Abstract
Background
Melanoma is a highly invasive and metastatic malignant tumor, with pathogenesis involving multiple signaling pathways and transcription factors. Current treatments face significant challenges, including drug resistance, toxic side effects, and recurrence.
Objectives
This review aims to evaluate the therapeutic potential of natural products in the treatment of melanoma and to identify the associated challenges and future research opportunities.
Methodology
A comprehensive literature search was conducted using PubMed and Web of Science to identify studies reporting on natural products with anti-melanoma activity.
Results
Natural products demonstrate diverse pharmacological activities, including antiproliferative, proapoptotic, and antimetastatic effects, highlighting their potential in melanoma treatment. However, challenges such as limited efficacy, safety concerns, and the lack of standardized formulations remain significant barriers.
Conclusion
This review provides insights into the therapeutic applications of natural products for melanoma, discusses existing limitations, and proposes future directions, contributing to the development of effective and safe anti-melanoma treatments.
Introduction
Melanoma, a malignant skin cancer arising from melanocytes, is highly invasive and metastatic, making it a leading cause of skin cancer-related deaths (Hyeraci et al., 2023; Switzer et al., 2022). Factors such as global climate change, increased ultraviolet (UV) radiation, and an aging population have led to rising melanoma incidence and mortality, posing a significant public health threat (Fadadu & Wei, 2022; Flynn et al., 2023). Current treatments include surgery (van Akkooi et al., 2022), radiation (Trappetti et al., 2022), chemotherapy (Tamura et al., 2022), immunotherapy (Huang & Zappasodi, 2022), and targeted therapy (Rager et al., 2022; Yang et al., 2024). However, due to melanoma’s biological traits and resistance, these treatments have limited efficacy, significant side effects, and high costs, highlighting the need for safer, more effective, and affordable alternatives (Xing et al., 2017).
Melanoma pathogenesis involves dysregulation of multiple signaling pathways and transcription factors, disrupting melanocyte proliferation, differentiation, survival, and metastasis. The wingless-type MMTV integration site (WNT)/β-catenin pathway plays a key role in melanoma initiation and progression (Lüönd et al., 2022; Wang, Zhang, et al., 2022; Wang, Zhao, et al., 2022; Wu et al., 2022) (Figure 1). This conserved signaling system regulates cell fate, proliferation, migration, polarity, and self-renewal. When WNT proteins bind to cell membrane receptors like frizzled receptors (FZD) and lipoprotein receptor-related protein (LRP), they inhibit glycogen synthase kinase (GSK)-3β-mediated β-catenin phosphorylation, preventing degradation. Accumulated β-catenin translocate to the nucleus, binds T-cell factor (TCF)/lymphoid enhancer-binding factor (LEF) transcription factors, and activates downstream genes such as c-Myc, cyclin D1 (Ccnd1), and axis inhibitor 2 (AXIN2), promoting cell cycle progression and proliferation. Additionally, the pathway’s role differs between early-stage and metastatic melanoma, where it is less active in the former and upregulated in the latter, complicating the therapeutic outcomes of natural products.
Signal Pathways in Melanoma.
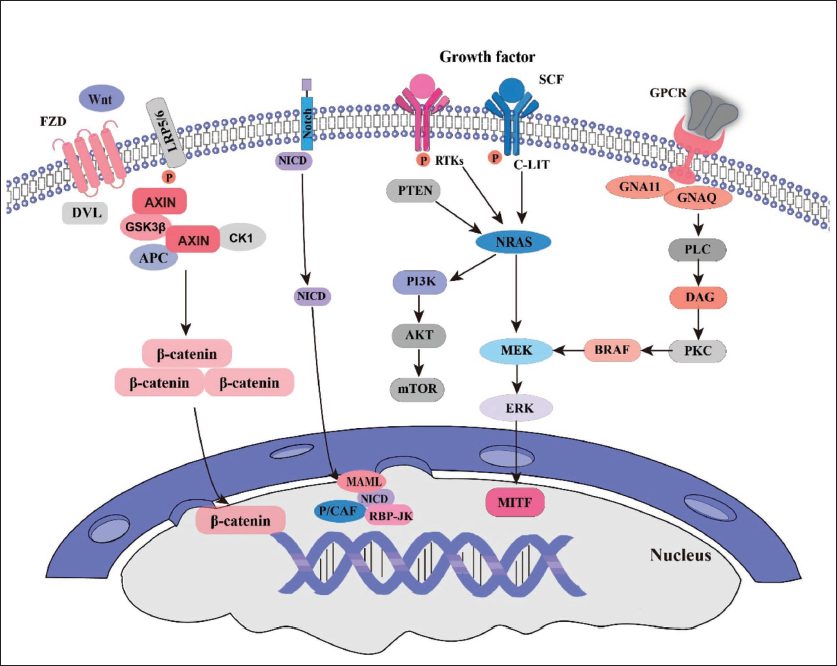
The expression and activity of the WNT/β-catenin pathway in melanoma are complex and vary across subtypes, molecular variants, and clinical stages, affecting the efficacy of natural products (Xue et al., 2016). For instance, in desmoplastic melanoma, WNT/β-catenin activity is lower compared to cutaneous melanoma, influencing responses to therapies targeting this pathway. Mutations in β-catenin or upstream regulators can also alter pathway sensitivity to treatments disrupting β-catenin-TCF/LEF interactions (Saikia et al., 2024; Yarza et al., 2023). Additionally, the pathway’s role differs between early-stage and metastatic melanoma, where it is less active in the former and upregulated in the latter, complicating the therapeutic outcomes of natural products (Duan & Bonewald, 2016; Vuong & Mlodzik, 2024).
The microphthalmia-associated transcription factor (MITF, Figure 1) is a key regulator in melanocytes, controlling melanin production, cell growth, and survival. It influences DICER for melanocyte survival and cyclin-dependent kinases (CDK)2 for cell growth, with its function affected by v-raf murine sarcoma viral oncogene homolog (BRAF) and neuroblastoma RAS viral (v-ras) oncogene homolog (NRAS) mutations (Chauhan et al., 2022; Gelmi et al., 2022). Notch1 promotes melanoma progression through β-catenin upregulation, while β-catenin suppression reverses this effect. Notch signaling, via RBP-J, directly enhances MITF-mediated gene transcription but competes with MITF upon activation, reducing MITF activity and contributing to melanoma invasiveness (Rigg et al., 2023). While β-catenin regulates MITF, melanoma cells use WNT/β-catenin for transformation and noncanonical pathways for metastasis. MITF activity is further modulated by transcriptional inhibitors like sex-determining region Y-box 10 (SOX10) and BRN2, making it a potential target for melanoma therapy (Capparelli et al., 2022; Simmons et al., 2022).
The interaction between MITF and Notch signaling is crucial in understanding melanoma progression. These pathways influence each other, forming a regulatory network that drives tumor growth (Hartman & Czyz, 2015). While WNT/β-catenin regulates MITF for proliferation, noncanonical signals facilitate metastasis, meaning β-catenin is not strictly an oncogene (Phelps et al., 2022). Targeting both MITF and Notch pathways simultaneously shows promise, as combined inhibition more effectively suppresses melanoma growth and metastasis than targeting each individually (Shinzawa et al., 2023). This approach may help overcome resistance seen with single-pathway treatments, offering a more comprehensive strategy for melanoma therapy (Pan et al., 2023). This approach may help overcome resistance seen with single-pathway treatments, offering a more comprehensive strategy for melanoma therapy (Luo et al., 2023).
Melanoma pathogenesis involves multiple signaling pathways, such as WNT/β-catenin, Notch, MITF, and SOX10, offering potential therapeutic targets. However, current treatments face limitations like drug resistance, side effects, and recurrence, highlighting the need for safer, more effective therapies. Traditional Chinese medicine and herbal remedies, with their multitarget and synergistic effects, offer promising alternatives. The following sections will explore pharmacological studies on their anti-melanoma potential and mechanisms.
Inhibition of Tyrosinase Activity Pathway
Green Tea
Green tea is widely used in food, cosmetics, and medicine due to its rich functional ingredients. The primary bioactive polyphenols, including epigallocatechin-3-gallate (EGCG) (Almatroodi et al., 2020; Zhang et al., 2021), gallocatechin-3-gallate (GCG) (Sato et al., 2022), epicatechin-3-gallate (ECG) (Chen et al., 2020; Tian et al., 2021), epigallocatechin gallate (EGC) (Zhan et al., 2020), and epicatechin (EC) (Farhan, 2022), are the main bioactive polyphenols in green tea and also known for their health-promoting effects. Studies show that catechins (ECG, EGCG, and GCG) in green tea inhibit tyrosinase activity, significantly reducing melanin synthesis in B16F10 melanoma cells by downregulating MITF expression through the cAMP signaling pathway (Zhang et al., 2020). Their low cytotoxicity and strong anti-melanoma effects suggest potential therapeutic value. Li et al. (2019) found that tea polyphenols, EGCG, and lanthanide metal ions (Sm3+) form nano complexes that activate caspase-3/7 in melanocytes, leading to poly ADP-ribose polymerase (PARP) cleavage and apoptosis, while effectively inhibiting wound-induced melanoma cell migration. In mouse melanoma models, these nanocomplexes demonstrated superior tumor inhibition compared to the anticancer drug 5-fluorouracil. Catechins exhibit varying inhibitory effects on melanoma cells depending on concentration and cell cycle stage. Future research integrating genomics, proteomics, and metabolomics will further elucidate how catechins regulate specific gene expressions and metabolic pathways, offering new insights for anti-melanoma therapies.
Polygonum cuspidatum
Traditional Chinese Medicine, specifically P. cuspidatum, is known for dispelling pathogenic wind and removing dampness. Its main components, such as polygonin and vanicosides A and B (Ahmad et al., 2022; Kim, Wang, et al., 2019), exhibit strong antimelanin activity. Jeong et al. (2010) extracted 5,4′-dihydroxystilbene-3-O-β-
The active components in green tea and P. cuspidatum have tyrosinase activity and melanin synthesis, but current research mainly focuses on in vitro studies, lacking in vivo data. Future studies should evaluate the metabolism and bioavailability of these compounds to determine their clinical efficacy. Additionally, the selective inhibitory effects on different melanoma types require further investigation. Integrating multiomics technologies can deepen our understanding of these mechanisms and lay the foundation for novel anti-melanoma therapies. Overall, these components show significant potential in anti-melanoma treatment, but more rigorous studies are needed to validate their clinical applicability.
Inducing Apoptosis
Curcumae longae Rhizoma
Curcumin was a kind of Traditional Chinese Medicine with the same origin as medicine and food, which had the effect of breaking blood and promoting qi and menses (Giordano & Tommonaro, 2019). In recent years, curcumin extracted from rhizomes has gradually shown antitumor effects (Mansouri et al., 2020; Passos et al., 2023). Tang and Cao (2022) stated that curcumin extracted from turmeric could inhibit the expression of SOX10, Notch1, and HIS-1, increase the expression of miR-222-3p, and ultimately inactivate the Notch pathway. The Notch pathway is a crucial pathway in the development of melanoma associated with traditional Chinese medicine, and its inhibition can reduce the proliferation, migration, and invasion of melanoma to some extent. The study by Szlasa et al. (2020) indicated that curcumin could serve as an effective photosensitizer. After curcumin photodynamic therapy (PDT), cancer cells lost their motility, resulting in deoxyribonucleic acid (DNA) breaks, increased expression of caspase-3, and actin cytoskeleton recombination. Curcumin-mediated PDT has proven to be a promising treatment for malignant melanoma. Another major component of turmeric, β-elemene, also demonstrated good anti-melanoma activity. Balavandi et al. (2020) found that β-elemene significantly reduced the viability of A375 melanoma cells and induced cell apoptosis. Combination therapy with β-elemene and radiation resulted in an increased apoptosis rate. These studies suggest that turmeric can be used as a potential drug for treating melanoma. Mechanisms by which curcumin and β-elemene exert their effects at different stages of the cell cycle and their interactions with other signaling pathways need further exploration. Integrating multiomics approaches could provide deeper insights into their anti-melanoma mechanisms and help identify potential biomarkers for treatment efficacy. Overall, while turmeric components hold significant potential for melanoma therapy, more rigorous studies are needed to validate their clinical applicability.
Arnebiae radix
A. radix, a common Chinese medicine, is widely recognized for its significant anti-inflammatory properties and effectiveness in promoting wound healing, particularly in skin diseases (Zhu et al., 2022). Research by Lee et al. (2021) demonstrated the anticancer effects of shikonin, an active compound derived from A. radix, using xenograft models and various assays, including MTT and annexin V/propidium iodide staining. Their findings indicated that shikonin inhibits the proliferation of human melanoma cells by inducing apoptosis via the mitogen-activated protein kinase (MAPK) pathway. Furthermore, Durchschein’s team (Durchschein et al., 2018) extracted and synthesized shikonin derivatives from Onosma paniculata, revealing their ability to cause double-stranded DNA breaks through histone 2A variant (H2AX) phosphorylation, along with increased PARP protein levels leading to caspase-induced cell apoptosis. Other studies indicated that shikonin triggered A375 cell apoptosis and autophagy by activating ROS-mediated endoplasmic reticulum stress and the p38 pathway, significantly upregulating P21 independent of P53 status while inhibiting growth-related genes (Liu et al., 2019; Wang et al., 2020).
These findings underscore the potential of A. radix and its components, particularly shikonin and its derivatives, in melanoma therapy. However, most current research is confined to in vitro studies, limiting their clinical application. Future investigations should focus on the pharmacokinetics and bioavailability of these compounds through animal models and clinical trials to assess their effectiveness and safety. Additionally, elucidating the mechanisms by which shikonin and its derivatives induce apoptosis and autophagy at different cell cycle stages is crucial. Employing multiomics approaches could enhance our understanding of these mechanisms and aid in identifying therapeutic targets and biomarkers. In summary, while the components of A. radix show promising potential in melanoma treatment, further in vivo studies and clinical trials are essential to validate their efficacy and safety for clinical use.
Coptidis rhizoma
Berberine, a key compound found in C. rhizoma, has garnered significant research interest due to its cytotoxic effects on various cancer cells and its role in preventing metastasis (Salek et al., 2022). Ren et al. (2020) demonstrated that berberine enhances the expression of miRNAs related to cell cycling while degrading their target genes, effectively blocking the cell cycle, inhibiting the growth and migration of cutaneous melanoma A375 cells, and promoting apoptosis. Additionally, Wang et al. (2021) explored berberine in chemo-PDT, finding that its combination with PDT significantly enhanced the effectiveness of cisplatin against cisplatin-resistant melanoma by activating the ROS/p38/caspase cascade. Despite these promising findings, there are substantial gaps in understanding the mechanisms of berberine, particularly its effects on the immune microenvironment and interactions with other targeted therapies. Future research should aim to explore berberine’s potential in multitarget therapies, especially in combination with immunotherapy and chemotherapy, to assess possible synergistic effects.
Ginseng radix et Rhizoma
Ginseng, a common Chinese medicine, is recognized for its vitality-boosting effects and contains various saponins with significant pharmacological activities, including immune regulation and antitumor effects (Kwon et al., 2023; Zhou et al., 2023). Kim et al. (2019) demonstrated that ginsenoside Rg3 inhibits the growth and migration of the A375.S2 melanoma cell line by inducing apoptosis. Meng et al. (2019) confirmed Rg3’s ability to suppress melanoma growth and angiogenesis by reducing matrix metalloproteinase (MMP)-2, MMP-9, and vascular endothelial growth factor (VEGF) expression. Wu et al. (2019) found that Siberian ginseng gold nanoparticles (SG-GNPs) induce melanoma cell apoptosis through ROS generation. Research has also focused on ginseng polysaccharides. Xie et al. (2023) isolated polysaccharides that exhibited anti-melanoma effects and induced tumor apoptosis, suggesting their potential as therapeutic agents. However, most studies emphasize individual components without examining their interactions. Future research should investigate the combined effects of ginseng’s active components and strategies to enhance their bioavailability and therapeutic efficacy.
Euphorbiae Ebracteolatae Radix
Euphorbiae Ebracteolatae Radix, a toxic antitumor drug, exhibits pharmacological activities in antiviral, anti-inflammatory, and antitumor domains, offering new avenues for anti-melanoma research (Yang et al., 2021). Si et al. (2020) isolated chamaejasmin B (CHB) from Stellera chamaejasme L., demonstrating that CHB induces G0-G1 cell cycle arrest in B16F0 cells by downregulating cyclin-dependent kinase 4 (Cdk4), Ccnd1, and proliferating cell nuclear antigen (Pcna), while upregulating p21. Additionally, CHB induces apoptosis through the mitochondrial pathway and inhibits the growth of B16F0 and B16F10 tumors in vivo. Xie et al. (2021) extracted 44 substances from Euphorbia fischeriana, all of which exhibited moderate cytotoxicity against the A375 melanoma cell line. However, despite the promising findings, current research lacks comprehensive insight into the full spectrum of molecular mechanisms underlying the anti-melanoma effects of these compounds. For instance, how these compounds might modulate the tumor microenvironment or interact with specific immune pathways remains unclear. Future studies should explore the combination of these compounds with other targeted therapies, such as immune checkpoint inhibitors, to better understand potential synergistic effects and broaden their therapeutic applications.
Rabdosiae rubescentis Herba
Rabdosia rabdosa was a plant in the Labiaceae family, which had the effect of clearing heat and detoxifying. Oridonin (Li et al., 2021), a tetracyclic diterpenoid compound, had the effect of inhibiting the proliferation of various tumor cells (Abdullah et al., 2021; Liu et al., 2021). Hua et al. (2021) treated choroidal melanoma cell lines MUM-2B and C918 cells with different concentrations of TNF-related apoptosis-inducing ligand (TRAIL) and laminarin, respectively, and found that the apoptosis rate of melanoma was significantly higher when TRAIL and laminarin acted on MUM-2B cells at the same time than when the two drugs acted alone. Moreover, the inhibitory effect on MUM-2B cells was stronger than that on C918 cells. While these findings are encouraging, they highlight the need to further explore the underlying mechanisms by which R. rubescentis herba compounds, such as oridonin, exert their anti-melanoma effects. Specifically, future research should focus on the interactions between oridonin and key apoptotic pathways, such as the TRAIL pathway, and the potential for its use in combination therapies to overcome resistance to existing treatments. Moreover, further in vivo studies are needed to validate these effects and assess their safety and efficacy in clinical settings.
Inhibition of Angiogenesis
Grewia tenax
G. tenax (Forssk.) Fiori, found in the Arabian Peninsula, Africa, and Southeast Asia, is utilized in various ethnopharmacological systems for conditions like jaundice and anemia (Mahunu et al., 2020; Rehman et al., 2022). AlQathama et al. (2023) demonstrated that the hexane extract of G. tenax significantly inhibits tumor progression in melanoma mice, primarily due to the compounds lupinol and β-sitosterol. Lupinol, a triterpenoid found in various Chinese herbs, has garnered attention for its antitumor properties, including inhibiting cell proliferation, inducing apoptosis, and reducing invasion and metastasis in multiple cancers. Bhattacharyya et al. (2019) reported that lupinol inhibits angiogenic mimicry and angiogenesis in melanoma models by altering cancer stem cells and endothelial progenitor cell populations. Additionally, β-sitosterol, a plant sterol with antioxidant and anti-inflammatory activities, has shown promise in anti-melanoma research by inhibiting tumor cell proliferation and inducing apoptosis (Bociort et al., 2021). Studies by Sundstrøm et al. (2019) confirmed that β-sitosterol reduces mitochondrial respiratory capacity by inhibiting complex I, which hinders melanoma cell growth and reduces brain metastasis formation. The exploration of β-sitosterol’s mechanisms provides a foundation for adjuvant therapy and combination treatments against melanoma.
Cinnamomi Cortex
Cinnamon was a kind of Chinese medicine to invigorate the spleen and replenishing qi, and was used as a spice in ancient times. The antityrosinase (Siew et al., 2022) and antioxidation (Lee et al., 2020) activities of various components of cinnamon made cinnamon another object of anti-melanoma research. Ullah et al. (2019) obtained a series of derivatives from cinnamic acid in Chinese herbal medicine cinnamon by structurally modifying it. By acting on α-melanocytotropic hormone (α-MSH)-stimulated B16F10 melanoma cells, they found four cinnamamides with significant antimelanin formation and tyrosine kinase inhibition activity in a dose-dependent manner. Patra et al. (2019) found that cinnamaldehyde, another important component of cinnamon, could inhibit angiogenesis and metastasis of B16F10 melanoma cells by inhibiting hypoxia-inducible factor-1α (HIF-1α) protein accumulation, and this process might be related to targeted inhibition of the PI3/Akt/mTOR pathway. Cappelli et al. (2023) extracted essential oil (CINN-EO) from Cinnamomum zeylanicum and evaluated its effects on the human metastatic melanoma cell line M14. CINN-EO exhibited cytotoxicity associated with increased ROS and Fe(II) levels, leading to reversible accumulation of cells in the G2/M phase of the cell cycle. By inducing an incomplete stress response, CINN-EO may enhance the antitumor effects of traditional drugs, positioning it as a candidate for combination therapy. While these natural compounds demonstrate potential in inhibiting angiogenesis and limiting melanoma progression, further studies are needed to understand their mechanisms fully. Future research should investigate the molecular pathways involved, explore synergistic effects with existing therapies, and conduct in vivo studies to establish safety and efficacy.
Regulating Immune Response
Astragalus (Salehi et al., 2021), a common tonic Chinese medicinal material, had the effect of invigorating the spleen and replenishing qi. In recent years, research on Astragalus has gradually shifted from antioxidation (Cui et al., 2022; Hou et al., 2023) and antiaging (Yan et al., 2023) to antitumor (Li et al., 2020; Wang, Cui, et al., 2022) research. A variety of antitumor drug components in Astragalus were found. The studies of Hwang et al. (2021) demonstrated that Astragalus polysaccharide (APS) enhances the anticancer effects of immune checkpoint inhibitors. Intranasal treatment with APS increased lineage−CD11c+ dendritic cells (DCs) in mesenteric lymph nodes (mLN) by upregulating CC-chemokine receptor 7 expression, thereby enhancing the activity of antiprogrammed death ligand 1 (PD-L1) monoclonal antibodies against pulmonary metastatic melanoma.
Ding et al. (2021) found that APS reduces the immunosuppressive activity of myeloid-derived suppressor cells (MDSCs) in melanoma mice by remodeling intestinal microbiota and fecal metabolites, revealing its therapeutic potential in tumor growth control. Gong et al. (2022) confirmed that APS downregulates PD-L1 and influences the phosphoinositide 3-kinase (PI3K)/protein kinase B (AKT) signaling pathway, indicating its potential as an adjuvant in chemotherapy. Additionally, various compound preparations containing Astragalus have attracted research interest. Wang, Bai, et al. (2022) showed that a mixture of Astragalus propinquus and Pinellia pedatisecta inhibits the PI3K/Akt signaling pathway, suppressing melanoma cell proliferation and inducing apoptosis. Zhang et al. (2022) found that combining Astragalus with trichosanthin effectively inhibits melanoma growth in mice. SH003 [a mixture of natural products derived from Astragalus membranaceus, Angelica gigas, and Trichosanthes kirilowii, and formononetin (FMN)] prepared by Han et al. (2023) were found to exert anti-melanoma effects through the PD-1/PD-L1 pathway.
Although studies indicate that APS enhance antitumor effects by regulating C-C motif chemokine receptor 7 (CCR7) and the PI3K/AKT signaling pathway, the specific mechanisms remain unclear. Future research should explore how Astragalus components affect interactions between immune and tumor cells, especially under immunosuppressive conditions. Additionally, compound preparations like A. propinquus Schischkin and P. pedatisecta Schott (A&P) and SH003 show promise against melanoma, yet their mechanisms are not well understood. Further investigation into the pharmacological actions of these compounds is essential, including their effectiveness in various therapeutic contexts and potential drug interactions and side effects, to better understand their clinical applications.
Other Potential Treatment Methods
Cannabis sativa
C. sativa was a cannabis plant of the mulberry family. The clinical application of this traditional Chinese medicine was limited due to its apparent addiction and withdrawal symptoms during the medication period. At present, studies have proved that C. sativa extract has obvious therapeutic value in many fields, such as anti-inflammatory (Cavalheiro et al., 2022), neuroprotection (Barbalace et al., 2023; Zagožen et al., 2021), and anticancer (Koltai & Shalev, 2022; Mukosi & Motadi, 2023). Schanknecht et al. (2023) and Simmerman et al. (2019) injected cannabidiol (CBD), which is one of the C. sativa extracts, intraperitoneally into the B16F10 melanoma model established under the skin of C57BL/6 mice. The results showed that CBD significantly reduced the growth rate of melanoma and prolonged the survival time of melanoma mice. Although mice treated with cisplatin showed the longest survival, quality of life and energy were likely better observed in mice treated with CBD. More studies (Schanknecht et al., 2023) have shown that the upregulation of cyclooxygenase (COX) pathway-related genes leads to the increase of prostaglandin E2, which is related to the increase of mutation and invasion of melanoma cells in vitro, while cannabis-specific allyl flavonoid compounds have been shown to directly inhibit the COX pathway, which undoubtedly provides a new target for anti-melanoma research. Despite concerns about addiction and withdrawal symptoms related to C. sativa, research highlights its therapeutic potential in anti-inflammatory, neuroprotective, and anticancer applications. Future studies should focus on the specific mechanisms by which C. sativa extracts, especially CBD, exert anti-melanoma effects. Simmerman et al. (2019) found that intraperitoneal CBD injection significantly reduced the growth rate of B16F10 melanoma models and prolonged the survival of melanoma mice. While cisplatin-treated mice showed the longest survival, those treated with CBD demonstrated better quality of life and energy levels. CBD may enhance patients’ quality of life, though its survival benefits might not match those of traditional chemotherapy. Moreover, upregulation of COX pathway-related genes increases prostaglandin E2 levels, linked to heightened mutation and invasion in melanoma cells. Certain allyl flavonoids from cannabis have been shown to directly inhibit the COX pathway, offering a new target for anti-melanoma research. Future studies should investigate the molecular mechanisms of these cannabis components and assess their potential synergistic effects with existing treatments.
Vassobia breviflora
Altevir Rossato Viana’s team verified the cytotoxicity against melanoma by studying the pharmacological activity of water extract (Viana et al., 2023), acetone extract (Rossato Viana, Godoy Noro, et al., 2023), and hexane extract (Rossato Viana, Nicola, et al., 2023) of V. breviflora, which is a Solanaceae plant. Hexane extract showed cytotoxicity in B16F10 melanoma cells and could increase the death frequency of melanoma cells by enhancing ROS production. The water extract of V. breviflora shows antioxidant effects. While only the highest concentration induced genotoxicity in piezoelectric composite based micromachined ultrasound transducer (pCMUT), lower concentrations reduced ATP release and enhanced P2X7 receptor activation in B16F10 melanoma cells, suggesting potential as a new drug target for melanoma through purinergic receptors. Our findings position V. breviflora as a promising candidate for treatment. Additionally, the acetone extract exhibited activity in the A375 melanoma cell line at 0.1 mg/mL, offering a fresh perspective on using Breviflorum japonicum extracts in cancer therapy.
The study found that the hexane extract of V. breviflora exhibited cytotoxicity in B16F10 melanoma cells, increasing cell death by enhancing ROS production. The water extract demonstrated antioxidant effects. Although only the highest concentration of V. breviflora initiated genotoxicity in pCMUT, lower concentrations decreased ATP release and enhanced P2X7 receptor activation in mouse melanoma B16F10 cells, suggesting that this natural compound may activate ADP/ATP analogs, possibly through purinergic P2X7 and P2Y1 receptors, which could serve as new drug targets for melanoma. While these findings provide preliminary evidence of V. breviflora’s anti-melanoma potential, its specific mechanisms require further exploration.
Paeoniae radix
Peony exhibits significant pharmacological activity against melanoma, with research focusing on the extraction and antitumor mechanisms of Paeonia sinensis (Baek et al., 2022; Wang, Li, et al., 2022). Xiang et al. (2020) found that Paeoniflorin inhibits gene transcription by suppressing the nuclear factor kappa-B (NF-κB) pathway and upregulating E-cadherin, thus reducing cell invasion and migration. It also induces apoptosis via death receptor and mitochondrial pathways. Wen et al. (2023) demonstrated that Paeoniflorin inhibits α-MSH-induced cytopigmentation in B16F10 melanoma cells by blocking cAMP-response element binding protein (CREB) activation and reducing TRP-1, TRP-2, and MITF expression. These findings contribute to understanding the mechanisms of Traditional Chinese Medicine in melanoma treatment.
Sarracenia purpurea
The purple carnivorous pitcher plant S. purpurea is a medicinal plant used by Canadian First Nations people to treat a wide variety of illnesses (Huang et al., 2022; Kannan et al., 2020). Due to its longstanding ethnomedicinal uses, the extracts of S. purpurea are safe as pharmaceuticals and are expected to have few side effects for human use. Liu et al. (2022) found that the acetone extract of S. purpurea exhibits significant anticancer potential and inhibits the Staphylococcus aureus SsbA protein. Key compounds in the extract include driman-8,11-diol (18.8%), deoxysericealactone (15.89%), and stigmast-5-en-3-ol (12.17%), which show inhibitory effects on melanoma cells. Studies have shown that purpurea was found to be able to promote the distribution of the G2 phase and decrease the cell proportion in the G1 and S phases in a concentration-dependent manner in B16F10 melanoma cells. Apocynin, a key component, exhibited antityrosine kinase activity in studies by Ashooriha et al. (2020), inhibiting melanin production without cytotoxicity. Further research into the specific mechanisms of S. purpurea’s components on melanoma could better assess its potential in cancer treatment.
Discussion
Melanoma is a highly invasive and metastatic tumor with rising global incidence, particularly among individuals of European descent. Its pathogenesis involves complex interactions of signaling pathways and transcription factors, including WNT/β-catenin, Notch, MITF, and SOX10, which regulate melanocyte proliferation, differentiation, survival, and metastasis.
This manuscript presents findings on 15 natural compounds demonstrating anti-melanoma activity, categorized into five pharmacological groups: tyrosinase inhibition, apoptosis induction, antiangiogenesis, immune regulation, and synergistic effects. Our experimental data provide insights into their mechanisms of action. We also discuss the historical use of these natural products in treating melanoma and other cancers, offering valuable context for modern research and highlighting the potential for innovative therapies. Our study identifies specific natural products that modulate the WNT/β-catenin and Notch pathways, leading to reduced melanoma cell proliferation, enhanced apoptosis, and inhibited metastasis. However, the variability in purity and stability of natural products limits their efficacy and safety. Standardized formulations are needed for reliable therapeutic outcomes. Future research should also explore these compounds’ precise molecular interactions and pharmacokinetics. The integration of genomic, proteomic, and metabolomic technologies could facilitate the discovery of additional natural products and novel targets. Combining high-throughput screening with advanced bioinformatics may lead to personalized treatment approaches for melanoma. Additionally, combining natural products with existing chemotherapeutic agents might improve efficacy and address drug resistance, pending validation through clinical trials.
In summary, while this research highlights the potential of natural products for melanoma treatment, future efforts should focus on refining formulations, elucidating molecular mechanisms, and exploring combination therapies to improve patient outcomes. These efforts will contribute to advancing melanoma treatment and guide future research in this area.
Abbreviations
α-MSH: α-Melanocytotropic hormone; A&P: Astragalus propinquus Schischkin and Pinellia pedatisecta Schott; AXIN2: Axis inhibitor 2; BRAF: v-raf murine sarcoma viral oncogene homolog; Ccnd1: Cyclin D1; CCR7: C-C motif chemokine receptor 7; CDK: Cyclin-dependent kinases; Cdk4: Cyclin-dependent kinase 4; COX: Cyclooxygenase; CREB: cAMP-response element binding protein; EC: Epicatechin; ECG: Epicatechin-3-gallate; EGC: Epigallocatechin gallate; EGCG: Epigallocatechin-3-gallate; FMN: Formononetin; FZD: Frizzled receptors; GCG: Gallocatechin-3-gallate; GLUT1: Glucose transporter type 1; GP-a: Acidic pectin; GP-c: Crude polysaccharide; GP-n: Neutral polysaccharide; GSK: Glycogen synthase kinase; H2AX: Histone 2A variant; HIF-1α: Hypoxia-inducible factor-1α; LRP: Lipoprotein receptor-related protein; MAPK: Mitogen-activated protein kinase; MDSC: Myeleloid-derived suppressor cells; MITF: Microphthalmia-associated transcription factor; mLN: Mesenteric lymph nodes; MMP: Matrix metalloproteinase; NF-κB: Nuclear factor kappa-B; NICD: Notch intracellular domain; NRAS: Neuroblastoma RAS viral (v-ras) oncogene homolog; PARP: Poly ADP-ribose polymerase; pCMUT: Piezoelectric composite based micromachined ultrasound transducer; Pcna: Proliferating cell nuclear antigen; PD-L1: Programmed death ligand 1; PDT: Photodynamic therapy; PI: Propidium iodide; PI3K/AKT: Phosphoinositide 3-kinase/protein kinase B; RBP-JK: Recombination signal binding protein for immunoglobulin kappa J region; ROS: Reactive oxygen species; SG-GNPs: Siberian ginseng gold nanoparticle; SOX10: Sex-determining region Y-box 10; TCF/LEF: T-cell factor/lymphoid enhancer-binding factor; TRAIL: TNF-related apoptosis-inducing ligand; VEGF: Vascular endothelial growth factor; WNT: Wingless-type MMTV integration site; WZB117: 2-Fluoro-6-(m-hydroxybenzoyloxy) phenyl m-hydroxybenzoate.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by the Key Laboratory of Green Chemistry of Sichuan Institutes of Higher Education (LYJ2202); Key Laboratory of Exploitation and Study of Distinctive Plants in Education Department of Sichuan Province (TSZW2023ZB-09, TSZW2104, TSZW2023ZB-12); Key Laboratory of Low-cost Rural Environmental Treatment Technology at Sichuan University of Arts and Science (XCH2023ZB-10).
