Abstract
Background
Breast cancer, a malignant tumor, is the most common cancer type among women worldwide. Conventional chemotherapy treatments for breast cancer often cause adverse side effects in normal tissues and can lead to treatment failure. Plant-derived natural compounds offer an effective alternative with fewer side effects for cancer patients. Phyllanthus amarus is a medicinal plant widely distributed in China, India, and Brazil and reported to be used for the treatment of various types of cancer.
Objectives
This study was aimed to investigate the anticancer efficacy of methanolic extract of P. amarus against MCF-7 breast cancer cells.
Materials and Methods
We probed the anticancer activity in MCF-7 breast cancer cells by isolating the methanolic extract of P. amarus leaf extract (MEPA). The standard chemotherapy drug docetaxel is combined with MEPA to increase the drug efficacy. Moreover, the intracellular reactive oxygen species (ROS), alterations in mitochondrial membrane potential (MMP), nuclear condensation, and apoptotic activation were investigated in MCF-7 cells. Additionally, the customized PCR array comprising of cell cycle, apoptosis, mTOR, and JAK-STAT-related genes was studied during different MEPA treatments.
Results
In the MCF-7 breast cancer cells, the MEPA and docetaxel combination demonstrated strong cytotoxicity, with IC50 values of 45 and 2.6 µg/mL, respectively. MEPA and docetaxel together exhibited enhanced alterations in MMP and increased generation of ROS in MCF-7 cells. Furthermore, the combination of docetaxel with MEPA induces nuclear condensation and apoptotic cell death. During MEPA treatment, the gene expression profiles in MCF-7 cells were accessed. The JAK-STAT, mTOR signaling pathway, apoptosis, and cell growth-related genes were strongly affected by the dose-dependent administration of MEPA.
Conclusion
This study suggests that MEPA could be a useful alternative treatment for breast cancer. When combined with traditional chemotherapy, MEPA might offer better results with fewer side effects. More in vivo studies are needed to confirm MEPA’s effectiveness in animal models.
Introduction
Breast cancer is the second most frequent type of cancer worldwide in women. Over the course of the last 40 years, there has been an increase in the incidence of breast cancer. From 2010 to 2019, the rate grew by 0.5% annually, with localized-stage and hormone receptor-positive illness being the main cause of this growth (Giaquinto et al., 2022). The long-term survival rates for breast cancer following surgical or chemotherapy treatment remain a major hurdle due to high rates of recurrence and metastasis. It is widely classified into four subtypes based on receptor status such as luminal A, luminal B, HER2-positive, and triple-negative (Turner et al., 2021). The luminal A type of breast cancer is most common due to its high hormone sensitivity, mainly attributed to its elevated expression of estrogen receptors (ER) (Holliday & Speirs, 2011). Conventional treatment options for breast cancer are radiotherapy, chemotherapy, surgery, and hormone therapy (Waks & Winer, 2019). Unfortunately, recurrence and treatment resistance are two difficulties in the breast cancer treatment (Burguin et al., 2021). Metastases account for 30% of recurrent conditions in early-stage breast cancer patients (Pisani et al., 2002).
In this regard, medicinal herbs have gained significant recognition worldwide and have emerged as crucial components in the treatments of various diseases and possess therapeutic properties (Koparde et al., 2019). Natural compounds have held a significant part in the treatment and inhibition of diverse health issues, forming the foundation of traditional healing systems (Yuan et al., 2016). Moreover, numerous terrestrial plants such as Taxus baccata, Ginkgo biloba L., Euphorbia peplus, Combretum caffrum, Curcuma longa L., Podophyllum peltatum L., Artemisia annua L., Taxus brevifolia, Camptotheca acuminata Decne, and others remain to play a vital role in modern therapeutic approaches and particularly in the field of chemotherapy (Dehelean et al., 2021). These natural compounds have demonstrated their efficacy and continue to be extensively explored for their potential in treating various diseases (Cragg & Pezzuto, 2016). In some cases, these compounds may use along with the standard chemotherapeutics to improve their efficacy in cancer cells (Lichota & Gwozdzinski, 2018). Several phytochemicals and plant extracts have been reported to regulate cancer-related cellular signaling pathways and their related gene expressions in cancer cells (Tiwari et al., 2023; Wang et al., 2018). Determining the gene expression pattern of signaling pathways under the treatment conditions of various plant-based compounds may define the way to treat the cancer cells in personalized way.
Phyllanthus amarus is a therapeutic plant, widely found in tropical and subtropical regions, and has a past of traditional use as an herbal remedy (Bose Mazumdar Ghosh et al., 2022). Extensive research has revealed that the extract of P. amarus contains a diverse array of bioactive compounds, including flavonoids, alkaloids, triterpenes, polyphenols, lignans ellagitannins, proanthocyanins, and saponins (Patel et al., 2011), which display potent pharmacological activities, antioxidant (Van Tang Nguyen Quan Van Vuong & Scarlett, 2015) and anticancer activities (Poompachee & Chudapongse, 2012). Since no clinical research has been done on P. amarus extract, it is uncertain how they regulate cellular signaling pathways associated with breast cancer. Therefore, in our study, we observed the anticancer activity of methanolic leaf extract of P. amarus against MCF-7 breast cancer cells. Furthermore, a gene expression analysis with major cell signaling pathways related genes was studied in response to the methanolic extract of P. amarus in MCF-7 breast cancer cells.
Materials and Methods
Chemicals
Methanol, Dulbecco’s modified Eagle’s medium (DMEM) (Thermo-Fisher), fetal bovine serum (FBS) (Sigma–Aldrich), Trypsin EDTA (Sigma–Aldrich), antibiotic and antimycotic solution (Hi-media), phosphate buffer saline (PBS) (Hi-media), dichloro-dihydro-fluorescein diacetate (DCHF-DA), rhodamine-123 (Rh-123), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide MTT, dimethyl sulfoxide (DMSO), and all fluorescent dyes such as acridine orange (AO), ethidium bromide (EtBr), Hoechst 33342 were purchased from Sigma–Aldrich.
Plant Materials
P. amarus plants were collected and cleaned with sterilized distilled water. Subsequently, they were allowed to dry in shaded areas for 1 week. The leaves were then transformed into a powdered form using an electric blender. This powder was carefully stored in sealed containers until it was needed for further use.
Preparation of Methanolic Plant Extract
To prepare the extract, a measured amount of the powdered P. amarus (500 g) was mixed with methanol (90%) in a Soxhlet apparatus. The mixture was subjected to extraction, and the resulting extract was concentrated using a water bath at a controlled temperature below 60°C. Subsequently, the methanolic extract was suspended in distilled water for further use (Yao et al., 2018).
Cell Line Maintenance
MCF-7 cell lines were procured and cultured in DMEM, supplemented with FBS (10% FBS), antibiotics, and antimycotic solution with 5% carbon dioxide at a temperature of 37°C.
Cell Viability Assay
The viability of the cell is examined using the colorimetric MTT analysis. The cells were put down in a 96-well plate at a density of 1 × 103 cells per well, and they were then incubated for 24 h at 37°C. The treatment was given with increasing concentration of methanolic extract of P. amarus (MEPA) (0, 1, 10, 20, 40, 60, 80, 100 µg), docetaxel (0, 0.5, 1, 2, 4, 6, 8, 10 µg/mL), and combination of MEPA (0–100 µg/mL)/docetaxel (2.7 µg/mL) for 48 h, respectively. After the treatment incubation, the MTT reagent is added in the concentration of 1 mg/mL for another 4 h at 37°C. Following the removal of medium 100 µL of DMSO is added to dissolve the formed formazan crystals. At 570 nm, the formazan reduction is read in a microplate reader (Shimadzu, RF-5301 PC).
Measurement of Intracellular Reactive Oxygen Species (ROS) Formation
The dichloro-dihydro-fluorescein diacetate (DCFH-DA) dye is used to measure the intracellular ROS concentration. In the 6-well plate, the cells were grown and incubated until they reached confluency. Then, the treatment was given with MEPA, docetaxel, and combination of MEPA and docetaxel for further 48 h. Then in each well 1 µL of DCFH-DA (1 µg/mL) is added and incubated for 30 min in dark condition. After that, the cells were examined using a fluorescent microscope (Nikon TS-100) after being twice cleaned with PBS. A spectrofluorometer (Shimadzu, RF-5301 PC) was used to determine the fluorescence intensity with excitation and emission at 485 ± 10 nm and 530 ± 12.5 nm, respectively.
Measurement of Mitochondrial Membrane Potential (MMP)
The Rh-123 is a mitochondrial-specific dye, which binds and accumulates only in the healthy mitochondria in the cells and emits green fluorescence. In the 6-well plate, the cells were grown and incubated until they reached confluency. Then, the cells were treated with MEPA, docetaxel, and combination of MEPA and docetaxel for another 48 h. Then in each well, 1 µL of Rh-123 (1 µg/mL) was added and incubated for 30 min in dark condition. After washed twice using PBS, the images were captured using a fluorescence microscope (Nikon TS-100). Then the green fluorescence intensity was estimated with using a spectrofluorometer (Shimadzu, RF-5301 PC) with the excitation at 450 nm and emission at 490 nm.
Assessment of Apoptotic Cell Population
AO, a dye that permeates cells, and EtBr, a nonpermeable dye, are utilized to quantify the morphological alterations of apoptosis in cells. In the 6-well plate, the cells were grown and incubated until they reached confluency. Then, the treatment was given with MEPA, docetaxel, and combination of MEPA for another 48 h. After 48 h of treatment condition, the cells were incubated with AO/EtBr (1:1 ratio) 30 min in the dark. After washed twice with PBS, the cells were seen under a fluorescence microscope (TS-100 with NIS-E v3.1, Nikon). By dividing the apoptotic cells (EtBr-stained cells) by entire number of cells in the field ×100, the apoptotic population was calculated.
Determination of Deoxyribonucleic Acid (DNA) Fragmentation by Hoechst 33342 Staining
The structural changes of DNA by fragmentation and nuclear condensations indicate apoptosis. Cells were seeded in a 6-well plate and allow it to reach confluency. Then the cells were treated with MEPA, docetaxel, and combination of MEPA for 48 h. After 48-h incubation, the cells were rinsed with PBS and then fixed with methanol in cold condition for 10 min. After this, the cells were rinsed again with PBS, and Hoechst 33342 dye (1 µg/mL) was added at 37°C for 30 min. Then the cells were seen in a fluorescence microscope with blue filter (TS-100 with NIS-E v3.1, Nikon).
Polymerase Chain Reaction (PCR) Array Analysis
The MCF-7 cells were seeded in 60 mm cell culture Petri dishes and treated with the different concentrations of MEPA (10, 25, and 50 µg/mL). Then, the total mRNA was isolated using PureLink RNA Extraction Mini Kit (Thermo Scientific, USA) and quantified with Eppendorf spectrophotometer. The isolated mRNA was further converted into first strand cDNA with RevertAid First Strand cDNA Synthesis Kit (Thermo Scientific, USA) as per the manufacturer’s instructions. The custom PCR array (Qiagen) was designed with genes related to cell cycle regulation (CYR61, BTG1, CRYAB, HIST2H3D), apoptosis (Bcl-2, Bcl-xL, BAX, THBS1), mTOR (HIF1A), and JAK-STAT (PIM1). The cell cycle regulation, mTOR, and JAK-STAT pathway-related genes were selected based on the microarray analysis conducted by Amid et al. (2012). The gene amplification was carried out by quantitative real-time PCR system (Applied Biosystems™ StepOne™ Real-Time PCR System) using QuantiTectTM SYBR Green RT-PCR Kit. The GAPDH was used to normalize the gene expression and the fold changes in gene expression were calculated with 2(–△△ct).
Statistical Method
The data in the entire manuscript was represented as mean ± standard deviation (SD), and the statistical significance was calculated by one-way analysis of variance (ANOVA) using SPSS Statistical Software Suite. The values not sharing a common superscript differ significantly at p ≤ 0.05 vs. control Duncan’s multiple range test (DMRT).
Results
Effect of MEPA and Docetaxel on Cell Viability of MCF-7 Breast Cancer Cells
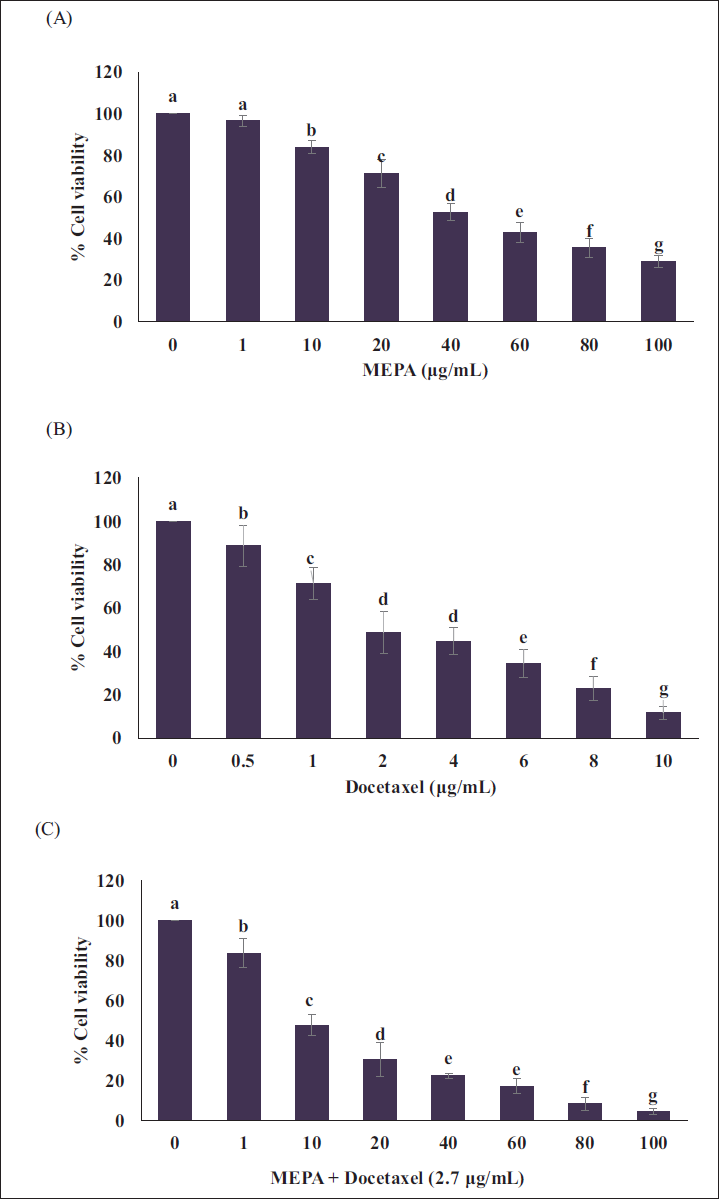
Initially, we have examined the cytotoxic effects MEPA on MCF-7 breast cancer cells (Figure 1A). The MCF-7 cells were treated with the increasing concentration of MEPA and found the IC50 value of 45 µg/mL. Further, we observed the cytotoxic effects of docetaxel in MCF-7 cells with IC50 of 2.7 µg/mL (Figure 1B). Interestingly, the cotreatment of MEPA and docetaxel at increasing concentration significantly decreased the viability of MCF-7 cells (Figure 1C).
Effect of (A) Methanolic Extract of Phyllanthus amarus (MEPA), (B) Docetaxel, (C) MEPA + Docetaxel (2.7 µg/mL) Cytotoxicity on MCF-7 Breast Cancer Cells for 48 h in a Dose-Dependent Manner. The Points with Error Bar are Represented as Mean ± Standard Deviation (SD) of Three Independent Experiments and Values Not Sharing a Common Superscript Differ Significantly at p ≤ 0.05 [Duncan’s Multiple Range Test (DMRT)].
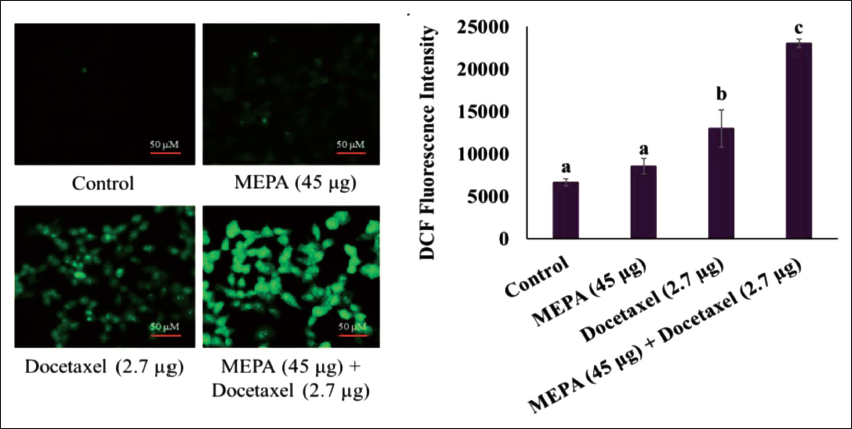
Effect of MEPA and Docetaxel on Intracellular ROS Formation in MCF-7 Breast Cancer Cells
Increased ROS levels are recognized to have a part in cell death brought on by a variety of stressors. We investigated whether MEPA-induced cell death correlated with increased ROS levels in MCF-7 cells. In this experiment, the standard chemotherapeutic, docetaxel treatment showed ROS production compared with MEPA alone and the control group. Interestingly in combination with MEPA (45 µg/mL) and docetaxel (2.7 µg/mL), we found increased production of ROS formation while compared to docetaxel alone treated group (Figure 2).
Effect of Methanolic Extract of Phyllanthus amarus (MEPA) and Docetaxel Combination in Intracellular Reactive Oxygen Species (ROS) Generation in MCF-7 Breast Cancer Cells for 48 h. The Fluorescence Images are Acquired Using Fluorescence Microscope and the Bar Diagram Depicts the Fluorescence Intensity of DCF Under MEPA and Docetaxel Treatment in MCF-7 Cells. The Points with Error Bar are Represented as Mean ± Standard Deviation (SD) of Three Independent Experiments and Values Not Sharing a Common Superscript Differ Significantly at p ≤ 0.05 [Duncan’s Multiple Range Test (DMRT)].
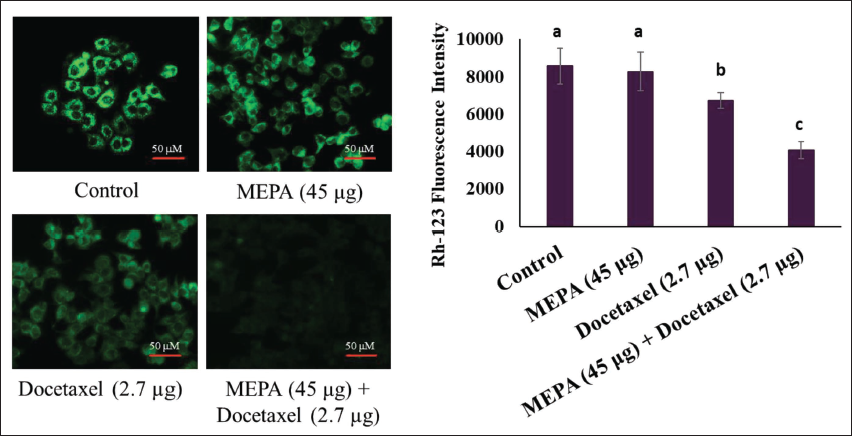
Effect of MEPA and Docetaxel on MMP in MCF-7 Breast Cancer Cells
The disruption of MMP is the early sign of apoptosis and cell death. We examined the role of MEPA on alterations in MMP in MCF-7 breast cancer cells. The MEPA (45 µg/mL) and docetaxel (2.7 µg/mL) alone treated group showed minimal alterations in MMP in breast cancer cells. Fascinatingly, the cotreatment of MEPA and docetaxel increased the alteration in MMP with a decrease in the accumulation of Rh-123 fluorescence intensity in MCF-7 breast cancer cells (Figure 3).
Effect of Methanolic Extract of Phyllanthus amarus (MEPA) and Docetaxel Combination on Mitochondrial Membrane Potential (MMP) Alterations in MCF-7 Breast Cancer Cells for 48 h. The Fluorescence Images Acquired Using Fluorescence Microscope and the Bar Diagram Depict the Fluorescence Intensity of Rhodamine Under MEPA and Docetaxel Treatment in MCF-7 Cells. The Points with Error Bar are Represented as Mean ± Standard Deviation (SD) of Three Independent Experiments and Values Not Sharing a Common Superscript Differ Significantly at p ≤ 0.05 [Duncan’s Multiple Range Test (DMRT)].
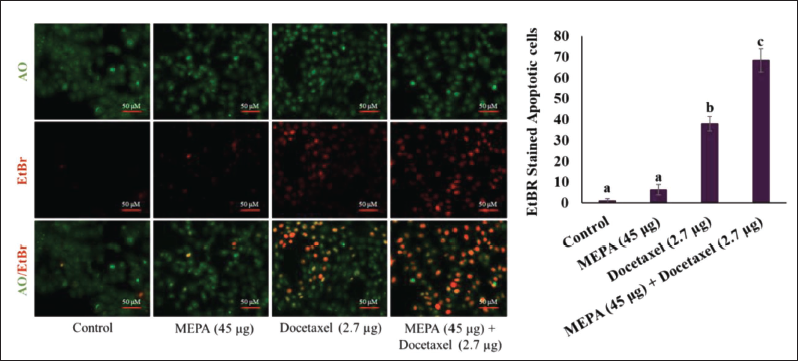
Effect of MEPA and Docetaxel on Apoptotic Cell Death in MCF-7 Breast Cancer Cells
Changes in the morphology of cell structure are a hallmark of cell apoptosis. The MEPA was extensively studied for the apoptotic-inducing property in MCF-7 cells. Our results showed that the MEPA (45 µg/mL) and docetaxel (2.7 µg/mL) treated cells showed 8.3 ± 2.08 and 38 ± 3.6 apoptotic cell population compared to the untreated control cells. Eventually, the cotreatment of MEPA and docetaxel treatment significantly increased the apoptotic cell population to 68.3 ± 5.6 in MCF-7 breast cancer cells and results showed significantly a greater quantity of apoptotic cells while compared to docetaxel combined group (Figure 4).
Effect of Methanolic Extract of Phyllanthus amarus (MEPA) and Docetaxel Combination of Apoptotic Cell Population in MCF-7 Breast Cancer Cells for 48 h. The Fluorescence Images Acquired Using Fluorescence Microscope and the Bar Diagram Depict the Fluorescence Intensity of Acridine Orange (AO)/Ethidium Bromide (EtBr) Under MEPA and Docetaxel Treatment in MCF-7 Cells. The Apoptotic Population was Calculated by Dividing the Apoptotic Cells by the Entire Cells in the Field ×100. The Points with Error Bar are Represented as Mean ± Standard Deviation (SD) of Three Independent Experiments and Values Not Sharing a Common Superscript Differ Significantly at p ≤ 0.05 [Duncan’s Multiple Range Test (DMRT)].
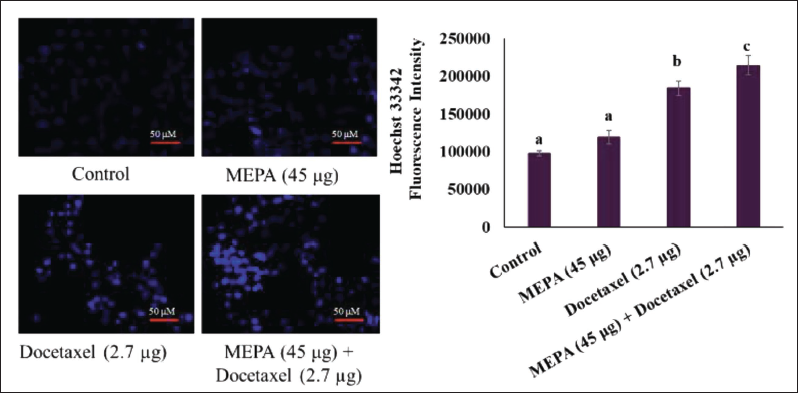
Effect of MEPA and Docetaxel on Nuclear Condensation in MCF-7 Breast Cancer Cells
Nuclear condensation and DNA fragmentation will eventually lead to the initiation of apoptosis in cancer cells. The amount of Hoechst 33342 fluorescence intensity levels indicates the levels of the nuclear fragmentation in the cells. We examined the DNA damaging effects of MEPA in MCF-7 breast cancer cells. It was found that MEPA (45 µg/mL) and docetaxel (2.7 µg/mL) treatment showed the basal fluorescence intensity of Hoechst 33342 in MCF-7 breast cancer cells. Interestingly, the combination of MEPA and docetaxel treatment significantly enhanced the fluorescence intensity of Hoechst 33342 compared to MEPA and docetaxel alone treated cells (Figure 5).
Effect of Methanolic Extract of Phyllanthus amarus (MEPA) and Docetaxel Combination of Nuclear Condensation in MCF-7 Breast Cancer Cells for 48 h. The Fluorescence Images are Acquired Using Fluorescence Microscope and the Bar Diagram Depicts the Fluorescence Intensity of Hoechst Under MEPA and Docetaxel Treatment in MCF-7 Cells. The Points with Error Bar are Represented as Mean ± Standard Deviation (SD) of Three Independent Experiments and Values Not Sharing a Common Superscript Differ Significantly at p ≤ 0.05 [Duncan’s Multiple Range Test (DMRT)].
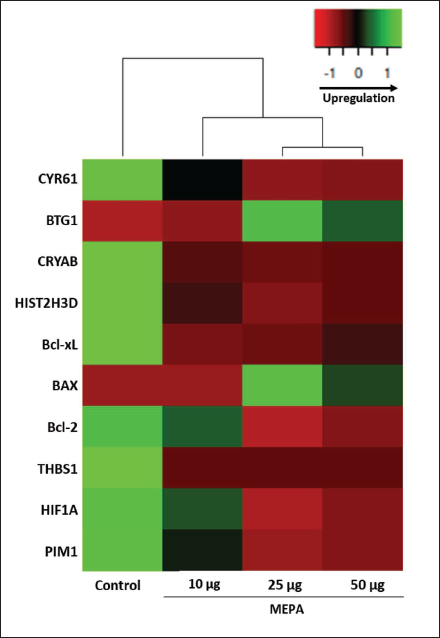
Effect of MEPA on Cancer Cell Proliferation-related Gene Expressions in MCF-7 Breast Cancer Cells
The gene expression pattern of apoptosis, JAK/STAT pathway cellular proliferation, mTOR, and cell cycle arrest was screened under the MEPA treatment conditions in MCF-7 breast cancer cells. We found that increasing concentration of MEPA significantly upregulated the BTG1 and BAX gene. In contrast, its doses dependently downregulated the genes of CYR61, CRYAB, HIST2H3D, Bcl-2, Bcl-xL, THBS1, HIF1A, and PIM2 in MCF-7 breast cancer cells (Figure 6).
Effects of Methanolic Extract of Phyllanthus amarus (MEPA) on Gene Expression Profile of Genes Related to Cellular Proliferation, Apoptosis, mTOR, and JAK-STAT Signaling Pathway. The mRNA was Isolated and Reverse Transcribed from MCF-7 Breast Cancer Cells Under Dose-Dependent Treatment of MEPA for 48 h. The Clustergram Heatmap was Generated Using Heatmapper.ca Online Tool.
Discussion
Breast cancer resists chemotherapy by maintaining continuous proliferative signals and bypassing mechanisms that suppress tumor growth. Cellular signaling pathways highly correlate with the proliferation of cancer cells. Various chemotherapeutics have been used to treat advanced breast tumors, but despite decades of efforts, they failed in clinical practice because of their adverse side effects (Anand et al., 2023). Alternatively, plant-based medicines are employed to reduce the possibility of adverse effects. Natural substances that are derived from plants are easily accessible and readily assimilated in the body. Moreover, they tend to have fewer adverse effects and lower toxicity compared to conventional treatments (Jain et al., 2021).
P. amarus and its phytocomponents have been extensively studied for their antitumor properties in several cancer cells (Ahmad et al., 2015; Paul et al., 2019). The methanolic crude extracts of P. amarus contain phenolics, flavonoids, saponins, proanthocyanins, gallic acid, rutin, and catechin in higher concentrations (Nguyen et al., 2017). These combinations of phytochemicals might be the reason for antitumorous properties of P. amarus. The extract of P. amarus has been shown to induce apoptosis and decrease breast cancer cell proliferation (Abhyankar et al., 2010). Additionally, it has been demonstrated to prevent migration, adhesion, and invasion in breast and metastatic lung cancer (Lee et al., 2011). Therefore, in our study, we examined the anticancer properties of MEPA against MCF-7 breast cancer cells. We found MEPA significantly increased the cytotoxicity in MCF-7 breast cancer cells. The secondary metabolites present in the dimethyl formamide extract of P. amarus decreased the cell viability of T47D breast cancer cells dose dependently (Pammi & Giri, 2021). Similar to this, lignans found in the nontransformed root of P. amarus have been shown to effectively inhibit the proliferation of HeLa cervical cancer cells (Sparzak et al., 2015).
Since plant-derived substances have lesser anti-cancer efficacy than traditional anticancer medications, numerous preclinical studies have shown that combining phytochemicals combined with conventional anti-cancer drugs is more effective than using phytochemicals alone in the treatment of cancer (Kumar et al., 2023). The continuous exposure of traditional chemotherapeutics develops chemotherapeutic resistance and modulates epigenetics in cancer cells. Thus, the minimal concentration of traditional anticancer drugs with the nontoxic effective plant-derived substances will be the novel treatment strategy to defend cancer. In our study, we combined the MEPA with docetaxel to effectively improve the antitumorigenic properties in breast cancer cells. Interestingly, we found the combination of MEPA and docetaxel efficiently lowered the cell proliferation of MCF-7 breast cancer cells than the MEPA and docetaxel alone treatments. Similarly, Phyllanthus niruri spray dried extract combined with cisplatin significantly displayed the cytotoxic effects in HT29 and HepG2 cancer cell lines (Araújo et al., 2012). Dirican et al. (2015) report that thymoquinone and docetaxel combination synergistically exerts cytotoxic activity and initiate apoptosis in DU-145 human prostate cancer cells.
Phytochemicals have been found to exhibit anticancer properties through a mechanism involving the production of oxidative stress (Fatima et al., 2021; Jang & Lee, 2023). In 2012, Suski et al. demonstrated the strong correlation between alterations in MMP and increased ROS generation (Suski et al., 2012). The increased ROS subsequently leads to the reduction in antioxidant levels and damage important biomolecules such as DNA, RNA, proteins, and lipids (Vallejo et al., 2017). In our investigation, we found that MEPA treatment altered the MMP level in MCF-7 cells when compared to the control. Furthermore, the MEPA and docetaxel combination dramatically decreased the cellular MMP, which ultimately caused the MCF-7 breast cancer cells to produce more ROS. The combination of MEPA and docetaxel treatment considerably increased the ROS generation than the MEPA and docetaxel alone treatment group. The different plant sources from Phyllanthus species reported to be induce intracellular ROS generation mediated by MMP in cancer cells (Abhyankar et al., 2010; Huang et al., 2014; Stefanowicz-Hajduk et al., 2016). Geraniin isolated from Phyllanthus urinaria significantly produced intracellular ROS in MCF-7 breast cancer cells and possess potent cytotoxicity. Similarly, phyllanthin, a lignan isolated from Phyllanthus species, caused loss of MMP and increased ROS generation in MOLT-4 T-lymphoblast cells (Wang et al., 2021). Foster et al. (2020) reported that docetaxel activity on DU-145 prostate cancer cell lines was improved by 50% when combined with ethyl acetate bark extract of Annona muricata L.
The cytotoxic drugs particularly mediate DNA damage and eventually promote apoptosis in cancer cells (Pfeffer & Singh, 2018). Docetaxel was found to damage DNA and cause senescence in TC-1 and TRAMP-C2 cell lines (Simova et al., 2016). Additionally, it can cause cancer cells that are either treated alone or in combination with medicinal plants to undergo apoptosis. The apoptotic morphological alterations in the MEPA and docetaxel combination group were observed in our studies using the AO/EtBR dual staining apoptosis assay. The hallmark alterations of apoptosis brought on by chemotherapy include nuclear condensation, apoptotic bodies, and membrane blebbing (Pfeffer & Singh, 2018). In MCF-7 breast cancer cells, we investigated the potential for DNA damage caused by the combination of MEPA and docetaxel treatment. We observed that MCF-7 cells had more nuclear condensed cellular structures than the cells treated with MEPA and docetaxel alone. Thus, we are showcasing MEPA’s ability to damage DNA and induce apoptosis in MCF-7 breast cancer cells. Similarly, Bahamin et al. (2021) reported that docetaxel combined with extracts from Amygdalus haussknechtii and Alhagi maurorum might induce apoptosis in 4T1 breast cancer cells. In prostate cancer cell lines, the combination of Ganoderma lucidum polysaccharide with Docetaxel markedly increased apoptotic cell death (Rahimnia et al., 2023).
Under various MEPA treatment conditions, the gene expression patterns of key cellular signaling pathways, including apoptosis, mTOR, JAK-STAT, and cell cycle arrest, were examined in MCF-7 breast cancer cells. For the gene expression experiments, markers including CYR61, BTG1, CRYAB, HIST2H3D, Bcl-2, Bcl-xL, BAX, THBS1, HIF1A, and PIM2 were employed. We observed that the genes linked to cell cycle arrest, such as CYR61, CRYAB, and HIST2H3D, were downregulated while BTG1 was upregulated in increasing concentration of MEPA in MCF-7 cells. Additionally, there was a considerable downregulation of apoptotic-related genes such Bcl-Xl, and Bcl-2, and an enhancement of BAX overexpression. The mTOR signaling pathway-related HIF1A gene and the JAK-STAT pathway-related PIM1 gene were found to be downregulated in the dose-dependent treatment of MEPA in the MCF-7 breast cancer cells. The downregulation of CYR61, CRYAB, and HIST2H3D promotes cell growth arrest and leads to apoptosis in various cancer cell lines (Ascenção et al., 2022). The different plant extracts control key apoptotic genes like Bcl-2, Bcl-xL, and BAX, which greatly accelerates apoptosis in breast cancer cells (Rai et al., 2023). The HIF1A gene related to hypoxia-mediated signaling through mTOR leads to tumor progression and tolerance toward hypoxia in cancer cells (Muz et al., 2015). Thus, downregulation of HIF1A might reduce the proliferate rate of breast cancer cells. The PIM1 regulated by JAK-STAT pathway involved in the tumor growth, cell survival, drug resistance, and differentiation (Zhang et al., 2018). Therefore, downregulating the expression of PIM1 can reduce breast cancer progression and metastasis. The gene expression analysis confirmed that the cellular signaling-related genes expressed differentially in MCF-7 cells and conversely regulated by MEPA treatment to inhibit cell growth activate apoptosis and prevent mTOR and JAK-STAT pathway initiation. Thus, the MEPA has the important role in the molecular mechanism to regulate cell signaling pathways to promote antitumorous activity in MCF-7 breast cancer cells.
Conclusion
Our investigation reveals that the MEPA exhibits significant anticancer effects on MCF-7 breast cancer cells. MEPA synergistically enhances the activity of docetaxel by increasing cellular cytotoxicity, altering MMP, and promoting ROS production. Additionally, the combination of MEPA and docetaxel induces DNA damage and apoptosis-mediated cell death in MCF-7 cells. MEPA also regulates cellular signaling pathways associated with cancer cell proliferation, thereby inhibiting cell growth. Collectively, these results highlight the potential of MEPA as an effective agent against MCF-7 breast cancer cells. However, further in vivo research is required to confirm MEPA’s effectiveness in animal models.
Footnotes
Abbreviations
AO: Acridine orange; DCFH-DA: Dichloro-dihydro-fluorescein diacetate; DMRT: Duncan’s multiple range test; EtBr: Ethidium bromide; MEPA: Methanolic extract of Phyllanthus amarus; MMP: Mitochondrial membrane potential; RH-123: Rhodamine-123; ROS: Reactive oxygen species.
Declaration of Conflicting Interest
The authors declared no potential conflicts of interest regarding the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable as there is no animal or human subjects involved in the experiments.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
