Abstract
Acetaminophen (APAP) overdose causes acute liver injury (ALI). Nicotinamide adenine dinucleotide (NAD) is an essential coenzyme, and NAD+ is oxidized type which synthesized from nicotinamide (NAM). The present study aimed to investigate the role of NAD+ in ALI and protective property of NAM. The mice were subjected to different doses APAP. After 8 hours, the serum activities of alaninetransaminase (ALT) and aspartate aminotransferase (AST), the hepatic NAD+ level and nicotinamide phosphoribosyltransferase (NAMPT) expression were determined. Then, the mice were pretreated with NAM (800 mg/kg), the hepatoprotective effects and the key antioxidative molecules were evaluated. Our findings indicated that APAP resulted in remarkable NAD+ depletion in a dose-dependent manner accompanied by NAMPT downregulation, and NAM pretreatment significantly elevated the NAD+ decline due to upregulation of NAMPT. Moreover, the downregulated Kelch-like ECH-associated protein-1 (Keap1), upregulated nuclear factor erythroid 2-related factor 2 (Nrf2) and its translocation activation after NAM administration were confirmed, which were in accordance with improved superoxide dismutase (SOD) and glutathione (GSH) levels. Finally, NAM dramatically exhibited hepatoprotective effects by reducing the liver index and necrotic area. This study has suggested that APAP impairs liver NAD+ level and NAM is able to improve hepatic NAD+ to activate antioxidant pathway against APAP-induced ALI.
Introduction
Acetaminophen (APAP) is one of the most commonly used analgesic and antipyretic drugs worldwide, which shows acceptable safety at therapeutic dose. However, an overdose due to suicide attempt or unintentional intake can induce acute liver injury, acute liver failure or even death. Thus, ALI induced by APAP is an obvious public health issue.
The pathogenesis of APAP-induced hepatotoxicity has been studied for decades. It is widely known that APAP overdose leads to excess production of reactive metabolite N-acetyl-p-benzoquinoneimine (NAPQI), which causes exhaustion of glutathione (GSH) and formation of protein adducts resulting in mitochondrial dysfunction, energy metabolism disturbance, oxidative stress, hepatocyte necrosis ultimately. Hence, developing novel therapeutic target that can mitigate APAP-induced ALI is urgent. 1
Nicotinamide adenine dinucleotide (NAD), including oxidized (NAD+) and reduced (NADH) forms, is a critical coenzyme involved in various vital biological processes, such as glycolysis, tricarboxylic acid cycle, oxidative phosphorylation, and fatty acid oxidation. Due to the central role, the deficiency of NAD+ has been found in a broad array of pathological situations, including heart dysfunctions,2,3 muscle dystrophy, 4 acute kidney injury, 5 neurodegenerative disorders 6 and metabolic diseases. 7 Moreover, declined NAD+ has been confirmed in senescent rodents8,9 and humans. 10 Meanwhile, these reports have also demonstrated that supplementation of NAD+ precursor is beneficial to diseases. Thus, NAD+ may be an attractive target to combat APAP-induced ALI, and replenishment with NAD+ precursors is suggested as a promising strategy.
NAD+ is yielded from two pathways, de novo and salvage, and the latter is the most dominant in mammals. 11 For salvage pathway, nicotinamide phosphoribosyltransferase (NAMPT) is the rate-limiting enzyme that catalyzes nicotinamide (NAM) to synthesize NAD+. As an important precursor of NAD+, NAM has been used to prevent and treat the disease pellagra in humans for many years. In addition, more extensive therapeutic effects have been confirmed in a diversity of diseases, including diabetes, 12 cancers,1,13 cerebral ischemia, 14 and Alzheimer disease. 15 Importantly, NAM has low toxicity, and the LD50 in mice is estimated about 2.5 g/kg when given intravenously. 16 Therefore, NAM may be an eligible precursor of NAD+ to protect against APAP-induced hepatotoxicity.
It has been widely acknowledged that oxidative stress plays a pivotal role in the pathogenesis of APAP overdose. Interestingly, NAM possesses the ability to reduce oxidative stress. 17 Hence, we postulate that NAM improves the NAD+ level, and then activates the antioxidative pathway to combat hepatotoxicity induced by APAP. In the present investigation, the influence and mechanism of different doses APAP on NAD+ levels were explored firstly. Subsequently, the alterations of NAD+ levels and protein expression of nuclear factor erythroid 2-related factor 2 (Nrf2) and its suppressor Kelch-like ECH-associated protein-1 (Keap1) were determined after NAM pretreatment. Finally, antioxidant markers and protective effects of NAM were investigated.
Materials and method
Materials
APAP and NAM were purchased from Sigma-Aldrich (St. Louis, MO, USA) and Beyotime Biotechnology (Shanghai, China), respectively. The commercial assay kits for measuring serum alaninetransaminase (ALT), aspartate aminotransferase (AST), superoxide dismutase (SOD) and glutathione (GSH) were obtained from Nanjing Jiancheng Bioengineering Research Institute (Nanjing, China). The total protein extraction kit, nuclear and cytoplasmic protein extraction kit were purchased from Beyotime Biotechnology (Shanghai, China). BCA Protein Assay Kit and enhanced chemiluminescence (ECL) reagent were from Pierce Biotechnology (Rockford, IL, USA). Antibodies used for immunoblotting including NAMPT, Nrf2, Keap1, β-actin, Lamin B, Peroxidase-conjugated goat anti-rabbit immunoglobulin IgG and anti-mouse IgG were obtained from Proteintech Group (Wuhan, China).
Animals and experimental settings
Male Balb/c mice, average body weight 18–20 g (aged 6–8 weeks), were purchased from the Experimental Animal Center of Chongqing Medical University. The mice were fed with a standard laboratory chow and water ad libitum. They were housed in specific pathogen-free conditions at a temperature of 20–25°C, 50 ± 5% relative humidity under a 12 h dark/light cycle. All animal experiments were approved by the Animal Care and Use Committee of Chongqing Medical University.
The experiments consisted of two scenarios (Figure 1). Firstly, to investigate whether APAP exposure affects hepatic NAD+ level, the mice were randomly allotted to four groups (n = 4/group): (1) control, (2) APAP 100 mg/kg, (3) APAP 300 mg/kg, (4) APAP 500 mg/kg. After overnight fasting (16 hours), the mice were subjected to different doses APAP by intraperitoneal injection, and the animals in the control group were given an equal volume of sterile phosphate-buffered saline (PBS). In the second scenario, the potential effects of NAM on APAP-induced acute liver injury and the underlying mechanisms were studied. Following the random principle, the mice were divided into four groups (n = 4/group): (1) control, (2) NAM, (3) APAP, (4) NAM+APAP. The mice in the NAM+APAP group were intraperitoneally injected with NAM (800 mg/kg) for three times at 24, 12, and 1 h before APAP (500 mg/kg) administration. Meanwhile, in APAP group, the mice were administered with normal saline (NS) three times prior to APAP exposure. The animals in the control group were given an equal volume of vehicle, while the mice in the NAM group were given an equal dose of NAM. All the mice were sacrificed at 8 h after APAP exposure, and blood samples and liver tissues were harvested. A small portion of the liver tissues was fixed in 4% paraformaldehyde solution, while the remaining tissues were stored at −80°C for further analysis.

Diagram of experimental settings.
Measurement of serum ALT and AST activities
The serum ALT and AST levels were assessed following the manufacturer’s instructions. In brief, the serum samples were diluted 10 times with pure water, and then 10 µl solution was added into the 96-well plate, and incubated with 50 µl ALT or AST matrix fluid at 37°C for 30 min. Secondly, 50 µl 2-4-dinitrophenylhydrazine was added into each well, and continuously incubated at 37°C for 20 min. Finally, 500 µl NaOH solution was added to each well at room temperature for 20 min, and then detected absorbance at 490 nm.
Determination of liver NAD+ levels
The levels of liver NAD+ were determined using commercial assay kits in accordance with the manufacturer’s protocols. Protein concentration was determined by a BCA protein assay kit.
Western blot analysis
The frozen livers were homogenized and centrifuged, and the supernatant was collected to extract total protein. Additionally, cytosolic and nuclear proteins were isolated according to the manufacturer’s instructions of nuclear and cytosolic extraction kit. Protein concentration was measured by a BCA protein assay kit. The protein samples were loaded into 10% SDS-PAGE polyacrylamide gels, and then electrophoretically transferred to nitrocellulose membrane. Blotted membranes were blocked with the buffer containing 5% (w/v) nonfat milk, and then incubated with the primary antibody overnight at 4°C. Next, the membrane was washed with TBST three times before incubation with the secondary antibody at room temperature for 2 h. After washing, proteins in the membranes were visualized by enhanced chemiluminescence kits.
Measurement of oxidative stress parameters
Mice liver tissues SOD and GSH levels were determined by the commercial kits according to the manufacturer’s instructions. All results were normalized by the total protein concentration in each sample.
Determination of liver index
The body mass of each mouse was weighed before being sacrificed. Furthermore, the wet mass of mouse liver was determined after sacrificed. The liver index is calculated by the formula, liver index = (liver mass)/(mouse body mass) × 100%.
Necrotic area analysis
The liver tissues were fixed with 10% neutral buffered formalin, embedded in paraffin, and cut into 5-mm thick sections. Then, the sections were stained with hematoxylin and eosin (H&E) according to standard protocols and subsequently analyzed with Image J. Necrotic area is the ratio of necrotic area to total area. 18
Statistical analysis
The experimental results were expressed as means ± standard deviation (SD), and multiple comparisons among different groups were performed by one-way analysis of variance with post hoc Dunnett’s C test. A value of p < 0.05 was known as statistically significant difference.
Results
Decreased hepatic NAD+ levels due to APAP exposure in a dose-dependent manner
To investigate the effects of APAP on liver NAD+ levels, we first established a dose-dependent ALI model in mice and detected the alterations of hepatic NAD+ contents. Overnight fasted mice were intraperitoneally injected with 100, 300, 500 mg/kg APAP for 8 hours. Then, serum ALT, AST and hepatic levels were determined. As shown in Figure 2, 100 mg/kg APAP didn’t affect the serum ALT and AST levels, but 300 and 500 mg/kg APAP significantly increased the values of the two enzymes in serum. Consistently, all the three doses impaired hepatic NAD+ contents in a dose-dependent manner, and the higher dose APAP caused lower NAD+ levels. These data shows that APAP induces NAD+ deficiency in a dose-dependent manner in mice.
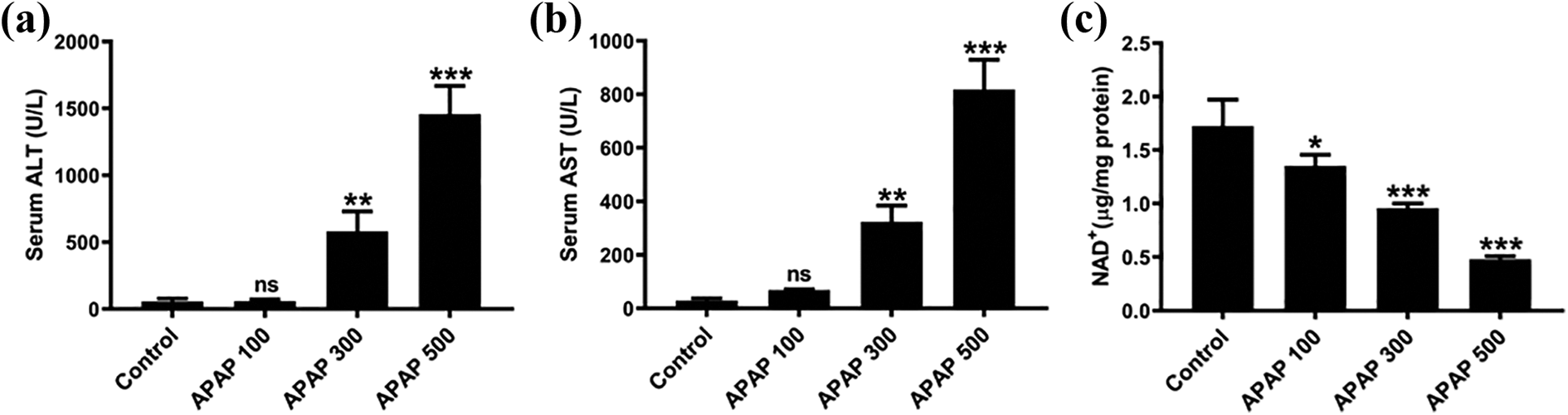
Effects of APAP on hepatic NAD+ levels (n = 4). Blood and liver tissues were collected after different doses APAP (100, 300, 500 mg/kg) for 8 hours. (a) Serum ALT level; (b) serum AST level; (c) hepatic NAD+ contents. All values are expressed as mean ± SD. *p < 0.05; **p < 0.01; ***p < 0.005: compared with control group.
Downregulation of NAMPT after APAP challenge
To probe the mechanism of NAD+ decrease, we tested protein expression of NAMPT the rate-limiting enzyme of NAD+ salvage biosynthesis pathway. The Western blot results showed NAMPT was downregulated after APAP administration (Figure 3). These results indicate that APAP hinders salvage synthesis being responsible for NAD+ deficiency. According to these results, we chose the dose 500 mg/kg to conduct subsequential experiments.

Mechanism of APAP on hepatic NAD+ levels (n = 4). (a) Protein expression of NAMPT was evaluated by Western blot; (b) semiquantitative analyses of NAMPT protein levels were conducted, and β-actin was deemed as the loading control. All values are expressed as mean ± SD, ***p < 0.005: compared with control group.
NAM improves hepatic NAD+ deletion induced by APAP
To discern whether NAM has the potency to reverse the deletion of NAD+ induced by APAP, mice were treated with NAM (800 mg/kg, i.p.) at 24, 12 and 1 h before APAP (500 mg/kg, i.p.) administration. As presented in Figure 4(a), APAP overdose resulted in a notable decrease of hepatic NAD+ level, which was in consistent with above data presented in Figure 2(c). After NAM pretreatment, the decreased hepatic NAD+ level was remarkably reversed. To investigate the underlying mechanism, we assessed the protein levels of NAMPT. Compared with APAP group, the NAMPT expressions were upregulated due to NAM pretreatment (Figure 4(b) and (c)). Taken together, NAM activates the NAD+ synthesis via upregulating its rate-limiting enzyme NAMPT, and then increases hepatic NAD+ levels.

Effects of NAM on hepatic NAD+ levels (n = 4). (a) Hepatic NAD+ levels were evaluated after administration with NAM (800 mg/kg) three times at 24 h, 12 h, and 1 h preceding APAP (500 mg/kg) exposure for 8 hours; (b) protein expression of NAMPT was analyzed by Western blot; (c) semiquantitative analyses of NAMPT protein levels were conducted, and β-actin was deemed as the loading control. All values are expressed as mean ± SD. *p < 0.05: compared with control group; ###p < 0.005: compared with APAP group.
NAM activates Nrf2 pathway
Given that NAD+ has a key role in redox reactions, we characterized the protein expression of Nrf2 and its repressor Keap1. As shown in Figure 5(a) to (c), the results displayed that APAP hindered Nrf2 protein levels, and its inhibitor Keap1 displayed increased expression conversely. However, NAM treatment improved Nrf2 downregulation and repressed Keap1 upregulation. We further investigated the translocation activation of Nrf2. As shown in Figure 5(d) to (f), NAM triggered Nrf2 translocation from cytoplasm to nucleus which evidenced Nrf2 activation. The data suggests that NAM promotes Nrf2 nuclear translocation and activation.
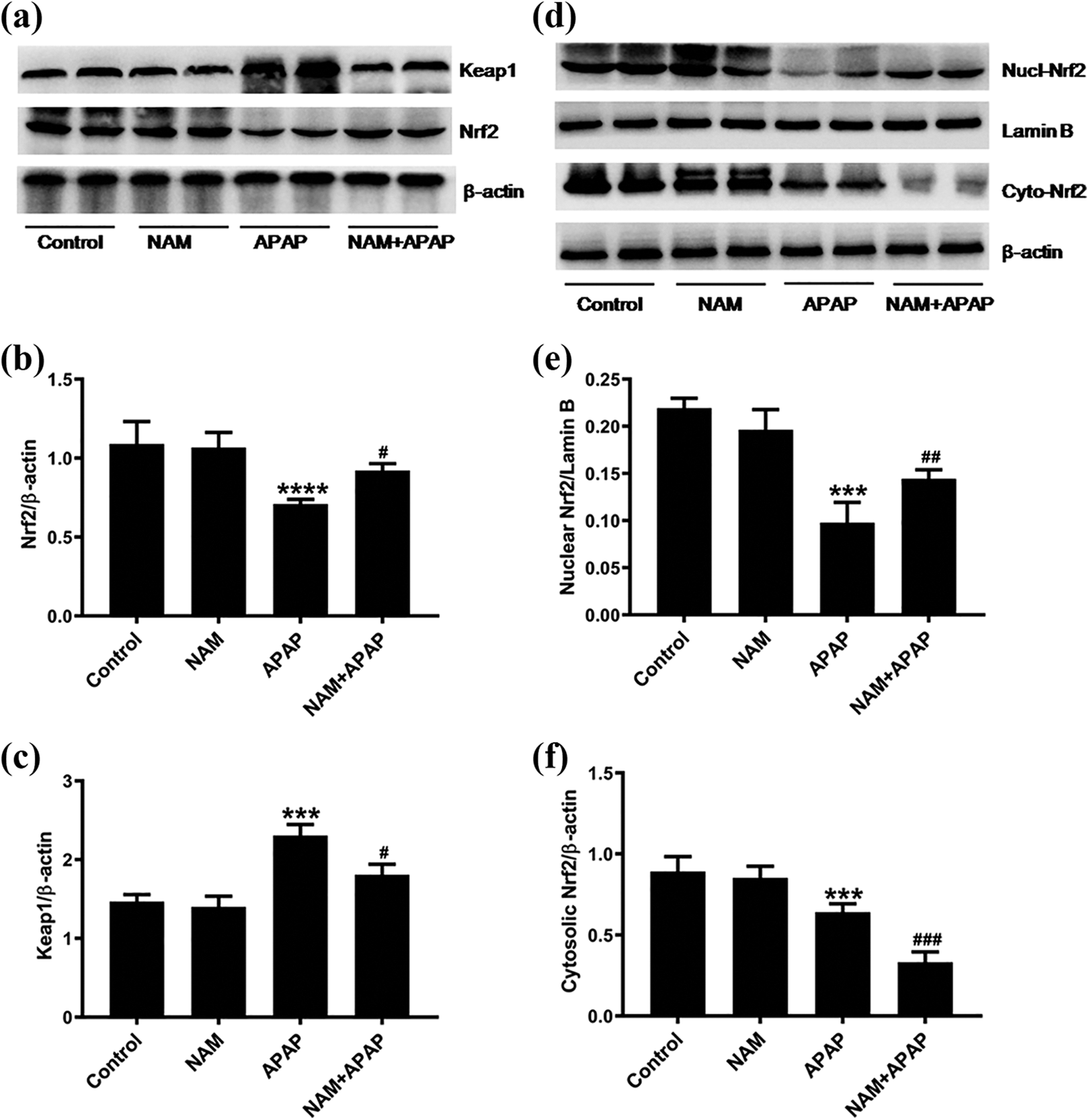
Effects of NAM on Nrf2 pathway (n = 4). The liver tissue homogenates were extracted for preparation of total protein, nuclear and cytosolic protein. Protein expressions were analyzed by Western blot, and β-actin or Lamin B was deemed as the loading control for semiquantitative analyses. (a) Protein expressions of Nrf2 and Keap1; (b) semiquantitative analyses of Nrf2; (c) semiquantitative analyses of Keap1; (d) protein expressions of nuclear and cytosolic Nrf2; (e) semiquantitative analyses of nuclear Nrf2; (f) semiquantitative analyses of cytosolic Nrf2. All values are expressed as mean ± SD. ***p < 0.005: compared with control group; #p < 0.05, ##p < 0.01, ###p < 0.005: compared with APAP group.
NAM activates antioxidant defense and attenuates APAP-induced ALI
Attributing to Nrf2 activation, we determined whether NAM pretreatment could enhance the antioxidant defense. So, the key antioxidant non-enzyme molecules glutathione (GSH) and enzyme superoxide dismutase (SOD) were characterized. As shown in Figure 6(a) and (b), pretreatment with NAM improved the reduced levels of GSH and SOD due to APAP exposure. Considering that NAM enhances antioxidant defense, we further confirmed its hepatoprotective property with the liver index and necrotic area via analyzing H&E staining (Figure S1). As shown in Figure 6(c) and (d), NAM alleviated the elevation of liver index and necrotic area caused by APAP challenge. In brief, these results reveal that NAM activates antioxidant defense and attenuates APAP-induced ALI.

NAM activates antioxidant defense and attenuates APAP-induced ALI (n = 4). NAM (800 mg/kg) were intraperitoneally injected three times before APAP (500 mg/kg) exposure for 8 hours. Liver samples were harvested to evaluate GSH levels, SOD activities, liver index and necrotic area. (a) Hepatic GSH levels; (b) hepatic SOD activities; (c) assessment of liver index; (d) measurement of necrotic area. All values are expressed as mean ± SD. ***p < 0.005: compared with control group; #p < 0.05, ###p < 0.005: compared with APAP group.
Discussion
As an essential redox carrier, NAD+ plays a fundamental role in the regulation of energy metabolism pathways, especially in mitochondria. It is well known that incomplete hydride and electron transfer can produce reactive oxygen species (ROS) in mitochondrial respiratory chain, which leads to oxidative stress. Therefore, NAD+ may be an important molecule involved in the oxidative stress induced by APAP overdose and is an appropriate antioxidative target. In this study, we firstly revealed that the contents of NAD+ fell in mice livers after APAP insult in a dose-dependent manner. NAD+ deficiency has been confirmed in a variety of pathological processes. However, little is known in APAP-induced ALI. Our data demonstrated APAP overdose caused NAD+ depletion, indicating that NAD+ plays a key role in the pathogenesis of APAP-induced ALI.
Seeing that NAD+ pool is determined by salvage biosynthesis pathway, we postulated that NAD+ decline after APAP exposure is due to decreased biosynthesis. Thus, we detected the protein expression of NAMPT governing NAD+ biosynthesis. The Western blot results displayed reduced NAMPT expression following APAP administration, which has been supported by a recent report. 19 Owing to the rate-limiting enzyme that catalyzes NAM to synthesize NAD+, downregulation of NAMPT represented the impaired salvage synthesis pathway. Therefore, APAP overdose elicits NAD+ decrease that is attributed to protein expression of downregulated NAMPT, which suggests the NAD+ disturbance suffering from less synthesis. This is the new mechanism to delineate the NAD+ deficiency.
NAM has been applied for treating multiple diseases for several decades of years in clinic. However, the beneficial effects of NAM on liver injuries have been evidenced only in research fields. In various animal models of liver injuries, which were induced by methotrexate, 20 ischemia-reperfusion, 21 alcohol, 22 lipopolysaccharide/D-galactosamine, cecal ligation and puncture, 23 and thioacetamide, 24 NAM exerted hepatoprotective effects, but the roles of NAD+ remained unclear in these processes. Recently, NAM was found to promote liver regeneration following partial hepatectomy in mice and activation of sirtuin 1 indicated that NAD+ was the critical factor in the process. 25 Nevertheless, it is uncertain whether NAD+ is involved in protective effects of NAM on APAP-induced hepatoxicity. In present study, we demonstrated that NAM restored the NAD+ levels impaired by APAP in mice livers. This was concomitant with the protein expression of increased NAMPT that exhibited activated salvage synthesis pathway. Actually, there were few reports on the NAMPT levels followed NAM treatment. One study found the steep increase of NAMPT levels after NAM administration in mice exposed to 6 Gy irradiation. 26 Another piece of evidence observed that 100 mg/kg NAM remarkably elevated hepatic NAMPT contents in high fat diet rats. 27 Both of the two results are consistent with our data, but the underling mechanism needs to be elucidated in detail.
As a transcriptional factor, Nrf2 activates the expression of hundreds of genes, such as phase II detoxification enzymes, antioxidant proteins and transporters. 28 Recently, NAD+ administration activating Nrf2 has been confirmed. 29 Our study demonstrated APAP suppressed Nrf2 expression and induced activation of its repressor Keap1 which caused oxidative stress. Indeed, it has been elucidated by a wealth of studies.30–32 Following NAM treatment, the declined expression of Nrf2 increased, accompanying with Keap1 downregulation. Further analysis of cytoplasm and nucleus expression of Nrf2 uncovered that NAM was responsible for the translocation activation of Nrf2. In recent reports, Nrf2 upregulated by NAM are also observed in hepatocellular carcinoma both in vivo and in vitro 1 and normal melanocytes. 33 Therefore, NAM elevates NAD+ to promote Nrf2 activation. Nevertheless, the mechanism needs to be further defined.
Taking into account Nrf2 activation, we further assessed the antioxidant effects of NAM with two critical molecules SOD and GSH widely used in variety of studies.34,35 It is known that GSH exerts pivotal role in the pathogenesis of APAP due to its exhaustion caused by NAPQI, and N-acetylcysteine, as the only antidote for APAP treatment via restoring GSH to remove toxic NAPQI. Additionally, SOD is directly involved in ROS elimination and it is regulated by Nrf2. 28 Thus, enhancement of GSH and SOD can promote the antioxidant defense. Our investigation revealed that NAM markedly restored GSH and SOD depletion insulted by APAP, which attenuated APAP-induced oxidative stress in mice. In the light of the antioxidant property of NAM, it is reasonable to confer hepatoprotective benefits. Finally, we observed the beneficial effects of NAM on APAP-induced ALI. After NAM administration, the liver index and necrotic area were improved confirming the hepatoprotective effects of NAM.
Our study clearly demonstrated that APAP overdose repressed NAMPT at protein levels, and thus resulted in NAD+ depletion owing to salvage synthesis inhibition. As a precursor of NAD+, NAM improved the NAD+ decline attributing to synthesis increase via upregulating protein expression of NAMPT. Restored NAD+ promoted the Nrf2 translocational activation. As a consequence, the antioxidant defense was strengthened and NAM exerted protective effects against APAP-induced hepatotoxicity.
Supplemental material
Supplemental Material, sj-docx-1-het-10.1177_09603271211014573 - Nicotinamide improves NAD+ levels to protect against acetaminophen-induced acute liver injury in mice
Supplemental Material, sj-docx-1-het-10.1177_09603271211014573 for Nicotinamide improves NAD+ levels to protect against acetaminophen-induced acute liver injury in mice by J Xu, L Zhang, R Jiang, K Hu, D Hu, C Liao, S Jiang, Y Yang, J Huang, L Tang and L Li in Human & Experimental Toxicology
Footnotes
Author contributions
J Xu and L Zhang contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Science and Technology Bureau of Yuzhong District [2015-16-19] and Basic Medical College Research Plan [2013-5].
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
