Abstract
Background
Ferroptosis is a novel type of regulated cell death and targeting ferroptosis may be a potential treatment strategy for lung cancer. Ziyuglycoside II (ZYG II) has a significant inhibitory effect on the growth of lung cancer cells. However, the selective anti-tumor effect of the ZYG II against lung cancer has not been systemically studied.
Objectives
We combined ferrostatin-1 and erastin to explore the potential therapeutic mechanism of the ZYG II for lung adenocarcinoma.
Materials and Methods
A549 and H1299 cells were randomly divided into the control, ZYG II, ferroptosis inhibitor group (ZYG II+ ferrostatin-1), and erastin group (ZYG II+ erastin). Cell proliferation was detected using the CCK-8 method. Cell migration and invasion were evaluated using the Transwell assay. The protein expression levels of Glutathione Peroxidase 4 (GPX4), Solute Carrier Family 7 Member 11 (SLC7A11), and Transferrin receptor 1 (TFR1) were measured using western blotting.
Results
Compared with the control group, the cell proliferation, migration, and invasion abilities of the ZYG II group significantly decreased, the protein expression levels of GPX4 and SLC7A11 in the ZYG II group declined significantly, and the expression of TFR1 increased significantly (p < 0.05). After adding ferrostatin 1 (ZYG II+ Ferrostatin 1), the cell proliferation, migration, and invasion abilities of the inhibited cells were significantly increased, the expression of GPX4 and SLC7A11 increased significantly and the expression of TFR1 decreased significantly (p < 0.05). However, after adding the erastin (ZYG II+ erastin), the cell viability was further inhibited in A549, the expression levels of GPX4 and SLC7A11 were further inhibited and the expression of TFR1 was further increased (p < 0.05).
Conclusion
ZYG II significantly inhibited the survival rate, proliferation, migration, and invasion ability of A549 and H1299 cells, possibly by inducing ferroptosis.
Introduction
Lung cancer is the second most commonly diagnosed cancer and the leading cause of cancer-related deaths worldwide, with age-standardized morbidity and overall mortality rates are 0.224% and 0.18% globally (Sung et al., 2021). Researchers have recently found that ferroptosis is generally inhibited in lung cancer cells. Ferroptosis was first coined in 2012 as a form of iron-dependent regulated cell death; it is characterized by an increase in intracellular free iron and accumulation of lipid peroxide (Hirschhorn & Stockwell, 2019). The lung tissue is in an environment of high oxygen concentration compared to other tissues. This environment causes lung tumors to experience high oxidative pressure. lung cancer cells increased the antioxidant capacity of the cells by upregulating the expression of System XC– (a cystine/glutamate antiporter) (Ji et al., 2018b). To avoid the facilitation and reinforcement of ferroptosis in the conversion process, lung cancer cells have taken various measures to improve the induction threshold of ferroptosis to inhibit ferroptosis and promote the occurrence and development of lung cancer (Wu et al., 2020).
As an adjuvant treatment for cancer, Chinese herbal medicine replacement therapy has long been widely used not only in China, but worldwide, and has an excellent reputation because of its efficacy, availability, and lack of side effects. Sanguisorba officinalis L.SP. (SA), a member of the rosaceous family, is a widely prescribed Traditional Chinese Medicine herbal product, exerting various pharmacological effects, including anti-inflammation (Yasueda et al., 2020), anti-cancer (Liao et al., 2020), antibacterial (Zhu et al., 2020), anti-lipid peroxidation (Lenzi et al., 2019), liver-protection (Meng et al., 2020), hypoglycemia (Son et al., 2015), and anti-obesity (Ji et al., 2018a). The major bioactive ingredients of SA are triterpenoid saponins, tannins and flavonoids, One of the main bioactive components of SA is ziyuglycoside II (ZYG II) (Zhao et al., 2017). ZYG II can protect against cancers by inducing apoptosis of cells and stagnation of the cell cycle (G0/G1 phase and S phase) arrest and can also inhibit the vitality of proliferative tumors (Jang et al., 2018). Ferroptosis is widely considered the key factor affecting the occurrence and development of various cancers. The induction of ferroptosis in non-small cell lung cancer (NSCLC) indicates that it can control tumor progression both in vivo and in vitro (Zou et al., 2021).
Glutathione peroxidase 4 (GPX4), the upstream activity of system XC– and the subsequent generation of glutathione (GSH, the GPX4 cofactor) reduce complex hydroperoxide to the non-toxic lipid alcohols, interrupting the chain reaction of lipid peroxidation. Therefore, XC–/GSH/GPX4 may be the main target for the induction of ferroptosis (Angeli et al., 2014; Dixon et al., 2012; Yang et al., 2014). The systemic XC-/GPX4 pathway is the main mechanism of cellular anti-oxidation, and its dysfunction can lead to intracellular ROS accumulation and lipid peroxidation, leading to ferroptosis (Lu et al., 2020). Solute Carrier Family 7 Member 11 SLC7A11 adjusts the major transport activities of cystine and glutamate, which in GSH biosynthesis is a rate-determining step (Verrey et al., 2004). Transferrin receptor 1 (TFR1) is a specific marker of ferroptosis, mainly located on the Golgi apparatus and cell membrane (Feng et al., 2020) leading to the accumulation of iron in the I/R (Ding et al., 2011). TFR1 can mediate the transfer of ferritin from extracellular to intracellular (Yang & Stockwell, 2008). Thus, the protein expression levels of GPX4, SLC7A11, and TFR1 were selected in this study.
Materials and Methods
Materials and Reagents
Cell lines
Human lung adenocarcinoma cell lines (A549 and H1299) (A549: BNCC337696, H1299: BNCC100859, BeNa Culture Collection).
Drug Formulation
ZYG II (A0626, Chengdu Main Biological Technology Co., LTD): 20 mg of ZYG II was dissolved in Dimethyl Sulfoxide (DMSO) (0.5 mL) to prepare a mother liquor with a concentration of 40 mg/mL and then diluted according to the experimental setup.
Ferroptosis inhibitor ferrostatin 1 (HY100579, MCE): 5 mg ferrostatin was dissolved in 1.9059 mL DMSO to prepare a mother liquor with a concentration of 10 mM, and the mother liquor was diluted 200 times to obtain a 0.05 mM solution, which was then diluted 100 times to obtain a working concentration of 0.5 µM.
Ferroptosis agonist erastin (HY15763, MCE): erastin (1 mg) was dissolved in DMSO (0.1828 mL) to prepare a mother liquor at a concentration of 10 mM, which was then diluted to 1,000 times to obtain a working concentration of 10 µM.
Methods
Cell Culture and Grouping
Human lung adenocarcinoma cell lines (A549 and H1299) were cultured in Roswell Park Memorial Institute 1640 supplemented with 10% fetal bovine serum at 37°C in 5% CO2, 95% air, and 55% humidity. A549 and H1299 cells were divided into four groups according to the intervention plan: blank control group, ZYG II group, erastin group (ZYG II+ erastin), and ferroptosis inhibitor group (ZYG II+ ferrostatin-1).
Cell Viability Was Analyzed Using the CCK-8
According to the manufacturer’s protocol. Cells were seeded and cultured at a density of 7 × 103/well in 100 µL of the medium in 96-well microplates. After treatment for 24 h, 10 µL of CCK-8 (KGA317, KeyGEN BioTECH) reagent was added to each well, and the cells were cultured for 2 h. The absorbance was analyzed at 450 nm using a microplate reader, with wells without cells as blanks. Cell proliferation was expressed as absorbance.
Cell Scratch Test by the Transwell Assay
When the cell density reached more than 90%, a single scratch was made using a sterile 10 µL pipette tip, as perpendicular to the bottom as possible. After the scratch was completed, the cell culture solution was discarded and the cells were washed gently with PBS three times. The cells were then incubated in a Fetal Bovine Serum-free culture medium. Images of scratches were captured at 0, 24, and 48 h. The healing rate of the scratch was calculated based on the width of the scratch corresponding to the two time periods, 0–24 h or 0–48 h. Scratch healing rate (%) = (0 h scratch width – 24 h/48 h scratch width)/0 h scratch width × 100%.
Cell Invasive Capability Was Detected by the Transwell Assay
After the culture medium was removed from the wells, the cells were washed with PBS for 5 min, and then stained with 0.1% crystal violet for 1 h. As instructed by the supplier, after dyeing, the cells were wiped off in the inner chamber with a cotton swab, and the chamber was inverted on a glass slide to capture a picture. After pictures were taken, the staining solution was removed from the wells, and 1 mL of 33% acetic acid was added to each well to dissolve the staining solution in the cells, mixed well, and allowed to stand, then 200 µL per well was pipetted into a 96-well plate, and the absorbance was analyzed at 570 nm using a multifunctional microplate reader using wells with 200 µL of 33% acetic acid as blanks. Finally, the invaded cells were observed and counted.
Western Blot
Cells were collected and lysed in Radioimmunoprecipitation Assay lysis buffer and the protein concentration was assayed using a BCA Protein Assay Kit (Mishra et al., 2017). Equal amounts of protein (10 µg) were separated using Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis and transferred to Polyvinylidene Fluoride membranes. After blocking with 3% non-fat milk for 1 h, and incubated overnight at 4°C with primary antibodies against mouse monoclonal anti-GAPDH (TA-08, ZSGB-BIO, 1/2,000). After three washes with Tris-buffered saline with Tween 20, the membranes were incubated with the secondary antibodies goat anti-mouse IgG/HRP (H+L) (ZB-2305, ZSGB-BIO, 1/2,000); rabbit anti-GPX4 (14432-1-ap, proteintech, 1/1000); rabbit anti-SLC7A11 (26864-1-ap, proteintech, 1/1,000) for 2 h, which were washed three times with Tris-buffered saline with Tween 20 afterward, wet with luminescent liquid, and then placed in the specimen storage area of the ultra-high sensitivity chemiluminescence imaging system.
Statistical Analysis
All data exhibited the average ± Standard Error of Mean of three independent experiments. GraphPad Prism 7.00 software was used for data analysis. A One-way Analysis of Variance was employed for multiple group comparisons. Statistical significance was set at p < 0.05.
Results
ZYG II Suppresses Cell Viability in A549 and H1299 Cells
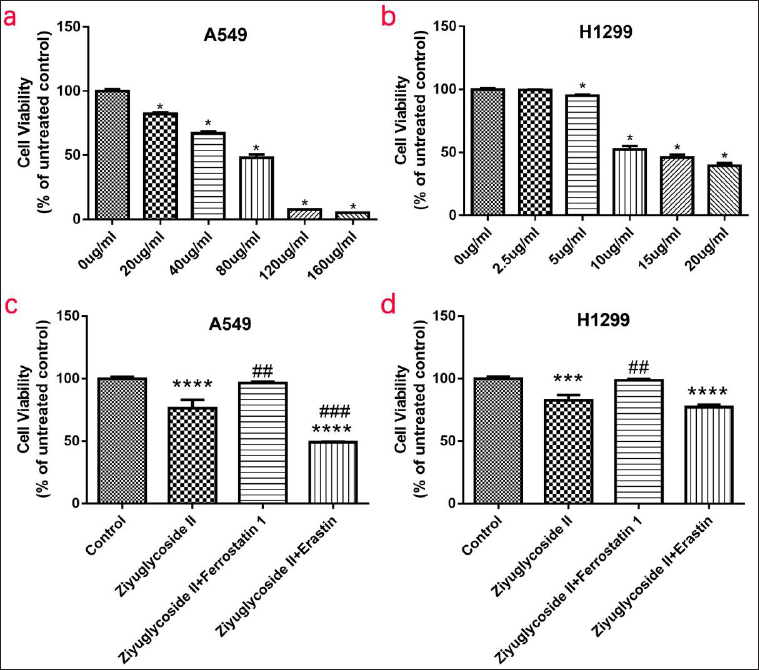
To investigate the effect of ZYG II on survival rate, A549, and H1299 cells were treated with different concentrations of ZYG II (A549: 0, 20, 40, 80, 120, and 160 µg/mL; H1299: 0, 2.5, 5, 10, 15, and 20 µg/mL) for 24 h. We found that the cell survival rate significantly decreased in the ZYG II group. The cell survival rate of A549 cells was approximately 80% in the ZYG II group (20 µg/mL) (Figure 1a), which was significantly lower than that in the control group. In addition, the survival rate of H1299 cells was 94% in the ZYG II group (5 µg/mL) (Figure 1b), which was significantly lower than that in the control group. The survival rates of H1299 cells were less than 50% in the ZYG II group (10 µg/mL). Therefore, the recommended intervention concentrations were as follows: A549 cells: 20 µg/mL, H1299 cells: 5 µg/mL. The viability of A549 (Figure 1c) and H1299 (Figure 1d) cells were significantly decreased in the ZYG II group compared with the control group (A549, p < 0.0001; H1299, p < 0.001), similar to the erastin group (A549, p < 0.0001; H1299, p < 0.0001). After adding ferroptosis inhibitor ferrostatin 1 (ZYG II+ Ferrostatin 1), the cell proliferation ability of the inhibited cells was significantly increased (A549, p < 0.001; H1299, p < 0.001). After adding the ferroptosis agonist erastin (ZYG II+ erastin), the cell viability was further inhibited in A549 (p < 0.001).
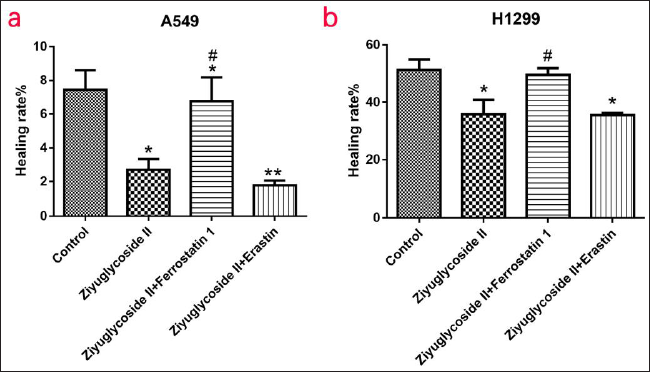
ZYG II attenuates the migration and invasion ability of A549 and H1299 cells.
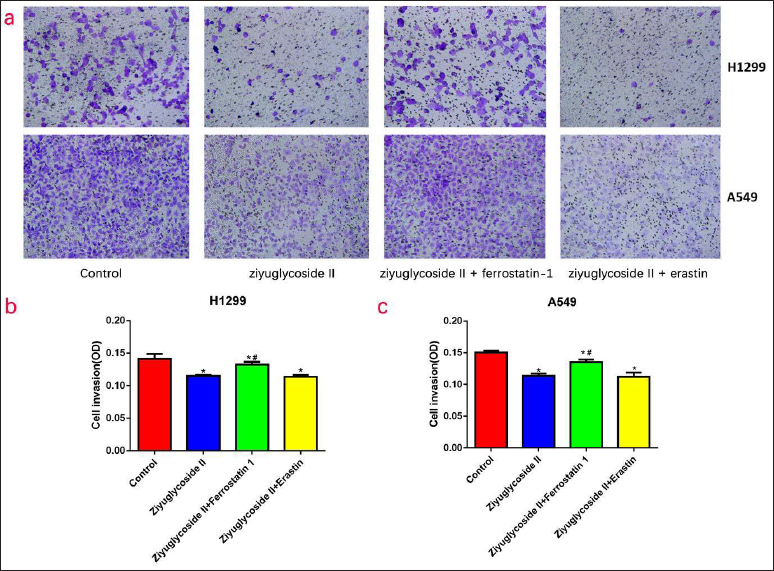
The migration ability of A549 cells (Figure 2a) and H1299 cells (Figure 2b) was significantly decreased in the ZYG II group compared with the control (A549, p < 0.05; H1299, p < 0.05), this was also observed in the ferroptosis inhibitor group (A549, p < 0.05) and the erastin group (A549, p < 0.01; H1299, p < 0.05). After the addition of ferrostatin 1, the cell migration ability of the inhibited cells was significantly increased (A549, p < 0.05; H1299, p < 0.05). Compared with the control group, the invasion ability of A549 cells (Figure 3c) and H1299 cells (Figure 3b) was significantly decreased in the ZYG II group (A549, p < 0.05; H1299, p < 0.05), and the ferroptosis inhibitor group (A549, p < 0.05; H1299, p < 0.05), which was the same in the ferroptosis agonist group (A549, p < 0.05; H1299, p < 0.05). After adding ferrostatin 1, the cell invasion ability of the inhibited cells was significantly increased (A549, p < 0.05; H1299, p < 0.05).
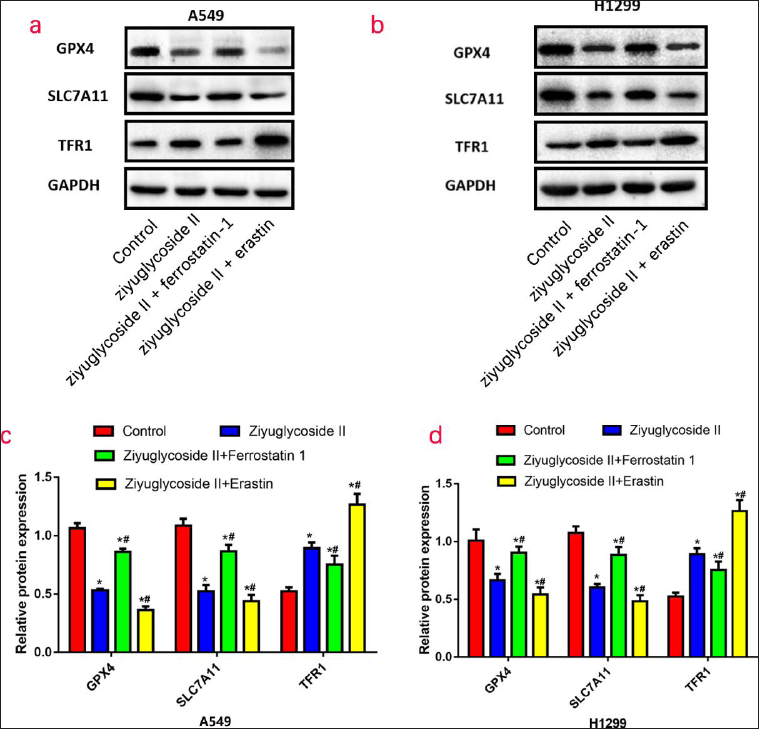
ZYG II suppresses the expression of ferroptosis-related proteins GPX4 and SLC7A11, but promotes the expression of TFR1 in A549 and H1299 cells.
In A549 cells (Figure 4c) and H1299 cells (Figure 4d), compared with the control group, the protein expression levels of GPX4 and SLC7A11 were significantly decreased (A549, p < 0.05; H1299, p < 0.05) and the TFR1 expression was increased (A549, p < 0.05; H1299, p < 0.05) in the ZYG II group. After the addition of ferrostatin 1, the expressions of GPX4 and SLC7A11 increased (A549, p < 0.05; H1299, p < 0.05) and the expression of TFR1 decreased (A549, p < 0.05; H1299, p < 0.05); on the contrary, after the addition of the agonist erastin, the expressions of GPX4 and SLC7A11 were further inhibited (A549, p < 0.05; H1299, p < 0.05), while TFR1 expression was further increased (A549, p < 0.05; H1299, p < 0.05).
Discussion
Ferroptosis, a novel form of non-apoptotic regulated cell death, was discovered through chemical screening (Dixon et al., 2012). GPX4 plays an important role in regulating classical ferroptosis by neutralizing lipid peroxides. Cysteine, taken up in its oxidized form as cystine by the system XC– cystine/glutamate antiporter (mainly including catalytic subunit SLC7A11) or generated by endogenous production via transsulfuration, is used for the biosynthesis of reduced GSH, which is then oxidized by nicotinamide adenine dinucleotide phosphate to promote GPX4 circulation (Yang et al., 2014). The absorption, consumption, storage, and turnover of iron all affect the sensitivity of ferroptosis. Transferrin (TRF) and TFR1 transport extracellular iron ions into cells. TFR has been identified as an inducer of ferroptosis and drives the internal metabolic pathway of cells, playing a vital role in the process of ferroptosis (Gao et al., 2015). Therefore, we explored the relationship between ZYG II and ferroptosis in lung cancer cells through the expression levels of the GPX4, SLC7A11, and TFR1 genes.
The iron dependence of cancer cells is significant in many cell death pathways, including ferroptosis, a form of iron-dependent cell death, and both iron accumulation and depletion are useful in anticancer therapy (Torti et al., 2018). Lung cancer cells directly target GSH by upregulating system XC– to increase cellular antioxidant capacity. SLC7A11 and SLC3A2 are centrally expressed in tumor cell lines, suggesting that the activity of system XC– may be higher in these cells (Huang et al., 2005; Ji et al., 2018b). Oligonucleotide microarray analysis revealed that among the nine lung cancer cell lines, SLC7A11 was highly expressed in A549, HOP-62, NCI-H226, NCI-H322M, and NCI-H460 cell lines, and the expression levels of SLC7A11 and SLC3A2 were the highest in A549 cell lines. Overexpression of GPX4 can also promote the proliferation of lung cancer cells and resist ferroptosis. Knocking down the expression of GPX4 with siRNA or inhibiting the activity of GPX4 with RSL3 can inhibit the proliferation, migration, and invasion activity of H1299, A549, and NCI-H460 cells, while ferrostatin-1, a ferroptosis inhibitor, can reverse the above phenomenon, (Lai et al., 2019) which means that inhibition of GPX4 can induce ferroptosis in lung cancer cells. The rapid growth and proliferation of cancer cells require large amounts of iron ions, leading to an increase in TRF receptors in malignancies such as lung, brain, breast, and colorectal cancers (Inoue et al., 1993). Elevated serum ferritin levels have been found in both NSCLC and small cell lung cancer (SCLC) patients, The expression of TFR1 was elevated in 88% of NSCLC patients (Kuang & Wang, 2019). In a case-control study of 200 patients, it was found that serum iron and ferritin levels in patients with lung cancer were significantly higher than those in healthy individuals, suggesting that the higher the iron content in the body, the higher the risk of lung cancer. The research data showed that the incidence of lung cancer was more than two times higher in people whose serum iron and ferritin content are abundant in the body, and ferritin was significantly positively correlated with the lung cancer stage (Sukiennicki et al., 2019). In the H1299 mouse xenograft model, it was verified that overexpression of TFR1 could accelerate the absorption of iron by lung cancer cells, promote tumor growth, and shorten the survival time of the mouse (Cai et al., 2016). Heat shock protein β-1 can reduce the concentration of intracellular iron ions by inhibiting the expression of TRF1. The inactivation of Heat shock protein β-1 contributes to the accumulation of iron ions to promote ferroptosis of tumor cells induced by erastin, a ferroptosis agonist (Sun et al., 2015). These researches show that researchers have used A549 and H1299 cell lines to explore the relationship between them and the expression levels of GPX4, SLC7A11, and TFR1. In addition, the cell culture cycle of A549 and H1299 cell lines is relatively short, which provides a favorable basis for our experimental material selection. In our experiments, we found that the expression levels of GPX4 and SLC7A11 in H1299 and A549 cells with the addition of ferrostatin 1 were significantly increased, and the expression of TFR1 was noticeably decreased. In addition, the activity, migration, and invasion ability apparently increased, while erastin may have contrasting effects, which further confirmed the existence of ferroptosis in NSCLC. Ferroptosis can affect the efficacy of chemotherapy, radiotherapy, and immunotherapy of cancers; therefore, combination therapy targeting ferroptosis signaling may improve outcomes (Chen et al., 2021). Some studies have shown that GPX4 is positively correlated with resistance to anticancer drugs, such as L-685458, palbociclib, topotecan, and lapatinib, which indicates that drugs targeting ferroptosis can overcome the drug resistance of tumor cells (Ni et al., 2021; Zhang et al., 2020). Ferroptosis inducers can inhibit the growth of drug-resistant tumor cells; for instance, they can be used for lung cancer cells that are resistant to epidermal growth factor receptor inhibitors (Zhang et al., 2021).
The molecular formula of ZYG II is C35H56O8, a triterpenoid saponin (Jang et al., 2018). Triterpene glycosides from the roots of SA play a crucial role in antitumor activity and have been reported to have significant cytotoxicity against various tumor cells, including BGC-823 (human gastric carcinoma cells), NCI-H460 (human large cell lung cancer cells), HepG2 (human hepatocellular carcinoma cells), MCF-7 cells (human breast cancer cells), SGC-7901 (human gastric adenocarcinoma cells), and human cervical cancer cells (HeLa) (Hu et al., 2015). Sanguisorba saponins can significantly inhibit the growth of tumor cells, and with an increase in the concentration of total S. saponins, the cell survival rate of human lung cancer cells (A549), HeLa, and human hepatocarcinoma cells (SK-HEP-1) gradually decrease (Wang et al., 2021). Gallic acid and ellagic acid, belonging to the S. saponins extract family, can activate the apoptotic pathways of mitochondria in cancer cells and inhibit the proliferation, vascularization, and migration of vascular endothelial cells. Moreover, S. saponins extract inhibited tumor growth and angiogenesis in vivo (Wang et al., 2012). S. saponins extract can also stimulate the immune system through the proliferation of spleen lymphocytes, increase the secretion of cytokines, and activate phagocytosis of macrophages to significantly inhibit the proliferation of cancer cells (Cai et al., 2012). To date, the relationship between S. saponins and ferroptosis has not been reported. However, in our study, it was demonstrated that ZYG II has a noticeable inhibitory effect on A549 and H1299 cells, and with the increase in the concentration of ZYG II, the inhibition of cell survival rate gradually increases. In addition, ZYG II cannot only inhibit the migration and invasion of lung cancer cells but also probably induce ferroptosis. The expression levels of GPX4 and SLC7A11 were significantly decreased, and TFR1 was noticeably increased in lung cancer cells treated with ZYG II, consistent with the antitumor effects of erastin. But in fact, we can better determine the experimental concentration of ZYG II by increasing the number of experiments and prolonging the experiment time to 48 h or even longer.
Conclusion
ZYG II can enhance the inhibitory effects on proliferation, invasion, and migration of lung adenocarcinoma cells, which may be related to the expression levels of GPX4, SLC7A11, and TFR1 during ferroptosis in lung adenocarcinoma cells. This study provides a new experimental foundation for further enriching the understanding of the antitumor effects of ZYG II.
Footnotes
Abbreviations
DMSO: Dimethyl sulfoxide; GPX4: Glutathione Peroxidase 4; GSH: Glutathione; NSCLC: Non-small cell lung cancer; SA: Sanguisorba officinalis L.; SLC7A11: Solute Carrier Family 7 Member 11; TFR1: Transferrin receptor 1; TRF: Transferrin; ZYG II: Ziyuglycoside II.
Authors’ Contributions
JHZ, LJX, and HLW contributed to the concept and writing-original draft. HC, XRM, and XL contributed to data curation and formal analysis. XL contributed to the analysis and interpretation of data. XFL and LSL contributed to the investigation and methodology. WS, JHZ, HLW, and LJX contributed to the visualization, resource, and project administration. LJX, LYH, TC, JWL, and XC contributed to funding acquisition, conceptualization, supervision, writing, reviewing, and editing. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
Not applicable.
Funding
This work was supported by grants from, the Fujian Province Natural Science Fund Project (2023J011846), the Fujian Provincial Youth Scientific Program on Health (2020QNA079), the Outstanding Youth Project of Fujian Provincial Hospital (2014YNQN31), and the Special Research Foundation of Fujian Provincial Department of Finance, China (2021-848, 2022-840), and National famous and old Chinese medicine experts (Xue-mei Zhang, Xiao-hua Yan) inheritance studio construction project, China.
