Abstract
Objective
To investigate the downstream target genes regulated by the nitric oxide–soluble guanylate cyclase–cyclic guanosine monophosphate (NO-sGC-cGMP) signal pathway and their possible roles in the pathogenesis of pulmonary hypertension (PH).
Methods
Digital gene expression tag profiling was performed to identify genes that are differentially expressed after activation of the NO-sGC-cGMP signal pathway in human pulmonary artery smooth muscles cells using 8-bromo-cyclic guanosine monophosphate, BAY 41-2272 and BAY 60-2770. Results were confirmed using real-time polymerase chain reaction. Gene ontology and signal transduction network analyses were also performed.
Results
A number of genes were differentially expressed, including
Conclusion
These results may provide new insights into the molecular mechanisms of PH.
Keywords
Introduction
Pulmonary hypertension (PH) is a serious disease characterized by elevated blood pressure in the pulmonary artery, pulmonary vein or pulmonary capillaries, leading to shortness of breath, dizziness, weakness and even symptoms of shock symptoms such as fainting. 1 Without treatment, the disorder will progress rapidly to right heart failure and death within 3 years of diagnosis.
The pathogenesis of PH is not completely understood, but multiple studies have suggested that an imbalance of vasoconstriction/vasodilation and proliferation/antiproliferation may be involved.
2
Nitric oxide (NO), which is produced by NO synthase from
Impairment of the NO-sGC-cGMP pathway, associated with dysregulation of NO production, sGC activity and cGMP degradation, has been shown to contribute to the progression of PH. 5 A number of sGC stimulators and activators that can bind to the β subunit of sGC and stimulate it in a direct or indirect way have been identified, including 3-(5′-hydroxymethyl-2′-furyl)-1-benzyl indazole (YC-1), 3-(4-amino-5-cyclopropylpyrimidin-2-yl)-1-(2-fluorobenzyl)-1H-pyrazolo[3,4-b]pyridine (BAY 41-2272), 2-[1-[(2-fluorophenyl)methyl]-1H-pyrazolo[3,4-b]pyridin-3-yl]-5-(4-morpholinyl)-4,6-pyrimidinediamine (BAY 41-8543) and riociguat (BAY 63-2521).6,7 They can act as potent pulmonary vasodilators, decreasing the mean pulmonary arterial pressure and vascular resistance, and have been shown to reverse structural lung vascular remodelling and right heart hypertrophy in PH. 8 Riociguat is the first sGC stimulator to undergo clinical study. In clinical trials it significantly improved the pulmonary vascular haemodynamics in pulmonary arterial hypertension and chronic thromboembolic PH with no serious adverse events. 9 In contrast, a specific inhibitor of sGC, 1H-[1,2,4]oxadiazolo[4,3-a]quinoxalin-1-one, has been shown to block the rise in cGMP and the antiproliferative action of the NO-sGC-cGMP pathway. 10
To date, studies suggest that once cGMP is produced, its effects could be executed though three main groups of cellular target molecules: cGMP-dependent protein kinases (PKGs), cGMP-gated cation channels and phosphodiesterases (PDEs). 11 The PDE inhibitor sildenafil is used to treat idiopathic PH.12,13 However, besides these three groups of molecules, little is known about the downstream target genes of the NO-sGC-cGMP pathway and the signalling events regulating gene expression. Using genome-wide technologies that can measure the expression of thousands of genes simultaneously, differences in gene expression in response to particular treatments can be measured. In the present study, digital gene expression tag profiling was used to measure differences in gene expression in human pulmonary artery smooth muscle cells treated with two sGC agonists 14 and a cGMP analogue. 15 Gene ontology analysis of the differential gene expression was then used to reveal the candidate genes involved in the activation of NO-sGC-cGMP pathway.
Materials and methods
Digital gene expression tag profiling
Human pulmonary artery smooth muscle cells isolated from patients with PH were generously provided by Dr Jun Wang from Capital Medical University, Beijing, China. They were cultured in smooth muscle cell medium (ScienCell Research Laboratories, Carlsbad, CA, USA) supplemented with 2% fetal bovine serum and 1% smooth muscle cell growth supplement (ScienCell Research Laboratories) and maintained at 37℃ in a humidified atmosphere of 5% carbon dioxide. The sGC agonists BAY 41-2272 (an NO-independent but haem-dependent sGC stimulator) and BAY 60-2770 (an NO- and haem-independent sGC activator) for digital gene expression tag profiling were generously provided by Professor Johannes-Peter Stasch from Bayer Schering Pharma AG, Cardiology Research, Pharma Research Centre, Germany. These were dissolved in dimethyl sulphoxide (DMSO) at a concentration of 100 mmol/l and stored at −20℃ until used.
Smooth muscle cells (1 × 106) were treated with 2 µl of 100 µmol/l BAY 41-2272 for 8 h, 0.2 µl of 10 µmol/l BAY 41-2272 for 24 h, 0.2 µl of 10 µmol/l BAY 60-2770 for 24 h, or 2 µl of 100 µmol/l 8-bromo-cGMP (8-Br-cGMP; Sigma-Aldrich, St Louis, MO, USA) for 8 h. Controls were treated with DMSO (2 µl or 0.2 µl as controls for BAY 41-2272 and BAY 60-2770, respectively) or 2 µl double-distilled H2O (controls for 8-Br-cGMP) for the same time periods. The cells (2 × 106) were then harvested at the indicated time points.
Total RNA was extracted using TRIzol reagent according to the manufacturer’s instructions (Invitrogen, Carlsbad, CA, USA). Magnetic oligo-beads were used to isolate the mRNA from 6 µg of total RNA. First- and second-strand cDNA was synthesized using oligo(dT) primers, purified using a Qiaquick PCR Purification Kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions, and digested with DNase I in order to produce a substantial enrichment of fragments about 200 bp in length. The samples (20 µl) were then loaded onto 1.2% agarose gel (Invitrogen) and fragments between 100 and 300 bp were extracted using a Qiaquick Gel Extraction Kit (Qiagen) according to the manufacturer’s instructions. The 5′ and 3′ ends of the fragments were end-repaired. The samples were purified and ligated to genomic adapters (Illumina, San Diego, CA, USA) and then loaded onto 2% agarose gel and separated by electrophoresis (120 V for 30 min), and the fragments between 100 and 300 bp were isolated from the gel and purified using the Qiaquick Gel Extraction Kit. A polymerase chain reaction (PCR) of the gel-purified cDNA (total volume 50 µl) was performed using DNA polymerase and primers provided by Illumina according to the manufacturer’s instructions. After 15 cycles of PCR amplification, the sample were loaded onto 1.2% agarose gels and fragments between 100 and 300 bp were purified from the gel again. The samples were then quantified spectrophotometrically using a NanoDrop spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA) and sequenced using a HiSeq™ 2000 system (Illumina).16,17
Determination of differentially expressed genes
To compare the differential expression of genes across samples, the raw data was collected and filtered to remove low quality tags, adaptor tags and tags with a single copy number. The gene expression levels were determined by the number of raw clean tags in each library normalized to the transcripts per million clean tags. A significant difference was defined as a
Quantitative real-time PCR
Quantitative real-time PCR was performed to validate the digital gene expression results. Human pulmonary artery smooth muscle cells for quantitative real-time PCR were purchased from ScienCell Research Laboratories and were cultured in the same smooth muscle cell medium as described above. Smooth muscle cells (1 × 106) were treated with 2 µl of 100 µmol/l BAY 41-2272 (Sigma-Aldrich) for 8 h. Controls were treated with 2 µl DMSO for the same time period. The cells (2 × 106) were then harvested.
Total RNA was extracted using TRIzol reagent according to the manufacturer’s instructions. First-strand cDNA was generated with random primers using a SuperScript™ First-Strand Synthesis System for RT-PCR (Invitrogen). Real-time PCR was performed using the iQ5 Multicolor Real-Time PCR Detection System (Bio-Rad Laboratories, Hercules, CA, USA) with SYBR® Premix Ex TaqTM II (Takara Biotech Co., Dalian, China) according to the manufacturer’s protocol. The target genes were
Gene ontology analysis of the differentially expressed genes
Gene ontology (GO) analysis is a commonly used technique to determine the main functions of differentially expressed genes according to the GO database.
18
A 
Pathway analysis of the differentially expressed genes
Pathway analysis was based on the Kyoto Encyclopedia of Genes and Genomes (KEGG), a comprehensive pathway-related database.
19
Statistical analyses
All data were expressed as the mean ± SD. Differential gene expression levels were based on log2 ratios among the different groups. Statistical comparisons were performed using the Student’s
Results
Differentially expressed genes
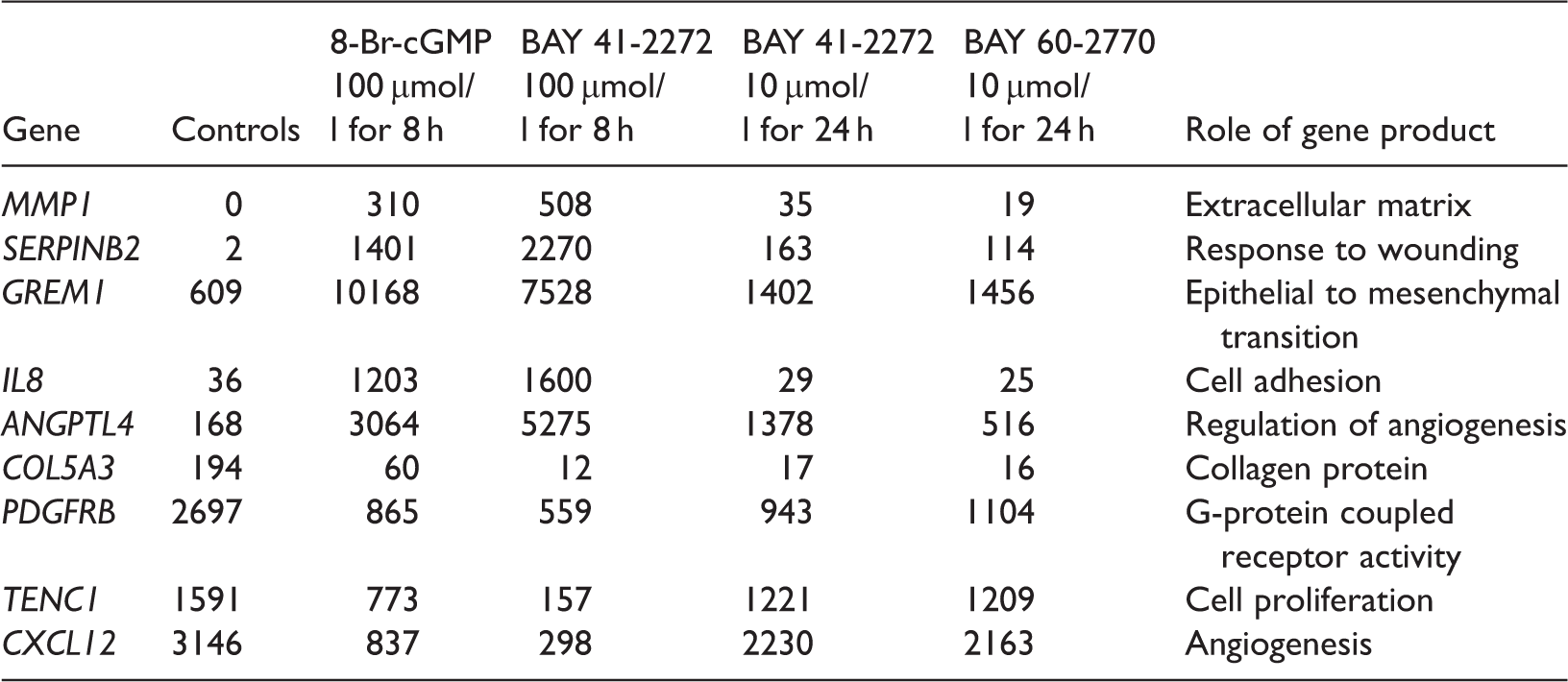
Compared with controls, 539 upregulated genes and 367 downregulated genes were identified in cells treated with 8-Br-cGMP, and 993 upregulated genes and 1002 downregulated genes were identified in cells treated with BAY 41-2272 (100 µmol/l for 8 h) (Figure 1). In addition, 219 and 143 genes were significantly upregulated and 353 and 351 genes were significantly downregulated after treatment with BAY 41-2272 (10 µmol/l for 24 h) and BAY 60-2770 (10 µmol/l for 24 h), respectively. A total of 84 of the upregulated genes and 87 of the downregulated genes showed similar variation trends in all four groups, suggesting that activation of the NO-sGC-cGMP pathway might affect expression of a group of important downstream factors. Nine genes that were significantly up- or downregulated compared with controls when the NO-sGC-cGMP pathway was activated are shown in Table 1. Interestingly, these differentially expressed genes are involved in the degradation of the extracellular matrix, the response to wounding and cell adhesion, which are closely related to the pathogenic processes of PH.
Differentially expressed genes (DEGs) in pulmonary artery smooth muscle cells from patients with pulmonary hypertension treated with 8-bromo-cyclic guanosine monophosphate (8-Br-cGMP), BAY 41-2272 or BAY 60-2770 compared with control cells. Significantly up- or downregulated genes in pulmonary artery smooth muscle cells from patients with pulmonary hypertension treated with 8-bromo-cyclic guanosine monophosphate (8-Br-cGMP), BAY 41-2272 or BAY 60-2770 compared with control cells measured using digital gene expression tag profiling. The values given are the number of unique reads, which is an indication of expression levels.
Validation of the differentially expressed genes
The potential candidate genes identified using digital gene expression tag profiling were validated using specific primer-based methods. Expression of mRNA levels for genes that were upregulated in the digital gene expression analysis and that may play important roles in regulating extracellular matrix degradation, cell proliferation and angiogenesis were measured using quantitative real-time PCR. Gene expression in human pulmonary artery smooth muscle cells treated with BAY 41-2272 (100 µmol/l for 8 h) is shown in Figure 2. Expression of Expression of genes 
GO analysis of differentially expressed genes
Gene ontology items associated with differentially expressed genes in pulmonary artery smooth muscle cells from patients with pulmonary hypertension treated with BAY 41-2272 (100 µmol/l for 8 h) and their corrected

Pathway analysis of differentially expressed genes
Identification of significant signal transduction pathways based on an analysis of differentially expressed genes in pulmonary artery smooth muscle cells treated with BAY 41-2272 (100 µmol/l for 8 h).

NOD, nucleotide oligomerization domain; MAPK, mitogen-activated protein kinase.
Discussion
Despite several advances in therapy, there is currently no curative treatment for PH and its pathophysiology is only partially understood. cGMP, produced by activation of the NO-sGC-cGMP pathway, is a key regulator of vascular smooth muscle cell contractility, growth and differentiation, and has been shown to be associated with PH. 4 However, the regulation of gene expression by cGMP and the physiological effects and functions of cGMP remain to be fully defined.
In the present study, human pulmonary artery smooth muscle cells were treated with 8-Br-cGMP, BAY 41-2272 and BAY 60-2770, activators of the NO-sGC-cGMP pathway, to investigate PH-related differentially expressed genes and signalling pathways. Using digital gene expression tag profiling, a group of downstream genes that are possibly regulated by cGMP were identified. Among the differentially expressed genes detected by Illumina sequencing and confirmed by quantitative real-time PCR,
In the light of the results of the present study, further investigations into the expression and functions of the protein products of these genes in PH using a greater number of samples are warranted.
To better understand the molecular events involved in the development of PH, functional enrichment analysis of the differentially expressed genes was performed. The results indicated that the enriched biological processes and pathways were very similar following activation of the NO-sGC-cGMP pathway irrespective of the activating agent. Using GO analysis, regulation of cellular process, biological process and cellular macromolecule biosynthetic process were the most significantly enriched items in the present study. Pathway analysis can reveal the distinct biological processes and identify signalling pathways; in the present study, enriched categories included pathways in cancer, p53 signalling pathway, NOD-like receptor signalling pathway, malaria, MAPK signalling pathway, base excision repair and basal cell carcinoma. Interestingly, a number of these pathways (pathways in cancer, p53 signalling pathway and MAPK signalling pathway) are involved in cell proliferation, cell differentiation and cell apoptosis,29,30 which have been reported to be associated with the vascular remodelling seen during PH progression.31,32 These differentially expressed genes may be potent regulators of pulmonary artery smooth muscle cells and dysregulation may lead to the development of pulmonary vascular remodelling.
The differentially expressed genes identified in the present study are probably important contributors to the pathogenesis of PH. However, how cGMP regulates these differentially expressed genes and how these genes regulate vascular remodelling needs to be further investigated. The functions of the other items identified by pathway analysis may also play a role in PH that has not been elucidated yet. Validation and investigation of the downregulated genes will be undertaken in the future.
In conclusion, the present study identified some differentially expressed genes and related pathways regulated by the NO-sGC-cGMP signal pathway, and found that most of the differentially expressed genes were involved in cell proliferation, cell migration and apoptosis, which might contribute to the pathological pulmonary vascular remodelling in PH. These results may provide a novel insight into the molecular mechanisms of PH, especially those involved in pulmonary vascular remodelling. Further studies on these identified genes and signalling pathways may lead to the discovery of new drug targets for PH therapy.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the National High Technology Research and Development Program (grant 2012AA02A511), the National Department Public Benefit Research Foundation (no. 201302008), the National Key Technology Research and Development Program (grant 2013BAI09B00) and the National Natural Science Foundation of China (grant 81400036).
