Abstract
Background and Objectives
Aconitine is a toxic substance capable of causing damage to the nervous system, digestive system, and gastric tissue. Wogonin exhibits significant antibacterial, antioxidant, and potential anticancer effects. The PI3K/Akt pathway is known to engage in the development of cancer cells. Consequently, it is of great significance to investigate the potential of wogonin on aconitine-induced gastric tissue damage in gastric cancer (GC) and its correlation with the signaling pathway.
Materials and Methods
Forty modelled mice were divided into a normal group, normal saline group, aconitine (low, high) dose group, wogonin (high, medium, low) dose group, celecoxib group, and Weifuchun group. After treatment, we detected the mouse’s tumor volume and survival day, as well as determined the expression of PI3K/Akt, LC3B, and p62.
Results
During modeling, HE staining revealed that the low-dose aconitine group exhibited improved pathological conditions, reduced tumor volume and mass compared to the saline group, and elevated survival days. After modeling, tumor volume and mass decreased concentration-dependent in each wogonin dose group. Wogonin treatment greatly prolongs survival days and effectively inhibits tumor growth. The expressions of LC3B, P62, PI3K, Akt, and mTOR mRNA were all decreased compared to the model group. Meanwhile, the treatment promoted the expression of LC3 and Beclin1 proteins related to the autophagy pathway.
Conclusion
Wogonin effectively ameliorates gastric tissue damage induced by aconitine in GC, prolongs the survival days of mice, and inhibits tumor growth through activation of the PI3K/Akt signaling pathway and reduction of P62 protein.
Introduction
Wogonin, primarily extracted from Scutellaria baicalensis, exhibits potent antioxidant properties, effectively scavenging free radicals and protecting cells from oxidative damage. With antioxidative, anti-inflammatory, and immunomodulatory activities, wogonin inhibits inflammatory reactions and hinders the growth of various bacteria. Therefore, it is utilized in treating some conditions such as acute and chronic hepatitis, cholelithiasis, hyperimmune liver disease, bronchitis, and enteritis (Guo et al., 2023). Studies have demonstrated that S. baicalensis can not only inhibit the growth of cancer cells but also induce apoptosis of cancer cells, particularly in malignant tumors. PI3K/Akt further inhibits cancer cell metastasis by suppressing MMP1 (Limanaqi et al., 2020). This article aims to investigate gastric tissue damage caused by aconitine in gastric cancer (GC) and the regulatory effect of wogonin on the PI3K/Akt signaling pathway.
Aconitine induces the activation of inflammatory cytokines, including interleukin (IL)-6, IL-8, and tumor necrosis factor (TNF)-α, and transcription factors such as nuclear factor-κB (NF-κB), AP-1, and Nrf2, thereby initiating inflammatory responses and promoting cell proliferation (Limanaqi et al., 2020; Zhang et al., 2022, 2023). Additionally, it might lead to substantial production of reactive oxygen species (ROS) and nitric oxide (NO), damaging the cell membrane and exacerbating gastric mucosal damage (Chung et al., 2021). Simultaneously, the activation of caspase-3 and caspase-9 is a crucial factor in programmed cell death (Zhang et al., 2021).
As Coptis chinensis strengthens the body’s ability to scavenge free radicals by inhibiting the activity of enzymes like NADPH oxidase (Min et al., 2023), rhubarb can partially inhibit the PI3K/Akt signaling pathway, exerting a negative impact on cell growth. Licorice further enhances the inflammatory response by inhibiting the TLR signaling pathway. Therefore, traditional Chinese medicine shows promising potential in alleviating gastric tissue damage caused by aconitine in GC (Case & Son, 2021; Khan et al., 2023).
Wogonin mitigates the inflammatory response by inhibiting the release of inflammatory cytokines such as TNF-α, IL-1, and MCP. Concurrently, it eliminates oxidative stress products like OH and HNO2, thereby preserving cell immunity against oxidative damage (Dai et al., 2021). On the other hand, wogonin promotes the formation of autophagosomes to clear damaged organelles by regulating LC3 and p62, thereby maintaining the stability of the intracellular environment to improve some diseases such as rheumatoid arthritis, enteritis, and respiratory infections (Wang et al., 2019). In this process, signaling pathways such as ERK1/2, JNK, and p38 influence cell growth. Under the regulation of the mTOR and ULK1/2 pathways, it modulates the programmed cell death process and the formation of autophagosomes (Yang et al., 2022).
In gastric tissue damage induced by aconitine in GC, the activated PI3K/Akt signaling pathway stimulates cell growth in response to growth factors like EGF, IGF-1, and FGF (Pearson & Temenoff, 2022). Additionally, hormones such as Ins and IGF-1 activate the PI3K/Akt signaling pathway upon binding to cell surface receptors, consequently influencing the cell’s metabolic capacity (Wang et al., 2022). The pathway also can be activated by cytokines like IL-10 and IL-13. This pathway is pivotal in the treatment of neurological diseases such as Alzheimer’s disease (Zhou et al., 2020).
Traditional Chinese medicines like Astragalus, Sophora flavescens, and Salvia miltiorrhiza have been found to modulate the activity of the PI3K/Akt signaling pathway, affecting cell growth and apoptosis processes. With anti-inflammatory and antioxidant activities, wogonin relieves gastric mucosal damage induced by aconitine through inhibition of the PI3K/Akt signaling pathway (Chen et al., 2023). Consequently, this study provides a novel insight into the impact of wogonin on aconitine-induced gastric tissue damage in GC.
Materials and Methods
Materials and Instruments
Experimental Animals and Grouping
MFC mice (batch number: PNS-MC-20) were purchased from Shanghai Yaji Biotechnology Co., Ltd. A total of 100 Male Kunming (KM) mice of SPF grade (half of which were male and female) were provided by Changsha Tianqin Biotechnology Ltd.
Instruments and Reagents
Wogonin (purity >98%) was provided by Chengdu Yirui Biotechnology Co., Ltd., RPMI 1640 culture medium by Beijing Solaibao Technology Co., Ltd., fetus bovine serum by Hyclone Company (USA), and 4% paraformaldehyde (Shanghai Yanmu Industrial Co., Ltd.). TNF-α, IL-6, GAG, and ELISA kits were purchased from research and development (R&D) Company (USA), rabbit antihuman pERK1/2 monoclonal antibodies from Icono Biomedicine Co., Ltd., secondary antibodies (Immunoway), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) antibody (Beijing Biolab Technology Co., Ltd.). Surgical microinstruments were purchased from Suzhou Medical Equipment Factory; vertical electrophoresis tank, electrophoresis instrument, and gel imager from Shanghai Heng Qin Instrument Equipment Co., Ltd.; and nucleic acid protein analyzer from Shanghai Jihui Scientific Analytical Instrument Co., Ltd.
Methods
Animal Model
MFC mouse GC cells were incubated in RPMI 1640 medium. After adjusting the cell density, 95 mice were inoculated with the single-cell suspension in their right armpit. Once palpable nodules with a diameter exceeding 5 mm appeared 5–7 days post-inoculation, 5 mice were chosen for low-dose aconitine injection, 90 mice for high-dose aconitine injection, and 5 mice for normal saline injection. The growth of tumors in mice was observed, and successful model construction was confirmed if the tumor size surpassed that in the saline group.
Grouping
Following the successful model establishment, mice were randomly assigned to seven groups: Model group, normal saline group, high-dose wogonin group, medium-dose wogonin group, low-dose wogonin group, celecoxib group, and Weifuchun group. After the injection of corresponding solutions of the same volume, various indicators were observed in the mice. Subsequently, the 50 successfully modeled mice were randomly divided into five groups: Normal saline group, celecoxib group, high-dose, medium-dose, and low-dose groups, and the survival period of the mice was observed.
Monitoring of Tumor Volume and Tumor Weight and Calculation of Tumor Inhibition Rate
We measured tumor volume using enhanced T1WI, manually tracking the lesions, multiplying by the slice thickness and gap, and summing all slices to obtain the tumor volume before and after treatment. After the mice were sacrificed, their GCs were dissected and weighted, and the tumor inhibition rate was calculated.
Survival Test of Tumor-bearing Mice under Various Doses of Wogonin
The survival rate of mice was detected based on the survival days and median survival period of mice.
Reverse Transcription Quantitative Polymerase Chain Reaction (RT-qPCR) of Messenger Ribonucleic Acid (mRNA) Expression in Mouse Tumor-bearing Cancer Cells
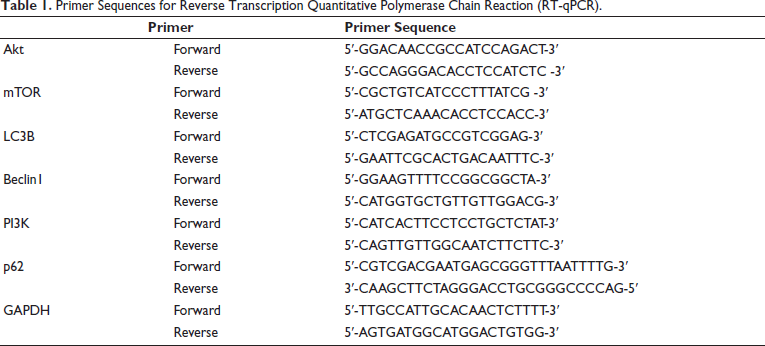
Ribonucleic acid (RNA) was extracted from mouse stomach tissue (100 mg) from each group. The concentration of the extracted RNA was measured, and reverse transcription was conducted through RNA. The relevant PCR primer sequences are shown in Table 1, with a primer concentration of 0.3 µmol L–1 complementary deoxyribonucleic acid (cDNA). The 2 µL reaction conditions were carried out according to the instructions using a two-step method. With GAPDH serving as the internal reference, the relative expression of mRNA was calculated using the 2−∇∇Ct method (Table 1).
Primer Sequences for Reverse Transcription Quantitative Polymerase Chain Reaction (RT-qPCR).
Western Blot Analysis
Mouse gastric tissue cell proteins were extracted, followed by concentration and purity measurements using a BCA protein assay kit. A 30 µ sample protein was taken from each group and administered with primary antibodies mTOR[EPR390(N)] (ab134903, 1:1000), LC3B[EPR18709] (ab192890, 1:1000), Beclin1[EPR19662] (ab302669, 1:1000), PI3K[ EPR25156-60] (ab302958, 1:1000), Akt[EPR18853] (ab285140, 1:1000), p62[EPR4844] (ab109012, 1:1000), and the protein expression levels were calculated with reference to GAPDH.
Hematoxylin and Eosin (HE) Staining
The tumor tissue was fixed in a 4% paraformaldehyde solution. Subsequently, it underwent processes including dehydration, embedding, dewaxing, gradient alcohol dehydration, xylene treatment, washing, differentiation, HE staining, and sealing with neutral resin before being observed under a microscope.
Statistical Analysis
The data were analyzed using SPSS 21.0 and GraphPad Prism software. If there are no special requirements, p < 0.05 is used as the test standard.
Results
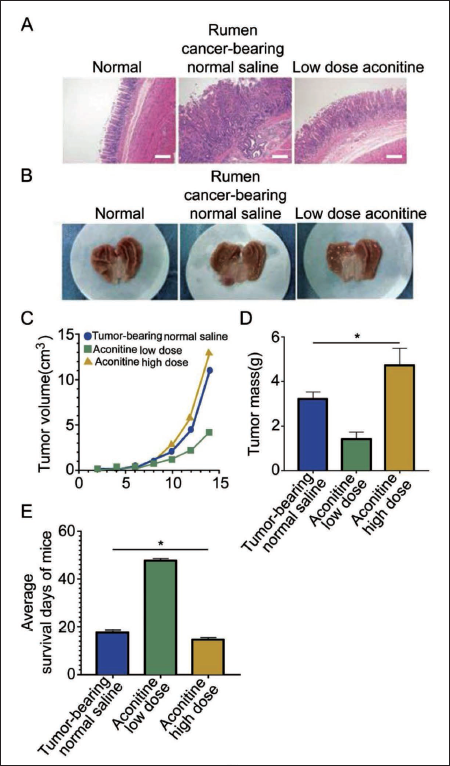
Low-dose aconitine inhibits tumor growth, while high-dose aconitine promotes tumor growth.
The results of HE staining demonstrated a significant improvement in the pathological morphology of GC tumors in the low-dose aconitine group (Figure 1A), as supported by the images of removed GC tissue (Figure 1B). Contrarily, the high-dose aconitine group had the largest tumor volume (Figure 1C) and the highest tumor mass (Figure 1D). Analysis of the survival days indicated that the high-dose aconitine group had the shortest survival period (Figure 1E), suggesting the potent tumor-promoting effects of high-dose aconitine in mice.
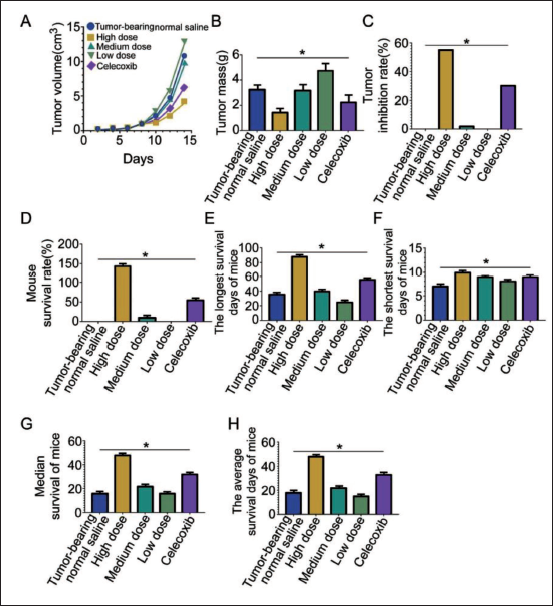
Wogonin has a Positive Antitumor Effect on Cancer-bearing Mice
In each wogonin group, tumor volume suppression was evident (Figure 2A), with the smallest tumors in the high-dose group. Notably, the tumor volume in the high-dose wogonin group remained smaller than that in the celecoxib group (p < 0.05), and the tumor mass was significantly lower than in other groups (Figure 2B). However, while the tumor inhibition rate was high in the high-dose group, the inhibition rate in the normal saline and low-dose group declined to zero. Intriguingly, wogonin treatment dose-dependently increased survival days, median survival period, and survival rate (Figure 2C–F). Remarkably, under high-dose wogonin conditions, these parameters surpassed those of the normal saline and celecoxib groups (Figure 2G–H).
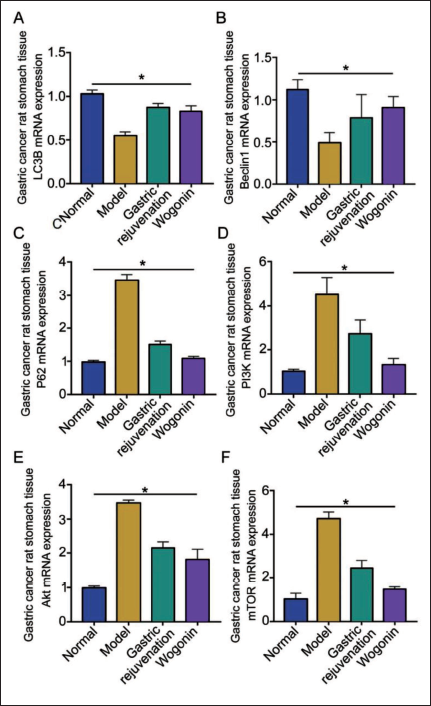
The Improvement Effect of Wogonin on GC is Related to the PI3K/AKt Pathway
To elucidate wogonin’s specific mechanism of action on tumor cells in mice with rumen cancer, a common adjuvant treatment drug, Weifuchun, was selected for comparison. The expression of LC3B, P62, PI3K, Akt, and mTOR mRNA in the wogonin-treated group all decreased compared to the Weifuchun treatment group but was higher than that of the model group (Figure 3A, C–F), while the expression of Beclin1 mRNA was increased (Figure 3B).
Discussion
Aconitine is a toxic substance, and there are many cases of aconitine poisoning, with even death in severe cases (Wang et al., 2016). Research has found that low-dose aconitine promotes tumor cells in GC mice, greatly shortening their survival time and aggravating the condition of GC mice. Through the PGE2/COX2 pathway, aconitine upregulated the expression of Treg, thereby unbalancing the immune system and leading to accelerated tumor growth (Thomas et al., 2023). Wogonin is a natural flavonoid extract from S. baicalensis root, characterized by antispasmodic, anti-inflammatory, antioxidant, and anticancer activity (Lei et al., 2021). Numerous experiments have shown its effects on the proliferation, migration, and apoptosis of various tumors (Xu et al., 2021). Wogonin is reported to inhibit the proliferation of colon cancer cells by activating the STAT3 signaling pathway, downregulating the expression of c-Myc, Mcl-1, and Bcl-xl, and promoting cell apoptosis (Zou et al., 2020). Additionally, wogonin can downregulate the activity of the JAK/STAT3 pathway by inhibiting JAK1/2, further inhibiting the malignant behaviors of GC cell lines (Tan et al., 2019). This evidence has depicted wogonin’s potential protective effect on GC progression. On the other hand, previous studies noted that downregulation of the PI3K/Akt signaling pathway hindered the proliferation of GC cells and induced cancer cell apoptosis (Xing et al., 2019). Therefore, wogonin might alleviate gastric tissue damage in GC caused by aconitine, and its effect is possibly associated with the PI3K/Akt signaling pathway.
In the present study, we established a mouse model of GC with gastric tissue damage caused by aconitine-induced aconitine and then administered various doses of wogonin. It was found that the tumor volume and tumor mass in the medium- and high-dose groups were decreased, whilst the high-dose wogonin group had the highest tumor inhibition rate among groups. High dosage of wogonin had the most significant effect on survival days and the survival period of mice. This suggests that wogonin alleviates gastric tissue damage caused by aconitine in GC. To a certain extent, its function is attributed to its inhibitory effect on vascular endothelial growth factor (VEGF) secreted by GC cells when wogonin downregulates the expression of B7H1 and MICA (RAE-1) on the surface of GC cells, suppressing tumor cell metastasis and the tumor immune microenvironment. The immunogenic death caused by tumor cells will not only cause dendritic cells to phagocytose but also stimulate tumor-specific immune responses, thereby protecting from GC and strengthening an individual’s immunity (Feng et al., 2022). Studies have found that wogonin can activate caspase-3 while increasing the level of ROS and inducing tumor cell apoptosis (Alonso Bellido et al., 2023). Additionally, wogonin can upregulate the expression of the p53 gene through the JAK/STAT signaling pathway, inhibit cyclin B1 expression, and induce cell G2/M phase arrest, thereby inhibiting tumor cell division (Li et al., 2022; Zeng et al., 2023). Consistently, the present study noted that the PI3K/Akt signaling pathway was significantly activated under the intervention of wogonin, suggesting the association between the protective effect of wogonin on gastric damage in GC and this signaling pathway.
Furthermore, the PI3K/Akt signaling pathway is capable of inhibiting the growth of GC cells. Through inhibition of this pathway, recombinant methioninase can induce GC cell apoptosis, downregulating the expression of PI3K, p-Akt/t-Akt, and GLUT-1 (Zhu et al., 2020). As such, RAD-9 activates p38 and MARK and inhibits the PI3K/Akt signaling pathway to suppress the proliferation of GC cells in a time- and concentration-dependent manner. Meanwhile, Bcl-2 protein, Akt, and p-Akt protein expression levels were downregulated, and Bax and caspase-3 protein expression levels were increased, thereby inhibiting the growth of GC cells (Kaufman et al., 2021; Pei et al., 2021). Therefore, effectively inhibiting the activation of PI3K/Akt is crucial to suppress GC cell growth.
In this animal experiment, in order to investigate the effect of wogonin on gastric tissue damage caused by aconitine, we compared the Weifuchun and wogonin groups and found that the wogonin group had lower expression levels of LC3B, P62, PI3K, Akt, and mTOR and a higher level of Beclin1. These results confirm the therapeutic effect of wogonin on GC, even better than the general adjuvant drug Weifuchun, and this therapeutic approach may be related to the PI3K/Akt signaling pathway.
Conclusion
In conclusion, wogonin effectively inhibits the proliferation and migration of GC cells and alleviates gastric tissue damage caused by aconitine. This finding might contribute to the treatment of GC and elucidate the potential of wogonin in the treatment of diseases. However, this study still has not studied the correlation between wogonin and other pathways, and further research is needed.
Footnotes
Abbreviations
GC: Gastric cancer; HE: Hematoxylin and eosin; LC3B: Microtubule-associated protein 1 light chain 3 beta; mTOR: Mammalian target of rapamycin; PI3K/Akt: Phosphatidylinositol 3-kinase/protein kinase B.
Acknowledgments
We gratefully acknowledge the Jinhua People’s Hospital Laboratory for providing the necessary equipment for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the ethnic committee of Jinhua People’s Hospital Wenzhou, Medical University Affiliated Jinhua Hospital.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
