Abstract
Objectives
The present study aims to examine the effects of baicalin on the human medullary breast carcinoma (MBC) cell line Bcap-37 and to determine whether baicalin regulates Bcap-37 cell apoptosis through the ERK/p38 mitogen-activated protein kinase (MAPK) signaling pathway.
Materials and Methods
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay and Transwell assay were utilized to measure the effects of baicalin on the proliferation and migration ability of Bcap-37 cells, respectively. Flow cytometer analysis was implied to detect the effects of baicalin on Bcap-37 cell apoptosis. Real-time quantitative polymerase chain reaction (RT-PCR) was conducted to observe the influence of baicalin on mRNA expressions of apoptosis-related genes. Western blot was executed to further explore the action of baicalin on apoptosis-related proteins. PD98059 (a specific inhibitor of ERK) and SB203580 (a specific inhibitor of p38 MAPK) were used to further clarify the intrinsic mechanism of baicalin regulating apoptosis in Bcap-37 cells.
Results
Compared with the control group, baicalin significantly inhibits the proliferation activity of Bcap-37 cells in a concentration- and time-dependent manner, with a p-value < 0.05. The transwell assay indicated that the migration viability of cells decreased further, followed by the increased concentration of baicalin, and the p-value had a statistical difference. Besides, flow cytometry was conducted to assess the effects of baicalin on the early and late apoptosis rates of Bcap-37 cells, and results showed that baicalin highly promoted the apoptosis level both at the early and late stages with a statistical difference in a concentration-dependent manner (p < 0.05). Results of RT-PCR presented that, compared with the control group, baicalin significantly activated the mRNA expression of Bax, p38, and p-ERK1 and abolished the mRNA expression of Bcl-2 in every dosing group in a concentration-dependent way (p < 0.05). Western blot exhibited that, compared with the control group, baicalin promoted the protein expression of caspase-3, caspase-9, Bax, p38, P-ERK, and p53 while playing an opposite function to Bcl-2 in each dosing group (p values have statistical differences). At last, PD98059 and SB203580 were applied to explore the potential mechanism of baicalin in apoptosis promotion. And results revealed that, compared with the group treated with baicalin alone, protein expression of Bax, p38, p-ERK, caspase-3, and caspase-9 was downregulated obviously in the group treated with both baicalin and PD98059 or SB203580 (p < 0.05). The p53 expression inhibition showed a difference only when compared to the group adding PD98059. Compared with the blank group, a statistical difference was only observed in Bcl-2 expression in the group treated with baicalin in combination with PD98059 or SB203580 (p < 0.05). Interestingly, a p53 expression difference was only displayed between the blank group and the group treated with baicalin and SB203580 (p < 0.05).
Conclusion
Our study observed that baicalin inhibits the proliferation and migration ability of Bcap-37 cells. In addition, baicalin induces cell apoptosis via the ERK/p38 MAPK signal pathway.
Introduction
According to the latest global cancer statistics, breast cancer remains the most commonly diagnosed cancer and the leading cause of cancer death in women in most countries worldwide (Sung et al., 2021). Medullary breast carcinoma (MBC) is a special type of infiltrating breast cancer, accounting for approximately 5%–10% of cases (Qin et al., 2021). MBCs display similar gene expression profiling to basal breast cancer and thus were clarified as a subgroup of triple-negative basal-like cancers. They are generally negative for estrogen receptor (ER), and HER2 is frequently associated with BRCA1 and p53 mutations (Bertucci et al., 2006; Lee et al., 2022; Romero et al., 2018). MBC has been sub-divided into typical medullary carcinoma and atypical MBC (Lee et al., 2022; Qin et al., 2021). MBC has been reported to have a favorable prognosis in general, while atypical MBC is characterized by a poor prognosis (Qin et al., 2021). Clinically, adjuvant therapies like chemotherapy, endocrine therapy, targeted therapy, radiation therapy, and Chinese medicine therapy can absolutely improve the prognosis of breast cancer. Nevertheless, deficiencies remain in adjuvant therapies for MBC.
Cell death is usually divided into two categories: apoptosis and necrosis. Apoptosis is a programmed cell death, a tightly regulated intracellular process that maintains organisms’ homeostasis and fine-tuned life cycle (Xu, et al., 2019). Aberrant apoptosis inhibition is involved in plenty of diseases, including cancer (Murray & Gibson, 2022). Moreover, numerous studies have shown that various chemotherapy drugs may obliterate cancer cells by inducing apoptosis (Wang et al., 2022). It has been reported that the mitogen-activated protein kinase (MAPK) signaling pathway plays a key role in cell apoptosis (Sun et al., 2019). Abnormal activation of MAPK signaling pathways leads to a series of unfavorable processes, including the impairment of apoptotic mechanisms, cell malignant transformation, and aberrant proliferation, as well as the facilitation of tumor development and progression and metastasis promotion (Jiang et al., 2020; Liu et al., 2022). Previous studies observed that the MAPK signaling pathway played a crucial role in breast cancer tumorigenesis and progression (Ventura-Bixenshpaner et al., 2018; Yi et al., 2020). Besides, research has indicated that one of three members of MAPK, the p38-MAPK-mediated signaling pathway, plays an indispensable role in bone metastasis for breast cancer (Jie et al., 2022).
Baicalin (BAI), a flavonoid isolated from Scutellaria baicalensis Georgi derived from the Shanxi and Shandong provinces of China, is known as a major Traditional Chinese Medicine herb. Baicalin exhibits a variety of pharmaceutical properties, including antioxidant, anti-inflammatory, antiallergic, embryo-protective, cardioprotective, hepatoprotective, renal-protective, neuroprotective, antimicrobial, antipyretic, detoxifying, and antipathogen (Ganguly et al., 2022; Wang et al., 2018). Also, it has a long history of use in the treatment of plenty of symptoms related to cancer (Cheng et al., 2018). Studies indicated that baicalin presents the ability to suppress the progression of cancer cells without systemic toxicity in plenty of cancers, including breast, colon, hepatic, leukemia, lung, and skin (Singh et al., 2021). Besides, literature also revealed that baicalin induces apoptosis in various organ-specific cell lines (such as breast, cervical, etc.) by modulating diverse relevant signaling pathways (Singh et al., 2021). For example, Zhou et al. (2009) observed that the combination of baicalin inhibits the growth of breast cancer cell lines (MCF-7 and MDA-MB-231) and enhances apoptosis of MCF-7 via ERK/p38 MAPK signaling pathways. As a special type of infiltrating breast cancer, MBC has limited adjuvant treatment, and there is little research on the effects of baicalin in it. Therefore, our research aimed to explore and clarify the potential function of baicalin in the MBC cell line Bcap-37.
Materials and Methods
Materials
Baicalin was purchased from Shanghai Yuanye Biotechnology Co. Ltd. The purity of baicalin used in this study was more than 99.90%, and the dry powder was added to dimethyl sulfoxide (DMSO) and was fully dissolved to formulate into a series concentration of 0 (NC), 50, 100, 150, 200, and 250 µmol/L, respectively. All antibodies were procured from Cell Signaling Technology (Boston, MA, USA). PD98059 and SB203580 were acquired from Biomol (Philadelphia, PA, USA).
Cell Culture
Human medullary breast cancer Bcap-37 cells used in this experiment were the 10th and 11th generations, with the same human leukocyte antigen (HLA) typing and no changes in HLA antigen phenotype. It was purchased from the Cell Bank of Shanghai Academy of Sciences and was cultured in Roswell Park Memorial Institute-1640 medium (Gibco) containing 10% fetal bovine serum (FBS) at 37℃ and 5% CO2 in a humidified atmosphere. When the cells are fused to 70%–80%, aspirate the culture solution, and wash the cells gently three times with normal temperature phosphate-buffered saline (PBS) water. Then, add 1 mL of 0.25% trypsin solution, place in an incubator for 2 min, and observe under a microscope. When most of the cells were free and fell off, the digestion was terminated with a 10% FBS culture solution. The cell suspension was collected into a centrifuge tube and centrifuged at 1000 rpm for 5 min, discarded the supernatant, and resuspended the cells in culture medium. Inoculate the cells in a new petri dish to reach the concentration required for subsequent studies.
Cell Growth Assay
Bcap-37 cells suspension was seeded into 96-well flat-bottom plates and incubated at 5% CO2 at 37℃ until a cell monolayer formed. Then, six concentration gradients (0, 50, 100, 150, 200, and 250 µmol/L) of baicalin were set up with three replicate wells per concentration. After incubating for 24, 48 and 72 h, cells were observed and counted using an inverted microscope. Ten microliters of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) solution (5 mg/mL, 0.5% MTT) was added to each well, and cells were incubated for an additional 2 h. Thereafter, the culture medium was aspirated, and the wells were washed with PBS three times, 100 uL DMSO was added to each well. The plates were shaken at low speed for 10 min to dissolve the crystals completely. Finally, the absorbance value of each well was detected at the 490 nm using enzyme-linked immunosorbent assay (ELISA) plate reader (BioTek, Winooski, VT, USA). The inhibition rate (IR) (%) was calculated as the following formula:
Inhibition rate (IR) (%) = (1 – ODSample/ODControl) × 100%
Transwell Assay
Bcap-37 cells (5×105) were plated into the upper side of a Transwell chamber, and each chamber was added with 200 uL serum-free medium. Meanwhile, 600 µL of culture medium was added to the bottom chamber. Then cells were cultured at 37°C in a 5% CO2 incubator for 48 h. Afterward, the small chamber was removed; the cells in the upper chamber were carefully wiped off with cotton swabs and were immersed in formaldehyde and fixed for 5 min at room temperature. After aspirating the fixative, the vignin was stained for 15 min and washed in tap water. The vignin-stained slides from the last water rinse were counterstained with eosin for 5 min and rinsed with tap water. The stained membranes were cut and placed on a glass slide, and the number of invading cells on the bottom surface of the membrane was observed and counted under an optical microscope, and pictures were taken under the microscope (Olympus Corporation Inc., Japan).
Flow Cytometric Analysis
The Bcap-37 cells were seeded in the six-well plates at a density of 4×105 cells per well and cultured for 24 h. They were then starved in a medium without FBS for 24 h followed. Then it was treated with a series of concentrations (0, 50, 100, 150, 200, and 250 µmol/L) of baicalin for 48 h. Subsequently, cells were incubated in PBS containing 100 ug/mL RNase and 40 ug/mL propidium iodide at room temperature for a duration ranging from half to 1 h before flow cytometry analysis. Cell cycle distribution and DNA content were assessed utilizing a Coulter Epics V instrument (Beckman Coulter, Inc., Fullerton, CA). The percentage of apoptotic cell population and cell cycle were carried out via Elite 4.0 software (Phoenix Flow System, San Diego, CA).
Quantitative RT-PCR
Bcap-37 cells were exposed to baicalin at a range of concentrations (0, 50, 100, 150, 200, and 250 µmol/L) for 48 h. The total RNAs were extracted and followed by reverse transcription, then two-step real-time quantitative polymerase chain reaction (RT-PCR) was utilized to detect the mRNA expressions of Bax, Bcl-2, p38, p-ERK1, and p-ERK2, normalized by the mRNA level of GAPDH. The primer sequences of genes are represented in Table 1. RT-PCR was implemented using the MasterCycler Gradient PCR machine as follows: predenaturation at 95℃ for 30 s; 40 cycles of denaturation at 95℃ for 5 s and annealing at 60℃ for 34 s; melting curves obtained at 65℃–95℃.
Primer Sequences of GAPDH and Five Signatures (Bax, Bcl-2, p38, p-ERK1, and p-ERK2).

Western Blot Analysis
Total proteins were extracted and quantified via a bicinchoninic acid kit, with equal amounts of proteins fractionated on 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinylidene fluoride membranes. The protein expression level was analyzed using a primary antibody (1:1000, except p53 1:200, Cell Signaling Technology) and a secondary antibody (1:800, Cell Signaling Technology) that conjugated with horseradish peroxidase. Relative expression levels of p53, caspase-3, caspase-9, Bcl-2, Bax, p-ERK, p38, and GAPDH (the loading control) were investigated and qualified for this approach. Followed by enhanced chemiluminescence (ECL) staining utilizing the ECL system (Amersham Biosciences). Quantitative analysis was conducted through Alpha Ease FC (FluorChem FC2) software.
Statistical Analysis
Data analysis was executed via SPSS 18.0 statistical software, and the measurement data was presented in the format of means ± standard deviation (x ± s). Group comparisons were executed by the Student’s t-test and one-way analysis of variance (ANOVA). A significance threshold of p < 0.05 was reckoned to be statistically significant.
Results
Baicalin Inhibits the Proliferation of Bcap-37 Cells
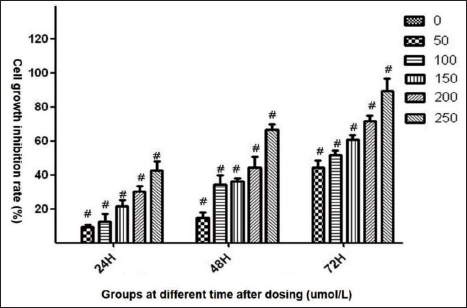
Baicalin at various concentrations of 0 (NC), 50, 100, 150, 200, and 250 µmol/L was used to treat breast cancer cell Bcap-37, and its ability to inhibit growth was explored in 24, 48, and 72 h, respectively. Figure 1 exhibited the condition of Bcap-37 cells treated with baicalin at a series of concentrations for 48 h, and results indicated that the proliferation of Bcap-37 was significantly inhibited, followed by an increase in concentration. Figure 2 suggests that cell growth IRs were significantly increased, followed by exposed time extension and gradually increased concentrations of baicalin, and the p-value had statistical significance in every dosing group compared to the control group (p < 0.05).
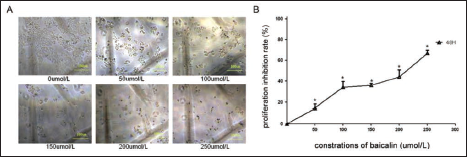
Baicalin Inhibits the Migration of Bcap-37 Cells
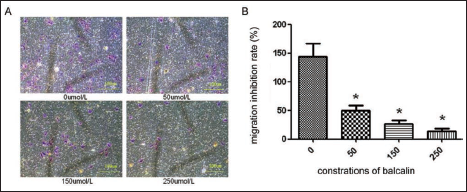
To evaluate whether baicalin affects the migration of Bcap-37 cells, a Transwell assay was conducted. Cells were treated with different concentrations of 0 (NC), 50, 150, and 250 µmol/L of Baicalin for 48 h. Under 100× high magnification, we found that the number of migrated cells passing through the polycarbonate membrane increased significantly with the increase in the concentration of baicalin, with a statistical difference (Figure 3). As shown in Table S1, the definite amount of each dosing group was 144.00 ± 27.22, 69.67 ± 9.76, 26.67 ± 6.88, and 14.00 ± 4.27, respectively. And statistically significant disparities were observed between the control group and the three dosage groups (*p < 0.05).
Baicalin Induces Apoptosis of Bcap-37 Cells
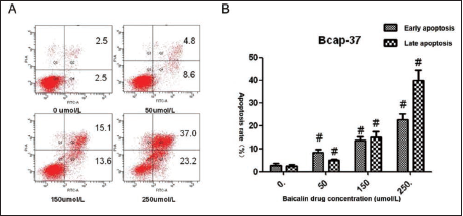
To further study the effects of baicalin on apoptosis of Bcap-37 cells, we treated cells with baicalin at various concentrations at 0 (NC), 50, 150, and 250 µmol/L for 48 h and detected the early and late apoptosis via flow cytometry. As Figure 4 shows, the number of apoptotic cells increased, obviously followed by the gradually raised concentration of baicalin. We also investigated the early and late apoptosis rates (%) of cells in the control and three dosing groups. Consistent with Figure 4, compared with the control group, the apoptosis rates of three dosing groups were higher not only in the early stage (2.58 ± 0.97 vs 8.5 ± 1.27, 13.6 ± 1.47, 22.7 ± 2.38, p < 0.05) but also in the late stage (2.38 ± 0.72 vs 5.0 ± 0.39, 15.0 ± 2.39, 39.8 ± 4.59, p < 0.05) of the period of cell apoptosis (Table S2).
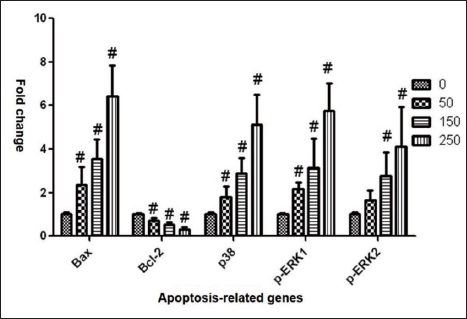
Baicalin Regulates the mRNA Expression of Apoptosis-related Genes in Bcap-37 Cells
To further probe the potential mechanism of baicalin in the apoptosis of medullary breast cancer cells (Bcap-37), first of all, we treated Bcap-37 cells with different concentrations of 0 (NC), 50, 150, and 250 µmol/L of baicalin for 48 h, extracted the total RNA, conducted reverse transcription, and PCR analysis followed. Results exhibited in Table S3, we found that in contrast to the control group, the mRNA expression level of Bax (1.00 ± 0.08 vs 2.37 ± 0.82&, 3.55 ± 0.91&, and 6.41 ± 1.45&, &p < 0.05), p38 (1.00 ± 0.10 vs 1.81 ± 0.49&, 2.88 ± 0.70&, and 5.11 ± 1.39&, &p < 0.05), p-ERK1 (1.00 ± 0.04 vs 2.16 ± 0.32&, 3.14 ± 1.35&, and 5.76 ± 1.25&, &p < 0.05), p-ERK2 (1.00 ± 0.09 vs 1.66 ± 0.45, 2.70 ± 1.09&, and 4.12 ± 1.82&, &p < 0.05) all were increased significantly in three dosing groups, while the mRNA expression of Bcl-2 was opposite (1.00 ± 0.05 vs 0.71 ± 0.12&, 0.51 ± 0.14&, and 0.29 ± 0.11&, &p < 0.05). Data on the mRNA expression of the genes above was visualized in Figure 5. Thus, we could conclude that, compared to the control group, all three concentrations of baicalin could reduce the expression of apoptosis-related signatures of Bcl-2 while promoting the levels of Bax, p38, p-ERK1 and p-ERK2, and most of the differences were statistically significant (p < 0.05).
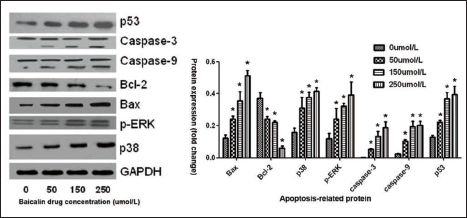
Baicalin Regulates the Protein Expression of Apoptosis-related Genes in Bcap-37 Cells
Next, various concentrations of 0 (NC), 50, 150, and 250 µmol/L of baicalin were applied to Bcap-37 cells for 48 h. Then, we conducted a Western blot to find out whether baicalin affects the protein levels of apoptosis-related genes, which encompassed p53, caspase-3, caspase-9, Bcl-2, Bax, p-ERK, and p38. From the results of the Western blot, we discovered that, compared with the control group, p53, caspase-3, caspase-9, Bax, p-ERK, and p38 protein expression were obviously activated, followed by an increase in the concentration of baicalin. On the contrary, with the increased dosage of baicalin, Bcl-2 expression decreases significantly. In contrast to the negative group, all of the dosage groups in every signature showed statistical significance (p < 0.05) (Figure 6). The statistical analysis is represented in Table S4.
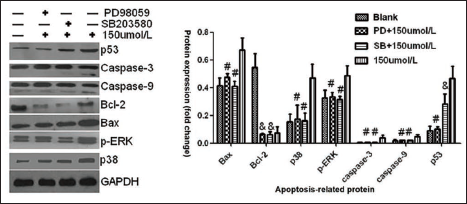
Baicalin Enhances Bcap-37 Cell Apoptosis via the ERK/p38 MAPK Pathway
Based on the results of the Western blot above, we aimed to further assess the intrinsic role of ERK and p38 MAPK in the baicalin treatment of breast medullary carcinoma PD98059 (an ERK-specific inhibitor) and SB203580 (a p38-specific inhibitor) were applied. Bcap-37 cells were treated with baicalin at 150 µmol/L alone or in combination with PD98059 or SB203580 for 48 h. As Figure 7 shows, compared with groups treated with baicalin alone, the protein expression of Bax, p38, p-ERK, caspase-3, and caspase-9 was upregulated obviously in groups treated with both baicalin and PD98059 or SB203580 (#p < 0.05). Interestingly, we observed that p53 expression showed a statistical difference only between the group treated with baicalin alone and the group treated with baicalin and PD98059. Bcl-2 displayed no statistical difference between any compared groups. In comparison to the reference group, the protein levels of Bax, p38, p-ERK, caspase-3, caspase-9, and p53 showed no statistical difference in the group treated with baicalin in combination with PD98059 and baicalin with SB203580 except Bcl-2 (
Discussion
Chinese medicine baicalin is widely used in clinical practice with various pharmacological functions, including anticancer (Huang et al., 2022; Zu et al., 2023). However, to our knowledge, regarding the role of baicalin as adjuvant therapy for MBC, few studies have been conducted. Hence, the present study aims to explore and clarify the underlying properties of baicalin in the treatment of MBC.
In the present study, we conducted cell growth assays and found both concentration-dependent and time-dependent cell growth capacity suppression in response to baicalin in Bcap-37 cells. As previous research has shown, baicalin promotes ferroptosis to display anticancer effects via attenuating FTH1 expression in bladder cancer cells 5637 and KU-19-19 (Kong et al., 2021). Next, we observed that, compared with the control group, baicalin absolutely reduced the migration rate of Bcap-37 cells in a concentration-dependent manner. Which is consistent with previous studies in breast cancer cells MCF-7 and MDA-MB-231 (Gao et al., 2018). Baicalin was also found to decrease the invasion and migration activity of pancreatic carcinoma SW1990 cells (Huang et al., 2019).
Inducing cancer cell apoptosis is a key mechanism for baicalin to antitumor.33−35 Similarly, studies indicated that baicalin triggers the apoptosis activity of breast cancer cells to enhance the chemosensitivity of docetaxel and doxorubicin (Lin et al., 2021; Zeng et al., 2020). In accordance with the preview studies, we found that baicalin significantly promoted both early and late apoptosis rates of Bcap-37 cells in a concentration-dependent manner. Apoptosis is a complex signaling pathway that is mediated by a series of apoptosis-related genes, including Bax, p38, p53, Bcl-2, and ERK1 (Lu et al., 2022; Wang et al., 2019). Also, a study revealed that baicalin promotes apoptosis of MCF-7 cells through up-regulating p53 and Bax (Wang et al., 2008). Hence, we carried out RT-qPCR to explore the underlying function of baicalin in apoptosis-related genes. And we found that, compared with the control group, the mRNA levels of Bax, p38, p-ERK1, and p-ERK2 all increased significantly in the three dosing groups, while the mRNA expression of Bcl-2 was opposite.
As a hub of two apoptotic pathways, the extrinsic (death receptor pathway) and the intrinsic pathway (mitochondrial pathway) (Zhang et al., 2022), caspase-3 assumes a vital role in the final stage of apoptosis (Xu et al., 2022). In the intrinsic pathway, caspase-3 is controlled by the upstream regulator caspase-9 (Deng et al., 2022). Bcl-2, which assumes a pivotal role in the mitochondrial pathway, suppresses cell apoptosis. On the contrary, Bax induces cell apoptosis if overexpressed (Sun et al., 2017). Our study indicated that baicalin upregulated the expressions of caspase-3, caspase-9, and Bax proteins while downregulating Bcl-2 protein expression in Bcap-37 cells in a concentration-dependent manner, which is in line with previous researches. For example, Pang et al. (2022) observed that baicalin induced apoptosis in osteosarcoma cells by enhancing Bax and cleaved caspase-3 expression and decreasing Bcl-2 expression. Besides, baicalin upregulated caspase-3 and caspase-9 expression, downregulated Bcl-2 and induced apoptosis in gastric cancer cells and osteosarcoma cells (Wan & Ouyang, 2018; Wang et al., 2017). p53, encoded by the TP53 gene, acts as one of the most frequently mutated tumor suppressors in human cancer and induces cell cycle arrest and apoptosis (Smolle et al., 2019). In MCF-7 cells, baicalin was found to promote apoptosis by upregulating the levels of p53 and Bax (Wang et al., 2008). Consistently, baicalin significantly upregulated the protein expression of p53, with a statistical difference in our research.
It has been reported that the ERK/p38 MAPK signaling pathway is involved in apoptosis in breast cancer and deeply affects its prognosis (Kumar et al., 2021; Wen et al., 2019; Xing et al., 2022). Furthermore, literature has demonstrated that baicalin combined with baicalein promotes apoptosis capacity in breast cancer cells of MCF-7 via the ERK/p38 MAPK pathway (Zhou et al., 2009). Therefore, we explored the effect of baicalin on the ERK/p38 MAPK signaling pathway in medullary breast cancer Bcap-37 cells and PD98059 (an ERK-specific inhibitor) and SB203580 (a p38-specific inhibitor) were utilized. Then, we observed that, compared with the group treated with baicalin alone, the protein expression of Bax, p38, p-ERK, caspase-3, and caspase-9 was downregulated obviously in the group treated with both baicalin and PD98059 or SB203580 (p < 0.05). p53 expression shows a statistical difference only between the group treated with baicalin alone and the group treated with baicalin and PD98059. Its mechanism needs more in-depth research. Compared with the blank group, the protein expression of Bax, p38, p-ERK, caspase-3, caspase-9, and p53 showed no statistical difference between the groups treated with baicalin in combination with PD98059 and baicalin with SB203580 except Bcl-2 (p < 0.05). Therefore, we thought that the apoptosis promotion of baicalin to apoptosis-related proteins was converted by PD98059 and SB203580.
Conclusion
In summary, baicalin inhibits the proliferation and migration abilities of medullary breast cancer cells Bcap-37. Besides, baicalin promotes Bcap-37 cell apoptosis via the ERK/p38 MAPK signaling pathway. Our research may contribute to the individual management of patients with medullary breast cancer.
Footnotes
Abbreviations
MBC: Medullary breast cancer; IR: Inhibition rate; RT-PCR: Real-time quantitative; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide.
Acknowledgments
We thank the editor and reviewers for critical review of the manuscript.
Authors’ Contribution
XL, QT and JL conceived and designed the study. XL cultured the cells, XL, QT, WL, DZ, and XF performed the experiments. XL, QT, WL, SH and XS analyzed data. XL and QT contributed to the writing of the manuscript. All above authors reviewed the manuscript.
Declaration of Conflicting Interests
The authors assert that the research was conducted without any commercial or financial relationships that could be interpreted as a potential conflict of interest.
Supplemental material
Supplemental material is available for this article online.
Statement of Ethical Approval and Informed Consent
Not applicable.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
