levels of above parameters in PCs group had no significant change, indicating PCs themselves did not cause liver injury in mice. Meanwhile, these liver function parameters had no notable difference between the low-dose PCs group and the APAP group, indicating that the low-dose PCs group exerted no significant protective effect on the liver injury in mice. However, serum levels of ALT, AST, T-Bil, TC, and TG in the medium- and high-dose PCs groups were significantly reduced, compared with the APAP group (P < .05). Besides, the reduction of ALT, AST, T-Bil, TC, and TG in serum exhibited dose-dependent induction with various concentrations of PCs pretreatment (Figures 2a–2e).
PCs Reduced Liver Inflammation Induced by APAP
Acute liver damage usually leads to inflammation. There was an obvious increase in the total expression of inflammation cytokines in liver induced by APAP. Serum levels of TNF-α, IL-1β, and IL-6 significantly elevated in the APAP group. All PCs-treated groups exhibited a decrease in TNF-α, IL-1β, and IL-6 compared with the APAP group (Figures 3a–3c).
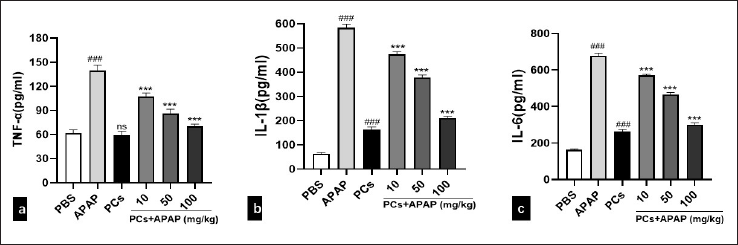
# indicates a significant difference from the PBS group. *indicates a significant difference from the APAP group. (###P < 0.001,***P < 0.001).
PCs Pre-treatment Alleviated Histological Damage of the Liver
Morphological features of hepatic changes caused by APAP toxicity at 8 hours after APAP exposure are exhibited in Figures 4a–4f. Normal hepatic structures were observed in the PBS control group, as normal central venous structure, and hepatic lobular structure (Figure 4a). Severe centrilobular necrosis and hemorrhage were observed in APAP-treated (400 mg/kg) Kunming mice, indicating successful APAP-induced ALI models (Figure 4b). No histological abnormality was observed in the PCs group (100 mg/kg), which exhibits hepatic lobule clear boundaries, clear cell outline, liver nuclei larger, rounded, central cells, and central venous structures normality (Figure 4c). In contrast, hepatic histopathology of APAP-treated mice with PCs (10 mg/kg) showed intense centrilobular necrosis, hemorrhage, and plenty of inflammatory cells (Figure 4d). However, mild cytoplasmic vacuoles of hepatocyte of early hydropic degeneration were observed in APAP-treated mice with PCs (50 mg/kg) (Figure 4e). Surprisingly, complete regeneration of hepatic cells and evidence of mitotic figures were observed in the APAP-treated Kunming mice group (Figure 4f). In particular, the area of necrosis in the group of APAP + PCs (10 mg/kg) was 32.3% ± 5.0%, compared with 17.5% ± 5.0% in the group of APAP + PCs (50 mg/kg), 8.1% ± 5.0% in the group of APAP + PCs (100 mg/kg), which increased in the APAP group with 46.8% ± 5.1% (P =.0002 < .05) (Figure 4g). Severity score of APAP + PCs (100 mg/kg) was significant and different from the group of APAP + PCs (10 mg/kg). Meanwhile, the severity of liver injury in the APAP + PCs (100 mg/kg) group was obviously lower than that in the APAP group (P < .0001) (Figure 4h). Furthermore, these findings revealed that APAP-induced liver injury was significantly ameliorated by PCs pretreatment in a dose-dependent manner.
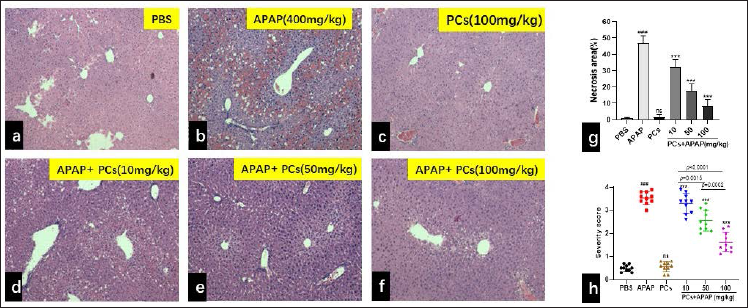
# indicates a significant difference from the PBS group.
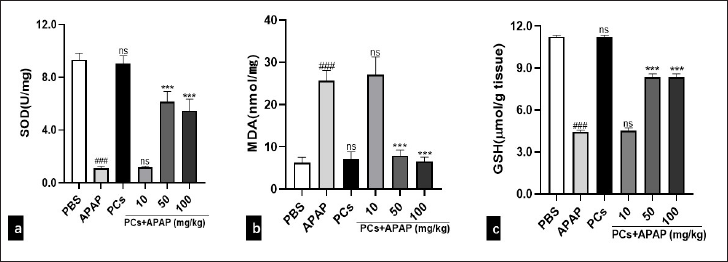
PCs Pre-treatment Mitigated Oxidative Stress
In this study, oxidative stress status, including MDA, SOD, and GSH in the liver tissues, was investigated. An overdose of APAP caused a remarkable decrease of SOD and GSH, and an increase of MDA in the liver. However, in the PCs control group, SOD activity was not significantly different from the PBS group (P =.2284 > .05). Besides, in the APAP plus low-dose PCs group, SOD activity did not change significantly (P =.0578 > .05). However, in the medium- and high-dose PCs group, SOD activity was significantly increased (P < .0001) compared with the APAP group (Figure 5a). What’s more, MDA (P =.4341 > .05) did not change significantly in the APAP + low-dose PCs group compared with the APAP group, whereas it was significantly lower in the APAP group pretreated with the medium- and high-dose PCs (P < .0001) (Figure 5b). However, compared to the APAP group, the levels of GSH (P =.4146 > .05) in the low-dose PCs group did not change significantly. Additionally, GSH was significantly increased (P < .0001) in the high-dose PCs group compared with the APAP group (Figure 5c). These results indicated that medium- and high-dose PCs pretreatment ameliorated oxidative stress.
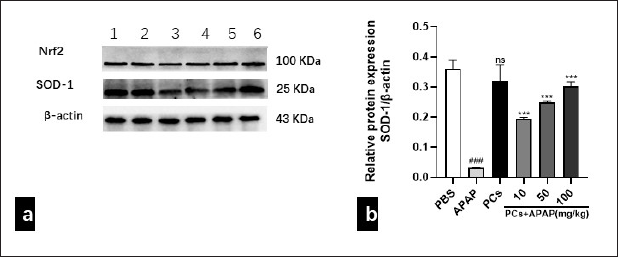
PCs Pretreatment Elevated Protein Expression of Nrf2 and SOD-1
Considering that oxidative stress, inhibiting by PCs, is an essential process in the pathogenesis of APAP overdose, we further investigated whether PCs pretreatment affects the protein expression of Nrf2 and SOD-1. The results indicated that protein expressions of Nrf2 and SOD-1 were inhibited due to APAP exposure. However, they were increased gradually in the PCs pretreatment group, especially at the medium and high doses. Compared to the APAP group, PCs directly elevated the protein expression of Nrf2 and SOD-1 (Figure 6a and 6b).
SOD-1 protein expression of PCs by the Western blot. (a) Western blotting bands and (b) relative protein levels of SOD-1 were indicated in each group. β-actin was used as an internal control. 1: PBS control; 2: PCs (100 mg/kg); 3: APAP model (400 mg/kg); 4: PCs (10 mg/kg) + APAP (400 mg/kg); 5: PCs (50 mg/kg) + APAP (400 mg/kg); 6: PCs (100 mg/kg) + APAP (400 mg/kg). All data are presented as mean ± SD (n = 10/group), Student’s t-test; ns = no difference.
Discussion
APAP is one of the most extensively used analgesic and antipyretic drugs. Its overuse, however, may cause hepatotoxicity and ALF. APAP-induced hepatotoxicity has already become the most dominant reason for ALF.[30] Multi-stages and multi-signal pathways have been proved in APAP-induced hepatotoxicity. For instance, APAP metabolism, endoplasmic reticulum (ER) stress, oxidative stress, autophagy, and microcirculatory dysfunction are involved in liver repair and regeneration.[31] Several plants have been testified for their capacities to prevent and treat ALI.[32,33] Accumulating studies have evidenced that PCs have a potential of antioxidative stress. According to the research, a low dosage of PCs can inhibit liver injury caused by ethanol or carbon tetrachloride and stimulate hepatic cells to proliferate.[34] One of the previous studies indicated that Nrf2 was a vital transcription factor not only for regulating response of intracellular antioxidant but also for maintaining homeostasis of intracellular redox.[35] The protective effects of Nrf2 from oxidative damage was confirmed by Nrf2-null mice that were more vulnerable to acetaminophen-induced ALI.[36] Nrf2 was also the critical regulator for SOD, GSH, and other antioxidants.[37,38] Therefore, activating the Nrf2-mediated antioxidative defense system might be an effective method for protecting against APAP-induced ALI.
In this study, we found that APAP (400 mg/kg) significantly induced the elevation of serum ALT, AST, T-Bil, TC, and TG. Reversely, PCs pretreatment could decrease these parameters, indicating liver functions. Besides, PCs also attenuated the APAP-induced elevation of inflammatory cytokines, such as TNF-α, IL-1β, and IL-6. The survival rates of mice pretreatment with PCs at the medium- and high doses were above 60%–70%, which indicated that PCs could improve survival rates in mice with APAP-induced liver injury. Apparently, APAP induced hepatotoxicity, including the vacuolar degeneration, and the necrosis of liver cells, which was significantly ameliorated by PCs pretreatment in a dose-dependent manner. Additionally, we analyzed the most common parameters (MDA, SOD, and GSH) indicating oxidative stress to explore the mechanism of the protective effects of PCs against ALI induced by APAP. The results showed the decreased hepatic SOD activity and level of GSH, while the increased content of MAD in the APAP group. However, PCs improved these parameters, indicating the inhibition of oxidative stress. Finally, we investigated the protein expression of Nrf2, vital transcriptional factor of antioxidative stress, and its target molecule SOD-1. The data indicated that PCs could increase the expression of Nrf2 and SOD-1, which further indicated that the Nrf2 signaling pathway was involved in the mechanism of antioxidative stress of PCs.
However, there are some limitations in the present study. First, we only carried out the pretreatment experiments. Actually, the posttreatment condition is more consistent with the clinical practice. Moreover, the detail of signaling pathways of Nrf2 needs to be further investigated. Finally, antiinflammation property of PCs in APAP-induced hepatotoxicity is interesting, and it is necessary to explore its mechanism.
Conclusion
Based on this study, the potential protective role of PCs against APAP-induced ALI was investigated. Kunming mice were peritoneally injected with PCs 30 min before the exposure of APAP. Survival rates of mice were scored every 12 hr for three days in succession, serum levels of ALT, AST, T-Bil, TC, TG, TNF-α, IL-1β, and IL-6, and histological analysis, hepatic oxidative stress, and protein expression were successively measured. PCs not only improved survival rates of APAP-induced liver injury in mice but also reduced the levels of ALT and AST in serum, attenuated liver histopathological damage, and activated protein expression of Nrf2 and SOD-1. The results evidenced that PCs ameliorated APAP-induced ALI, and Nrf2 signaling pathway modulating antioxidative stress might be involved in it.
Footnotes
Acknowledgements
The financial assistance comes from Chengdu’s second people’s hospital, and professor Li Liu is gratefully acknowledged. This research was supported by vital research and development technology innovation research and development project of Chengdu Science and Technology Bureau key research (2019YF05-00140SN).
Declaration of Conflicting Interests
We promise that we do not have any commercial interest in connection with the work submitted.
Funding
Chengdu Science and Technology Bureau key research (2019YF05-00140SN).
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
