Abstract
Background
Over the years, plants have been utilized worldwide for the treatment of cancer in the traditional system of medicine, notably in the majority of developing nations. The use of chemotherapeutic drugs is frequently associated with lethal side effects when treating cancer. Therefore, the panacea to the vicious side effects linked to synthetic pharmaceuticals is the alternative usage of widely accessible and affordable therapeutic approaches. Chicoric acid, a major constituent of Cichorium intybus, has recently been shown to exhibit antioxidant, anti-inflammatory, antidiabetic, antiviral, neuroprotective, hepatoprotective, and anticancer properties.
Objectives
Except for a few preliminary studies, no detailed study on the chemotherapeutic efficacy of chicoric acid against cancer cells has been done till now. Therefore, the aim of this study is to assess the potential of chicoric acid against colon cancer cells.
Materials and Methods
Different in vitro assays like MTT, DAPI staining, rhodamine staining, H2DCFDA staining, caspase activity, and cell analysis were performed on colon cancer HCT116 cells to assess their growth inhibitory properties.
Results
According to the results, chicoric acid dose-dependently suppressed the growth of colon cancer HCT116 cells by inducing cell cycle arrest at G0/G1 phase and apoptosis via both intrinsic and extrinsic pathways. In silico findings unveiled the cell cycle regulatory capacity of chicoric acid through binding CDC25 phosphatases, implying its potential as an antiproliferative agent capable of suppressing cell growth.
Conclusion
Collectively, the results suggested strong chemopreventive potential of chicoric acid against colon cancer cells. Thus, chicoric acid may turn out to be an excellent anticancer agent, which may be explored in the future for the therapeutic drug development against cancer.
Introduction
Globally, colorectal cancer (CRC) is regarded as the third most prominent cancer. By the year 2040, it is predicted to increase with an incidence of 3,200,000 new cases and 1,600,000 deaths (Morgan et al., 2023). CRC is a significant cause of morbidity and mortality in the United States, with 153,000 new cases and 52,000 deaths in 2023 alone (Siegel et al., 2023). Among all the diagnosed cases of CRC, around 25% of patients have been found to exhibit metastasis, while 20% of them progress to advanced metastatic stage IV, with no cure available so far (National Cancer Institute, 2018; Riihimäki et al., 2016; Siegel et al., 2023). The 5-year survival rate is found to be 12.5% in the United States in patients with stage IV CRC. Thus, it is urgently required to develop safe and effective therapy for the advanced stage disease (Siegel et al., 2023).
Chemotherapy and surgery are the mainstays of current CRC treatment. The chemotherapy treatments consist of combinations like 5-fluorouracil (5-FU), oxaliplatin with or without leucovorin, or a combination of 5-FU, leucovorin, and irinotecan (FOLFIRI). Despite the progress made in medical research, approximately 50% of CRC patients experience a recurrence of tumors. Moreover, the nonspecific activity of chemotherapeutic agents is linked to a variety of adverse effects as well as drug resistance. Along with the tumor cells, it also affects nonmalignant cells. It was reported that a multicomponent therapeutic approach involving plant-based compounds has the potential to enhance treatment while delaying side effects (Rejhová et al., 2018). As further explained, combination therapy often results in a superior outcome by enhancing the cytotoxic impact of the standard medication on cancer cells (Bayat Mokhtari et al., 2017). In this context, there is an urgent need to develop drugs that are based on plants to minimize drug-associated toxicity.
The use of herbal medicine has grown in importance worldwide, affecting both global trade and health. Large segment of the healthcare system still heavily relies on medicinal plants (Zhang, Leung, et al., 2015; Zhang, Wang, et al., 2015). Chicoric acid is one of the predominant constituents derived from Cichorium intybus, recently recognized for its diverse medicinal properties, including antioxidant, anti-inflammatory, antidiabetic, antiviral, neuroprotective, hepatoprotective, and anticancer (Park et al., 2011; Lee & Scagel, 2013; Yang et al., 2022). However, except for a limited number of preliminary studies, no detailed work on the therapeutic efficacy of chicoric acid against cancer cells has been done till now. Moreover, the impact of chicoric acid against colon cancer cells also remains unexplored. That is why the aim of the current study was to delineate the therapeutic potential of chicoric acid against colon cancer cells.
Materials and Methods
Chemicals
Chicoric acid was purchased from Cayman Chemical, USA. Hoechst 33342, Rhodamine123, MTT, ethidium bromide (EtBr), DAPI, trypan blue, PI, H2DCFDA, acridine orange (AO), and caspase assay kits were procured from Sigma–Aldrich. McCoy’s medium, FBS, trypsin-ethylenediaminetetraacetic acid (EDTA) solution, antibiotic-antimycotic solution, and lactate dehydrogenase (LDH) cytotoxicity assay kit were procured from Gibco.
Cell Culture
Human colon cancer HCT116 cells were procured from NCCS, Pune, India. The cells were maintained by using McCoy’s cell growth medium supplemented with 10% FBS and 1% antibiotic-antimycotic solution in a CO2 incubator supplied with 5% CO2 and 37℃ temperature.
Assessment of Cytotoxicity
MTT colorimetric assay was performed to evaluate the cell viability of HCT116 cells treated with chicoric acid (25–150 µM). Briefly, 5 × 103 cells per well in 96-well plate were grown overnight, followed by coculturing with varied doses of chicoric acid (25–150 µM) for another 24 h. Subsequently, 10 µL of MTT (5 mg/mL) dye was added to each well, followed by an incubation of 4 h at 37°C in a CO2 incubator. The purple crystals of formazan were then dissolved in 100 µL of dimethyl sulfoxide and incubated in the dark for 15 minutes. The intensity of the color was then recorded at 570 nm using a microplate reader. The result was expressed as percent cell viability relative to control.
LDH Cytotoxicity Assay
Briefly, HCT116 cells (5 × 103/well) were grown in a 96-well plate, followed by coculturing with chicoric acid (50–150 µM) for 24 and 48 h. Later, LDH activity was measured as per the manufacturer’s protocol.
Assessment of the Morphological Changes
Briefly, HCT116 cells (5 × 103/well) were grown overnight in a 96-well plate, followed by treatment of chicoric acid (50–150 µM). The changes in the morphological appearance of cells were then examined using a Floid imaging station, ThermoScientific, USA.
Evaluation of the Nuclear Changes
DAPI staining was carried out for the assessment of nuclear fragmentation and condensation within HCT116 cells. HCT116 cells (5 × 103/well) were grown overnight in a 96-well plate. Further, the cells were given treatment with chicoric acid (50–150 µM) for another 24 h. Then, the cells were fixed with chilled methanol for 10 minutes, along with washing with 0.2% of triton buffer (permeabilizing buffer). Later, the cells were stained with DAPI and incubated for 30 min at 37°C. Finally, the cells were observed under a fluorescence microscope.
Qualitative Assessment of Apoptosis
DAPI/PI double staining was done in order to assess the apoptosis. HCT116 cells were grown as mentioned above, followed by the treatment of chicoric acid (50–150 µM) for 24 h. Imaging was done via FLoid imaging station using blue and red filters for DAPI and PI, respectively. Additionally, AO/EtBr dual staining was also performed to examine apoptosis in HCT116 cells. Briefly, the cells were grown as described above, followed by treatment with chicoric acid at the indicated doses for 24 h. The treated cells were then washed and exposed to a mixture of AO and EtBr dye. Thereafter, the cells were quickly analyzed under a fluorescence microscope using green and red filters for AO and EtBr, respectively.
Qualitative Estimation of Reactive Oxygen Species (ROS) Generation
To measure the intracellular ROS generation in HCT116 cells, staining with H2DCFDA dye was performed. In a 96-well plate, HCT116 cells (1.5 × 104 cells/well) were seeded and incubated overnight at 37°C. After treatment of chicoric acid (50–150 µM), cells were incubated at 37°C for another 6 h. Later, the cells received 10 µM H2DCFDA and were incubated for another 30 minutes in the dark. Finally, the cells were observed under a fluorescence microscope after removing excess dye by washing.
Assessment of Cytotoxicity in the Presence of N-Acetyl-l-Cysteine (NAC)
The viability of HCT116 cells was also assessed in the presence of NAC. To summarize, the cells were initially exposed to 1 mM NAC for 2 h, followed by varying concentrations of chicoric acid (50–125 µM) for 24 h. Subsequently, cell survival was determined by the MTT assay.
Qualitative Evaluation of Mitochondrial Membrane Potential (∇Ψm)
Rhodamine123 staining was done to evaluate ∇Ψm of HCT116 cells. Briefly, cells were grown and exposed to chicoric acid at various doses (50–150 µM) for 24 h in a 96-well plate. Subsequently, each well received 100 µg/mL of Rhodamine123, and the plate was then incubated at 37°C for 30 minutes. Finally, stained cells were observed under a fluorescence microscope using an appropriate excitation filter. Additionally, ∇Ψm was also analyzed by using another dye, JC-1. Briefly, cells (1 × 105/well) were cultured as mentioned previously, followed by treatment of varying doses of chicoric acid for another 24 h. Subsequently, cells were stained with JC-1 dye and observed under a fluorescence microscope.
Estimation of Caspase Activities
Caspase activities in HCT116 cells were determined using colorimetric test kits in accordance with the manufacturer’s instructions. Briefly, HCT116 cells (3 × 107) were treated with chicoric acid (50–150 µM) for 24 h. Later, caspase activities were estimated in the cell lysates of all the treatment groups and were represented as fold change relative to the untreated control.
Assessment of the Cytotoxicity in the Presence of a Caspase Inhibitor
The viability of HCT116 cells was also assessed in the presence of a pan-caspase inhibitor (Z-VAD-FMK). Concisely, the cells were initially exposed to 50 µM Z-VAD-FMK for 2 h, followed by varying concentrations of chicoric acid (50–125 µM) for 24 h. Subsequently, cell survival was determined by the MTT assay.
Cell Cycle Analysis
Briefly, HCT116 cells (1 × 106) were grown and treated with chicoric acid (50–150 µM) for 24 h as described previously. Subsequently, cells were washed with PBS, spun down, and fixed in 4 mL of 70% ice-cold ethanol by incubating them at 4°C for an hour. Following centrifugation, cells were resuspended in PBS, followed by treatment of RNase A (200 µg/mL) for 30 min at 37°C to digest the ribonucleic acid (RNA). After incubation, propidium iodide was added in each tube, and cell cycle analysis was done on FACSLyric machine (BD Bioscience, USA).
qPCR Analysis
HCT116 cells were cultured in a 25 cm2 flask for 24 h, followed by the treatment of chicoric acid (50–125 µM) for 24 h. Subsequently, the total RNA of the cells was extracted using the PureLink RNA Mini Kit (ThermoScientific). First-strand complementary deoxyribonucleic acid (cDNA) was synthesized from total RNA (2 µg) using a Verso cDNA synthesis kit (ThermoScientific). Quantitative polymerase chain reaction (qPCR) was performed on the ABI 7500 Real-Time PCR Machine (Applied Biosystems) using DyNAmoColorFlash SYBR Green qPCR Kit (ThermoScientific) as per the manufacturer’s instructions. The primers were designed using NCBI Primer-BLAST. The primers used were: Bax (Forward: 5′-AAGAAGCT-GAGCGAGTGT-3′; Reverse: 5′-GGAGGAAGTCCAA-TGTC-3′), Bcl-2 (Forward: 5′-TCCATGTCTTTGGA-CAACCA-3′; Reverse: 5′-CTCCACCAGTGTTCCCATCT-3′), cyclin D1 (Forward: 5′-ATGTGTGCAGAAGGAGGTCC-3′; Reverse: 5′-CCTTCATCTTAGAGGCCACG-3′), CDK4 (Forward: 5′-CAGTGTACAAGGCCCGTGATC-3′; Reverse: 5′-ACGAACTGTGCTGATGGGAAG-3′), p21 (Forward: 5′-AGCAGAGGAAGACCATGTGG-3′; Reverse: 5′-TGTG-ATGATGGTGAGGATGG-3′), p27 (Forward: 5′-ATGTC-AAACGTGCGAGTGTCT-3′; Reverse: 5′-TTACGTTTGA-CGTCTTCTGA-3′). GAPDH was used as a housekeeping gene to normalize gene expression. Fold change change was calculated by ∇∇Ct method.
Preparation of Ligand and Protein for Molecular Docking
The 3D structure of chicoric acid (CID-5281764) and vitamin K3 (CID-4055), serving as the ligands, was downloaded from the PubChem database, and the targets Cdc25A (PDB ID-1C25), Cdc25B (PDB ID-1CWS3), and Cdc25C (PDB ID-3OP3) downloaded from RCSB-PDB database, underwent preparation steps for molecular docking utilizing Autodock 4.2.6 software. During the ligand preparation process, the ionization state of the ligands remained unaltered, no tautomers were generated, and the specific chirality of the ligands was maintained. Energetic minimization was conducted using the Merck Molecular Force Field (MMFF94). Subsequently, employing PyRx version 0.8, the chemical structures of each ligand were additionally optimized. The protein structure underwent preparation steps, including the removal of water molecules, the addition of polar hydrogen atoms, and the assignment of Kollman’s charges, followed by conversion into PDBQT format using Autodock 4.2.6 software.
Statistical Analysis
Data shown in this study are the mean ± standard error of the mean (SEM) of three individual experiments performed in triplicates. One-way analysis of variance (ANOVA), followed by Dunnett’s post hoc multiple comparison test, was performed to compare between the means of treatment groups. p < 0.05 was considered significant.
Results
Chicoric Acid Inhibited Cell Growth and Induced Cytotoxic Effect in HCT116 Colon Cancer Cells
The initial investigation of the effect of chicoric acid in HCT116 was done by MTT assay. Chicoric acid exhibited strong dose- and time-dependent suppression of the cell viability (Figure 1A). The cell viability of HCT116 cells post-treatment was found to be 94.44 ± 2.85%, 78.39 ± 3.45%, 50.53 ± 2.41, 32.14 ± 4.13%, and 14.77 ± 3.53% at doses of 50, 75, 100, 125, and 150 µM, respectively. Moreover, at 48 h of treatment, the cell viability was found to be 79.29 ± 4.37%, 61.85 ± 3.91%, 38.98 ± 2.57%, 21.25 ± 1.65%, and 9.48 ± 1.08% at the aforementioned doses. The IC50 of chicoric acid was found to be 92.92 µM and 77.16 µM after 24 and 48 h of treatment (Figure 1B). The results signified that chicoric acid has a strong potential to reduce the growth of colon cancer cells.
Chicoric Acid-mediated Cytotoxicity against HCT116 Cells. (A) The Percentage of Cell Viability of HCT116 Cells treated with Chicoric Acid (25–150 µM) for 24 and 48 h. (B) Graph showing IC50 Value of Chicoric Acid (92.92 µM at 24 h and 77.16 µM at 48 h) against Colon Cancer Cells. Results shown are the Mean ± Standard Error of the Mean (SEM) of Three Independent Experiments Performed in Triplicate (ns > 0.05, *p < 0.05, **p < 0.01, and ***p < 0.001 represent Significant Difference Compared with Control).
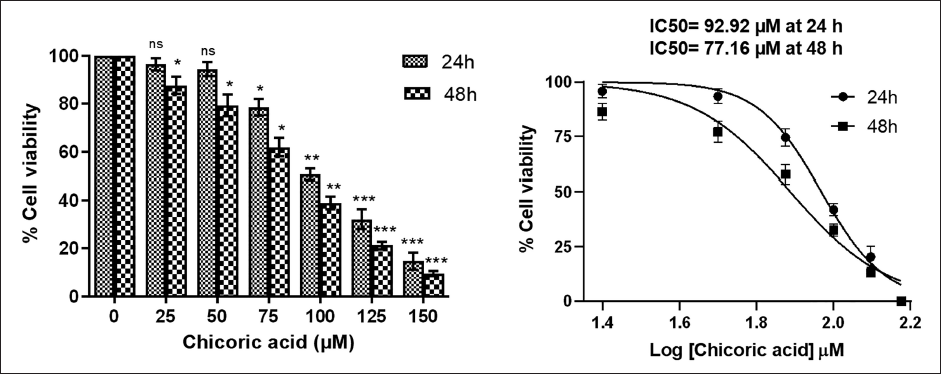
The cytotoxic impact of chicoric acid was also assessed using the LDH release assay, which gauges the extent of LDH release into the medium. An elevated LDH release into the media is indicative of cellular damage. According to the results, chicoric acid-induced cytotoxicity in HCT116 cells was dose- and time-dependent, as shown in Figure 1C.
Chicoric Acid-induced Cytotoxicity in HCT116 Cells was Determined by the Lactate Dehydrogenase (LDH) Release Assay. Results shown are the Mean ± Standard Error of the Mean (SEM) of Three Independent Experiments Performed in Triplicate (ns > 0.05, *p < 0.05, **p < 0.01, and ***p < 0.001 represent Significant Difference Compared with Control).
Furthermore, under a phase-contrast microscope, observable morphological changes were also seen in chicoric acid-treated HCT116 cells. Intact cell morphology was observed in the untreated control cells (Figure 1D). However, severe morphological alterations were found in chicoric acid-treated HCT116 cells in a dose-dependent manner. These alterations included an increase in cell shrinkage and detachment from the surface, leading to a greater number of floating cells. Thus, the results reveal that chicoric acid induces cytotoxicity in HCT116 cells.
Phase Contrast Micrographs of HCT116 Cells Treated with Chicoric Acid (50–150 µM) for 24 h. The Images shown are Representative of Three Independent Experiments (Scale Bar: 100 µm).

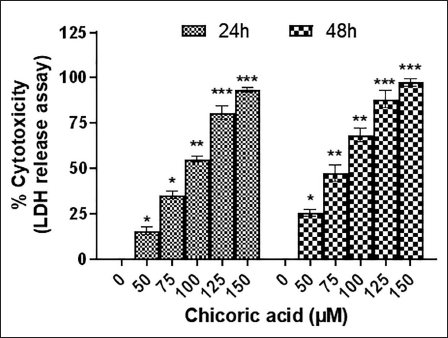
Chicoric Acid Caused Nuclear Condensation in HCT116 Cells
Cell nuclei morphology was examined using DAPI staining to evaluate whether chicoric acid causes death in colon cancer cells through apoptotic pathways. The untreated HCT116 cells exhibited consistent staining during all of the treatment hours, and there was no evidence of fragmentation in the nucleus (Figure 2A). However, after 24 h of treatment, a dose-dependent increase in the bright blue fluorescence was found in the nuclei of the cells, which indicated the presence of chromatin condensation.
Nuclear Morphology of DAPI-stained Nuclei in HCT116 Cells Cocultured with Chicoric Acid (50–150 µM) for 24 h was Observed under Fluorescence Microscopy. The Images shown are Representative of Three Independent Experiments (Scale Bar: 100 µm).
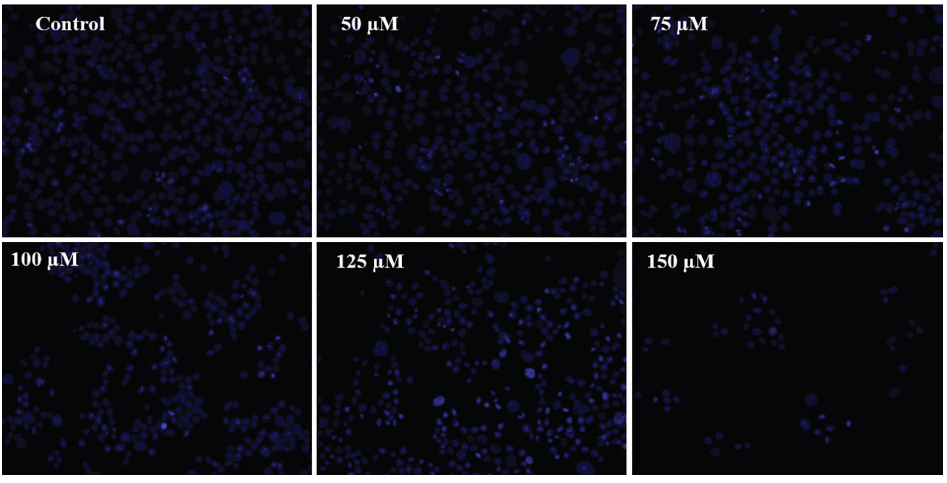
Chicoric Acid-induced Apoptosis in HCT116 Cells
In order to assess apoptotic cell death in HCT116 cells after treatment with chicoric acid, dual staining with DAPI and PI was performed. As can be seen in Figure 2B, a dose-dependent increase in the blue and red florescent nuclei in chicoric acid-treated HCT116 cells was observed, whereas the untreated HCT116 cells had diffuse blue and red nuclei. Additionally, chicoric acid-induced apoptosis was also noted in HCT116 cells in a dose-dependent manner by AO/EtBr staining.
DAPI/PI Merge Staining to Assess Phenotypic Features of Cell Death in HCT116 cells. The Images shown are Representative of Three Independent Experiments (Scale Bar: 100 µm).
As depicted in Figure 2C, live cells displayed a consistent green appearance with bright green nuclei. Early apoptotic cells exhibited irregularly shaped green nuclei, indicating chromatin condensation, evident through green patches and apoptotic bodies. Late apoptotic cells showed extensively fragmented chromatin and nuclei with an orange-to-red hue. Necrotic cells could be identified by uniformly orange-to-red nuclei with a structured appearance. Thus, the quantity of early and late apoptotic cells increased in chicoric acid-treated HCT116 cells in direct proportion to the dose.
Acridine Orange (AO)/Ethidium Bromide (EtBr) Dual Staining in HCT116 Cells Treated with 50–150 µM Chicoric Acid for 24 h. The Images shown are Representative of Three Independent Experiments (Scale Bar: 100 µm).
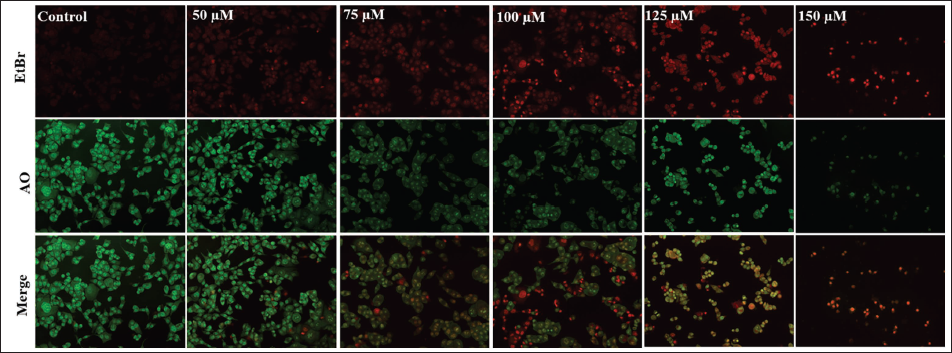
Chicoric Acid-induced ROS Generation in HCT116 Cells
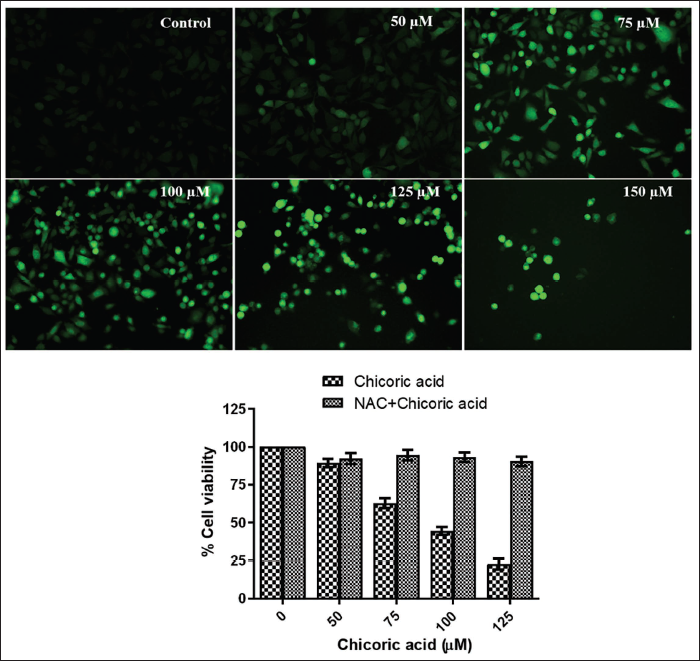
The fluorescence intensity of dichlorofluorescein (DCF) is a direct measure of ROS generation. As illustrated in Figure 3A, the chicoric acid-treated HCT116 cells displayed a gradual increase in green fluorescence as the doses were raised from 50 to 150 µM, indicating a rise in ROS generation. To demonstrate that the reduction in cell survival caused by chicoric acid is attributed to ROS, HCT116 cells were pretreated with NAC. The results showed that NAC pretreatment led to amelioration in the chicoric acid-induced cytotoxicity (Figure 3B). This suggests that the generation of ROS played a critical role in the cytotoxicity induced by chicoric acid in HCT116 cells.
(A) Augmentation of Reactive Oxygen Species (ROS) Generation. H2DCFDA-stained HCT116 Cells Cocultured with Chicoric Acid (50–150 µM) depict Enhanced Intracellular ROS Generation. The Images shown are Representative of Three Independent Experiments (Scale Bar: 100 µm). (B) N-Acetyl-l -Cysteine (NAC) leads to Amelioration in the Chicoric Acid-induced Cytotoxicity in HCT116 Cells. Data represent the Mean ± Standard Error of the Mean (SEM) of Three Independent Experiments Performed in Triplicate.
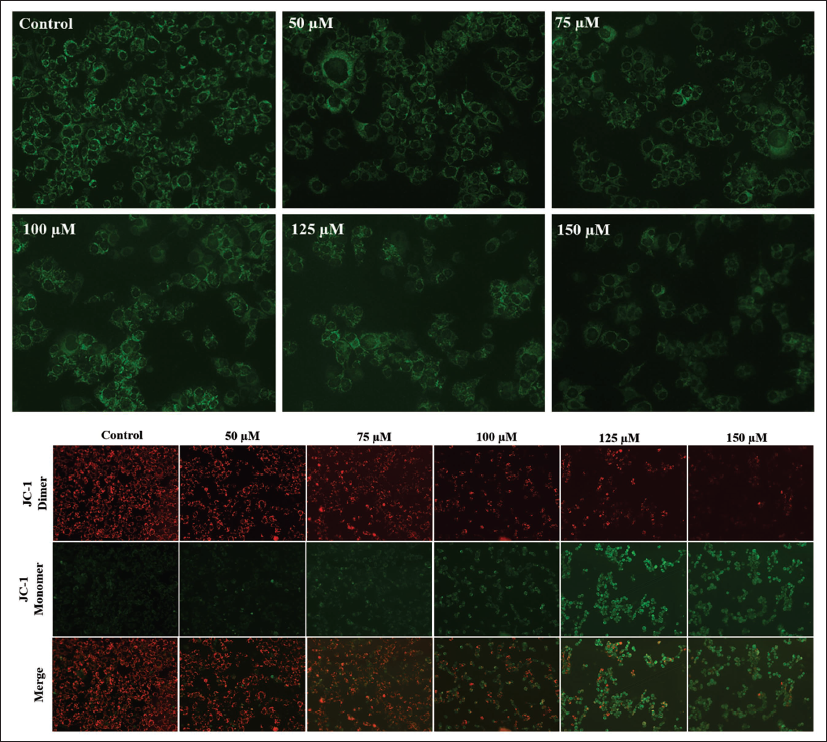
Chicoric Acid Triggered Loss of ∇Ψm in HCT116 Cells
Rh123 staining was done to assess the ∇Ψm in HCT116 cells treated with chicoric acid. The level of rhodamine fluorescence is inversely related to the ∇Ψm. As the dose of chicoric acid was raised from 50 to 150 µM, the fluorescence was found to be consistently decreased (Figure 3C). These findings demonstrate that chicoric acid reduces the ∇Ψm in HCT116 cells in a dose-dependent manner. Another lipophilic dye, JC-1, was also used to evaluate the reduction in the ∇Ψm in HCT116 cells. JC-1 can enter mitochondria and undergo a reversible color change from green to red as the ∇Ψm increases. In untreated control HCT116 cells, JC-1 dimerizes to form J-aggregates. This resulted in intense red fluorescence, indicating high ∇Ψm in control cells (Figure 3D). Conversely, in the chicoric acid-treated HCT116 cells, JC-1 remained in monomeric form, displaying a dose-dependent increase in the green fluorescence characteristic of the loss of ∇Ψm.
Reduction in the Mitochondrial Membrane Potential in HCT116 Cells Cocultured with 50–150 µM of Chicoric Acid Evaluated by (C) Rh123 Staining (D) JC1 Staining. The Images Shown are Representative of Three Independent Experiments (Scale Bar: 100 µm).
Chicoric Acid Caused Activation of Caspases in HCT116 Cells
A significant activation of caspases-3, -8, and -9 were found in HCT116 cells treated with chicoric acid for 24 h. As evident in Figure 4A, the percent caspase-3 activity was found to be 109.89 ± 1.41%, 121.75 ± 1.84%, 140.15 ± 3.11%, 175.12 ± 3.78%, and 193.44 ± 1.45% at doses of 50, 75, 100, 125, and 150 µM, respectively. Similarly, a noticeably increased activation of initiator caspase-8 was also observed, and percent activity was found to be 106.73 ± 1.89%, 112.31 ± 2.17%, 123.23 ± 1.12%, 148.81 ± 1.33%, and 170.56 ± 1.14% at the aforementioned doses. Moreover, dose-dependent activity of initiator caspase-9 was likewise noted and percent activity was 105.26 ± 1.21%, 112.15 ± 2.89%, 130.14 ± 2.64%, 153.29 ± 4.12%, and 167.81 ± 1.41% at the aforesaid doses. Thus, our findings suggest that apoptosis’s extracellular and mitochondrial pathways are activated in HCT116 cells treated with chicoric acid. The observed escalation in caspase activities may indicate a positive response of the HCT116 cancer cells to the treatment, leading to apoptosis.
(A) The Percentage of Caspase Activities in HCT116 Cells Cocultured with Chicoric Acid (50–150 µM) for 24 h. (B) Caspase Inhibitors (Z-VAD-FMK) lead to Amelioration in the Chicoric Acid-induced Cytotoxicity in HCT116 Cells. Data represent the Mean ± Standard Error of the Mean (SEM) of Three Independent Experiments Performed in Triplicate (ns > 0.05, *p < 0.05, **p < 0.01, and ***p < 0.001 represent Significant Difference Compared with Control).
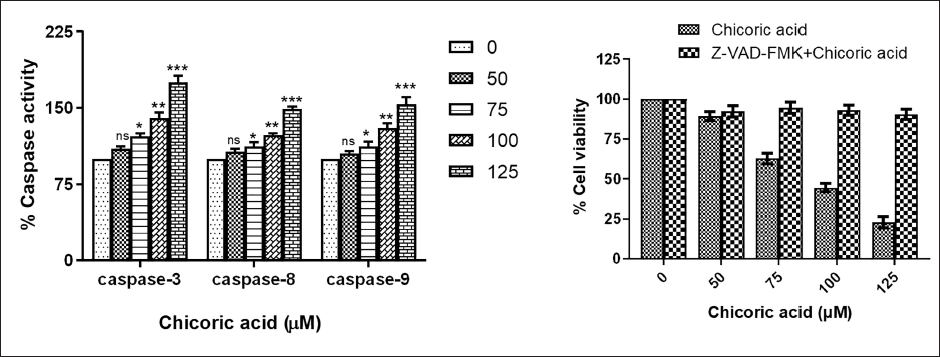
Additionally, to demonstrate that chicoric acid’s inhibition of cell growth is attributed to caspase activation, HCT116 cells were pretreated with a pan-caspase inhibitor (Z-VAD-FMK). The results showed that Z-VAD-FMK pretreatment led to a reversal in the chicoric acid-induced cytotoxicity (Figure 4B). This finding strongly supports that chicoric acid-induced apoptosis in HCT116 cells is primarily mediated through the activation of caspases.
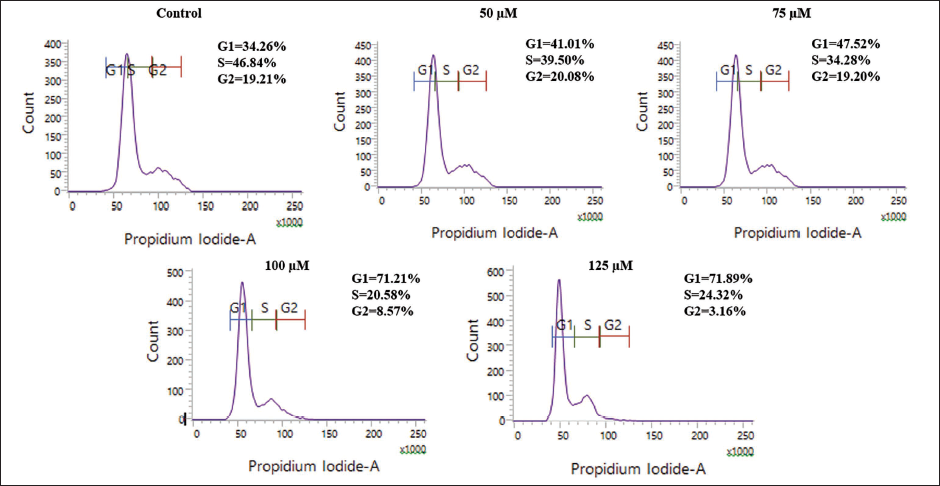
Chicoric Acid-induced G0/G1 Arrest in HCT116 Cells
The result revealed that chicoric acid treatment led to a notable G0/G1 arrest in HCT116 cells when compared to the untreated control. The G0/G1 population of cells was found to be 34.26% in the untreated control (Figure 4C). However, upon treatment with increasing doses of chicoric acid, the G0/G1 phase population showed a steady increase to 41.01%, 47.52%, 71.21%, and 71.89% at 50, 75, 100, and 125 µM, respectively. The effects of chicoric acid on the cell cycle demonstrate their distinct mechanisms of action in HCT116 colon cancer cell lines.
Cell Cycle Analysis of HCT116 Cells Cocultured with Chicoric Acid (50–125 µM) for 24 h. The Images shown are Representative of Two Independent Experiments.
Chicoric Acid Modulated the Expression of Bcl2 Family Proteins in HCT116 Cells
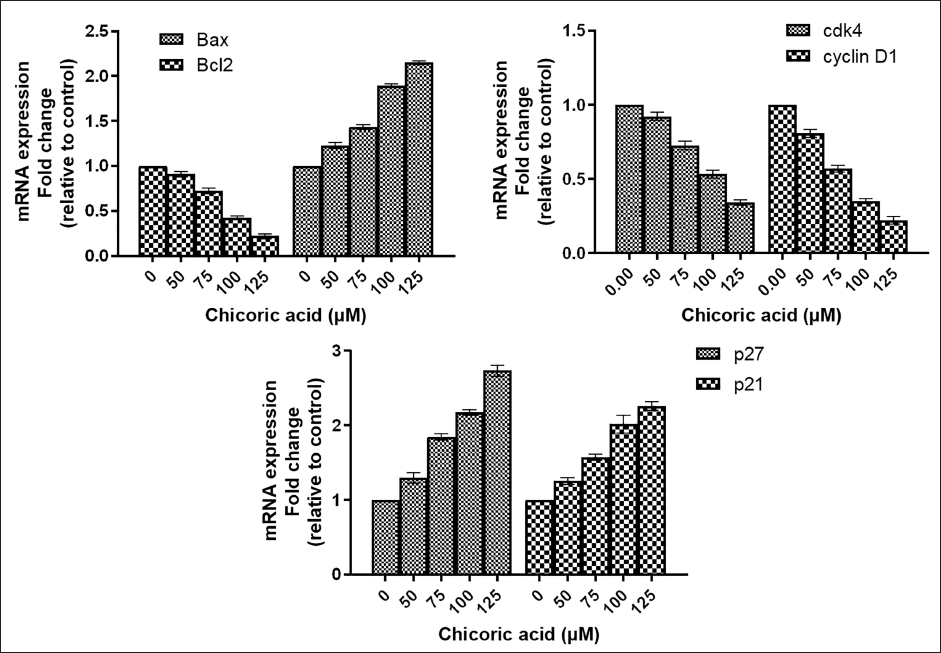
To assess the modulatory effect of chicoric acid on the gene expression of Bcl2 family proteins, qPCR was performed in chicoric acid-treated HCT116 cells. The qPCR results demonstrated a substantial increase in the expression of proapoptotic Bax, whereas a significant decline in the expression of antiapoptotic Bcl2 was observed in chicoric acid-treated HCT116 cells (Figure 4D).
Chicoric Acid-Mediated Cell Cycle Arrest through Regulation of Cyclin D1, cdk4, p21, and p27 Expression in HCT116 Cells
Through quantitative real-time PCR analysis (qPCR), messenger ribonucleic acid (mRNA) expression of the G1 phase proteins such as cyclin D1, CDK4, p21, and p27 were quantified in order to shed light on the mechanism of G0/G1 arrest by chicoric acid. Our findings demonstrated that chicoric acid downregulated mRNA expression of cyclin D1 and cdk4 in a dose-dependent manner in HCT116 cells, whereas the expression of p21 and p27 mRNA in HCT116 cells was considerably enhanced in comparison to the untreated cells (Figure 4E and F).
Modulation of Gene Expression in the Apoptosis Family and Cell Cycle Regulatory Proteins. Fold Change in the Messenger Ribonucleic Acid (mRNA) Expression of (A) Bax and Blc2, (B) cdk4 and cyclin D1 (C) p27 and p21. Data represent the Mean ± Standard Error of the Mean (SEM) of Three Independent Experiments Performed in Triplicate.
Inhibition of Cdc25 A, B, C by Chicoric Acid Assessed by Molecular Docking Analysis
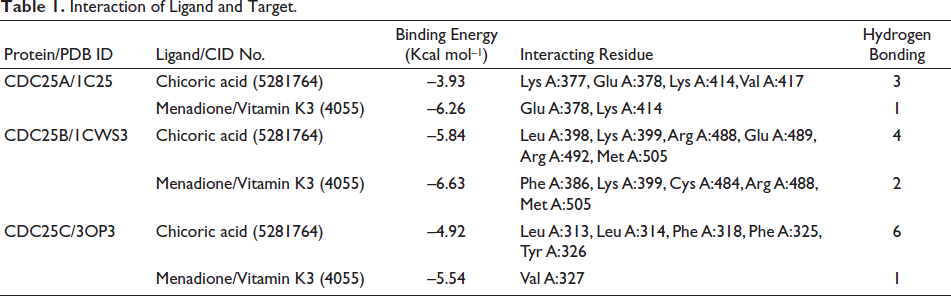
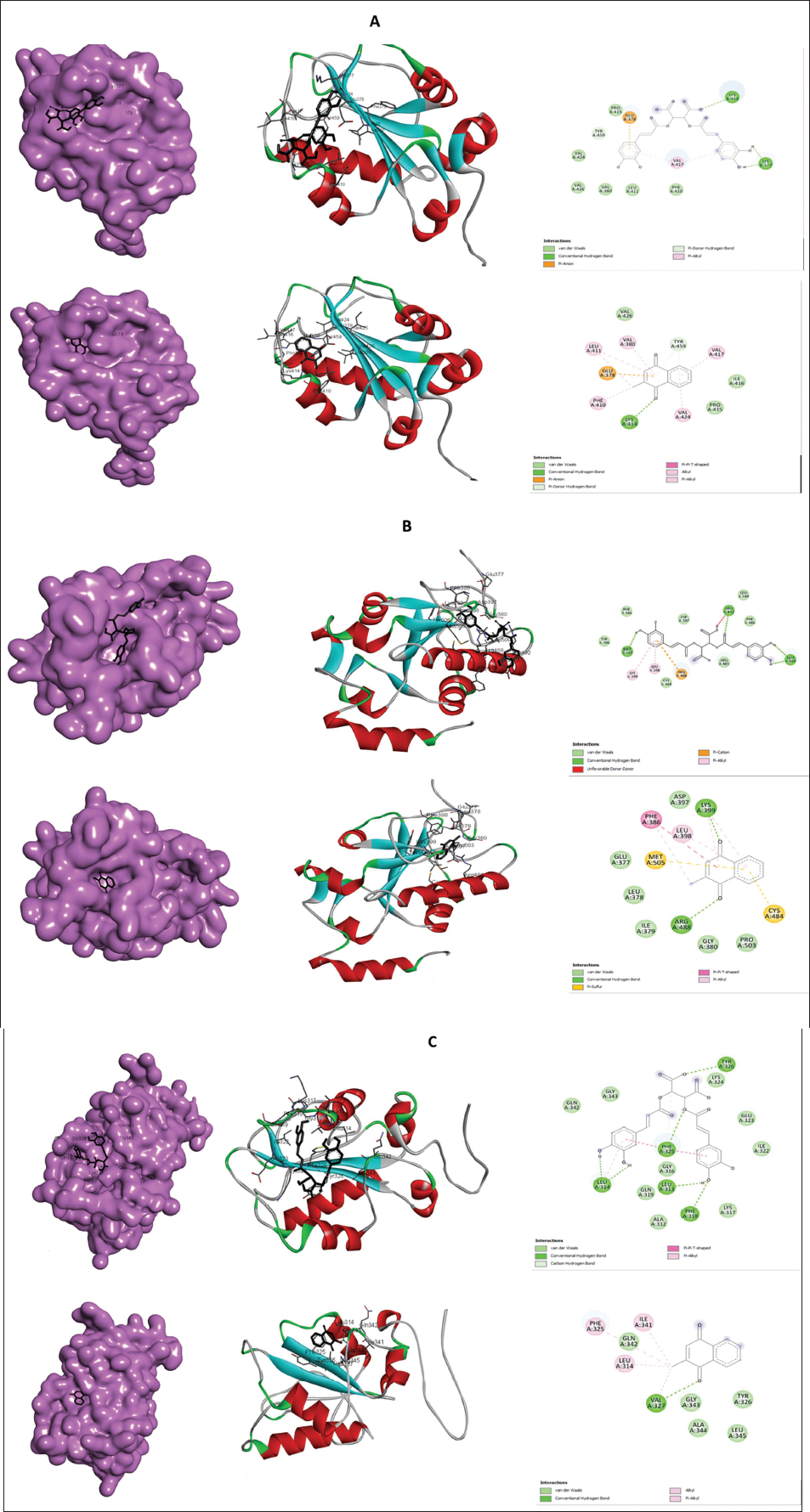
To delineate the regulatory effect of chicoric acid on the cell cycle and to identify its potential targets, in silico molecular docking studies were undertaken. Consequently, Cdc25 family phosphatases were identified as potential targets, as members of this family, Cdc25A, Cdc25B, and Cdc25C play very important role in overall cell cycle regulation. The findings from the docking study revealed that chicoric acid exhibits the potential capacity to bind and inhibit all the Cdc25 phosphatases, which have been found to be elevated in the colon as well as other cancers. This was evidenced by its significant binding affinity and the formation of numerous hydrogen bonds, as presented in Table 1 and depicted in Figure 5A–C. Among all three cell cycle regulators docked with chicoric acid, the highest binding energy was scored by the interaction with Cdc25 B (–5.84 Kcal mol–1) in comparison to its known irreversible inhibitor menadione (also known as vitamin K3), which scores –6.63 Kcal mol–1. The interacting amino acid residues are listed in Table 1. These findings indicate that chicoric acid is likely to engage with various cell cycle mediators; particularly, phosphatases, which are responsible for activating cyclin-dependent kinases by dephosphorylating specific residues at various points during the cell cycle progression.
Interaction of Ligand and Target.
2D and 3D Interaction of Chicoric Acid with (A) CDC25A, (B) CDC25B, and (C) CDC25C.
Discussion
The prevalence of colon cancer has been gradually rising because of recent changes in lifestyle, such as eating less or no fruits and vegetables, doing minimal physical activity, drinking too much alcohol, and being exposed to potentially harmful chemicals (Puzzono et al., 2021). Early-stage colon cancer patients undergo surgery as their primary treatment, whereas those with later stages (metastatic stage) receive chemotherapy. However, chemotherapy is also accompanied by side effects such as nephrotoxicity, hepatotoxicity, diarrhoea, low blood counts, nausea, nerve damage, exhaustion, discomfort aging, hair loss, and skin issues (Chibaudel et al., 2012). Identification of innovative treatments or medication candidates that selectively target cancer cells without having an adverse effect on healthy cells is therefore urgently needed. Due to their anticancer, antiangiogenic, and antimetastatic capabilities without producing any substantial adverse effects, naturally occurring plants/plant-based chemicals are receiving a lot of interest in current cancer research (Dehelean et al., 2021).
Chicoric acid, a natural dicaffeyl-tartaric acid, has been found in large number of edible plants and vegetables, but C. intybus, along with Echinacea purpurea and few others, are the primary source of this important chemical (Yang et al., 2022). Chicoric acid is a major and valuable functional food ingredient in many plants that are consumed as alternative medicines or food supplements (Peng et al., 2019). Although few preliminary reports regarding its pharmacological properties exist, but the anticancer effect of chicoric acid has not been explored much. Moreover, the potential of chicoric acid against colon cancer cells has not been delineated in detail, with only a single study published to date (Tsai et al., 2012). Hence, the primary objective of this study was to elucidate the antiproliferative and cytotoxic potential of chicoric acid against colon cancer.
First, we aimed to assess the efficacy of the chicoric acid on the HCT116 cells. Chicoric acid appeared to significantly reduce the growth of HCT116 cells as assessed by MTT and LDH release assay. Furthermore, morphological study using phase-contrast microscopy revealed that the morphologies of the cells were considerably altered in the chicoric acid-treated groups, which were distinguished by cellular shrinking, detachment from the surface, becoming spherical, and the formation of clumps. These findings indicated a dose-dependent reduction in the cell viability of HCT116 cells after treatment with chicoric acid. Our findings were in accordance with previous reports, where chicoric have been shown to exhibit antiproliferative and cytotoxic potential against gastric and colon cancer cell lines (Peng et al., 2019; Tsai et al., 2012). To the best of our knowledge, these are only two reports that describe the anticancer potential of chicoric acid. However, C. intybus extracts and its compounds other than chicoric acid have been reported to exhibit anticancer potential in previous studies (Al-Snafi, 2016; Conforti et al., 2008; Gospodinova & Krasteva, 2015; Kandil et al., 2019; Khan et al., 2020; Rahimipour et al., 2017; Saleem et al., 2014).
Evading apoptosis is a characteristic feature during the development of cancer, and thus, induction of programmed cell death is a significant therapeutic approach against cancer. Apoptosis is attributed to many morphological and physiological events, such as loss of membrane integrity, chromatin condensation, caspase activation, and modulation in the expression of pro and antiapoptotic genes (Pistritto et al., 2016). According to our findings, chicoric acid promoted cell death in HCT116 cells by inducing nuclear fragmentation and condensation, confirmed by DAPI staining. The DAPI/PI and AO/EB double staining findings also revealed unusual apoptotic characteristics as said above.
The large amounts of ROS produced by cancer cells result in a condition of enhanced basal oxidative stress. Intriguingly, pro-oxidant compounds can be used to target malignant cells with precision since this state of oxidative stress makes cancer cells susceptible to compounds that increase ROS levels (Liou & Storz, 2010). According to our findings, chicoric acid upsurged the ROS level in HCT116 cells in a dose-dependent manner, which could promote these cells to undergo ROS-mediated apoptosis. Moreover, NAC pretreatment confirmed that the production of ROS is a key event in chicoric acid-mediated growth inhibition of HCT116 cells. In a recent investigation, it was found that lactucin, a key compound of Chicorium, plays a significant role in generating intracellular ROS within caki-1 human renal cancer cells. Thus, the results suggested that ROS serves a crucial function in the process of lactucin-induced apoptosis (Jang et al., 2021).
The activation of apoptotic pathways involves a loss of ∆Ψm (Zhang, Leung, et al., 2015; Zhang, Wang, et al., 2015). Certainly, the activation of the mitochondrial permeability transition pore has been shown to disrupt the ∆Ψm, which releases factors that trigger apoptosis and the cessation of oxidative phosphorylation (Forrest, 2015). In chicoric acid-treated HCT116 cells, there was a significant dose-dependent loss in the ∆Ψm, which must have caused the apoptotic signal to be sent through the mitochondrial pathway.
The induction of extrinsic and intrinsic pathways of apoptosis is characterized by the activation of caspase-8 and -9, respectively. Apoptosis-associated morphological and biochemical features are caused by the activation of caspases-3, -6, and -7 (Khaw-On & Banjerdpongchai, 2012). The outcomes demonstrated that the activation of caspase-8, -9, and -3 activities was concentration-dependent, which indicated that chicoric acid-mediated apoptosis occurred simultaneously by death receptors and mitochondria. This proved that chicoric acid-treated HCT116 cells encountered apoptosis through both intrinsic and extrinsic mechanisms. Moreover, Z-VAD-FMK pretreatment substantiated that the activation of caspases is a crucial phenomenon in chicoric acid-mediated growth suppression of HCT116 cells. Interestingly, qPCR results showed that the treatment of chicoric acid led to a concomitant reduction in the Bcl2 mRNA expression and an increase in Bax expression. In a single previous study, chicoric acid demonstrated a notable induction of apoptosis in colon cancer cells via the activation of caspase-9 (Tsai et al., 2012). In a recent study, (3S)-1,2,3,4-tetrahydro-β-carboline-3-carboxylic acid, isolated from Cichorium endivia, was shown to exert cytotoxic effect on CRC HCT-8 cell line (Wang et al., 2012). This compound caused HCT-8 cells to undergo apoptosis in a dose-dependent manner characterized by the loss of ∆Ψm, activation of caspase-3, -8, and -9, upregulation of Bax, and downregulation of Bcl-2.
It is generally recognized that an unregulated cell cycle is strongly associated with carcinogenesis. The G1 and G2 stages of the cell cycle are the main checkpoints and are crucial for the progression of the cell cycle. The cell cycle analysis showed that chicoric acid prevented the transition from G1 to the S phase, which in turn abrogated cell cycle advancement at G0/G1. Moreover, qPCR analysis showed that the treatment of chicoric acid triggered the downregulation of cyclin (cyclin D1) and cyclin-dependent kinase (cdk4), along with a concomitant upregulation of cyclin-dependent kinase inhibitors (p21 and p27). G0/G1 arrest in HCT116 cells by chicoric acid could be attributed to the modulation of these genes. Therefore, our results showed that chicoric acid could cause apoptosis and cell cycle arrest and in colon cancer cells. In a recent study, lactucin, a sesquiterpene from the plant C. intybus, has been shown to exert cytotoxicity in A549 (IC50 = 79.87 µM) and H2347 (IC50 = 68.85 µM) cells by inducing apoptosis, cell cycle arrest at G0/G1 phase, and modulating expression of apoptosis and cell cycle regulatory proteins such as p53, Bax, Bcl2, p21, and PTEN (Imam et al., 2022).
Among the pivotal regulators governing normal cell division, the cell division cycle 25 (Cdc25) protein family emerges as a group of highly conserved dual-specificity phosphatases, crucial in the activation of Cdks (Aressy & Ducommun, 2008; Gabrielli & Burgess, 2016; Kristjánsdóttir & Rudolph, 2004). Within the human genome, three Cdc25 homologs (A, B, and C) have been identified thus far. Notably, Cdc25A and B exhibit proto-oncogenic properties, the dysregulation of which can lead to aberrant cell proliferation and contribute to the development of various diseases, including cancer. Their overexpression is documented across various malignancies such as prostate, breast, colorectal, ovarian, hepatocellular, and nonsmall cell lung cancers (Sur & Agrawal, 2016). Henceforth, Cdc25 phosphatases have arisen as promising chemotherapeutic targets within the field of oncology (Abdelwahab et al., 2022). Numerous compounds, including quinonoid and nonquinonoid structural variants exhibiting potential inhibitory activity against Cdc25A and B phosphatases, have undergone thorough review and analysis for their potential (Tao et al., 2021). Furthermore, an ample body of in vitro and in silico literature exists, demonstrating the potential of phytochemicals to target Cdc25 in numerous cancer cell lines, rendering it an appealing target for future drug development endeavors through targeting the cell cycle (Dakilah et al., 2024; Kabakci et al., 2019; Kolb et al., 2009; Sarkis et al., 2017).
An Illustration depicting the Possible Mechanism of Action of Chicoric Acid Predicted by In Silico Analysis, showcasing its Potential Binding and Blocking the Actions of CDC25 Phosphatases.
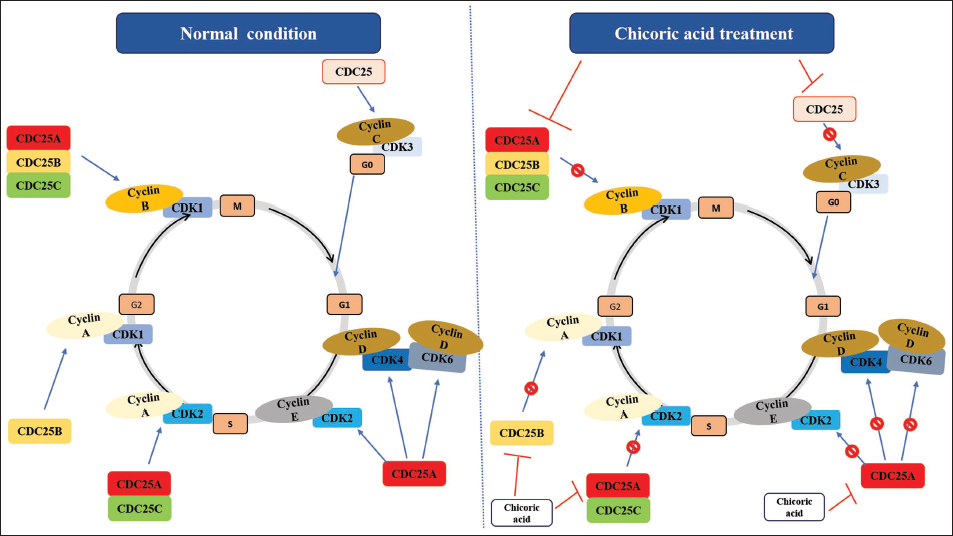
CDC25A governs the G1/S transition by dephosphorylating CDKs, enabling progression into the S phase and contributing to DNA integrity checkpoints. CDC25B and CDC25C primarily oversee the G2/M transition by activating CDK1, pivotal for mitotic entry (Dakilah et al., 2024). CDC25C, regulated by upstream pathways such as the DNA damage checkpoint, ensures precise cell cycle progression. Dysregulation of these isoforms can disrupt the cell cycle, leading to genomic instability and contributing to diseases like cancer (Kolb et al., 2009; Sadeghi et al., 2019). These observations suggest that targeting CDC25 could potentially regulate aberrant cell cycle progression, particularly by utilizing phytochemicals (Unger et al., 2012). In this investigation, docking simulations revealed significant binding of chicoric acid to the active sites of human CDC25A, CDC25B, and CDC25C phosphatases. Notably, chicoric acid exhibited the highest binding energy with CDC25B, indicating strong affinity compared to the established CDC25 phosphatase inhibitor, menadione. These findings unveil the cell cycle regulatory capacity of chicoric acid by targeting CDC25 subgroups, implying its potential as an antiproliferative agent capable of suppressing cancer cell growth (Figure 5D). The in vitro findings corroborate the in silico outcomes, thereby establishing a robust foundation for its efficacy.
Overall, the current study has established the cytotoxic and antiproliferative potential of chicoric acid against colon cancer cells. Previous studies on C. intybus have revealed that various parts of C. intybus contained caffeic acid derivatives, including chicoric acid, chlorogenic acid, isochlorogenic acid, and sesquiterpene lactones, with a notable presence of lactucin, lactucopicrin, 8-desoxylactucin, guaianolid glycosides encompassing chicoroisides B and C, sonchuside C, and so on. Since chicoric acid has not been fully explored yet for its therapeutic efficacy against cancer, the current study could be a significant contribution in the field of therapeutic drug development against colon as well as other cancers. However, further preclinical and clinical investigations are needed to fully explore the potential of chicoric acid as a leading drug candidate.
Conclusion
The findings of this study demonstrate that chicoric acid exerts an inhibitory solid impact on the growth of colon cancer cells by inducing apoptosis and cell cycle arrest. Chicoric acid-mediated growth suppression involved disruption of cellular and nuclear morphology and inner mitochondrial membrane potential, along with significant ROS generation and caspase activation in colon cancer cells. Moreover, chicoric acid led to programmed cell death in colon cancer cells by both intrinsic and extrinsic pathways, concomitantly with substantial cell cycle arrest at G0/G1. In silico analysis indicated that chicoric acid strongly binds to CDC25 phosphatases, which could be one of its possible mechanisms of action. The findings from this study propose that chicoric acid holds potential to be explored further in the drug development process for the treatment and management of cancer. Thus, further in vitro and in vivo studies will be essential to solidify its therapeutic role in combating cancer.
Footnotes
Abbreviations
None.
Author Contributions
M.A., M.V, S.F., and I.A.A. conceived and designed the project. I.A.A, N.A., and M.S. collected data from the literature. M.A., N.A., M.S., and I.A.A. analyzed the data and wrote the manuscript. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
NA.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors extend their appreciation to the Deputyship for Research and Innovation, Ministry of Education in Saudi Arabia for funding this research project through the project number RI-44-0849.
