Abstract
Background
Breast cancer, originating in the cells of the breast, involves a significant role of oxidative stress and inflammatory reactions in tumor cell proliferation. In this study, we investigate the chemoprotective impact of bryodulcosigenin against 7,12-dimethylbenz(a)anthracene (DMBA)-induced breast cancer in rats, aiming to elucidate the underlying mechanisms.
Materials and Methods
Female Wistar rats were used in this study, with breast cancer induced by intraperitoneal injection of DMBA at a dose of 80 mg/kg body weight. The rats were then treated orally with bryodulcosigenin at doses of 5, 10, and 20 mg/kg body weight for 22 weeks. Body weight, tumor marker, glycoprotein, mitochondrial, antioxidant, cytokines, and inflammatory parameters were estimated. Apoptosis parameters messenger ribonucleic acid (mRNA) expressions were estimated.
Results
Bryodulcosigenin significantly (p < 0.001) downregulated the tumor weight and incidence. Bryodulcosigenin significantly (p < 0.001) repressed the tumor markers like cancer antigen 15-3 (CA 15-3), alpha-fetoprotein (AFP), carcinoembryonic antigen (CEA), total sialic acid (TSA), and lipid-bound sialic acid (LSA); glycoprotein such as hexose, hexosamine, and sialic acid improved the level of mitochondrial parameters like succinate dehydrogenase (SDH), isocitrate dehydrogenase (ICDH), alpha-ketoglutarate dehydrogenase (α-KGDH), and malate dehydrogenase (MDH); cytokines, namely, interleukin-1β (IL-1β), tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and inflammatory parameters such as nuclear factor kappa B (NF-κB), prostaglandin (PGE2), cyclooxygenase-2 (COX-2), respectively. Bryodulcosigenin significantly (p < 0.001) altered the level of lipid parameters such as triglyceride, total cholesterol, high-density lipoprotein (HDL); antioxidant parameters like thiobarbituric acid reactive substances (TBARS), glutathione peroxidase (GPx), superoxide dismutase (SOD), catalase (CAT), glutathione (GSH), respectively. Bryodulcosigenin significantly (p < 0.001) altered the mRNA expression of apoptosis markers such as B-cell lymphoma-2 (Bcl-2), Bcl-2-associated protein x (Bax), and caspase-3.
Conclusion
We can conclude that bryodulcosigenin protects against DMBA-induced breast cancer by reducing inflammatory reactions and apoptosis.
Introduction
Cancer manifests as a diverse group of diseases marked by a multi-stage progression, encompassing complex processes such as migration, uncontrolled proliferation, metastasis, and invasion (Zingue et al., 2021). Cancer is a significant health issue that increases due to an increase in exposure to carcinogens (Kumar et al., 2016). Common types of cancer vary between genders. In males, common types include stomach, prostate, colorectal, renal, hepatic, and lung cancers. In females, common types include breast, colorectal, cervical, and lung cancers (Kumar et al., 2021). Breast cancer is acknowledged worldwide as the second most frequently diagnosed cancer across all types and is a major health issue in both lower-middle income and high-income countries (Zingue et al., 2021). It is estimated that one in every eight women will be diagnosed with breast cancer during her lifetime (Karnam et al., 2017). Various mammary carcinogenesis, such as Dimethylbenz(a)anthracene (DMBA), involves the physiological and cellular changes that lead to a tumor or lump in the breast (Kumar et al., 2023; Zingue et al., 2021). The causes of breast cancer are not yet fully understood, but several risk factors have been recognized. These include environmental factors such as polycyclic aromatic hydrocarbons (PAHs), hormonal factors like estrogen, age, and a family history of cancer. These factors have shown statistical correlations with the incidence of breast cancer (Afzal et al., 2021; Kumar et al., 2021).
Carcinogenesis is a multi-stage process that includes initiation, promotion, and progression, involving various genetic and epigenetic changes. Cells transition into a cancerous state when they lose the ability to control cell division, adhere to their original location, and undergo programmed cell death at the appropriate time (Karnam et al., 2017). Normal cells undergo apoptosis, a programmed cell death process when no longer needed. This well-regulated process involves multiple proteins and pathways, including nuclear factor kappa B (NF-κB) transcription pathway (Afzal et al., 2021; Patel et al., 2023). These protective mechanisms ensure the proper functioning and controlled life cycle of cells in the body. Mutations in genes such as putative protein tyrosine phosphatase (PTEN), breast cancer gene 1 (BRCA1), and breast cancer gene 2 (BRCA2) due to epigenetic factors are considered potential origins of breast cancer. Epigenetic changes can impact the expression of these genes, playing a role in the development and progression of breast cancer (Dillon et al., 2014; Karnam et al., 2017). Epigenetic changes can influence the expression of these genes, potentially leading to disruptions in normal cellular processes and contributing to the development of breast cancer. Indeed, the PTEN gene is involved in regulating cell growth and division, while BRCA1 and BRCA2 are recognized for their crucial roles in deoxyribonucleic acid (DNA) repair mechanisms. Dysregulation or mutations in these genes can lead to the development of various cancers, including breast cancer. Such mutations can increase the risk of developing breast cancer (Dillon et al., 2014; Mendes-Pereira et al., 2012).
PAHs, including 7,12-dimethylbenz(a)anthracene, are environmental carcinogens from human activities (Gelboin, 1980). PAHs are chemically stable compounds that can endure in the environment for extended periods (Zingue et al., 2021). DMBA is a site-specific carcinogen frequently employed to induce breast cancer in rodents. It operates by initiating or promoting gene mutations that play a role in carcinogenesis (Brinton et al., 2017; Zingue et al., 2021). Upon entering the body, DMBA is metabolized into epoxides. These epoxides can interact with DNA, forming PAH-DNA adducts, which are linked to the development of breast cancer (Brinton et al., 2017). Furthermore, DMBA activates the cytosolic aryl hydrocarbon receptor (AhR). Upon activation, AhR translocates into the nucleus and forms a complex with the AhR nuclear translocation protein (Karnam et al., 2017).
In recent decades, significant progress has been made in breast cancer treatment, primarily due to advancements in early diagnosis and improvements in hormonal and adjuvant chemotherapies. Notably, the United States Food and Drug Administration has approved two selective estrogen receptor modulators, raloxifene and tamoxifen, for breast cancer treatment. However, both drugs are associated with side effects similar to menopausal symptoms, including night sweats, hot flashes, weight gain, vaginal dryness, and swelling of the legs, feet, and ankles (Jordan et al., 2001).
Recently, various therapies have shown a protective effect against breast cancer, but still, these therapies are not 100% successful due to enhancing the morbidity linked with breast cancer (Arnold et al., 2022). Chemotherapy resistance is a common challenge, and there are limited effective treatments for advanced disease conditions (Martín et al., 2013). However, some chemotherapeutics, such as doxorubicin (DOX), showed effectiveness against breast cancer with the induction of side effects such as cardiotoxicity. Given the significant side effects associated with chemotherapy, there is an urgent need to develop novel therapeutic approaches for the treatment of breast cancer.
The World Health Organization (WHO) reports that more than 80% of the global population relies on conventional medicine as their main form of healthcare. The phytoconstituents extracted from plant sources were initially used to examine their pharmacological effects. Phytoconstituents are compounds found in plants that do not directly contribute to the plant’s normal physiological functions.
Despite this, they contribute significantly to both human and animal health and are essential in the treatment and prevention of diseases. These compounds are frequently investigated for their potential therapeutic properties and medicinal benefits.
A wide range of phytoconstituents are available and utilized for treating various diseases, including cancer, inflammation, diabetes, arthritis, obesity, and others (Adeoye et al., 2023; Plotnikoff et al., 2022). Bryodulcosigenin is a naturally occurring compound classified as a cucurbitane-type triterpenoid, commonly found in plants like cucumbers, melons, and gourds (Li et al., 2022). Bryodulcosigenin has been isolated from the roots of Bryonia dioica, a medicinal plant used in traditional European and Asian medicine (Ding et al., 2022; Li et al., 2022). Bryodulcosigenin has shown anti-inflammatory effects in numerous animal models of inflammation, including cotton pellet-induced granuloma, acetic acid-induced peritonitis, and carrageenan-induced paw edema (Ding et al., 2022; Li et al., 2022; Ukiya et al., 2002). It has also been reported to inhibit the production of pro-inflammatory cytokines by suppressing the activation of the NF-κB and mitogen-activated protein kinase (MAPK) pathways in macrophages. Bryodulcosigenin has shown significant anti-inflammatory properties.
It can inhibit the production of pro-inflammatory cytokines and reduce inflammation in various tissues. Studies have indicated that bryodulcosigenin may also possess anticancer properties (Li et al., 2022). Bryodulcosigenin has the ability to induce apoptosis (programmed cell death) in cancer cells and inhibit their proliferation. This effect is beneficial in potentially reducing tumor growth and preventing the spread of cancer. Bryodulcosigenin has been found to have cardioprotective effects, potentially by improving cardiac function and reducing myocardial injury (Ding et al., 2022). This can be particularly useful in managing heart diseases and conditions related to cardiac stress (Ukiya et al., 2002). Evidence suggests that bryodulcosigenin may protect neuronal cells from damage. Its neuroprotective properties could potentially be beneficial in managing neurodegenerative diseases such as Alzheimer’s and Parkinson’s diseases. A recent study suggests that bryodulcosigenin may have a positive impact on intestinal disorders, including inflammatory bowel disease and colitis-associated cancer. It is believed to achieve this by modulating the gut microbiota and reducing intestinal inflammation (Ding et al., 2022; Li et al., 2022). We are examining the chemoprotective effects of Bryodulcosigenin against DMBA-induced breast cancer in rats.
Materials and Methods
Experimental Design
Swiss Albino rats, females aged 10–12 weeks, weighing 150 ± 20 g, were employed in the current experimental study. Rats were procured from the departmental animal house and individually housed in polyethylene cages. Standard laboratory conditions were upheld, including a temperature of 22 ± 5°C, 65% relative humidity, and a 12/12-h dark/light cycle. The rats were fed a standard soy-free rat diet and had unlimited access to water. All the experimental procedures adhered to the principles and protocols established by the institutional ethical committee (SGYY2024039).
Diet
The following diets included bone flour (14.5%), fish flour (4.8%), corn (36.7%), sodium chloride (0.3%), wheat (36.6%), crushed palm kernel (7.3%), and vitamin complex (0.01%) and were given to the rats.
Toxicant
DMBA was employed to induce breast cancer in the rats. DMBA (80 mg/kg b.wt) was intraperitoneally used for the induction of breast cancer (Kumar et al., 2021). DMBA was solubilized in 1 mL of olive oil.
Oral Suspension
The oral suspension of bryodulcosigenin (>=98%, ChemFaces, Wuhan, Hubei 430056, PRC) (Supplementary Figure 1) was prepared via dissolving the 1% carboxyl methylcellulose (CMC) and prepared the suspension to give to the animal.
Experimental Protocol
The rats were categorized into distinct categories as outlined:
Group I: Normal control,
Group II: DMBA,
Groups III–V: DMBA + Bryodulcosigenin (5, 10, and 20 mg/kg b. wt), respectively.
The rats underwent the designated treatment for 22 weeks. Throughout the study, regular monitoring was conducted for water intake, food intake, and body weight across all groups of rats. Upon study completion, tumor latency was noted. The rats were then given anesthesia, and a retro-orbital vein puncture was used to obtain blood samples. To separate the serum, the blood samples were centrifuged for 10 min at 12,000 rpm.
Tumorgenicity Markers
Tumorigenicity markers such as cancer antigen 15-3 (CA 15-3), alpha-fetoprotein (AFP), and carcinoembryonic antigen (CEA) were assessed using enzyme-linked immunosorbent assay (ELISA) kits following the manufacturer’s instructions (UBI Magiwell, USA). The previously reported method was employed to estimate the levels of total sialic acid (TSA) and lipid-bound sialic acid (LSA) with minor modifications (Plucinsky et al., 1986).
Estimation of Glycoprotein
Hexose, hexosamine, and sialic acid were among the glycoprotein properties that were estimated using a previously published technique that was slightly modified (Singh et al., 2024).
Mitochondrial Parameter
The method used to estimate mitochondrial parameters, such as malate dehydrogenase (MDH), isocitrate dehydrogenase (ICDH), succinate dehydrogenase (SDH), and alpha-ketoglutarate dehydrogenase (α-KGDH), was slightly modified from a previously published approach (Bell et al., 1960; Mehler et al., 1948; Reed et al., 1969; Singh et al., 2024).
Hepatic and Lipid Parameters
Hepatic parameters such as aspartate aminotransferase (AST) and alanine aminotransferase (ALT), lipid parameters, namely, total cholesterol, triglyceride, and high-density lipoprotein (HDL) were estimated using the available kits following the manufacturer’s instruction (Biosource, Camarillo, CA).
Antioxidant, Phase I and II Enzymes
Antioxidant parameters like catalase (CAT), glutathione (GSH), superoxide dismutase (SOD), thiobarbituric acid reactive substances (TBARS), and CAT were estimated with slight modifications from the previously published approach. With a few modest adjustments, phase I and phase II enzyme levels were approximated using the previously described methodology (Kakkar et al., 1948; Kumar et al., 2023; Singh et al., 2024).
Cytokines and Inflammatory Parameters
Inflammatory cytokines such as tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and interleukin-6 (IL-6) were measured using ELISA kits according to the manufacturer’s guidelines (Cell Application, Inc., San Diego, CA, USA). Inflammatory parameters like NF-κB, cyclooxygenase-2 (COX-2), and prostaglandin (PGE2) were estimated using the manufacturer’s instructions of ELISA kits (Biosource, Camarillo, CA).
Reverse Transcription Polymerase Chain Reaction (RT-PCR)
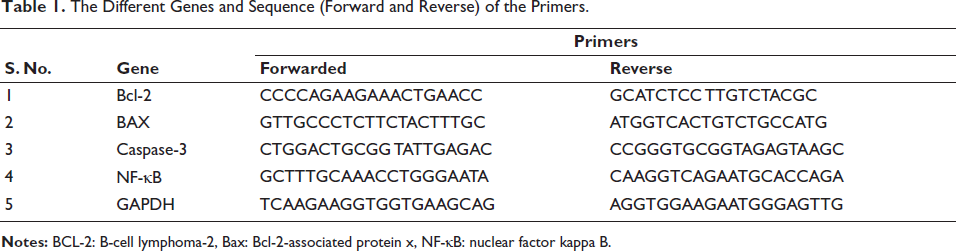
To extract total RNA from breast tissue, the Trizol reagent kit was employed. RNA levels were then assessed using RT-PCR with the complementary DNA (cDNA) Verso kit. The protocol involved 32 cycles of 94°C for 30 s and 72°C for 30 s, followed by reverse transcription for 30 min at 42°C. The primer sequences are listed in Table 1.
The Different Genes and Sequence (Forward and Reverse) of the Primers.
Statistical Analysis
Statistical analysis was conducted using graphpad prism 8 (St. Louis, USA). Data are presented as mean ± standard error of the mean (SEM). Statistical comparisons were performed with Dunnett’s test, and a significance level of p < 0.05 was used.
Results
Body Weight
A decrease in body weight is commonly observed in cancer patients. Similarly, the group of rats treated with DMBA exhibited a reduction in body weight and bryodulcosigenin treatment significantly (p < 0.001) enhanced body weight (Supplementary Figure 2).
Tumor Incidence
DMBA-induced group rats presented 9 rats out of 10 with 85.5 g tumor burden with 90% tumor incidence. Bryodulcosigenin (5 mg/kg b. wt) treated rats showed the 7 number of rats with 51.3 g tumor burden and 70% tumor incidence. Bryodulcosigenin (10 mg/kg b. wt) treated rats showed the 4 number of rats with 29.4 g tumor burden and 50% tumor incidence. Bryodulcosigenin (20 mg/kg b. wt) treated rats showed two rats with 11.2 g tumor burden and 28.57% tumor incidence (Table 2).
The Total Number of Rats/Rats with Tumors, Tumor Burden, and Tumor Incidence of Bryodulcosigenin on DMBA-induced Mammary Glands in Rats.

Tumor Marker
Rats with DMBA-induced breast cancer showed increased levels of AFP (Supplementary Figure 3a), CEA (Supplementary Figure 3b), CA 15-3 (Supplementary Figure 3c), and bryodulcosigenin treatment significantly (p < 0.001) repressed the level tumor marker.
TSA and LSA
The rats in the DMBA-induced group exhibited elevated levels of TSA (Supplementary Figure 4a) and LSA (Supplementary Figure 4b), and bryodulcosigenin significantly (p < 0.001) downregulated these levels.
Glycoprotein
Rats in the DMBA-induced group exhibited boosted levels of hexose (Supplementary Figure 5a), hexosamine (Supplementary Figure 5b), and sialic acid (1Figure 5c). Bryodulcosigenin significantly (p < 0.001) reduced the levels of these glycoprotein parameters.
Mitochondrial Parameter
DMBA-induced group rats exhibited improved levels of ICDH (Supplementary Figure 6a), SDH (Supplementary Figure 6b), MDH (Supplementary Figure 6c), α-KGDH (Supplementary Figure 6d), and bryodulcosigenin significantly (p < 0.001) upregulated the level of mitochondrial parameters.
Hepatic Parameters
The rats in the DMBA-induced group exhibited elevated levels of AST (Supplementary Figure 7a) and ALT (Supplementary Figure 7b). Bryodulcosigenin significantly (p < 0.001) reduced the levels of these hepatic parameters.
Lipid Parameters
The rats in the DMBA-induced group showed increased levels of total cholesterol (Supplementary Figure 8a) and triglyceride (Supplementary Figure 8b) and a decreased level of HDL (Supplementary Figure 8c). Bryodulcosigenin treatment significantly (p < 0.001) restored the levels of these lipid parameters.
Phases I and II
Rats induced with DMBA demonstrated reduced levels of GST (Supplementary Figure 9a), cytochrome B5 (Supplementary Figure 9b), and cytochrome P450 (Supplementary Figure 9c). Bryodulcosigenin significantly (p < 0.001) improved the levels of these phase I and II enzymes.
ATPase Enzymes
DMBA-induced rats exhibited a decreased level of Na+K+ATPase (Supplementary Figure 10a), Ca2+ATPase (Supplementary Figure 10b), and Mg2+ATPase (Supplementary Figure 10c), and bryodulcosigenin significantly (p < 0.001) improved the ATPase level.
Antioxidant
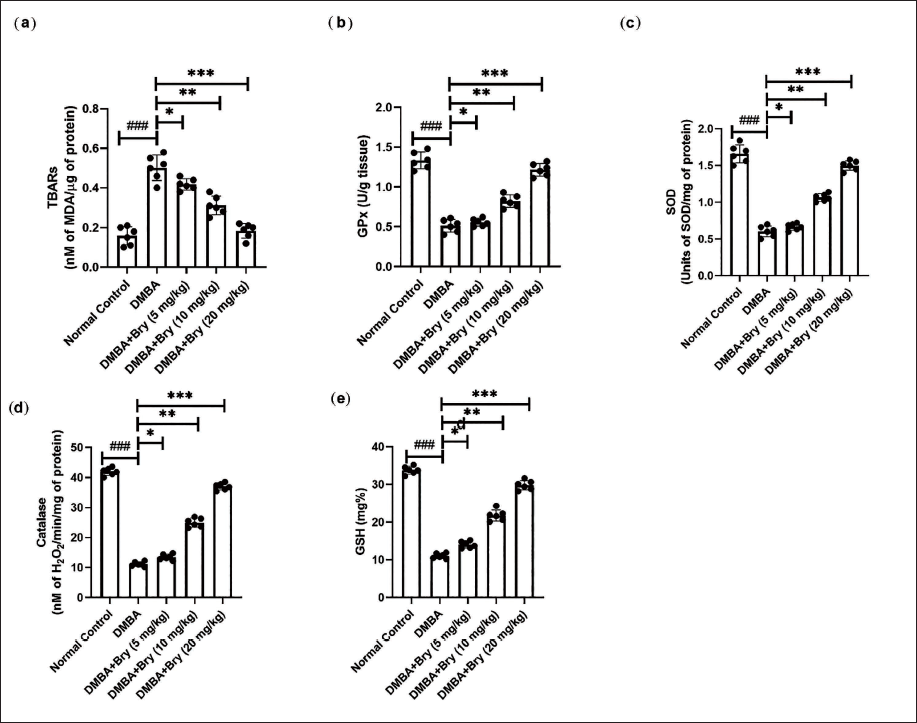
The rats in the DMBA-induced group demonstrated altered levels of TBARS (Figure 1a), glutathione peroxidase (GPx) (Figure 1b), SOD (Figure 1c), CAT (Figure 1d), and GSH (Figure 1e). Bryodulcosigenin significantly (p < 0.001) altered the levels of these antioxidant parameters.
The Level of Antioxidant Parameters Against Dimethylbenz(a)anthracene (DMBA)-induced Breast Cancer Rats. (a) Thiobarbituric Acid Reactive Substances (TBARS), (b) Glutathione Peroxidase (GPx), (c) Superoxide Dismutase (SOD), (d) Catalase (CAT), and (e) Glutathione (GSH). Each Value shown Means ± Standard Error of the Mean (SEM); n = 10, Where Normal Control is Compared with DMBA Control and ###p < 0.001 is Considered Extremely Significant. The Tested Group is Compared with DMBA Control, and *p < 0.05 was Significant, **p < 0.01 More Significant, and ***p < 0.001 Extremely Significant.
Cytokines and Inflammatory Parameters
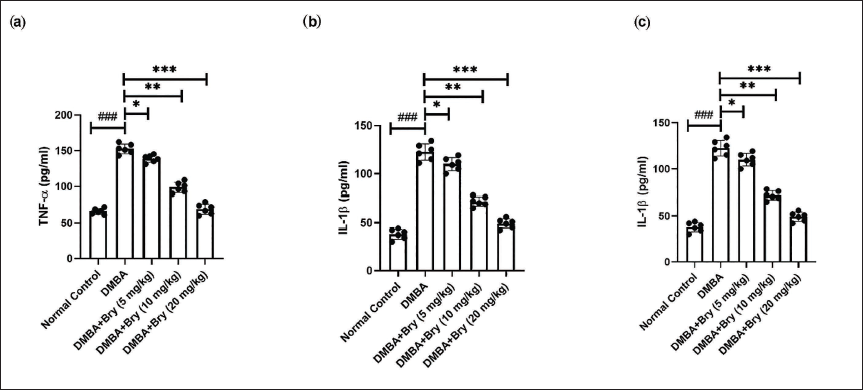
Rats in the DMBA-induced group showed the boosted level of TNF-α (Figure 2a), IL-1β (Figure 2b), IL-6 (Figure 2c), and bryodulcosigenin significantly (p < 0.001) reduced the cytokines level.
The Level of Inflammatory Cytokines Against DMBA-induced Breast Cancer Rats. (a) Tumor Necrosis Factor-α (TNF-α), (b) Interleukin-1β (IL-1β), and (c) Interleukin-6 (IL-6). Each Value shown Means ± Standard Error of the Mean (SEM); n = 10, Where Normal Control is Compared with Dimethylbenz(a)anthracene (DMBA) Control and ###p < 0.001 is Considered Extremely Significant. The Tested Group is Compared with DMBA Control, and *p < 0.05 was Significant, **p < 0.01 More Significant, and ***p < 0.001 was Highly Significant.
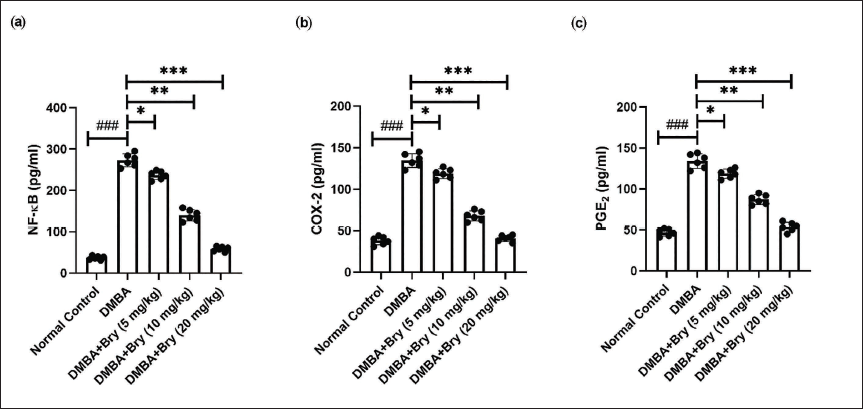
Rats in the DMBA-induced group showed the boosted level of NF-κB (Figure 3a), COX-2 (Figure 3b), PGE2 (Figure 3c), and bryodulcosigenin significantly (p < 0.001) reduced the level of inflammatory parameters.
The Level of Inflammatory Parameters Against DMBA-induced Breast Cancer Rats. (a) Nuclear Factor Kappa B (NF-κB), (b) Cyclooxygenase-2 (COX-2), and (c) Prostaglandin (PGE2). Each Value shown Means ± Standard Error of the Mean (SEM); n = 10, Where Normal Control is Compared with Dimethylbenz(a)anthracene (DMBA) Control and ###p < 0.001 is Considered Extremely Significant. The Tested Group is Compared with DMBA Control, and *p < 0.05 was Significant, **p < 0.01 More Significant, and ***p < 0.001 Extremely Significant.
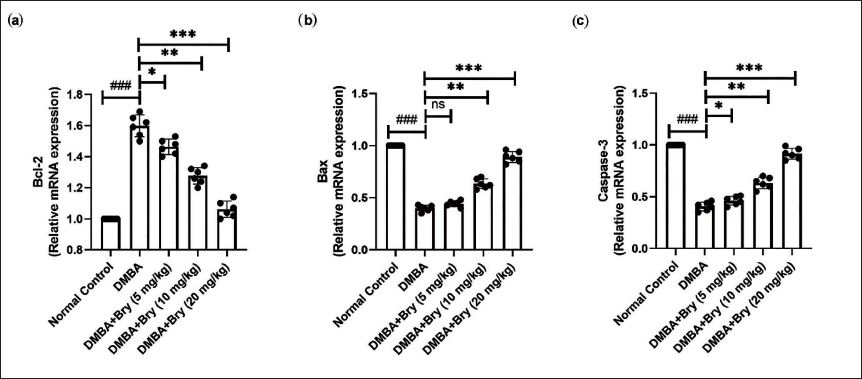
mRNA Expression
Rats in the DMBA-induced group demonstrated altered levels of B-cell lymphoma-2 (Bcl-2) (Figure 4a), Bcl-2-associated protein x (Bax) (Figure 4b), and caspase-3 (Figure 4c). Bryodulcosigenin significantly (p < 0.001) modulated the mRNA expression of these genes.
The messenger Ribonucleic Acid (mRNA) Expression of Apoptosis Parameters Against DMBA-induced Breast Cancer Rats. (a) B-cell lymphoma-2 (Bcl-2), (b) Bcl-2-associated protein x (Bax), and (c) Caspase-3. Each Value shown Means ± Standard Error of the Mean (SEM); n = 10, Where Normal Control is Compared with Dimethylbenz(a)anthracene (DMBA) Control and ###p < 0.001 is Considered Extremely Significant. The Tested Group is Compared with DMBA Control and *p < 0.05 was Significant, **p < 0.01 More Significant, and ***p < 0.001 Extremely Significant.
Discussion
Following the initiation of carcinogenesis, cancer cells undergo progressive division and growth, resulting in a substantial tumor burden (Haque et al., 2018). The rapidly proliferating neoplastic cells undergo alterations in various metabolic pathways, including those related to lipogenesis and energy regulation. This metabolic reprogramming is a hallmark of cancer cells, allowing them to adapt and support their uncontrolled growth and survival (Kumar et al., 2023; Patel et al., 2023). The DMBA-induced breast cancer model has become popular in research due to its high relevance, providing a platform for the scrutiny and exploration of the underlying mechanisms of various compounds, especially regarding their anti-inflammatory, antioxidant, and chemotherapeutic potentials (Kumar et al., 2016). DMBA, a powerful carcinogen, generates several reactive intermediates during metabolism, including hydroxyl radicals (OH), superoxide radicals (O2), and hydrogen peroxide (H2O2). These intermediates mainly bind to nucleophilic sites on cellular macromolecules in human breast cancer, which increases oxidative stress (Anbuselvam et al., 2007; Kumar et al., 2016).
Tumor parameters such as CA 15-3, AFP, and CEA are used as identification markers to detect cancerous cells. These biomarkers are often measured in blood tests. They can provide valuable information about the presence and progression of certain types of cancers, including breast cancer (CA 15-3), liver cancer (AFP), and colorectal cancer (CEA) (May, 1959). Elevations in these markers may indicate the presence of cancer or other medical conditions (Kumar et al., 2023). CA 15-3 is a biomarker isolated from Mucin-1 and is commonly used as an indicator for identifying breast cancer. AFP is used to identify tumor subgroups, and CEA is a glycosylphosphatidylinositol (GPI) glycoprotein (Konstantopoulos & Thomas, 2009). CEA functions as the functional carcinoma E and L selectin ligands in cancer cell adhesion and metastasis. These tumor markers provide valuable information for diagnosing and monitoring various types of cancers (Hao et al., 2019). During cancer progression, these parameters showed a significant increase. Similar results were observed in the DMBA group rats, and bryodulcosigenin significantly suppressed these levels, indicating its potential chemoprotective effect against breast cancer.
A reduction in mitochondrial enzymes associated with the TCA cycle, such as MDH, α-KGDH, and SDH, is regarded as a hallmark of breast cancer (Seyfried et al., 2020). Alterations in the activity of TCA cycle enzymes can indicate changes in the energy metabolism of cancer cells. The TCA cycle, commonly referred to as the Krebs cycle, is essential to cellular metabolism. Various factors in cancer contribute to the reduction in ATP molecule synthesis (Lee et al., 2022). These factors encompass changes in the morphological structure of the mammary gland, uncoupling of oxidative phosphorylation in the electron transport chain, modifications in membrane permeability (such as increased proton permeability), enhanced negative surface charge distribution, and a reduction in mitochondrial membrane fluidity (Fiorillo et al., 2021). These changes collectively lead to modifications in the oxidative phosphorylation mechanism, ultimately impacting the production of ATP, the primary energy currency of the cell (Lee et al., 2022; Seyfried et al., 2020). Rats in the DMBA group exhibited reduced levels of TCA cycle enzymes, and bryodulcosigenin significantly improved the levels of these enzymes.
Free radicals contribute to adenocarcinoma development in the mammary gland by inducing oxidative stress through metabolism and binding to nucleophilic sites on macromolecules (Patel et al., 2021; Zingue et al., 2021). The persistent generation of these mediators leads to the peroxidation of cellular lipids, culminating in malondialdehyde (MDA) expression in both tissue and serum samples. The levels of MDA can be correlated with the metastasis (invasion) and proliferation of breast cancer cells (Zingue et al., 2021). External antioxidant systems are crucial to assist lessen the impacts of oxidative stress in the body because the body’s natural antioxidant defense system might not be able to fully offset the effects of reactive oxygen species and oxidative stress (Kumar et al., 2016; Zingue et al., 2021). The DMBA-induced group exhibited elevated levels of conjugated dienes and TBARS, indicating a distinctly high level of lipid peroxidation (LPO). Bryodulcosigenin significantly suppressed these levels, suggesting protection against LPO. Endogenous antioxidants, such as CAT and SOD, are vital in combating oxidative injury. SOD and CAT work sequentially, with SOD catalyzing the conversion of superoxide radicals into hydrogen peroxide, and CAT further breaking down hydrogen peroxide into molecular oxygen and water (Pandey et al., 2018; Rajendran et al., 2019). This enzymatic antioxidant defense mechanism is essential for preserving the redox equilibrium inside cells and shielding them from the damaging effects of reactive oxygen species (Zingue et al., 2021).
GPx is a major enzyme that removes cellular peroxides, carcinogens, and xenobiotics. GPx achieves this by catalyzing reactions involving the conjugation of toxic substances with GSH as substrates, ultimately detoxifying these harmful compounds. The process involves the consumption of H2O2, and GPx plays a crucial role in maintaining cellular redox balance, protecting cells from oxidative stress, and preventing damage (Chandra Jagetia et al., 2021; Kumar et al., 2016). The reduction in GPx levels can have significant consequences for the body. GPx is essential for detoxifying intracellular peroxides, and a decrease in its activity may lead to the accumulation of reactive oxygen species (ROS) like H2O2 (Holley et al., 2017). Elevated levels of ROS can contribute to the degradation of polyunsaturated fatty acids, cross-linking of proteins, and damage to nucleic acids and lipids. This oxidative stress can have detrimental effects on cellular structures and functions, potentially contributing to various pathological conditions, including cancer. Maintaining optimal GPx activity is crucial for cellular defense against oxidative damage (Kumar et al., 2016). Bryodulcosigenin exhibited an improvement in the level of SOD, GSH, GPx, and CAT and suggesting the antioxidant effect.
ROS can significantly affect normal physiology by influencing various cellular processes (Karimi et al., 2019). The regulation of gene expression depends on ROS-responsive proteins’ roles as transcription factors and signaling mediators. Specifically, they are involved in regulating cytokines that promote inflammation (Sun et al., 2019). An imbalance in ROS levels and the inflammatory reactions that follow contribute to the pathophysiology of a number of diseases, including heart disease and cancer. Cellular homeostasis and the prevention of oxidative damage rely on maintaining a balance between the antioxidant defense system and ROS generation (Karimi et al., 2019; Sun et al., 2019). Under normal circumstances, the NF-κB transcription factor remains inactive in the cytoplasm due to its association with the inhibitory protein IκBα (inhibitor of κB alpha). This complex prevents NF-κB from translocating to the nucleus, which is essential for controlling the expression of genes related to inflammation, the immune system, cell survival, and other biological functions (Sun et al., 2019). ROS can impact this process by promoting the degradation of IκBα. Under oxidative stress or inflammation conditions, ROS can activate signaling pathways that result in the phosphorylation and subsequent degradation of IκBα (Morgan et al., 2018). This allows NF-κB to move to the nucleus and regulate the expression of its target genes. The activation of NF-κB by ROS is a crucial step in the cellular response to various stimuli, and disruption of this pathway is associated with inflammatory disorders and cancers. The intricate nature of cellular responses to oxidative stress and inflammation is underscored by the delicate balance between ROS and NF-κB signaling (Lingappan et al., 2018; Morgan et al., 2018).
Comprehending the molecular intricacies that govern apoptosis in cancer cells is imperative for developing targeted therapies. Such therapies aim to restore the delicate balance of apoptosis, offering a promising avenue for inhibiting tumor growth and progression (Wong et al., 2021). The primary cause of breast cancer development is widely acknowledged to be the dysregulation of apoptosis. The apoptosis cycle is primarily guided by two pathways: the death receptor-mediated extrinsic apoptosis pathway and the intrinsic mitochondrial pathway. Apoptosis family proteins, such as Bcl-2, work by neutralizing pro-apoptotic proteins like Bax. Bax is crucial in protecting tumor cells from apoptosis; it initially resides in the cytosol but translocates to the mitochondria to induce apoptosis. However, similar to Bcl-2 and zBcl-xl, Bax can also exhibit anti-apoptotic functions (Garner et al., 2017; Qian et al., 2022). The mitochondrial system, which encompasses both pro- and anti-apoptotic proteins, is regulated by Bcl-2. Bax and Bid move to the outer mitochondrial membrane during apoptosis, which makes Cytochrome C easier to release (Garner et al., 2017; Kim & Tilly, 2004; Qian et al., 2022). In various cells, apoptosis is orchestrated by an increased expression of Bcl-2 and Bcl-xl. Rats with DMBA-induced breast cancer exhibited reduced expression of Bax and caspase-3, alongside an increase in the expression of Bcl-2. Bryodulcosigenin treatment remarkably altered the mRNA expression of apoptosis parameters.
Conclusion
Bryodulcosigenin demonstrates breast protective effect via down-regulation of tumor weight and incidence. It effectively represses tumor markers such as AFP, CA 15-3, CEA, TSA, and LSA. Additionally, it improves mitochondrial function and regulates various cytokines and inflammatory parameters. Bryodulcosigenin modulates lipid and antioxidant levels and alters the mRNA expression of key apoptosis markers like Bcl-2, Bax, and caspase-3. The limitation of the study is that the long-term effects and safety of bryodulcosigenin are not fully understood. In the future, we will conduct extensive clinical trials to validate the efficacy and safety of bryodulcosigenin in humans.
Footnotes
Abbreviations
α-KGDH: Alpha-ketoglutarate dehydrogenase; AhR: Aryl hydrocarbon receptor; AFP: Alpha-fetoprotein; ALT: Alanine aminotransferase; AST: Aspartate aminotransferase; BRCA1: Breast cancer gene 1; BRCA2: Breast cancer gene 2; CA 15-3: Cancer antigen 15-3; CAT: Catalase; CEA: Carcinoembryonic antigen; CMC: Carboxyl methdylcellulose; COX-2: Cyclooxygenase-2; DMBA: Dimethylbenz(a)anthracene; DOX: Doxorubicin; GPx: Glutathione peroxidase; GSH: Glutathione; HDL: High density lipoprotein; H2O2: Hydrogen peroxide; ICDH: Isocitrate dehydrogenase; IL-1β: Interleukin-1β; IL-6: Interleukin-6; LPO: Lipid peroxidation, LSA: Lipid-bound sialic acid; MDA: Malondialdehyde; MDH: Malate dehydrogenase; NF-κB: Nuclear factor kappa B; O2: Superoxide radical; OH: Hydroxyl radical; PAHs: Polycyclic aromatic hydrocarbons; PGE2: Prostaglandin; PTEN: Protein tyrosine phosphatase; RNS: Reactive nitrogen species; ROS: Reactive oxygen species; SDH: Succinate dehydrogenase; SOD: Superoxide dismutase; TBRAS: Thiobarbituric acid reactive substances; TNF-α: Tumor necrosis factor-α; TSA: Total sialic acid; WHO: World Health Organization.
Acknowledgments
The authors are very thankful to the affiliated hospital of Jiaxing University for providing the necessary facility.
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All the experimental procedures adhered to the principles and protocols established by the institutional ethical committee (SGYY2024039).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
Supplementary Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
