Abstract
Background
Autophagy has always been a current research hotspot during lung cancer treatment.
Objectives
This study aimed to explore the molecular mechanism by which cucurbitacins loaded in liposome nanoparticles (Cu B-NPs) inhibit the development of lung cancer through Beclin 1 (BECN1) regulation.
Materials and Methods
Cu B-NPs were prepared, and the A549 group, Cu B group, and Cu B-NPs group were set up to assess the mechanism of Cu B-NPs in regulating autophagy and inhibiting the development of lung cancer. Cu B-NPs+si-BECN1 group and Cu B-NPs+pc-BECN1 group were also set up to analyze whether Cu B-NPs regulate autophagy through BECN1 to inhibit the development of lung cancer.
Results
Cu B-NPs were successfully prepared and had good dispersion stability. Cu B-NPs reduced cell viability, promoted apoptosis and the accumulation of autophagosomes, and regulated apoptosis-related proteins, thereby inhibiting lung cancer development. After activating/inhibiting BECN1 expression, Cu B-NPs promoted cell autophagy and apoptosis by upregulating BECN1 expression, thereby inhibiting the development of lung cancer.
Conclusion
In this study, Cu B-NPs were successfully prepared, and the particles promoted autophagy and induced apoptosis in A549 cells by upregulating BECN1, thereby inhibiting the development of lung cancer. This provides strong evidence and clues for further research and development of lung cancer treatment strategies.
Introduction
Although some progress has been made in the diagnosis and treatment of lung cancer in the past few decades, early-stage lung cancer usually has no obvious symptoms (Neugebauer et al., 2022; Zeng et al., 2024a) and is therefore often ignored and not discovered until it progresses to an advanced stage (Schabath & Cote, 2019). Moreover, the survival rate for advanced lung cancer is relatively low because the tumor may have spread to other sites. In addition, drugs for the treatment of lung cancer have major side effects, so it is particularly critical to find more effective and safe drugs and treatment methods. Autophagy is generally considered to have an important impact on the growth and survival of tumor cells (D’Arcy, 2019). Studies have shown (Qian & Yang, 2016; Xu et al., 2023) that autophagy helps maintain the health of cells by clearing damaged organelles and proteins, thereby playing an important role in gastric cancer cells. Abnormal regulation of autophagy is related to lung cancer. BECN1/Beclin 1 is closely related to autophagy (Kang et al., 2011), and it plays a key role in regulating the autophagy process. Autophagy can remove damaged organelles and cellular components and reduce the accumulation of harmful molecules in cells, thereby reducing damage to the genetic stability of cells and reducing the incidence of mutations. It can even reduce the presentation, system identification, and attack potential of antigens, thereby reducing the immunity of tumor cells (Cheng et al., 2024; Valivand et al., 2024). In some breast cancers, the expression of BECN1 is reduced, thereby reducing the occurrence of autophagy. After restoring the expression of BECN1, cellular autophagy is promoted, leading to the death of breast cancer cells (Aglan et al., 2024; Yan et al., 2024). Another study found (Tedesco et al., 2024) that the deletion of BECN1 was associated with the progression and recurrence of colorectal cancer. Therefore, regulating the expression of BECN1 may help improve the therapeutic effect of colorectal cancer. Meanwhile, BECN1 expression is related to lung cancer cell autophagy. In some cases, restoring the expression of BECN1 can promote lung cancer cell autophagy, thereby increasing the sensitivity of chemotherapy (Pagani et al., 2024).
Cucurbitacin (cucurbitacin B, Cu B) has been found to have anticancer potential in many studies (Li et al., 2024). Studies have shown (Peri et al., 2023) that Cu B can inhibit breast cancer cell growth, induce apoptosis, and interfere with cell migration and invasion. Other studies have shown (Hheidari et al., 2024) that Cu B can inhibit lung cancer cells and reduce the risk of tumors spreading to other parts. In addition, Cu B can also affect Wnt/β-catenin signaling to mediate spread and metastasis of liver cancer cells. However, its mechanism of action on lung cancer is still unclear. Whether it regulates autophagy through BECN1 to affect lung cancer requires further study. In addition, although Cu B is effective in treating cancer, it has many limitations in treating diseases due to low bioavailability and obvious toxic and side effects. Therefore, it is very important to find methods that can improve its shortcomings. Liposome nanoparticles are drug delivery systems commonly used to carry and deliver biologically active substances. It can effectively encapsulate drugs, stabilize them, and increase their bioavailability, thereby improving drug delivery (Zeng et al., 2024b). Studies have shown (Haciosmanoglu Aldogan et al., 2024) that liposome nanoparticles can not only achieve directional delivery of drugs through surface modification, allowing drugs to act more accurately on target tissues or cells, but can also reduce the toxicity of certain drugs and improve drug efficacy. Therefore, this study uses cucurbitacins loaded in liposome nanoparticles (Cu B-NPs) to treat lung cancer, aiming to explore the mechanism by which it regulates autophagy in gastric cancer cells through BECN1 and thereby inhibits the development of lung cancer.
Materials and Methods
Experimental Materials
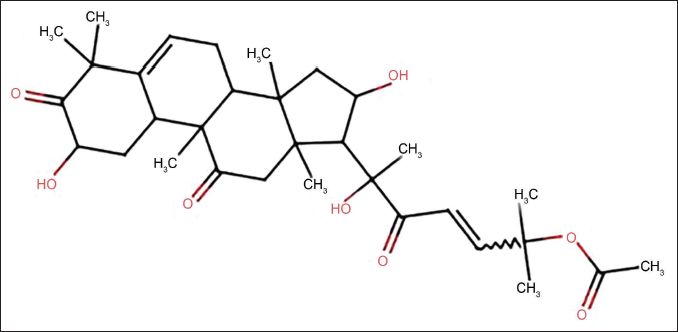
A549 cells (Shenzhen Haodihuatuo); Cu B (specification: 5 mg, chemical formula: CHO7; molecular structure shown in Figure 1), Hoechst staining kit (Shanghai Yuanye); BECN1 antibody (Wuhan Yipu); LC3-II/I (Wuhan Boote); p62 antibody (Guangzhou Bohui).
Molecular Structural Formula of Cucurbitacin.
Preparation and Characterization of Cu B-NPs
Preparation of Cu B-NPs
By mixing liposome components such as phospholipids and cholesterol in a certain proportion, and then adding an appropriate amount of methanol, a liposome solution was formed. Dissolve Cu B in an appropriate amount of methanol to form a Cu B solution. Then, the Cu B solution was added dropwise to the liposome solution while stirring thoroughly. The Cu B and liposome solution were uniformly mixed to prepare liposome nanoparticles, and then the solvent was removed by rotary evaporation to obtain a dry Cu B-NPs complex.
Characterization of Cu B-NPs
Dip a small amount of the nanoparticle dispersion liquid and evenly drop it on the conductive adhesive. After natural drying, spray gold for 1 min, and then observe the morphology of the nanoparticles with transmission electron microscopy (TEM).
Cell Culture
The A549 cells were cultured in RPMI 1640 medium at 37°C with a CO2 concentration of 5% until the cells grew well. This group of cells was labeled as the A549 group and used for subsequent experiments. Cu B was added to the A549 cells to make it fully contact with the cells, and set it as the Cu B group. Cu B-NPs were added to the A549 cell culture medium and incubated, set as the Cu B-NPs group, and analyzed the oxidative effects of Cu B-NPs on 549 cells. In addition, a Cu B-NPs+si-BECN1 group and a Cu B-NPs+pc-BECN1 group were set up to analyze whether Cu B-NPs regulate autophagy through BECN1 to inhibit the development of lung cancer.
Detection of Survival Rate of A549 Cells by 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyl-2H-Tetrazolium Bromide (MTT) Method
A549 cells were cultured in a culture medium until the cells grew well, and then cells were selected for subsequent experiments. After the cells were trypsinized into single-cell suspension, they were distributed into culture dishes to allow the cells to grow under different intervention effects. Add the MTT solution to the culture dish and incubate together for about 4 h. Remove the MTT solution, dissolve the purple product with a dissolving agent, immediately transfer the solution to a 96-well plate, and measure absorbance.
Hoechst Staining to Detect Apoptosis of A549 Cells
48 h after drug treatment, after all the culture medium was absorbed, fix with 0.5 mL fixing solution for 10 min, then rinse twice with phosphate buffered saline (PBS), add 0.5 mL Hoechst staining solution to the sample, stain for about 5 min, and then rinse it twice with PBS solution for 3 min each time, then add fluorescence quenching mounting medium and cover with a cover glass, and finally observe the sample under a fluorescence microscope.
Polymerase Chain Reaction (PCR) Detection of BECN1 Expression
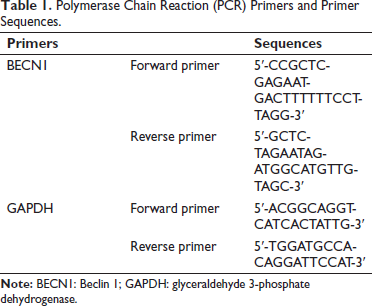
Lung cancer tissue was ground and homogenized using liquid nitrogen, total messenger ribonucleic acid (mRNA) was extracted, and reverse transcribed into complementary deoxyribonucleic acid (cDNA) using TaqManTM advanced miRNA cDNA synthesis tool. Real-time analysis was performed using a PCR system, and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was selected as the internal reference during the analysis process. Relative levels were estimated using the 2−∇∇Ct method. Table 1 lists the primer sequences (Shanghai Unico).
Polymerase Chain Reaction (PCR) Primers and Primer Sequences.
Western Blot Detection of Autophagy-related Protein Expression
Protein was extracted and quantified. The electrophoresed total protein was heated to 100°C and incubated for 5 min, then electrophoresed using an SDS-polyacrylamide gel (120 V, 100 min). The separated proteins were transferred to polyvinylidene difluoride (PVDF) membrane, which was blocked and incubated with rabbit antimouse BECN1, LC3-II, LC3-I, autophagy marker protein P62, and GAPDH overnight at 4°C (concentration 1:5,000). The next day, the samples were incubated, and the membrane washing step was performed. This process was repeated three times. They were incubated with secondary antibodies (concentration 1:10,000) for 1 h at 37°C.
Plasmid Transfection to Observe Autophagosomes
The next day, perform experiments on well-growing A549 cells according to the reagent instructions. Add Lipofectamine 2000 transfection reagent and GFP-LC3 plasmid to serum-free RPMI-1640 medium respectively, let it stand at room temperature for 5 min, and then mix. Let it sit for another 20 min before adding them to the cells to disperse evenly.
Conditions: Experiment at 37°C and 5% CO2 concentration. The cells were incubated in the incubator for 4 h, then the medium changed. After another 4 h, the cells were removed for further study and then the cells were cultured for 48 h. Then take out the A549 cells from the culture medium, wash them three times, and observe the fluorescence changes of GFP-LC3 (concentration 1:5,000) under a fluorescence microscope. If there is autophagy, there is an accumulation of green dots in the cells, which means that the cells have undergone autophagy. If there is diffusion, the green fluorescence without aggregated dots indicates that there is no autophagy.
Statistical Analysis
The data were analyzed using SPSS26.0 and GraphPad Prism software. If no special requirements exist, p < 0.05 is used as the test standard.
Results
Morphology and Characterization of Cu B-NPs
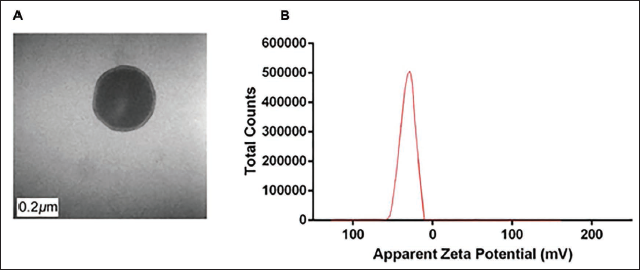
The prepared Cu B-NPs had an irregular spherical shape, an average particle size of (129.000 ± 0.030) nm, and a low degree of aggregation (Figure 2A). The Zeta value is −28.9 mV, indicating that Cu B-NPs have better dispersion stability (Figure 2B).
Morphology and Characterization of Cucurbitacins Loaded in Liposome Nanoparticles (Cu B-NPs) Composites. (A) Transmission Electron Microscopy (TEM) image of Cu B-NPs (Magnification ×30,000). (B) Zeta Potential Distribution of Cu B-NPs.
Cu B-NPs Promote Cell Autophagy and Inhibit the Development of Lung Cancer
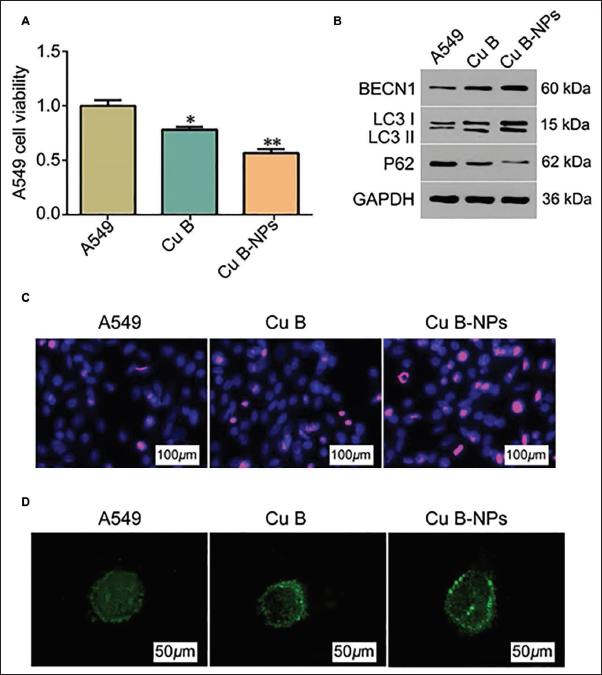
To explore the anticancer effect of Cu B-NPs, we used it to culture with A549. We found that under Cu B intervention conditions, cell viability decreased (Figure 3A), suggesting that Cu B has a specific anti-A549 effect. Further observation displayed that the intervention effect of NPs was the most prominent (vs. Cu B group, Figure 3A). In the apoptosis staining results, this group showed abnormal nuclear condensation and A549 cells shrank significantly (Figure 3C), further illustrating the tumor suppressor effect of Cu B-NPs. In addition, through the observation of autophagosomes, we found that under the intervention of Cu B-NPs, intracellular autophagosomes were highly aggregated and distributed in a dot-like manner (Figure 3D). LC3-I and LC3-II expression were both higher, while P62 showed a lower expression (Figure 3B). BECN1 was highly expressed, suggesting that anticancer effect of Cu B-NPs is related to BECN1. Compared with the Cu B group, * p < 0.05, ** p < 0.01.
Cucurbitacins Loaded in Liposome Nanoparticles (Cu B-NPs) Can Promote Cell Autophagy and Inhibit the Development of Lung Cancer (n = 5). (A) Viability of A549 Cells in Three Groups; (B) Western Blot to Detect the Expression of P62, LC3-I, and LC3-II Ratios; (C) Hoechst Staining to Detect Apoptosis of A549 Cells in Three Groups (×40); (D) The Production of Autophagosomes in A549 Cells in Three Groups; Compared with Cu B Group, *p < 0.05, ** p < 0.01.
Cu B-NPs Promote Cell Autophagy and Apoptosis and Inhibit the Development of Lung Cancer by Upregulating BECN1 Expression
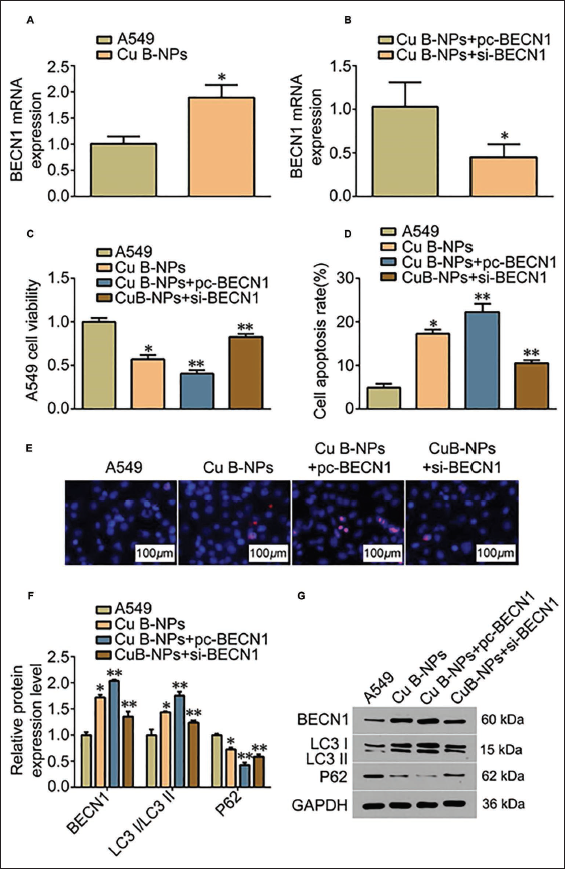
We further tested the expression of BECN1 mRNA and found that under the intervention of Cu B-NPs, its expression was abnormally high (Figure 4A), reflecting that Cu B-NPs can mediate the increase in BECN1 mRNA levels. After activating this gene, the cell viability of A549 was lost (Figure 4C). The apoptosis rate also increased sharply (Figure 4D). During this process, we found that the cell nuclei were highly condensed in Hoechst staining (Figure 4E), indicating that pc-BECN1 can promote cell apoptosis and phagocytosis amplification (vs. Cu B-NPs group). Silencing BECN1 significantly reversed this phenomenon (Figure 4C–D). The levels of apoptosis-related proteins, such as LC3-I and LC3-II, also changed accordingly (Figure 4E–F). Compared with the Cu B group, * p < 0.05, ** p < 0.01.
Cucurbitacins Loaded in Liposome Nanoparticles (Cu B-NPs) Promote Autophagy and Apoptosis and Inhibit the Development of Lung Cancer by Upregulating Beclin 1 (BECN1) Expression (n = 5). (A) Polymerase Chain Reaction (PCR) Detection of BECN1 Expression; (B) PCR Detection of BECN1 Expression in Two Groups after Inhibition/Activation of BECN1; (C) A549 Cell Viability of Four Groups by MTT; (D) A549 Cell Apoptosis by Flow Cytometry; (E) Hoechst Staining of Four Groups of A549 Cells (×40); (F) Autophagosome Production of A549 Cells in Four Groups; (G) Western Blot Detection of BECN1, LC3-II/I Ratio and Expression of Autophagy Marker Protein P62; Compared with Cu B Group, * p < 0.05, ** p < 0.01.
Discussion
Cucurbitacin is a natural product substance that has been shown to have potential antitumor activity (Liang et al., 2019). Cucurbitacin B can inhibit JAK2/STAT3 signaling to a certain extent and downregulate the expression of IRAK1, thereby inhibiting the biological processes of A549 cells and lung cancer cells. Moreover, cucurbitacin B changes the distribution and composition of MDA-MB-231 and SKBR-3 cytoskeleton and downregulates Rac1, CDC42, and RhoA. GTPases Rac1/CDC42/RhoA signaling can reduce levels of F-actin, FAK, and so on, thus achieving the effect of actively treating breast cancer to a certain extent (Zarezadeh et al., 2024). Furthermore, Cu B can also affect cell cycle regulatory proteins such as p21 and p27, thereby inhibiting uncontrolled cell proliferation. In addition, Cu B can also downregulate Bax, cyclin D1, and so on to reduce the blood supply of tumors, thereby preventing abnormal tumor growth. These mechanisms are often intertwined and act synergistically through multiple pathways. At the same time, the effect of cucurbitacin may vary due to factors such as its concentration, mode of use, and cellular environment (Jia et al., 2021; Mao et al., 2019). Due to the low bioavailability and targeting of cucurbitacins, to improve its shortcomings, researchers are developing various delivery systems such as nanoparticles, liposomes, and nanoemulsions to improve the distribution and targeting of drug in body. Therefore, in this study, lipid nanobodies were used as the drug-carrying system to successfully prepare Cu B-NPs.
To clarify the anticancer effect of Cu B-NPs, we found that Cu B inhibited viability of A549 cells, which indicates that Cu B-NPs may interfere with the survival and proliferation of cells in some way. In the experimental results, the cell shrinkage and abnormal condensation of cell nuclei further implied that Cu B-NPs had an impact on cell structure and morphology. By observing autophagosomes, we found that they promoted cell autophagy. With the intervention of Cu B-NPs, the results showed consistent changes and their corresponding inhibitory and promotional effects were more significant. This consistency shows that the effect of Cu B-NPs is reliable, and the antitumor potential of Cu B-NPs may directly or indirectly affect the survival, proliferation, and reversion of A549 (Levine & Kroemer, 2019).
Autophagy regulates cellular stress and survival (Lendvai et al., 2021). Overexpression of BECN1 may increase the formation of autophagosomes, thereby enhancing the autophagy ability of cells (Kang et al., 2011). The abnormal activation state of BECN1 mRNA in cells may promote autophagosomes formation and initiate autophagy, leading to competitive binding between miR-124 mimic and BECN1 mRNA and the mRNA of other target genes, reducing the number of miR-124 mimics. It has a regulatory effect on other target genes and reversely affects the gene regulation effect of miR-124 mimic on autophagy-related signaling pathways, thereby reducing P62 expression and further increasing LC3 protein levels. In addition, some studies have pointed out (Zhang et al., 2020) that BECN1 inhibits growth of MG803 gastric cancer cells and promotes autophagy. Our further Western blot results showed that under the intervention of Cu B-NPs, LC3-I and LC3-II levels increased while P62 decreased, and BECN1 was highly expressed. This suggests that the intervention of Cu B-NPs promotes the autophagy process of A549, possibly by affecting the formation of autophagosomes and related autophagy pathways, mainly through BECN1. The results of this study may be that cucurbitacin affects the transcription of the BECN1 gene and thereby regulates the stability of the BECN1 protein, or it may directly interact with the BECN1 protein to regulate autophagy.
Studies have shown that cucurbitacin B can affect cell autophagy by affecting the ubiquitination of SQSTM1, and it can also affect E1, E2, and E3 enzyme genes, thereby regulating the selective degradation of autophagy substrates. In addition, cucurbitacin B can inhibit mTORC1 complex and activate AMPK, thereby affecting the signal transmission of the mTOR pathway.
To verify our speculation that Cu B regulates autophagy in A549, we detected BECN1 mRNA and found that Cu B-NPs increased its expression, thereby promoting apoptosis in A549 cells. In addition, with the intervention of pc-BECN1, the pro-autophagy effect of Cu B-NPs was significantly improved, and the levels of apoptosis-related proteins such as LC3-I and LC3-II showed an upward trend. Analysis shows that under the condition of Cu B-NPs intervention, pc-BECN1 leads to the overexpression of the BECN1 gene, thereby increasing the content of BECN1 protein in cells, causing a more active start of the autophagy pathway and thus strengthening the autophagy process of A549 cells. The reversal effect of inhibiting BECN1 further confirmed the mechanism by which Cu B-NPs enhanced cellular autophagy. In the study, we significantly reversed this phenomenon after using si-BECN1. However, the optimal dose of Cu B-NPs still needs to be further studied in current research.
Conclusion
In summary, Cu B-NPs combine the advantages of nanotechnology and natural compounds. Cu B-NPs may be a potential lung cancer therapeutic drug or therapeutic adjuvant by regulating autophagy and apoptosis pathways. They can produce dual inhibitory effects on tumor cells, thereby providing a new treatment strategy. Cu B-NPs promote cell autophagy by improving drug delivery efficiency and upregulating the expression of BECN1. This mechanism provides a new therapeutic strategy for lung cancer treatment. Second, BECN1, as a key factor in regulating autophagy, may involve in lung cancer treatment. By increasing or decreasing the expression of BECN1, we can regulate the degree of autophagy, which affects the survival and proliferation of tumor cells to a certain extent. This provides a theoretical basis for the development of treatments targeting BECN1. It can also reduce the progression of lung cancer and is expected to improve patient’s survival rate and quality of life.
Footnotes
Abbreviations
BECN1: Beclin 1; Cu B: Cucurbitacin; Cu B-NPs: Cucurbitacins loaded in liposome nanoparticles; GAPDH: Glyceraldehyde 3-phosphate dehydrogenase; MTT: 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide; PBS: Phosphate buffered saline; PCR: Polymerase chain reaction; PVDF: Polyvinylidene difluoride
Acknowledgments
The authors gratefully acknowledge the Chongqing Jiangjin District Third People’s Hospital Laboratory for providing the necessary equipment for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the Ethics Committee of Chongqing Jiangjin District Third People’s Hospital.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
