Abstract
Background
Activation of PRR11 contributes to the progression of lung cancer and is related to its methylation status. However, the regulatory mechanism of luteolin-zinc (Lu-Zn) on PRR11 methylation-mediated lung cancer progression under hypoxic conditions remains to be explored.
Objectives
This study aims to investigate the inhibitory effect of Lu-Zn on lung cancer cells, its regulation of ferroptosis, and the role of PRR11 in lung cancer progression under hypoxia.
Materials and Methods
In vitro experiments were conducted to evaluate the effect of Lu-Zn on lung cancer cells, focusing on its impact on invasion, migration, and ferroptosis. The expression levels of PRR11, its methylation status, and microRNA-6769b-3p (miR-6769b-3p) were also analyzed.
Results
Under hypoxic conditions, Lu-Zn significantly inhibited the invasion and migration abilities of lung cancer cells and promoted ferroptosis. Additionally, Lu-Zn reversed the pro-cancer effects induced by PRR11. At the molecular level, Lu-Zn promoted the methylation of PRR11 and regulated miR-6769b-3p.
Conclusion
Our study demonstrates that Lu-Zn enhances the methylation of PRR11 and reduces its expression under hypoxic conditions through miR-6769b-3p. This leads to inhibition of the downstream PI3K/AKT/mTOR signaling pathway, promoting ferroptosis and exerting anti-lung cancer effects.
Introduction
Lung cancer is a prevalent malignancy and a major cause of patient mortality (Santucci et al., 2020). The high mortality rate is often due to the aggressive invasion and metastasis of tumor cells. The tumor microenvironment, a complex system, plays a crucial role in the onset and progression of tumors. The microenvironment comprises a network of Sertoli cells, fibroblasts, immune cells, endothelial cells, adipocytes, and the extracellular matrix (ECM), collectively creating a dynamic and functional milieu that regulates various physiological processes and cellular interactions (Abushukair & Saeed, 2022). In the late stages of tumor development, the surrounding tissues undergo significant changes, releasing various factors that promote tumor growth, leading to increased malignancy and metastasis (Li et al., 2020). Solid tumors can become hypoxic for various reasons, and hypoxia is not only a critical factor for poor prognosis but also closely associated with tumor progression, invasion, and migration (Terry et al., 2020; Zhang et al., 2024). Our experiments involve creating a hypoxic environment to simulate conditions in advanced tumors, aiming to study the effects of drugs on tumor invasion and metastasis.
DNA methylation typically involves the addition of a methyl group to the 5′ cytosine of CpG dinucleotides (Nasrullah et al., 2022). This process promotes gene silencing, transposon silencing, X-chromosome inactivation, and epigenetic reprogramming. Aberrant DNA methylation patterns have been linked to cancer development through two main mechanisms: hypomethylation of oncogenes, resulting in their activation, and hypermethylation of tumor suppressor genes, leading to their inhibition. We previously discovered that luteolin-zinc (Lu-Zn) significantly inhibits the expression of PRR11, leading us to speculate about its relationship with PRR11 methylation levels.
Luteolin, a well-studied plant compound, is commonly found in various vegetables and herbs (Caporali et al., 2022). Recent studies suggest that luteolin has the potential to impede cancer development by disrupting the cell cycle, suppressing proliferation, inducing apoptosis, and inhibiting the migration and invasion of cancer cells (Çetinkaya & Baran, 2023; Hussain et al., 2021). We synthesized a new derivative by combining luteolin with zinc to explore whether this derivative possesses enhanced biological activity. This study aims to investigate whether the anti-lung cancer effects mediated by the methylation status of PRR11 are related to Lu-Zn.
Materials and Methods
Materials
Luteolin was obtained from Sichuan Chengdu Manster Biotechnology Co., Ltd. MHY1485 (HY-B0795) was purchased from MedChemExpress. Anti-PRR11 (PA5113175) was purchased from Thermo Fisher Scientific. Fetal bovine serum (FBS) was provided by Senbeijia Biotechnology Co., Ltd., and ABclonal supplied antibodies against MMP2 (A6247), MMP9 (A0289), E-cadherin (A3044), N-cadherin (A3045), phosphorylated AKT (AP1208), phosphorylated PI3K (AP0427), phosphorylated mTOR (AP0115), and β-actin (AC026). Kits for detecting iron, lipid reactive oxygen species (ROS), malondialdehyde (MDA), and glutathione (GSH) were used to assess the expression of these molecules. The miRNA negative control (miR-NC), miR-6769b-3p mimics/inhibitors, Ad-PRR11, and Ad-Ctrl were all obtained from GenePharma Biotechnology Co., Ltd. (Shanghai, China).
Cell Culture
LLC (CL-0140) mouse lung cancer cells were obtained from Procell Life Science & Technology Co., Ltd. The cells were cultured in DMEM/high glucose medium supplemented with 10% FBS, 100 U/mL penicillin, and 100 µg/mL streptomycin at 37°C in a 5% CO2 atmosphere. For hypoxic treatment, cells were placed in a hypoxic incubator (37°C, 1% O2) for 24 hours.
CCK8
During the logarithmic growth phase, cells were harvested and seeded into 96-well plates at a density of 3.0 × 103 cells per well. Each well was treated with 10 µL of CCK-8 solution and incubated in darkness for 2 hours. Optical density at 450 nm was measured using a microplate reader.
Cell Scratch Assay
After treatment, cells were seeded in six-well plates. When the cells reached approximately 80% confluence, a 200 µL pipette tip was used to create a straight scratch in each well. The medium was replaced with a serum-free medium. Scratch closure was observed and photographed under a microscope at 0 and 24 hours, with different areas chosen for photography at each time point.
Clone Formation Assay
Cells were seeded at a density of 800–1,000 cells per well in six-well plates and incubated at 37°C. Cell growth was monitored daily, with medium changes every 3 days. After 2 weeks, cells were fixed, stained, and photographed.
Transwell Assay
For migration assays, the upper chamber of the Transwell was left uncoated with Matrigel (Corning). For invasion assays, the upper chamber was coated with Matrigel. Cells were placed in the upper chamber and treated accordingly, while the lower chamber contained a medium with serum. Cells were cultured in a hypoxic incubator, then fixed, stained, and observed under an optical microscope.
TCGA Database Lung Cancer Analysis
RNAseq data and corresponding clinical data for lung cancer were analyzed using the Cancer Genome Atlas (TCGA) (
Molecular Docking
AutoDock software was used to process and dock luteolin with PRR11, while Discovery Studio software was used for Lu-Zn docking with PRR11. The best docking sites and optimal binding energies were identified, and visualizations were created using PyMOL software.
Western Blotting (WB)
Protein concentration was determined using the BCA method after sample lysis. Following electrophoresis, membrane transfer, and blocking, primary antibodies and β-actin antibody were incubated overnight at 4°C. The next day, membranes were washed and exposed to secondary antibodies at room temperature for 1 hour. Detection was performed using the ECL Enhanced Kit (ABclonal).
Real-time Polymerase Chain Reaction (PCR)
RNA was converted to cDNA using the ABclonal ABScript II cDNA One-Step Reverse Transcription Kit. Real-time PCR amplification was performed on the Applied Biosystems StepOne system using TB Green® Premix Ex Taq™ (Tli RNaseH Plus). Primer sequences are listed in Table S1 in the supplemental material. Relative expression levels of target genes were determined using the 2-∇∇Cq method.
Cell Immunofluorescence
After processing, samples were fixed with 4% paraformaldehyde, permeabilized with 0.5% Triton X-100, and blocked with goat serum. Primary antibodies were added and incubated overnight. Fluorescently labeled secondary antibodies were then added, followed by DAPI staining. Images were captured using a fluorescence microscope.
Methylation Analysis
CpG islands of PRR11 were visualized using the UCSC Genome Browser. Methylation levels of PRR11 were assessed following established protocols (Veland et al., 2017).
Statistical Analysis
Data analysis was conducted using SPSS 25.0. In instances where the data exhibited a normal distribution, independent samples t-tests or paired t-tests were employed to compare groups.
Results
Lu-Zn Inhibits Proliferation, Invasion, and Migration of Lung Cancer Cells Under Hypoxia
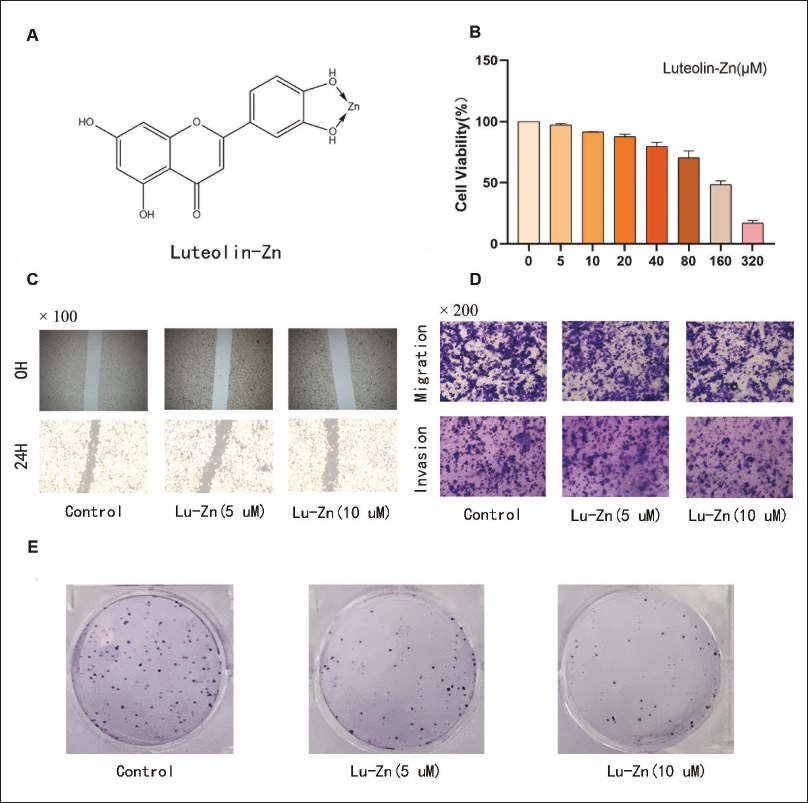
Lu-Zn was shown to reduce lung cancer cell viability in a dose-dependent manner, as demonstrated by the CCK8 assay (Figure 1A and B). Based on these findings, we selected 5 and 10 µmol/L concentrations of Lu-Zn for further experiments. In scratch tests, Lu-Zn significantly inhibited the movement of lung cancer cells (Figure 1C). Moreover, the Transwell assay confirmed Lu-Zn’s anti-invasion and anti-migration capabilities (Figure 1D). The colony formation assay revealed that Lu-Zn effectively suppressed lung cancer cell growth (Figure 1E). These results collectively affirm that Lu-Zn impedes the proliferation, invasion, and migration of lung cancer cells under hypoxic conditions, exhibiting potent anti-cancer properties.
Impact of Luteolin-Zinc (Lu-Zn) on Cell Viability, Proliferation, Invasion, and Migration Under Hypoxia. (A) Molecular Structure of Lu-Zn. (B) Treatment of LLC Lung Cancer Cells with Varying Concentrations of Lu-Zn. (C) Comparative Images of the Scratch Test at 0 and 24 Hours for Each Treatment Group. (D) Representative Images from Migration and Invasion Assays for Each Treatment Group. (E) Colony Formation Assay Results Following Lu-Zn Treatment.
Lu-Zn Inhibits the Expression of Pro-invasion and Pro-migration Factors in Lung Cancer Cells Under Hypoxia
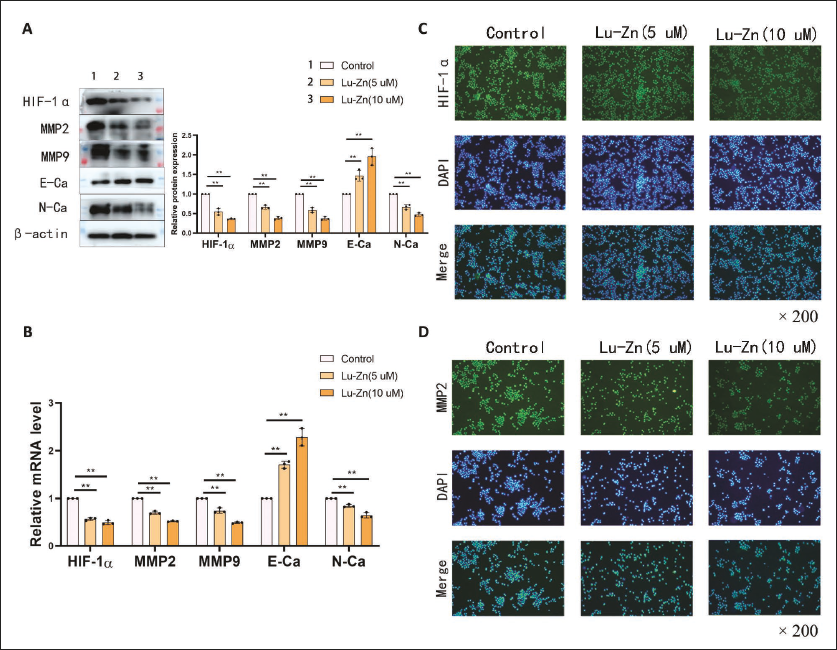
Under hypoxic conditions, Lu-Zn effectively suppressed the expression of HIF-1α (Figure 2A). WB and PCR assays revealed significant inhibition of pro-invasion and pro-migration factors MMP2, MMP9, and N-cadherin, along with upregulation of E-cadherin (Figure 2A). Immunofluorescence assays corroborated these findings, showing marked decreases in HIF-1α and MMP2 levels post Lu-Zn treatment (Figure 2C and D). These findings confirm that Lu-Zn obstructs the movement and spread of lung cancer cells in hypoxic conditions.
Lu-Zn Controls the PI3K/AKT/mTOR Signaling Pathway Under Hypoxia
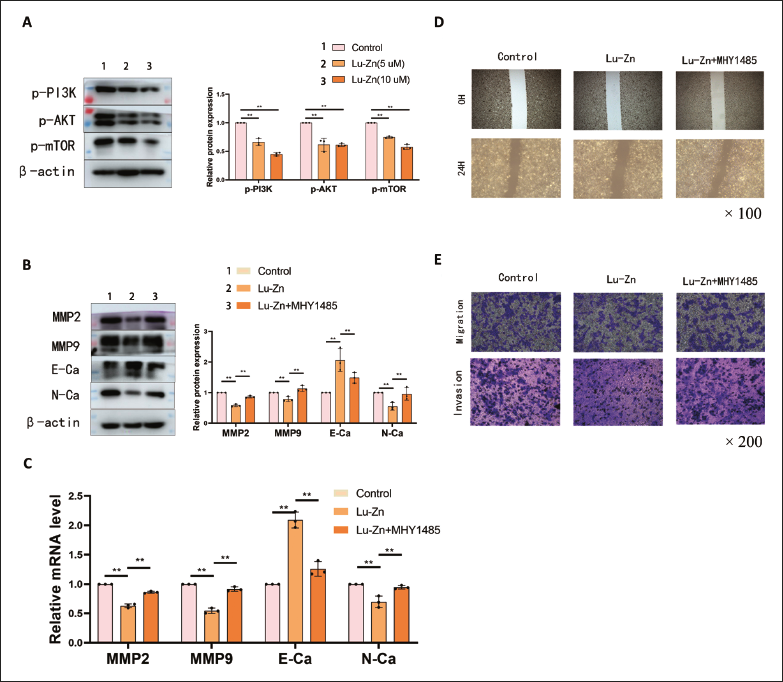
Lu-Zn significantly reduced the expression of p-AKT, p-PI3K, and p-mTOR in WB assays (Figure 3A). The addition of the mTOR agonist MHY1485 (20 µmol/L) notably increased the levels of MMP2, MMP9, and N-cadherin, while decreasing E-cadherin expression compared to the Lu-Zn group (Figure 3B and C). Additionally, MHY1485 attenuated Lu-Zn’s anti-invasion and anti-migration effects (Figure 3D and E). These results indicate that Lu-Zn modulates the PI3K/AKT/mTOR pathway, affecting lung cancer cell invasion and migration.
Lu-Zn Promotes Ferroptosis
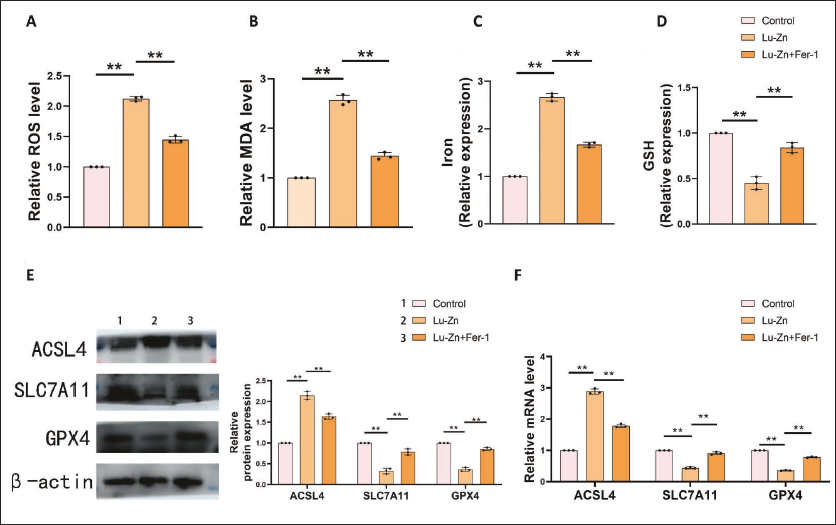
In tumors, the inhibition of the PI3K/AKT/mTOR signaling pathway can promote the progression of ferroptosis (Chen et al., 2024, 2023). Knowing that Lu-Zn can inhibit the PI3K/AKT/mTOR signaling pathway, we also examined ferroptosis-related indicators. Lu-Zn can significantly increase the levels of ROS, MDA, and iron while inhibiting the expression of GSH, an effect that is reversed upon the addition of a ferroptosis inhibitor (Figure 4A–D). Ferroptosis-promoting factor ACSL4 and ferroptosis-inhibiting factors SLC7A11 and GPX4 further suggested Lu-Zn’s role in promoting ferroptosis (Figure 4E and F). These findings suggest that Lu-Zn promotes ferroptosis, potentially through regulation of the PI3K/AKT/mTOR pathway.
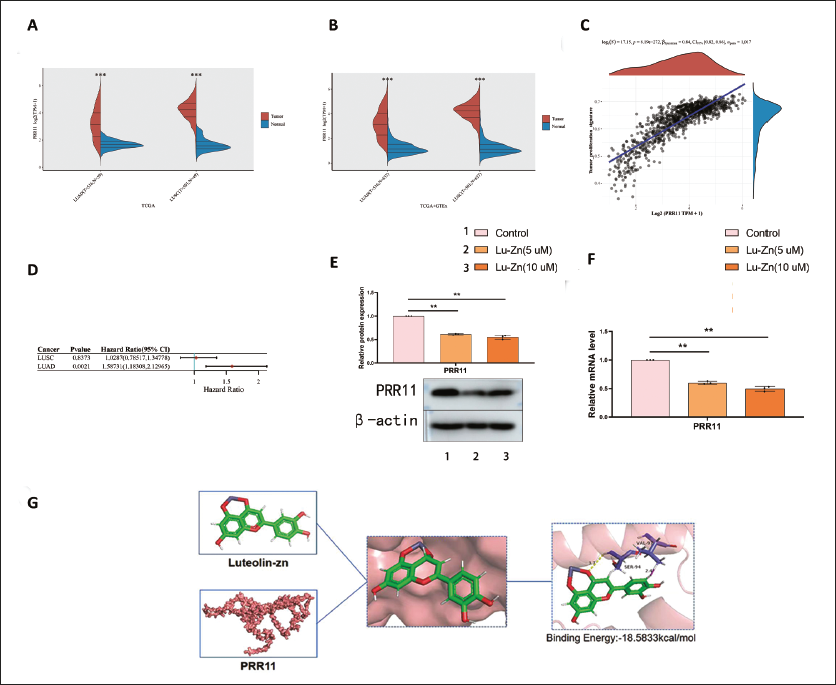
PRR11’s Role in Lung Cancer
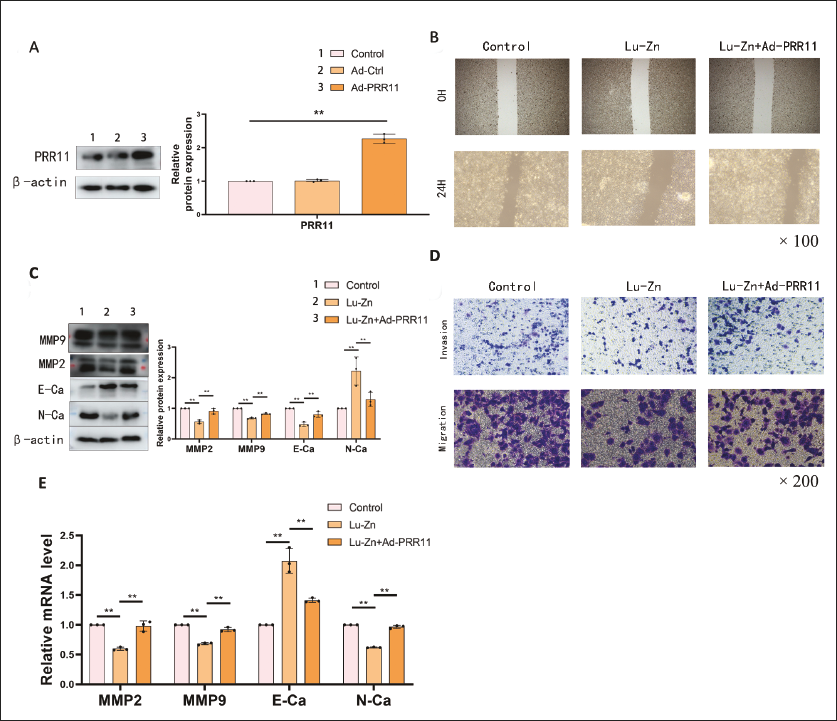
Using the TCGA database, we conducted expression, prognosis, and tumor proliferation analyses of PRR11. Non-small cell lung cancer (NSCLC) tumors exhibited significant upregulation of PRR11 (Figure 5A and B). Higher PRR11 expression correlated with increased tumor proliferation (Figure 5C) and poorer prognosis (Figure 5D). Lu-Zn treatment downregulated PRR11 expression, consistent with bioinformatics data (Figure 5E and F). Binding energy analysis revealed SER94 and VAL97 as Lu-Zn binding sites on PRR11, with a binding energy of –18.5833 kcal/mol (Figure 5G). Upregulation of PRR11 reversed the anti-invasion and anti-migration effects of Lu-Zn (Figure 6A–E), indicating that Lu-Zn’s anti-cancer effects are closely linked to PRR11.
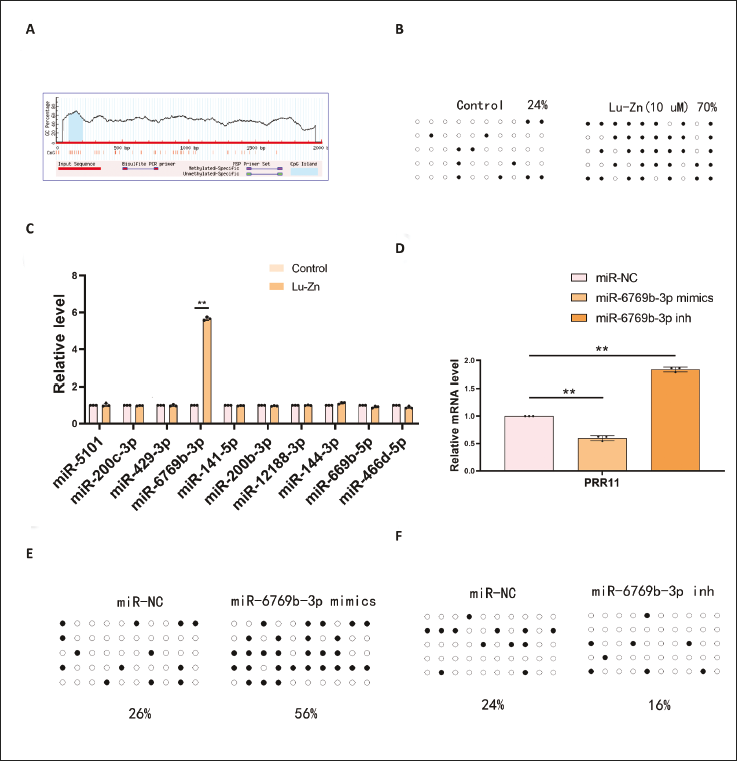
Methylation and MicroRNA Regulation of PRR11
Bioinformatics analysis identified a CpG island with 10 CpG sites in the PRR11 promoter (Figure 7A). Lu-Zn treatment significantly increased PRR11 methylation from 24% in controls to 70% (Figure 7B). Bioinformatics analysis predicted 10 potential miRNAs targeting PRR11, with miR-6769b-3p specifically increased by Lu-Zn (Figure 7C). miR-6769b-3p mimics reduced PRR11 expression and increased its methylation levels, while miR-6769b-3p inhibitors had the opposite effect (Figure 7D–F). These findings suggest that Lu-Zn modulates PRR11 expression and methylation through miR-6769b-3p.
Discussion
The development and occurrence of tumors are intricately linked to the tumor microenvironment. This dynamic environment consists of various supportive cells, including fibroblasts, immune cells, endothelial cells, and adipocytes, as well as the ECM, which regulates communication between cells and their surroundings (Goenka et al., 2023). In deep solid tumors, limited oxygen diffusion due to insufficient angiogenesis results in hypoxia, a condition where oxygen cannot reach cells far from the tumor center (Yang et al., 2024). Hypoxia is a significant factor associated with poor prognosis, tumor deterioration, infiltration, and migration (Tinganelli & Durante, 2020). In our study, we created a moderately hypoxic environment and observed that the addition of Lu-Zn significantly inhibited the expression of the key hypoxic factor HIF1-α, suggesting its potential to regulate hypoxic conditions and counteract tumor-promoting responses. The microenvironment significantly influences cancer cell migration and invasion, as seen in colorectal and breast cancers, where collective cell migration is common (Rabelo et al., 2023). Metastasis involves tumor cells invading through the ECM, traveling via blood or lymphatic vessels, and establishing growth at distant sites (Zanotelli et al., 2021). Matrix metalloproteinases (MMPs), which degrade the ECM, play a crucial role in this process. Our research demonstrated that Lu-Zn significantly inhibited MMP2 and MMP9, indicating strong anti-lung cancer properties.
Ferroptosis, an iron-dependent form of cell death distinct from apoptosis, necrosis, and autophagy, is characterized by redox imbalance and elevated ROS within cells (Zhang et al., 2022). Ferroptosis is characterized by an imbalance in redox state and elevated levels of ROS within cells. Ubellacker et al. (2020) found that melanoma cells metastasize more through the lymphatic system to avoid ferroptosis. Our findings suggest that Lu-Zn may enhance ferroptosis, contributing to its anti-lung cancer effects.
NSCLC is the most common type of lung cancer (Socinski et al., 2016). Despite recent advancements in treatment, early metastasis often leads to poor prognosis (Bansal et al., 2016; Friedlaender et al., 2020). Thus, discovering new therapeutic targets is crucial. PRR11, located on chromosome 17q22, has been identified as a gene involved in NSCLC progression and cell cycle regulation, influencing growth, motility, cell division, and cell death (Zhang et al., 2018; Zhao, 2015). Increased PRR11 expression in NSCLC tissues compared to normal tissues has been noted (Wang et al., 2023). PRR11 activates the Akt/mTOR signaling pathway, promoting tumor growth (Zhang et al., 2020). Our results also found that Lu-Zn inhibits the PI3K/AKT/mTOR signaling pathway, consistent with this. Subsequently, in our experiments, we upregulated the PRR11 gene and confirmed it through WB experiments. After upregulating the PRR11 gene, the proliferation, invasion, and migration abilities of Lu-Zn anti-lung cancer cells were significantly inhibited. Through bioinformatics and other studies, PRR11 has been identified as a biomarker for lung adenocarcinoma (Hu et al., 2023). In summary, PRR11 is the therapeutic target of Lu-Zn in the treatment of lung cancer.
DNA methylation, an epigenetic mechanism, plays a crucial role in gene expression regulation (Vrtačnik et al., 2015). High levels of methylation downregulating ZNF582-AS1/ZNF582 have been linked to esophageal cancer progression (Li et al., 2024). Similarly, DNMT1-induced hypermethylation of the SOCS1 promoter enhances the stemness of liver cancer stem-like cells (Lou et al., 2024). Our study showed that Lu-Zn promotes PRR11 methylation, inhibiting its expression. miRNAs, which regulate various diseases, including liver metabolism and tumor development, also play a significant role in lung cancer progression (Chen et al., 2022; Wang et al., 2021). For instance, altered 3′UTR regions of EGFR and KRAS oncogenes can prevent miRNA binding, promoting lung cancer (Bagci, 2024). miR-21 upregulation by stereotactic body radiotherapy (SBRT) may inhibit MDSC expansion in lung cancer models. Our research identified miR-6769b-3p as an inhibitor of lung cancer, with Lu-Zn enhancing its expression. Subsequent experiments showed that miR-6769b-3p targets PRR11. Importantly, PRR11 methylation mediated by miR-6769b-3p is involved in the inhibitory effect of Lu-Zn on lung cancer.
Conclusion
Our study indicates that Lu-Zn promotes PRR11 methylation and reduces its expression under hypoxia conditions through miR-6769b-3p. This reduction inhibits the downstream PI3K/AKT/mTOR signaling pathway, promotes ferroptosis, and exerts significant anti-lung cancer effects.
Footnotes
Acknowledgments
The authors would like to thank Taizhou University for providing facilities and technical support and Figdraw for providing drawing convenience.
Authors’ Contribution
Jiayong Huang: Writing – review & editing, Writing – original draft, Visualization, Validation, Supervision, Software, Resources, Methodology, Investigation, Funding acquisition, Formal analysis, Data curation, Conceptualization. Xiang Li: Writing – review & editing, Project administration, Methodology, Investigation, Funding acquisition, Conceptualization. Binbo Fang, Ting Guo, Yiyang Li, Yilin Yang, Jiongcheng Ying, and Haoyun Zhou: Methodology, Investigation.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This article does not involve human participants or animals. Therefore, informed consent and animal ethics approval are not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Taizhou Municipal Science and Technology Bureau (23ywb151).
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
