Abstract
Background
Drug-induced liver injury (DILI) is extremely common in clinical practice and needs to be focused. Anwulignan is a unique lignan monomer of Schisandra Sphenanthera Fructus and is used as the main active ingredient of Wuzhi tablet, which is used for liver protection in clinical practice. In this study, an acute liver injury mouse model induced by acetaminophen and paracetamol (APAP) was used to observe its effects.
Materials and Methods
An acute liver injury mouse model was established by intraperitoneally injecting APAP. Mice were divided into blank control (CON) group, APAP model (MOD) group, Anwulignan CON group, and Anwulignan MOD group. Anwulignan (6 mg/kg) or sodium carboxymethylcellulose (10 mL/kg) was intragastrically given to mice once a day, successively for 14 days. On day 14, 250 mg/kg APAP solution (10 mL/kg) or saline was injected intraperitoneally, and 12 h later, the mice were killed. alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels in the serum of mice were tested. The liver index of mice was calculated, and the pathology of liver injury was evaluated by HE staining in mice. The activities of glutathione peroxidase (GSH-Px) and superoxide dismutase (SOD) as well as the content of malondialdehyde (MDA) in the liver tissue were tested. The levels of kelch-like ECH-associated protein 1 (Keap-1), nuclear factor erythroid-2-related factor 2 (Nrf2), heme oxygenase 1 (HO-1), BCL2 associated X protein (Bax), B-cell lymphoma-2 (Bcl-2), caspase-3 and tumor suppressor protein (P53) mRNAs in the liver tissues of mice were detected by reverse transcription-PCR (RT-PCR), and the expression levels of Keap-1, Nrf2, HO-1, Bax, Bcl-2, caspase-3 and P53 proteins in the liver tissue of mice were detected by Western blot.
Results
Anwulignan can alleviate the acute liver injury of mice. The down-regulation of the cyclophosphamide (CYP2E1) expression in the liver tissue of mice to reduce the hepatotoxicity of APAP, and the regulation of Nrf2-ARE signaling pathway-related gene expressions to play an antioxidant role and apoptosis-related gene expressions to inhibit the liver cell apoptosis may be involved in the mechanism through which Anwulignan can exert its effect.
Conclusion
Anwulignan can alleviate the liver injury induced by APAP in mice, and this study may provide evidence for researching and developing some drugs and nutraceuticals used for preventing and treating liver injury.
Introduction
Liver injury is one of the major issues worldwide. The main types of liver injury include chemical, drug-induced, immune-mediated, and alcoholic liver injuries, and non-alcoholic fatty liver (Bajaj, 2019; Chen et al., 2020). Drug-induced liver injury (DILI) is caused by an abnormal accumulation of drugs or their metabolites in the liver, leading to liver failure or even death. In recent years, the incidence of DILI has been rising worldwide, and DILI has become a challenging problem for doctors to use drugs (Larson et al., 2005). Therefore, the research and development of effective and safe drugs or heath foods have become the focus currently.
Wuzhi Tablet, herbal medicine of Schisandra sphenanthera (Schisandrae Sphenantherae Fructus, Schisandra) ethanol extract can improve liver damage and metabolism, and reduce the activity of transaminase to protect against liver injury (Chen et al., 2020), and Anwulignan, a unique lignan monomer of Schisandra, is used as one of the main components in Wuzhi tablet (Wei et al., 2010). It was indicated in our previous studies that Anwulignan could improve liver injuries induced by D-galactose and carbon tetrachloride significantly through antioxidant effect (Gao et al., 2018). However, its effect on DILI has not been reported. It is well known that the long-term or excessive use of acetaminophen and paracetamol (APAP) can induce liver injury (Bunchorntavakul & Reddy, 2018). Acute liver injury mouse model induced by APAP is often used as DILI in research (Jiang et al., 2019; Kolaric et al., 2021). Therefore, the protective effect of Anwulignan against liver injury was observed using this model, and it was hypothesized that its hepatoprotective mechanism would be based on its antioxidation and anti-apoptosis in this study.
Materials and Methods
Animal Feeding
Male ICR mice, weighing 20 ± 2 g, were purchased from Changchun Yisi Experimental Animal Research Center (Changchun, China) and the license No was SCXK (Ji)-2020-0002)). The mice were raised in separate cages at 25 ± 1℃C, in 55% ± 5% humidity and 12 h light/dark cycle, and with free access to water and food. The mice were acclimatized to the laboratory environment for 3 days before experiments.
Agnets and Animals
Anwulignan (purity >99%) was provided by Chengdu Pufei De Biotech Co., Ltd. (Chengdu, China). APAP (purity >99%) was purchased from Shanghai Aladdin Reagent Co., Ltd. (Shanghai, China).
Forty ICR mice (male) were divided into 4 groups randomly (n = 10), Control (CON) group (carboxymethylcellulose sodium by gavage + saline in intraperitoneal injection), model (MOD) group (carboxymethylcellulose sodium by gavage + 250 mg·kg–1 APAP in intraperitoneal injection), Anwulignan (CON) group (6 mg·kg–1 Anwulignan by gavage + saline in intraperitoneal injection), and Anwulignan (MOD) group (6 mg·kg–1 Anwulignan by gavage + 250 mg·kg–1 APAP in intraperitoneal injection). Anwulignan or carboxymethylcellulose sodium was given once a day for 14 days, and APAP or saline was given intraperitoneally only once one hour after the last administration of the above agents on the fourteenth day.
Preparation of Samples
The mice were weighed at the 12th hour after they were administered with APAP, and then they were anesthetized with pentobarbital sodium to collect their blood samples by removing their eyeballs. The serum was obtained by centrifuging the blood at 3500 rpm for 10 min and kept at –80℃. The liver was removed, weighed, and observed with the naked eye. The liver tissue for the histopathological examination was fixed in formaldehyde, and the rest of the liver tissue was kept for detecting the activities of superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px), and the contents of GSH and malondialdehyde (MDA). Relative RNAs and proteins were analyzed by reverse transcription-PCR (RT-PCR) and Western blot, respectively.
Determination of Liver Indexes
Liver indexes of the mice (%) were determined according to the formula: liver weight/body weight × 100%.
Measurement of ALT and AST Levels in Serum
Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels in the serum of mice were tested according to the instructions of the kits (Nanjing Jiancheng Biotechnology Research Institute, Nanjing, China).
H&E Staining of Liver Tissue
The fixed liver tissue with formaldehyde was dehydrated with alcohol, embedded in paraffin, cut into 4 µm thick sections, dewaxed with xylene, stained with H&E (Hematoxylin & Eosin), washed with gradient ethanol, and mounted with neutral gel. The sections were examined under an optical microscope.
Detection of SOD, GSH-Px, GSH, and MDA in the Liver Tissue
The pieced liver tissue in saline was homogenated, and the homogenate was centrifuged at 4℃ and 3500 revolve per minute for 10 min for collecting the supernatant. The supernatant protein concentration was determined by the BCA method (Beyotime Biotechnology Research Institute, Shanghai, China). SOD and GSH-Px activities as well as MDA and GSH levels in the liver tissue of mice were measured according to the instructions of the kits (Nanjing Jianjian Institute of Biotechnology, Nanjing, China).
RT-PCR Analysis
An RNA extraction kit was used to extract the total RNA in the liver tissue (Vazyme Company, China). The instructions of the kits were followed to synthesize the cDNA and carry out the RT-PCR amplification. The gene sequence number was found in Genebank, and the primers were designed by Beijing Dingguo Changsheng Biotechnology Co., Ltd., Beijing, China with Primer Premier 6. β-Actin was used as the reference gene, and sequences of all primers are listed in Table 1. PCR amplification conditions included 94℃ and 3 min for pre-denaturation, 94℃ and 30 sec for denaturation, 30 sec for annealing (The annealing temperatures were 59℃ for tumor suppressor protein (P53), nuclear factor erythroid-2-related factor 2 (Nrf2) and heme oxygenase 1 (HO-1), 55℃ for β-actin, Keap1, BCL2 associated X protein (Bax), B-cell lymphoma-2 (Bcl-2), and caspase-3), and 72℃ for 30 sec for an extension. The amplification was repeated 30 times, and the samples were kept at 72℃ for 5 min and stored at 4℃. The PCR products (10 µL) were separated by gel electrophoresis, and the electrophoresis images were photographed and analyzed by a gel imaging system (Tanon 1600 gel Image system; Shanghai Tianneng Technology Co., Ltd., Shanghai, China).
Primer Sequences.

Western Blot Analysis
Protein expressions in the liver tissue of mice were measured by Western blot. The liver tissue was cut into pieces and the pieced tissue was placed in lysis buffer, and the tissue-lysis buffer solution was incubated on ice for 1 h to prepare the liver tissue homogenate. The supernatant was obtained by centrifuging the homogenate at 12,000 rpm and at 4℃ for 10 min. The supernatant protein concentration was determined by the BCA method. The 10% SDS-polyacrylamide gel electrophoresis on the target proteins in samples was performed. The proteins were transferred onto PVDF membranes for 2 h, and then TBST buffer containing 5% skim milk powder was used to block the non-specific sites of proteins on the membranes for 1 h. The proteins were incubated with the primary antibodies of cyclophosphamide (CYP2E1), Nrf2, Keap1, HO-1, P53, Bax, Bcl-2, and caspase-3 (1:1000, ABclone Company, USA) at room temperature, and incubated overnight. Then, the membranes were washed three times with TBST buffer, 10 min each time, then incubated with the second antibody HRP Goat anti-rabbit IgG (H+L) (1:5000) at room temperature for 1 h (ABclone Company, USA) and washed in the same way, and ECL developer (Vazyme Company, China) was used for the development of color. The chemiluminescence method was applied to analyze the protein bands.
Statistical Methods
The data were expressed as mean ± SD and analyzed with SPSS 20.0 software. The data among multiple groups were compared by one-way analysis of variance, and those between groups were compared by Tukey test. p < 0.05 indicates a significant difference.
Results
Body Weight, Liver Weight, Liver Index, and Serum ALT and AST Levels
The body weights of the mice were not significantly different among all groups (Figure 1A). The liver weight (Figure 1B), liver index (Figure 1C), and serum ALT (Figure 1D) and AST (Figure 1E) levels in MOD group were significantly higher than those in CON group (p < 0.01), while the liver weight, liver index, and serum ALT and AST levels were not significantly different between Anwulignan (CON) group and CON group, and all these indicators in Anwulignan (MOD) group decreased significantly (p < 0.01) compared with those in MOD group, indicating that Anwulignan could improve the APAP-induced acute liver injury.
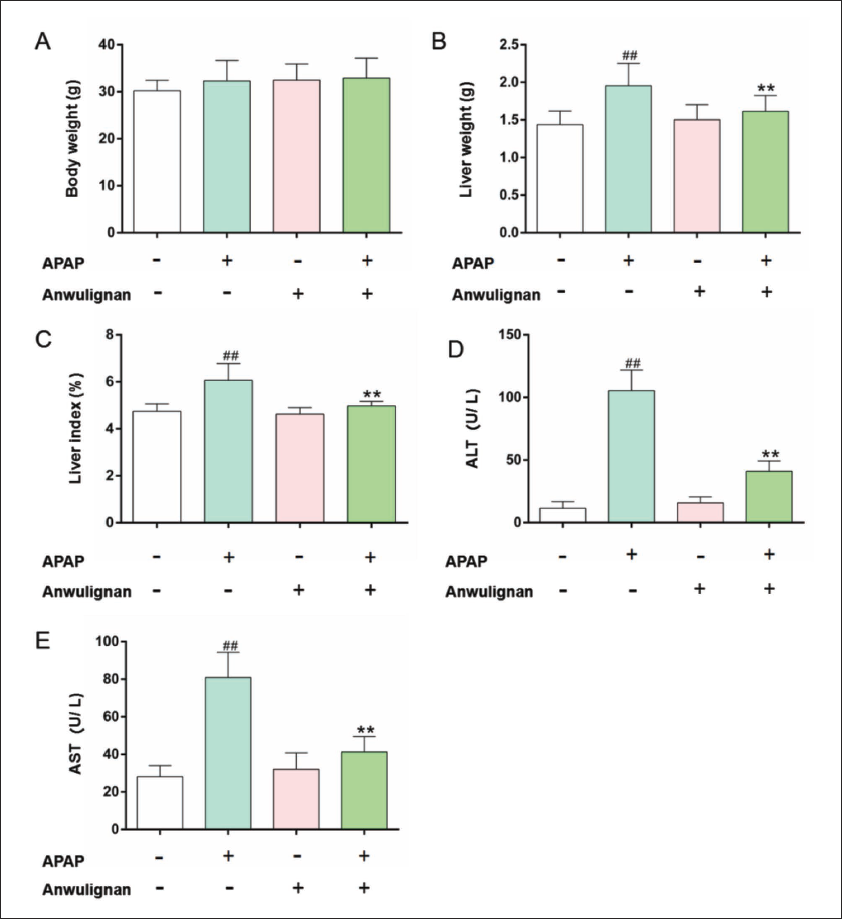
The H&E staining results showed that hepatocytes were normal in morphology, lobular structures were complete, and hepatic cords were arranged in order in the CON group; In the MOD group, around the central vein, there were cytoplasmic loosening, karyopyknosis, and light-stained chromatin; The morphology of hepatocytes was almost similar to that in CON group, but was improved in Anwulignan (MOD) group; In contrast to MOD group, hepatic cords were more regularly arranged, hepatic lobules near the central vein were less light-stained, and there were fewer nuclear divisions in Anwulignan (MOD) group (Figure. 2). These results indicate that Anwulignan could morphologically ameliorate the APAP-induced acute liver injury.
HE Staining of Liver Tissue. The Arrow Points are the Central Veins (×100 and ×200, n = 3).
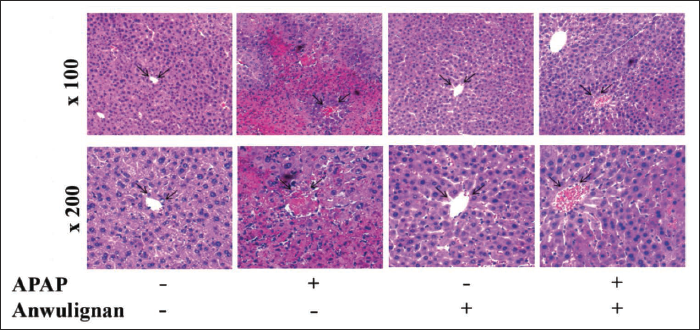
GSH-Px and SOD Activities, and GSH and MDA Levels
The results (Figure 3) showed that compared with that in CON group, the activity of GSH-Px (Figure 3C) and SOD (Figure 3D), as well as the content of GSH (Figure 3A) was significantly lower than those in MOD group (p < 0.01), but the content of MDA in MOD group (Figure 3B) was significantly higher than that in CON group (p < 0.01); however, compared with those in MOD group, all these parameters were reversed in Anwulignan (MOD) group; All these parameters were not significantly different between Anwulignan (CON) and CON groups (p > 0.05), suggesting that Anwulignan can exert its antioxidant effect to alleviate the APAP-induced acute liver injury.
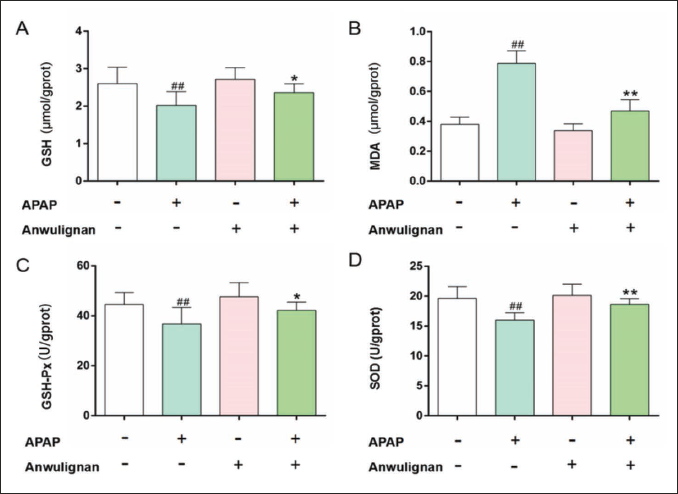
Expression of CYP2E1 in Liver Tissue
As shown in Figure 4, compared with that in the CON group, the CYP2E1 protein expression level decreased significantly in the MOD group (p < 0.01), but compared with that in the MOD group, the CYP2E1 protein expression level decreased significantly in Anwulignan (MOD) group (p < 0.01), and the CYP2E1 protein expression level was not significantly different between Anwulignan (CON) group and CON group (p > 0.05), demonstrating that Anwulignan could reduce the hepatotoxicity of APAP by lowering CYP2E1 level in the liver tissue.
Nrf2-ARE Signaling Pathway-related Gene Expressions in Liver Tissue
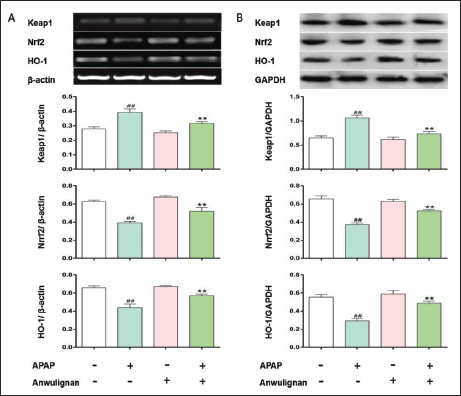
The results (Figure 5) showed that Keap1 mRNA and protein expression levels in the liver tissue of mice increased significantly compared with those in the CON group (p < 0.01), and Nrf2 and HO-1 mRNA and protein expression levels decreased significantly (p < 0.01) in MOD group; Expression levels of all these parameters were significantly reversed in Anwulignan (MOD) group compared with those in MOD group; Expressions of all these mRNA and proteins were not significantly different between Anwulignan (CON) and CON groups (p > 0.05), confirming that Anwulignan may alleviate APAP-induced oxidative stress via its regulating Nrf2-ARE signaling pathway-related gene expressions.
Expressions of Apoptosis-related Factors in Liver Tissue
Expression levels of P53 and Bax mRNA and proteins as well as caspase-3 mRNA and cleaved caspase3 proteins in the liver tissue of mice increased significantly (p < 0.01), and those of Bcl-2 mRNA and protein as well as the ratio of Bcl-2/Bax expressions in MOD group decreased significantly (p < 0.01) compared with those in CON group. Expression levels of all of above parameters increased significantly in Anwulignan (MOD) group (p < 0.01). Expression levels of these mRNA and proteins were not significantly different between Anwulignan (CON) and CON groups (p > 0.05) compared with those in MOD group (Figure 6), demonstrating that Anwulignan may meliorate APAP-induced acute liver injury by regulating the expression of P53, Bax, Bcl-2 and caspase-3/cleaved caspase3, and then reducing the apoptosis of hepatocytes in APAP-induced acute liver injury mice.
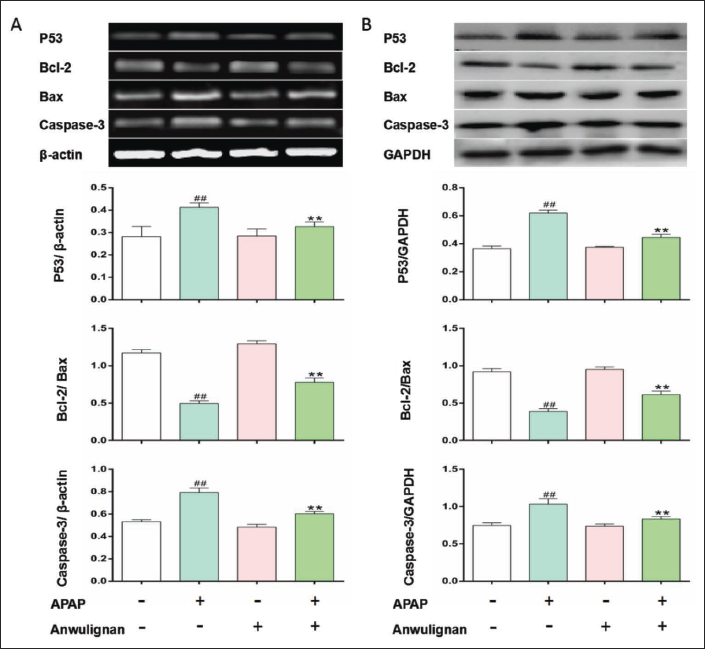
RT-PCR (A) and Western Blot (B) were used to Detect the Expressions of P53, Bax, Bcl-2, and Caspase-3 mRNA and Proteins.
Discussion
The liver is a vital organ for metabolism and detoxification in the body. Once the liver is damaged, various liver diseases even cirrhosis and liver cancer will occur (Go & Jones, 2017; Zhao et al., 2018). APAP is often used as a tool drug for inducing acute liver injury in research. Excessive APAP is metabolized to NAPQI in the CYP system, which reduces the antioxidant capacity of the liver, inducing oxidative stress, and eventually the injury of liver cells (Jiang et al., 2019; Ni et al., 2016). Liver index can be used to evaluate liver injuries. The size of the whole liver increases, and the liver index increases. ALT and AST levels are sensitive indicators to judge liver cell injury. When ALT and AST are elevated, liver cells are damaged (Kelava et al., 2010; Hyeon et al., 2004). The results showed that the liver weight and liver index were significantly increased, ALT and AST levels in serum also increased significantly, and the liver tissue was significantly damaged in mice treated with APAP. The pretreatment with Anwulignan significantly improved the above indicators and tissue damage, indicating that Anwulignan may alleviate the APAP-induced liver injury.
Toxicity of APAP arises from the formation of cytochrome P450 enzyme, especially reactive metabolite NAPQI formed by CYP2E1, which accumulates in the liver and binds with cysteine residues of cellular proteins, causing cell damage in liver tissues. Many studies have shown that the expression of CYP2E1 in the liver increases after APAP treatment (Cho et al., 2023; Masubuchi & Mikami, 2023). Whereas the inhibition of CYP2E1 expression can reduce the formation of NAPQI and thus alleviate liver injury, which is consistent with those reported above, CYP2E1 expression increased significantly, while decreased significantly in the liver tissue of mice treated with APAP after the administration of Anwulignan, so we hypothesized that Anwulignan might reduce liver tissue damage by inhibiting CYP2E1 expression in the liver of APAP-treated mice, thereby reducing the production of toxic substances such as NAPQI. Currently, targeting CYP2E1 has become one of the focuses for developing protective drugs against APAP-induced liver injury.
When a large amount of NAPQI is produced, excessive ROS is also produced in the liver, which then results in oxidation. Oxidative stress injury can be caused by the hepatotoxicity induced by high-dose APAP (Adamson & Harman, 1993). The Nrf2-ARE pathway is an endogenous anti-oxidative stress pathway in cells. Nrf2 is involved in ameliorating and alleviating acute liver injury as a key coordinator of oxidative stress (Nguyen et al., 2009; Reziwan et al., 2019). Keap1, a member of the Kelch family, is a negative regulatory protein of Nrf2, and Nrf2 can also induce antioxidant gene expressions, for example, HO-1, to reduce oxidative stress injury (Cheng et al., 2020; Kaspar et al., 2009). Studies have shown that Keap1 expression levels increase, and Nrf2 and HO-1 expression levels decrease after APAP treatment (Güvenç et al., 2020; Wang et al., 2018). It was found in this study that APAP could inhibit the accumulation of Nrf2 and HO-1 by increasing Keap1 expression. In contrast, Anwulignan treatment reduced Keap1 expression level and increased Nrf2 accumulation as well as expression content of HO-1, mitigating this inhibitory effect. Under the stimulation of oxidative stress, Nrf2 can also bind to ARE to activate SOD (Dong et al., 2017). SOD and GSH-Px are the key antioxidant enzymes in the main defense system against ROS when oxidative stress occurs. The increase of ROS can lead to cell membrane lipid peroxidation, inducing an excessive MDA production. The results showed that APAP increased MDA contents, but decreased GSH-Px and SOD activities, and Anwulignan significantly up-regulated GSH-Px and SOD activities, and reduced MDA contents, suggesting that Anwulignan might protect against the liver injury induced by APAP by inhibiting the activation of the oxidative stress response pathway Nrf2-ARE signaling pathway in mice. However, the precise mechanism related to the Nrf2-ARE signaling pathway needs to be further elucidated.
Oxidative stress injury can also induce cell apoptosis. Studies have shown that hepatocyte apoptosis is involved in APAP-induced acute liver injury (Atteya et al., 2019). APAP can induce the expression of Bax, a key pro-apoptotic factor, and inhibit the expression of Bcl-2, an important anti-apoptotic factor (Oz & Chen, 2008). P53, a tumor-suppressive gene, can induce the release of cytochrome C and promote caspase-3 activation, thereby leading to apoptosis (Amaral et al., 2010; Li et al., 2019). It was found in this study that Bax, caspase-3, and P53 expressions were significantly increased, while the level of Bcl-2 as well as Bcl-2/Bax ratio was significantly decreased in APAP-induce model mice, which is consistent with the findings reported in other studies in both mouse and rat models, APAP promoted apoptosis (Senocak et al., 2023; Che et al., 2019; Su et al., 2019). In contrast, Anwulignan significantly inhibited the up-regulation of Bax, caspase-3, and P53, and increased the expression of Bcl-2 as well as the ratio of Bcl-2/Bax, thus inhibiting apoptosis. It is suggested that Anwulignan alleviates APAP-induced liver injury in mice by regulating apoptosis-related factors. It has been found that after Nrf2 is activated, the expression of Bcl-2 in cells increases (Niture & Jaiswal, 2012). In our previous work, we also found that Anwulignan could simultaneously up-regulate Nrf2 and Bcl-2 expression levels in muscle tissues of chronic fatigue mice (Zhang et al., 2019). Therefore, it is suggested that Anwulignan may play an antiapoptotic role by regulating the Nrf2 pathway in APAP-induced liver injury.
The activation of oxidative stress and increased levels of apoptosis-related factors exacerbate the extent of DILI (Ezhilarasan, 2023; Pomacu et al., 2023). The results of our previous studies indicated that Anwulignan had relatively good anti-oxidative stress and anti-apoptotic effect in all other tissue injuries (C. Liu et al., 2021; J. Liu et al., 2022; Yuchong et al., 2020), suggesting that Anwulignan can play a role in clinical diseases. Furthermore, it is currently unknown whether the prophylactic effect of Anwulignan is dose-related and whether there exist additional protective mechanisms against acute liver injury, further studies are required to fully understand these aspects.
Conclusion
In conclusion, Anwulignan has a protective effect on APAP-induced liver injury in mice. Its underlying mechanisms are related to the regulation of the Nrf2 pathway, the inhibition of oxidative stress response, and the regulation of apoptosis-related factors.
Abbreviations
APAP: Acetaminophen; ALT: Alanine aminotransferase; AST: Aspartate aminotransferase; CYP: Cyclophosphamide; MDA: Malondialdehyde; RT-PCR: Reverse transcription-PCR; SOD: Superoxide dismutase; DILI: Drug-induced liver injury; GSH-Px: glutathione peroxidase; Keap-1: Kelch-like ECH-associated protein 1; Nrf2: Nuclear factor erythroid-2-related factor 2; HO-1: heme oxygenase 1; Bax: BCL2 associated X protein; Bcl-2: B-cell lymphoma-2; P53: Tumor suppressor protein.
Summary
Anwulignan can protect APAP-induced acute liver injury in mice by lowering serum ALT and AST levels, increasing GSH-Px and SOD activity in liver tissues, lowering liver weight and liver index and MDA content, regulating the antioxidant effect mediated by the Nrf2-ARE pathway, and regulating apoptosis-related genes.
Footnotes
Acknowledgments
We thank Professor Wei Xia from the College of Medical Technology and Professor Wenyue Zhuang from the College of Medical Technology for their critical comments on the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project was supported by the Department of Science and Technology of Jilin Province (YDZJ202301ZYTS152, YDZJ202301ZYTS168, 20200404053YY), Jilin Innovation and Entrepreneurship Talent Funding Programme (2023RY12), “Mindray” research and innovation projects of College of Medical Technology, Beihua University(2022), Jilin Provincial Development and Reform Commission (2020C033-2), National Natural Science Foundation of China (82274081).
Statement of Ethical Approval and Informed Consent
All experiments were carried out by the Guidelines for the Care and Use of Laboratory Animals in China, and all procedures were approved by the Animal Ethics Committee of Beihua University (No. 2023092101).
