Abstract
Objectives
This research explored the efficacy of valtrate (Val) in lung cancer (LC).
Methods
A549 and H1299 cells were dealt with Val. SLC7A11 was overexpressed by cell transfection. Xenograft model was established in nude mice. Cell viability, proliferation and apoptosis were measured by CCK-8, EdU and Annexin-V and propidium iodide staining, respectively. Ferroptosis was assessed by iron assay kit. reactive oxygen species level was tested by ROS assay kit. Histopathological changes in tumor tissues were analyzed by HE staining. Protein expression was analyzed employing Immunohistochemical staining and western blot.
Results
A549 and H1299 cell viability were notably lowered by Val in a concentration-dependent way. Cell proliferation was markedly repressed by 10 and 20 μM of Val, while apoptosis and ROS generation were markedly augmented. SLC7A11 and GPX4 expression was both distinctly lowered, while Fe2+ level was remarkably improved by 10 and 20 μM of Val. However, 20 μM of Val-triggered effects were distinctly counteracted by SLC7A11 overexpression. In vivo assay revealed that both tumor volume and weight were distinctly declined by 10 mg/mL Val. The variations of proliferation, apoptosis and ferroptosis-related proteins were in line with the results in vitro assay.
Conclusion
Val might antagonize malignant phenotypes of LC cells by reducing SLC7A11.
Introduction
Lung cancer (LC) has become the dominant reason for cancer-associated deaths worldwide in 2020. 1 As reported, the 5-year relative survival rate for LC is only 6%. 2 It is predicted that the mortality of LC in China may increase by about 40% between 2015 and 2030. 3 Currently, enormous improvements in treatment strategies have been achieved in LC, which mainly contains lung surgery, radiation, chemotherapy, immunotherapy and targeted therapy. 4 Despite of the great progress in the management of LC, the outcomes are still limited because of severe adverse effects, poor selectivity and drug resistance. 5 Hence, there is still urgent need to develop novel effective drugs for LC treatment.
Ferroptosis refers to an iron-dependent and atypical cell death that triggered by accumulation of lipid-based reactive oxygen species (ROS).6,7 Previous researchers have found that solute carrier 7A11 (SLC7A11) and GPX4 are crucial regulators of ferroptosis.8,9 SLC7A11 was verified to be upregulated in LC. 10 It was demonstrated that erastin could induce ferroptosis by inhibiting GPX4 expression, while ferrostatin-1 could in other way restrain ferroptotic cell death. 11 Mounting investigations have demonstrated that ferroptosis exert crucial roles in tumor growth inhibition. It was recorded that ferroptosis could repress cell proliferation in various cancers, such as glioma cancer, pancreatic cancer (PC), hepatocellular carcinoma and lung cancer.12–16 Researchers have discovered that some malignancies are vulnerable to ferroptosis intrinsically, 17 so targeting ferroptosis might be a prospective strategy for treatment of LC in the future.
Valeriana jatamansi Jones is a Chinese herbal medicine, which was previously verified to be used for the treatment of nervous disorders.18,19 Valtrate (Val), a new epoxide cycloallyl ether terpene that extracted from the root of V. jatamansi Jones,20,21 was discovered to possess cytotoxic and anti-tumor activities in previous studies.22,23 For instance, Lou et al. reported that Val exerted anti-tumor efficacy in breast cancer (BC) via repressing migration, while enhancing apoptosis of BC cells. 24 Besides, it was recorded that Val performed anti-PC activity through repressing Stat3 signaling pathway. 21 Another study demonstrated that IVHD-Val inhibited proliferation, arrested cell cycle progression and induced apoptosis of ovarian cancer (OC) cells. 25 However, whether it possess the same effect in LC remains uninvestigated.
In this research, series concentrations of Val treatment were carried out on A549 and H1299 cells to figure out the appropriate concentration to perform following experiments. Then, we preliminary studied the influences of Val treatment on the proliferation, apoptosis, ferroptosis and ROS generation of LC cells both in vitro and in vivo. Besides, the underlying regulating mechanism was also explored.
Methods and materials
Cell culture and treatment
A549 and H1299 cell lines were purchased from ATCC (Rockville, USA). A549 cells was cultured in DmEM (Thermo Fisher Scientific, MA, USA), while H1299 cells was maintained in RPMI-1640 medium (Thermo Fisher Scientific). The cell cultures were supplemented with 10% (v/v) FBS and 1% (v/v) streptomycin/penicillin. Cell culture was carried out under 5% CO2 and 37°C conditions. Culture medium was refreshed 3 times each week.
Val (purity >98%, molecular weight = 422.47 kDa) was obtained from Abmole Bioscience Inc. (Houston, USA), and its 2- and 3-dimensional structure were displayed in Figure 1(a). Cells were treated with different concentrations of Val (1.25 μM, 2.5 μM, 5 μM, 10 μM, 20 μM and 40 μM) that prepared in DMSO for 48 h.
21
For analysis of ferroptosis, A549 and H1299 cells were dealt with 20 μM of Val plus erastin (Sigma-Aldrich, USA) or ferrostatin‐1 (MedChemExpress, USA) for 48 h. Valtrate treatment lowered lung cancer cell viability.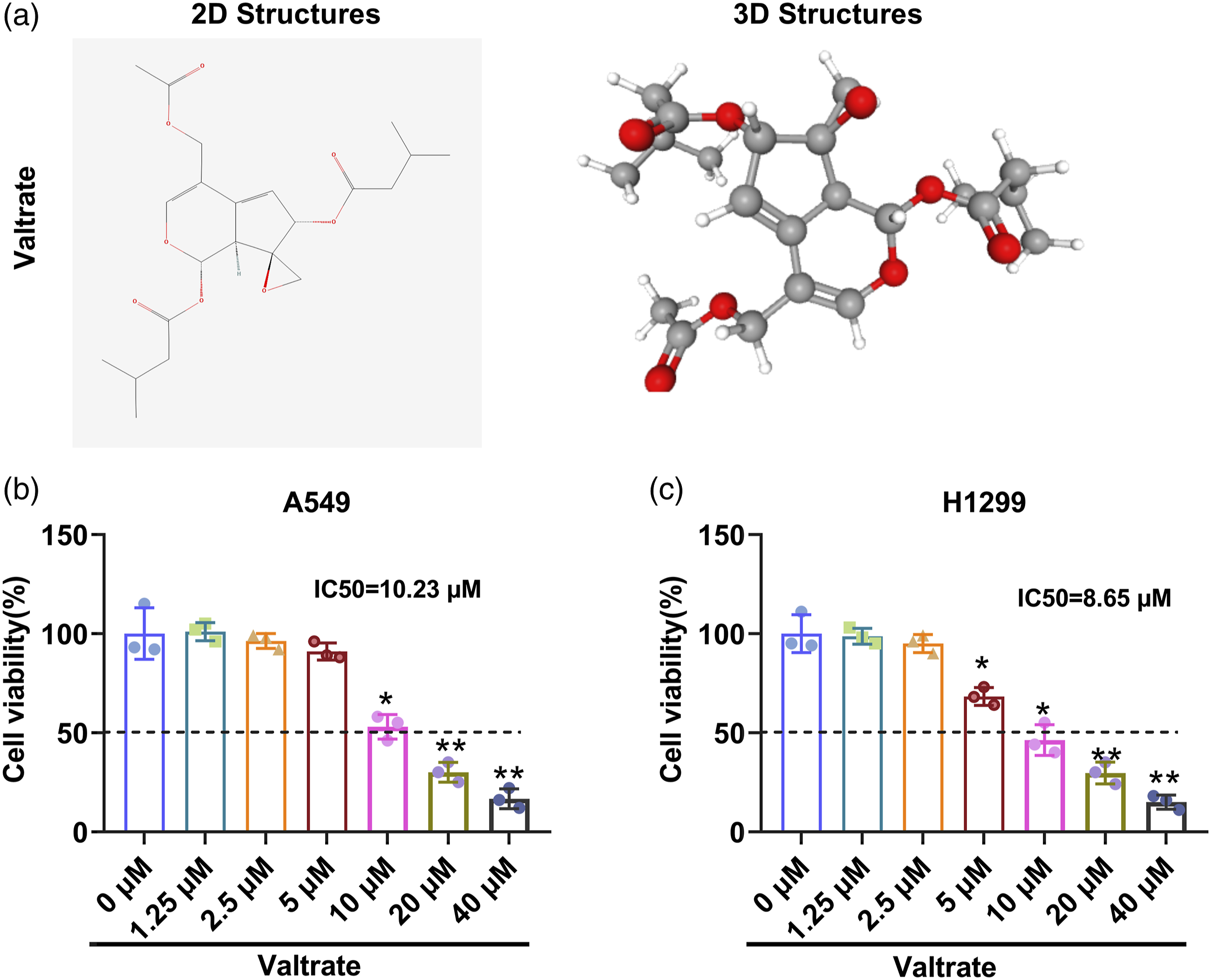
A549 and H1299 cells were exposed to various concentrations of Val (1.25, 2.5, 5, 10, 20 and 40 μM) for 48 h. A. The 2- and 3-dimensional structures of Val. B and C. Cell viability was detected by CCK8 assay in Val-treated A549 and H1299 cells. *p < 0.05, **p < 0.01. Each experiment has three replications.
Cell transfection
The overexpression plasmids SLC7A11 were constructed by Synbio Technologies (Suzhou, China). Then, A549 and H1299 cells were transfected with the recombinant SLC7A11 plasmids and the empty vector using Lipofectamine 2000 (Life Technology) for 48 h, respectively. After that, cell collection was performed.
Cell viability
Cell viability was assessed via Cell Counting Kit-8 (CCK-8, Dojindo) following the product instructions. Briefly, the transfected and non-transfected H1299 and A549 cells were plated into 96-well plates (1 × 105/ml) and cultured under 5% CO2 and 37°C conditions for 24 h. Afterwards, cells were treated with different concentrations of Val for another 24 h. Then, 10 μL of CCK-8 reagent was provided and the mixtures were maintained at the same conditions for 1 h. Finally, the absorbance of each well at 450 nm was measured using a microplate reader (Bio-Rad, Hercules, CA).
Cell proliferation
Cell proliferation was assessed using EdU incorporation assay (RIBOBIO, Guangzhou, China). H1299 and A549 cells at logarithmic growth phase were plated into 96-well plates (1 × 104/ml) and cultured in a 5% CO2 and 37°C incubator for 24 h. Afterwards, cells were exposed to different concentrations of Val for another 24 h. Then, the culture medium was replaced with 100 μL of 50 mM EdU medium and maintained under the same condition for 2 h. Thereafter, cells were fixed in 50 μL of 4% paraformaldehyde and stained in 100 μL of 1 × Apollo solution in dark condition at room temperature for 30 min, respectively. The nuclei were counterstained with DAPI reagent (Thermo Fisher Scientific). Finally, image observation was completed using a fluorescence microscope (Olympus, Japan).
Cell apoptosis
Cell apoptosis was tested by Annexin-V and propidium iodide (PI) staining (BD Biosciences, CA, USA). After treatment, A549 and H1299 cells were trypsinized and resuspended in 100 μL of binding buffer. Afterwards, 5 μL of Annexin-V-FITC and 5 μL of PI were respectively added and incubated with cells at room temperature for 15 min in dark condition. Thereafter, 400 μL of Annexin-V binding buffer was provided, and then flow cytometry analysis was conducted with a flow cytometer (Life Science, USA).
Determination of the intracellular iron level
The intracellular iron level was detected via an iron assay kit (Abcam, MA, USA) following the product instructions. After appropriate treatment, cells were harvested, homogenized in iron assay buffer on i.e. and centrifuged at 13,000 × g for 10 min at 4°C. Then, the supernatant was collected. Each 100 μL of the supernatant samples were mixed and incubated with 5 μL of assay buffer at 37°C for 30 min. After that, 100 μL of iron probe was supplied to each sample and incubated at 37°C for 60 min in dark condition. Absorbance of each well was measured with a microplate reader at 593 nm (Bio-Rad).
Measurement of the intracellular ROS level
The level of intracellular ROS was tested by ROS assay kit (Nanjing Jiancheng Bioengineering Institute, China). After appropriate treatment, A549 and H1299 cells were harvested and rinsed with pre-cold PBS for 3 times. Afterwards, cells were incubated with the corresponding culture medium replenished with 10 μM DCFH-DA (Invitrogen, USA) for 30 min under 37°C and dark conditions. After centrifuging at 1000 × g for 5 min at 4°C, the supernatant was discarded, and cells were resuspended in PBS. Finally, the results were observed and photographed by a fluorescence microscope (Olympus, Japan). Quantification of cellular fluorescence was performed using the ImageJ software (Bio-Rad).
Western blot
Expression of cell proliferation, apoptosis and ferroptosis-associated proteins was measured using western blot assay. After appropriate treatment, the cellular proteins of H1299 and A549 cells were extracted with BeyoLytic™ Mammalian Active Protein Extraction Reagent (Beyotime, Shanghai, China). Protein concentration was assessed by BCA Protein Assay Kit (Beyotime). After that, the interested proteins were separated by SDS-PAGE, transferred onto the PVDF membranes and blocked in 5% BSA at room temperature for 1 h, respectively. Then, incubations of the interested proteins and the corresponding primary antibodies were conducted at 4°C overnight. The primary antibodies included anti-Ki-67 (1:5000, ab92742, Abcam, UK), cleaved caspase 3 (1:500, ab32042, Abcam), SLC7A11 (1:5000, ab175186, Abcam), GPX4 (1:5000, ab125066, Abcam) and β-actin (1:4000, ab8227, Abcam). After rinsing with Tris Buffered Saline Tween (TBST), the interested protein and HRP-conjugated secondary antibody Goat Anti-Rabbit IgG H&L (HRP) (1:5000, ab6721, Abcam) incubation was performed at room temperature for 40 min. Then, the signals were developed using enhanced chemiluminescence (ECL) reagent. Results were observed and photographed using a LAS-3000 Imager (FUJIFILM, Japan).
Tumor xenograft formation
The male BALB/c nude mice (10, aged 5–6 weeks) were purchased from the Laboratory Animal Center of Zhejiang University (Zhejiang, China) and were reared in a pathogen-free, humidity- and temperature-controlled house under 12 h light and 12 h dark cycle for 1 week. All animal experiments were in accordance with the guidelines of Hangzhou Cancer Hospital (SYXK (ZHE)2019-0034).
Xenograft model was established to explore the anti-cancer efficacy of Val. A549 cells (1 × 107) were subcutaneous injected into the right flank of nude mice. Animal treatment was performed referencing to an earlier investigation. 21 After the tumor reached about 100 mm3, the animals were randomized divided into two groups: control group and Val (10 mg/kg) treatment group. Val was dissolved in vehicle, which containing 10% DMSO, 50% PBS and 40% Kolliphor/ethylene glycol, and was intraperitoneally injected twice daily every third day per week for the treatment group. Meanwhile, the control group were treated with the same volume of vehicle. The length and width of the tumor were measured every 7 days to calculate the tumor volume using the formular listed below: tumor volume (mm3) = length × width 2 × 0.5. Besides, mice weight was also determined every 7 days. After 35 days, the mice were sacrificed for euthanasia by injecting 150 mg/kg of Pelltobarbitalum Natricum, and then the tumors were harvested and weighed. Tumor tissues were fixed in 4% paraformaldehyde (PFA) for further analysis.
Haematoxylin and eosin staining
Observation of the histopathological changes in the tumor tissues was carried out via using HE Staining Kit (Beyotime) following the product instructions. Briefly, the fixed tumor tissues were then dehydrated with gradient ethanol, embedded in paraffin and cut into serial 4 μm sections. After that, the sections were deparaffinized, hydrated, stained with hematoxylin for 5 min and eosin for 2 min. Then, the slides were dealt with gradient alcohol and xylene, and finally sealed with neutral balsam (Solarbio, Beijing, China).
Immunohistochemical staining
Identification of the expression of proliferation, apoptosis and ferroptosis-associated factors was completed via performing IHC staining. Briefly, after deparaffinization and antigen retrieval, the tumor tissue sections were incubated with the primary antibodies, including anti-Ki67 (ab92742, Abcam), anti-cleaved caspase 3 (9661S, Cell Signaling Technology, USA), anti-GPX4 (ab125066, Abcam) and anti-SLC7A11 (ab37185, Abcam), at 4°C overnight. After rinsing with TBS 0.025% Triton, HRP-conjugated secondary antibody incubation was performed at room temperature for 1 h. Afterwards, color was developed with diaminobenzidine (DAB) at room temperature for 10 min, and the nucleus were counterstained with hematoxylin. Finally, results were observed under a microscope. Image quantitative analysis was conducted with ImageJ software (National Institutes of Health, USA).
Statistical analysis
Each experiment has three replications. Data were shown as mean ± standard deviation (SD). Comparison between two groups were carried out with two-tailed unpaired Student’s t-test. Comparison between more than two groups were conducted using One-way ANOVA followed by Tukey’s post hoc test. Graphpad Prism software 8 (Graphpad software, Inc.). p < 0.05 indicates statistically significant.
Results
Val treatment repressed LC cell proliferation, while enhanced cell apoptosis
For determination of the appropriate working concentration of Val in LC cells, H1299 and A549 cells were dealt with various concentrations of Val for 48 h. Afterwards, cell viability was determined. Results shown in Figure 1(b) and (c) revealed that H1299 and A549 cell viability were markedly inhibited by Val treatment in a concentration dependent way (p < 0.05 or p < 0.01). Considering that the IC50 values of A549 and H1299 cells were respectively 10.23 μM and 8.65 μM, so we finally selected three most representative concentrations of Val (5 μM, 10 μM and 20 μM) to carry out following experiments. Then, cell proliferation and apoptosis of H1299 and A549 cells were also assessed. Data displayed in Figure 2(a) and (b)manifested that the proliferation of A549 and H1299 cells were both notably repressed, while the apoptosis of these two cell lines were markedly enhanced by 10 and 20 μM of Val treatment (p < 0.05 or p < 0.01). Although the proliferation and apoptosis of H1299 cells were distinctly suppressed or promoted by 5 μM of Val treatment, while the A549 cells were not significantly influenced (both p < 0.05). The variations of cell proliferation and apoptosis were verified by the decreased expression of Ki-67 and increased expression of cleaved caspase 3 (Figure 2(c), p < 0.05 or p < 0.01), which variation trend was consistent with data displayed in Figure 2(a) and (b). For instance, it showed that 20 μM of Val treatment resulted in significant decrease of Ki-67 expression (fold change = 0.21 and 0.16, both p < 0.01) and notable increase of cleaved caspase three expression (fold change = 3.04 and 3.26, both p < 0.01) both in A549 and H1299 cells relative to control group. Combination of these results demonstrated that Val treatment distinctly repressed growth, while enhanced apoptosis of lung cancer cells. Valtrate treatment repressed lung cancer cell proliferation, while induced cell apoptosis.
A549 and H1299 cells were exposed to various concentrations of Val (5, 10 and 20 μM) for 48 h. A. Cell proliferation of Val-treated A549 and H1299 cells were assessed by EdU assay. B. Cell apoptosis of Val-treated A549 and H1299 cells were evaluated by Annexin-V/PI staining. C. Expression of proliferation-associated Ki-67 and apoptosis-related cleaved caspase three was measured by western blot, and β-actin was set as internal reference. *p < 0.05, **p < 0.01. Each experiment has three replications.
Val treatment induced ferroptosis of LC cells
SLC7A11 and GPX4 were identified as crucial regulators in cancer cell ferroptosis.
8
Researchers have found that SLC7A11 and GPX4 could regulate ferroptosis both at transcriptional level and post transcriptional level.26–29 In this study, we found that the protein expression of SLC7A11 and GPX4 in A549 and H1299 cells was both distinctly declined by 10 and 20 μM of Val treatment (Figure 3(a), p < 0.05 or p < 0.01). SLC7A11 expression was significantly decreased by 20 μM of Val treatment both in A549 and H1299 cells (fold change = 0.22 and 0.14, both p < 0.01), as well as GPX4 expression (fold change = 0.21 and 0.17, both p < 0.01). Meanwhile, the expression of SLC7A11 and GPX4 in H1299 cells was also markedly lowered by 5 μM of Val treatment (both p < 0.05). Considering that Fe2+ is common indicator of ferroptosis progression, so we detected its content in lung cancer cells. Results displayed that the level of Fe2+ in H1299 cells were markedly improved by 5, 10 and 20 μM of Val treatment, while Fe2+ level in A549 cells was distinctly elevated by 10 and 20 μM of Val treatment (Figure 3(b), p < 0.05 or p < 0.01). It showed that Fe2+ levels in A549 and H1299 cells were both markedly improved by 20 μM of Val treatment (fold change = 4.55 and 4.91, both p < 0.01). For further confirmation of the efficacy of Val treatment on ferroptosis, co-incubations of Val and ferroptosis inducer erastin or inhibitor ferrostatin-1 were respectively conducted on H1299 and A549 cells. Results displayed that, compared with 20 μM of Val treatment group, erastin treatment remarkably augmented Fe2+ level (Figure 3(c), fold change = 1.5, p < 0.05), while ferrostatin-1 treatment markedly lessened Fe2+ level in A549 cells (fold change = 0.51, p < 0.05). Meanwhile, the same trends were also discovered in H1299 cells (fold change = 1.43 and 0.5, p < 0.05). These outcomes indicated the elevating ferroptosis in Val-treated H1299 and A549 cells. Additionally, the lipid ROS levels in Val-treated H1299 and A549 cells were also assessed. Data displayed that 10 and 20 μM of Val treatment resulted in significantly increased contents of ROS in A549 and H1299 cells (Figure 3(d), p < 0.05 or p < 0.01). Besides, 5 μM of Val treatment also elevated ROS level in H1299 cells (p < 0.05). Collectively, these results manifested that Val treatment induced ferroptosis in LC cells. Valtrate treatment induced ferroptosis of lung cancer cells.
A549 and H1299 cells were exposed to various concentrations of Val (5, 10 and 20 μM) for 48 h. A. Expression of ferroptosis-associated SLC7A11 and GPX4 was tested by western blot, and β-actin was set as internal reference. B and C. Intracellular Fe2+ contentwas tested by iron assay kit. D. ROS level was measured via ROS assay kit. *p < 0.05, **p < 0.01. Each experiment has three replications.
Val treatment repressed cell proliferation and induced ferroptosis of LC cells via reducing SLC7A11 expression
Considering that SLC7A11 is a critical regulator in cancer cell ferroptosis, and it was markedly repressed by Val treatment in this study, we performed SLC7A11 overexpression in following experiments to explore the potential regulating mechanism. Data displayed in Figure 4(a) demonstrated that SLC7A11 was successfully overexpressed by 3.18-fold in A549 cells and 3.09-fold in H1299 cells relative to vector group (both p < 0.01). Following experiments revealed that SLC7A11 overexpression notably counteracted 20 μM of Val treatment-triggered repressing effects on cell viability and enhancing effects on cell apoptosis (Figure 4(b) and (c), all p < 0.05). Besides, 20 μM of Val treatment-induced elevations of Fe2+ and ROS levels were also notably offset by SLC7A11 overexpression in A549 and H1299 cells, exhibiting as decreasing Fe2+ levels by 0.42 and 0.45-fold, while reducing ROS levels by 0.31 and 0.4-fold, respectively (Figure 4(d) and (e), all p < 0.05). Besides, the inhibiting effects on SLC7A11 and GPX4 expression triggered by 20 μM of Val treatment were also markedly reversed by SLC7A11 overexpression, showing that SLC7A11 expression was remarkably increased by 3.9 and 6.19-fold (Figure 4(f), both p < 0.05), while GPX4 was notably elevated by 3.69- and 4.64-fold relative to 20 μM of Val treatment group (both p < 0.05). These data hinted that Val treatment might have carried out its repressing roles in LC cell proliferation and inducing roles in ferroptosis via declining SLC7A11 expression. Valtrate treatment repressed cell proliferation and induced ferroptosis of lung cancer cells via reducing SLC7A11 expression.
A549 and H1299 cells were transfected with the overexpressing plasmids SLC7A11 and empty vector, respectively. Then, the transfected cells were exposed to 20 μM of Val for 48 h. A. SLC7A11 expression in Val-treated A549 and H1299 cells were assessed by western blot, and β-actin was set as internal reference. B and C. Cell viability and apoptosis of Val-treated A549 and H1299 cells were assessed by CCK-8 assay and Annexin-V/PI staining, respectively. D and E. The levels of intracellular Fe2+ and ROS were tested by iron assay kit and ROS assay kit, respectively. F. Expression of ferroptosis-related SLC7A11 and GPX4 in Val-treated A549 and H1299 cells were measured via western blot. β-actin was set as internal reference. *p < 0.05, **p < 0.01. Each experiment has three replications.
Val treatment suppressed tumor growth in vivo
In order to disclose the potential roles of Val treatment in tumor growth, we constructed a subcutaneous xenograft tumor model in nude mice. Determination of tumor volume at each time point disclosed that 10 mg/mL of Val treatment markedly decreased the generated tumor volume compared with control group (Figure 5(a), p < 0.01), while no significant influence on mice weight was observed (Figure 5(b)). Besides, 10 mg/mL of Val treatment remarkably decreased the generated tumor weight (Figure 5(c), p < 0.01). Western blot results showed that SLC7A11 and GPX4 expression was both distinctly decreased by 10 mg/mL of Val (fold change = 0.21 and 0.17, both p < 0.01). HE staining displayed that 10 mg/mL of Val notably diminished typical characteristics of tumor cells (Figure 5(e)). Besides, IHC analysis revealed that the expression of proliferation-associated Ki-67 and ferroptosis-related GPX4 and SLC7A11 was all markedly repressed by 10 mg/mL of Val (Figure 5(e), fold change = 0.31, 0.26 and 0.22, all p < 0.01), while the expression of apoptosis-associated cleaved caspase three was distinctly enhanced (fold change = 3.16, p < 0.01). These outcomes indicated that Val treatment impeded tumor growth in vivo. Valtrate treatment suppressed tumor growth in vivo.
Xenograft model was established by subcutaneous injecting A549 cells (1 × 107) into the right flank of nude mice. A. Tumor volume was calculated using the formular listed below: tumor volume (mm3) = length × width2 × 0.5. B and C. Mice weight and tumor weight were both weighed. D. Expression of ferroptosis-associated SLC7A11 and GPX4 was tested by western blot. β-actin was used as internal reference. E. Histological changes in the control and 10 mg/kg of Val-treated tumor tissues were assessed by HE staining. Expression of proliferation-, apoptosis- and ferroptosis-associated proteins (Ki-67, cleaved-caspase 3, SLC7A11 and GPX4) were tested by immunohistochemical method. F. The graphical abstract of this study. **p < 0.01. Each experiment has three replications.
Discussion
Val was discovered to exert anti-tumor efficacies in PC and BC21,24 But until now, there is still no report recorded its efficacy in LC. Herein, we preliminary explored its roles in LC both in vitro and in vivo. We discovered that H1299 and A549 cell proliferation and ferroptosis-associated SLC7A11 and GPX4 expression were obviously repressed by Val treatment, while cell apoptosis, and intracellular Fe2+ and ROS levels were markedly augmented. Besides, SLC7A11 overexpression distinctly counteracted Val treatment-induced effects. Meanwhile, results of tumor xenograft formation assay demonstrated that tumor volume and weight were both distinctly inhibited by Val treatment. Therefore, these outcomes indicated that Val treatment could have antagonized malignant growth and induced ferroptosis of LC cells by reducing SLC7A11 expression.
In recent years, emerging investigations have explored the efficacies of Val treatment on malignant tumors. For instance, Chen et al. 21 discovered that Val treatment remarkably enhanced PC cell apoptosis, while inhibited cell proliferation and reduced tumor volume that generated in nude mice . Besides, Tian et al. 24 recorded that Val treatment observably induced BC cell apoptosis by elevating the expression of apoptotic proteins, such as cleaved caspase 3 and 7 . Meanwhile, as one of the V. jatamansi derivatives, IVHD-Val also played anticancer roles in OC, exhibiting as inhibiting proliferation, inducing apoptosis of A2780 and OVCAR-3 cells and repressing growth of the generated xenograft tumors. 25 Additionally, as a new active fraction of V. jatamansi Jones, F3 observably inhibited BC cell proliferation, and enhanced apoptosis. 30 In this research, we found that Val treatment notably repressed the proliferation, while augmented the apoptosis of A549 and H1299 cells. Besides, 10 mg/mL of Val treatment also markedly decreased the generated tumor volume and weight in the established nude mice. Obviously, these outcomes were in high accordance with the aforementioned literature, which indicated that Val treatment effectively antagonized malignant growth of LC cells both in vitro and in vivo.
Accumulating literature have proved that ferroptosis exert crucial roles in tumor growth inhibition. 15 For instance, researchers discovered that nanomedicine repressed tumor proliferation in vivo via promoting ferroptosis. 14 Another study revealed that amentoflavone (AF) impeded glioma cell proliferation by inducing ferroptosis. 16 In this research, we found that Val treatment distinctly improved the level of intracellular Fe2+, a common indicator of ferroptosis progression, which indicated that Val might induce ferroptosis in H1299 and A549 cells. Combined with the aforementioned efficacy of Val treatment on LC cell growth, we deduced that Val treatment could have antagonized malignant growth of LC cells via inducing ferroptosis.
ROS accumulation is deemed as a sign of ferroptosis. A previous report proposed that erianin, a dibenzyl compound extracted from Dendrobium, triggered ferroptosis in LC cells along with ROS accumulation. 31 Another study revealed that ruscogenin treatment triggered ferroptosis and elevated ROS production in PC cells. 32 More importantly, Gu et al. declared that Val treatment augmented ROS accumulation in PC cells. 21 Interestingly, we reached similar results in this study, demonstrating that Val treatment remarkably facilitated ROS accumulation in H1299 and A549 cells. Combination of the aforementioned results and literature demonstrated that Val treatment could have induced ferroptosis accompanied by augmenting ROS accumulation in LC cells.
SLC7A11 and GPX4 are proved to be suppressors of ferroptosis, and repressing their expression could trigger ferroptosis.7,33 Besides, SLC7A11 was demonstrated to be involved in the progression and prognosis of non-small-cell lung cancer (NSCLC). 34 Investigators discovered that postnatal deletion of GPX4 resulted in adult lethality. 35 Conversely, SLC7A11 KO mice are viable with no obvious phenotype. 36 Since repressing SLC7A11 is probable to induce less toxicity in patients relative to repressing GPX4, so SLC7A11 might be a more prospective target for cancer treatment than GPX4. Therefore, we performed SLC7A11 overexpression in subsequent experiments to explore the underlying mechanisms by which Val carrying out its efficacy in LC cells. Previous investigators found that LINC00618 enhanced ferroptosis via promoting ROS accumulation and repressing SLC7A11 expression in human leukemia. 37 Besides, Dong et al. proposed that Tanshinone IIA (Tan IIA) repressed gastric cancer (GC) cell growth and triggered ferroptosis by reducing SLC7A11 expression. 38 Similar results were reached in curcumin-treated NSCLC cells. 39 In this research, we discovered that SLC7A11 overexpression markedly counteracted Val treatment-triggered repressing effects on tumor growth and inducing effects on ferroptosis, which were in high accordance with aforementioned literature. Collectively, these discoveries hinted that Val treatment might achieve its efficacy through reducing SLC7A11 expression.
In conclusion, these findings manifested that Val treatment could have antagonized malignant growth of LC cells and induced ferroptosis by declining SLC7A11 expression. This study explored the main regulating mechanisms of Val in LC treatment, but its specific regulating mechanisms still needs further investigation, which will be continued in future research.
Supplemental Material
Supplemental Material - Valtrate antagonizes malignant phenotypes of lung cancer cells by reducing SLC7A11
Supplemental Material for Valtrate antagonizes malignant phenotypes of lung cancer cells by reducing SLC7A11 by W Xu and H Yu in Human & Experimental Toxicology
Footnotes
Author contributions
Wei Xu performed experimental studies, data acquisition and analysis, as well as manuscript drafting. Huan Yu contributed to study concepts and design, manuscript editing and submission.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
The full photograph of western blot data was supplied in supplementary material.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
