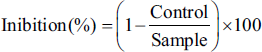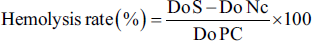Abstract
Background:
Juniperus turbinata Guss. is a medicinal mountain plant used by the Indigenous population in the traditional pharmacopeia.
Objectives
This study aimed at investigating the phytochemical analysis, antimicrobial, antioxydant and hymolytic activities of Juniperus turbinata Guss. leaves extracts.
Materials and Methods
The essential oils of Juniperus turbinata Guss. (EOJL) leaves were extracted, and their phytochemical composition was characterized by gas chromatography–mass spectrometry (GC–MS). EOJL was evaluated for its anti-free radical (2,2-diphenylpicrylhydrazyl (DPPH), ferric reducing antioxidant power (FRAP), and total antioxidant capacity (TAC)) properties, as well as antibacterial, antifungal, and hemolytic activities.
Results
The chemical study showed that EOJL contains 14 chemicals, with a 99.98% identification rate. The most abundant compounds in EOJL are linalool (45%), borneol (12%), and cineole (11%). The EOJL exhibited a 50% inhibition concentration of DPPH radicals value of 23.56 ± 0.72 µg/mL (DPPH) for its antioxidant activity, an effective concentration equivalent of 0.50 nm value of 239.08 ± 5.13 µg/mL (FRAP), and a total antioxidant activity of 735.03 ± 21.25 µg AAE/mg (TAC). The antibacterial efficacy of EOJL on a solid substrate demonstrated a range of 10–15 mm in diameter for bacterial strains, 14.33 ± 1.15 mm for C. albicans, and 56.68 ± 1.19% for Fusarium oxysporum. The bacterial strains exhibited a minimum concentration range of 6.15–11.26 µg/mL on the solid medium, whereas the range for the strains was between 5.11 and 10.23 µg/mL. The molecular docking analysis demonstrated that terpineol has potent antioxidant activity against NADPH oxidase, as shown by a shift score of –4.055 kcal mol–1. Eugenol also showed significant antioxidant activity with a shift value of –4.003 kcal mol–1. Camphor exhibited significant efficacy against S. aureus nucleoside diphosphate kinase, as demonstrated by a slip score of –5.156 kcal mol–1.
Conclusion
The findings highlight the significance of EOJL as a natural antioxidant and its antibacterial potential against pathogenic strains that are therapeutically relevant.
Keywords
Introduction
Plants serve as medicinal resources for a significant number of individuals worldwide. The utilization of traditional Moroccan medicine has expanded and become widespread across the entire nation. Extensive ethnobotanical and ethnopharmacological studies conducted in all regions of Morocco have identified over 500 plant species. Herbal medicines play a vital role in modern medicine, particularly in the creation of synthetic pharmaceuticals. Recent years have seen researchers make significant advancements in studying the biological effects of natural compounds derived from plants. Numerous studies have demonstrated the potent antibacterial properties of essential oils against various clinically relevant microorganisms. These organic items are expected to offer solutions to the growing problem of microbial resistance, which poses a significant burden on global healthcare systems (El Moussaoui et al., 2020; Yu et al., 2020). The World Health Organization conducted a study in 2008 and released the results of that poll. The survey found that more than 80% of the world’s population receives the majority of their medical care from traditional medicine. According to a study published by the World Health Organization in 2008, more than 80% of the world’s population obtains the bulk of their medical treatment from traditional medicine.
Because it is one of the most diverse conifer genera, Juniperus conifers play an important part in the dry and semi-arid habitats that are found in the northern hemisphere of Africa. Juniperus conifers include around 67 different conifer species and 28 different variants. Caryocedrus, oxycedrus, and sabina are the three subgroups that make up the Juniperus genus. Evergreen leaves that are fine, linear, prickly, and needle-like in appearance are found on this tree or shrub, which may grow to a height of 5–10 m. This plant’s blossoms yield fleshy, globular fruits that are usually referred to as berries, even though this appellation is inaccurate. In its natural environment, Juniperus flourishes on rocky, arid soil that is subject to drought.
The Juniperus turbinata, also known as J. turbinata, is a species that belongs to the Cupressaceae family and is found in abundance in the Canary Islands and North Africa, notably in Algeria, Morocco, and Tunisia. Other members of this family include around 130–140 species. In Morocco, most of its development happens at low altitudes along the beach; however, it may be found at heights of up to 2,400 m in the southern part of the Atlas Mountains. This species can exhibit a significant amount of genetic variation due to its widespread geographical range. There are many different applications for the important medicinal plant known as J. turbinata, which is utilized extensively in traditional medicine. It has earned a well-deserved reputation for being a good treatment for a variety of illnesses, including diabetes, diarrhea, and rheumatism. To manage blood sugar levels, it is also used as an oral hypoglycemic, and it is also used as a diuretic, which increases the amount of urine that is generated.
The usefulness of J. turbinata as a rich natural resource for the synthesis of antioxidant and antibacterial medicines that are effective against pathogenic strains that are regularly seen in clinical situations has been brought to light by several results (Chelouati et al., 2023). To cure illnesses such as rheumatism and diarrhea, a decoction made from the leaves of the J. turbinata plant is often employed (Lafraxo, El Barnossi et al., 2022). Gas chromatography–mass spectrometry (GC–MS) was used to identify the volatile terpenic compounds of J. turbinata. These compounds have been shown to possess a variety of pharmacological activities, such as antibacterial, anti-inflammatory, antiviral, expectorant, sedative, herbicidal, insect-repellent, and aromatic qualities.
Native American communities in the area make regular use of these herbs in their traditional medical practices. Within the framework of the process of enhancing the value of this medicinal plant, we carried out a phytochemical analysis of the essential oils using GC–MS, which was then followed by a theoretical investigation of the phytochemical composition of the essential oils by the use of molecular docking. The anti-free radical, antibacterial, and antifungal characteristics of these essential oils were evaluated in vitro, and the hemolytic activity of these essential oils was used to evaluate the toxicity of these essential oils in vivo.
Materials and Methods
Extraction of Terpene Compounds
The leaves of this plant were collected from a mountainous population (33.75750029; –4.32896628). The plant was registered and deposited in the herbarium under a voucher number (LSNA/TC/JP-22). The plant material (leaves) was dried in the dark for 2 days, and 200 g were prepared using an electric blender. The volatile compounds were extracted using a Clevenger apparatus (2,000 mL), in which 100 g of powdered leaves of the plant under study were added to 800 mL of distilled water. This mixture was heated to 120°C for 150 minutes. The EOJLs were recovered in stained vials to assess the biological activities after identification by GC–MS.
Phytochemical Analysis of EOJL by GC–MS
A Thermo Fischer Polaris QS/N 210729 mass spectrometer was used in gas chromatography together with a nonpolar HP-5MS column (60 m × 0.32 mm, film thickness 0.25 mm) and GC-FID (flame ionization detector). A 1 µL portion of EOJL was introduced onto the column after being diluted in hexane (10/90:v/v). The carrier gas was helium, flowing at a rate of 1 mL/min. The temperature profile called for an initial 2-minute heating to 40°C, followed by a 2-degree rise in temperature each minute up to 250°C, which was sustained for 10 minutes to guarantee excellent GC–MS performance. A 1:10 ratio was used to keep the injector and detector both at 260°C. Compound identification was done using mass spectra that were obtained in electron-impact mode at an ionization energy of 70 eV. For thorough compound characterization, retention indices (Ris) were created using homologous n-alkanes and cross-referenced with mass spectra from the NIST library and Adams database (Zouirech et al., 2022).
In vitro Antioxidant Activity of EOJL
The antiradical activity was determined by 2,2-diphenylpicrylhydrazyl (DPPH), ferric reducing antioxidant power (FRAP), and phosphomolybdate (total antioxidant capacity (TAC)) methods. EOJL and positive controls (butylated hydroxytoluene (BHT) or quercetin) were prepared in methanol (0.001–1 mg/mL).
DPPH Test
This evaluation was started by preparing DPPH free radicals in methanol (4 mg in 100 mL). 200 µL of EOJL was mixed with 800 µL of DPPH, and after incubation in the dark for 30 minutes at 30°C, the optical densities were calculated in a photometric spectrum at 517 nm. Antioxidant efficiency was determined by calculating 50% inhibition concentration of DPPH radicals (IC50); the lower the IC50, the higher the antioxidant efficiency. The inhibition (%) was determined according to the formula.
FRAP Test
200 µL of EOJL was added to 500 µL of phosphate buffer and 500 µL of K3Fe(CN)6 (1%). The mixture was incubated at 50°C/30 min with 500 µL of trichloroacetic acid (10%) and centrifuged at 3,000 rpm/10 min. The supernatant was isolated, and 0.5 mL of the supernatant was added to 0.5 mL H2O and 0.1 mL iron III chloride (FeCl3) (1 mg/mL). The negative and positive controls (BHT and quercetins) were prepared under EOJL conditions, just the negative control by replacing the EOJL with methanol. The optical densities of the mixtures were measured at 700 nm (Oyaizu, 1986).
TAC Test
This assay began with preparing a reagent medium: 600 mmol sulfuric acid, 4 mmol ammonium molybdate, and 28 mmol sodium phosphate. 2,000 µL of this reagent was added to 50 µL of each sample (EOJL, BHT, and quercetin), and the mixtures were incubated at 95°C/90 min. After cooling, the optical density (OD) of the solution tubes was determined using a spectrophotometer with a wavelength of 695 nm. The results of the antiradical power of the samples were expressed in micrograms of ascorbic acid equivalent (µg EAA/mg) (Prieto et al., 1999).
Hemolytic Activity
The hemolytic power of the essential oils studied was carried out in vitro at different concentrations (3, 6, 12, 25, 50, and 100 mg/mL) on a suspension of blood erythrocytes in phosphate-buffered saline (PBS) at pH = 7.4 ± 0.2 (sodium chloride (137 mM), potassium chloride (2.7 mM), sodium hydrogen phosphate (8 mM), and potassium dihydrogen phosphate (2 mM)).
Blood collected in heparin tubes from a healthy donor is used to prepare the erythrocyte suspension. It is centrifuged at 2,500 rpm for 10 minutes and, after the removal of the plasma, the pellet is washed three times with PBS, then re-suspended in the same volume of discarded plasma. The resulting erythrocyte suspension is diluted 20-fold with PBS.
The hemolytic effect test is performed according to the method of Guo-Xiang and Zai-Qun (Li & Liu, 2008). In hemolysis tubes, 20 µL of EOJL at different concentrations is added to 1,980 µL of the prepared erythrocyte suspension. These tubes are then incubated at 37°C for 60 minutes. After incubation, 250 µL samples are taken from each tube to be resuspended in 750 µL of PBS. These are gently mixed and placed in an ice bath to stop the reaction, then centrifuged at 2,500 rpm for 10 minutes.
Absorbance readings are taken at 548 nm using a spectrophotometer, against a blank containing PBS. Under the same conditions and using the same experimental procedures, we prepared a total hemolysis tube containing 100 µL of erythrocyte suspension and 1,900 µL of distilled water, and a negative control tube made up of 250 µL of erythrocyte suspension and 750 µL of PBS buffer solution. The hemolysis rate of the different extracts is calculated as a percentage of total hemolysis, after 60 minutes of incubation, according to the following formula:
Do S = optical density of the sample (EOJL); Do Nc = negative control optical density; Do PC = positive control optical density.
Antimicrobial Power of EOJL
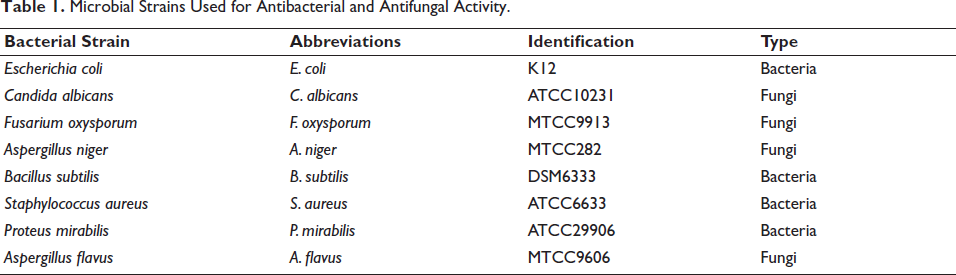
Evaluation of the antimicrobial power of EOJL was carried out on fungal and bacterial strains (Table 1). These strains were supplied by the Laboratory of Biotechnology, Faculty of Sciences, Fez, Morocco (Zouirech et al., 2022).
Microbial Strains Used for Antibacterial and Antifungal Activity.
The disc method was used as a first step on a solid medium to measure the zone of inhibition of the growth of microbial strains. 0.1 mL of the suspension (106–108 colony-forming unit (CFU)/mL) was added to 5 mL of agar (0.5%). Petri dishes were loaded with 20 mL of solid medium (Muller–Hinton and malt extract). After solidification of this medium, 6 mm diameter discs (Wattman No. 4) were disposed on the surface of the solid medium and filled with 15 µL of EOJL, oxacillin, or fluconazole. After incubating the Petri dishes at 28°C for 48–72 hours, the zone of inhibition was measured (Chebbac et al., 2022; El Barnossi et al., 2020).
The micro-dilution method was used to determine the growth inhibition concentration of microbial strains on a liquid medium. A 96-well microplate was used for this test. 100 µL of EOJL diluted in DMSO (10%) was added to the first column, and then 50 µL of liquid medium (Muller–Hinton for bacteria and malt extract for fungi) was added to all the wells. Finally, 30 µL of the microbial suspension was added to each well, after which the microplates were incubated at 28°C for 24 hours. The minimum concentration was determined by adding 20 µL of 2,3,5-triphenyl tetrazolium chloride (TTC) (0.2%) (22. 24) (El Barnossi et al., 2020; Sarker et al., 2007).
Molecular Docking
From the Protein Data Bank (PDB), the (3D) crystal structures of E. coli beta-ketoacyl-[acyl carrier protein] synthase (PDB; 1-FJ4), NADPH-oxidase (PDB; 2-CDU), and S. aureus nucleoside diphosphate-kinase (PDB; 3-Q8U) were obtained. The Schrödinger-Maestro-v11.5 protein preparation wizard was used to improve the structures. This involved adding hydrogen atoms to heavy atoms, converting seleno-methionines to methionines, and removing all water molecules. The structures were minimized using the optimized potential for liquid simulations (OPLS)-3 force field, with a maximum root mean square displacement of 0.30 Å for heavy atoms. A grid box with dimensions of 20 and a volumetric spacing of 20 connected the ligand to the protein-derived grid box. The standard precision (SP) method was used for this coupling, and the resulting outcomes were evaluated using the SP-G-Score. Flexible ligand docking was performed using the SP method in Schrödinger-Maestro v11.5’s Glide. Penalties were applied to non-cis/trans amide bonds. A partial charge cutoff of 0.15 and a Van der Waals scaling factor of 0.80 were given to the ligand atoms. Based on energy-minimized postures, the G-score was computed and recorded as the final result. For each ligand, the best-docked position with the lowest glide score value was preserved (Chelouati et al., 2023).
Statistical Analysis
The results of the tests were presented in the form of means and standard deviations (mean ± SD) based on repetitions. Statistical evaluations (Analysis of Variances (ANOVA)) were carried out using GraphPad Prism software (Version 7), with a significance threshold set at 0.05.
Results and Discussion
Phytochemical Identification of EOJL by GC–MS
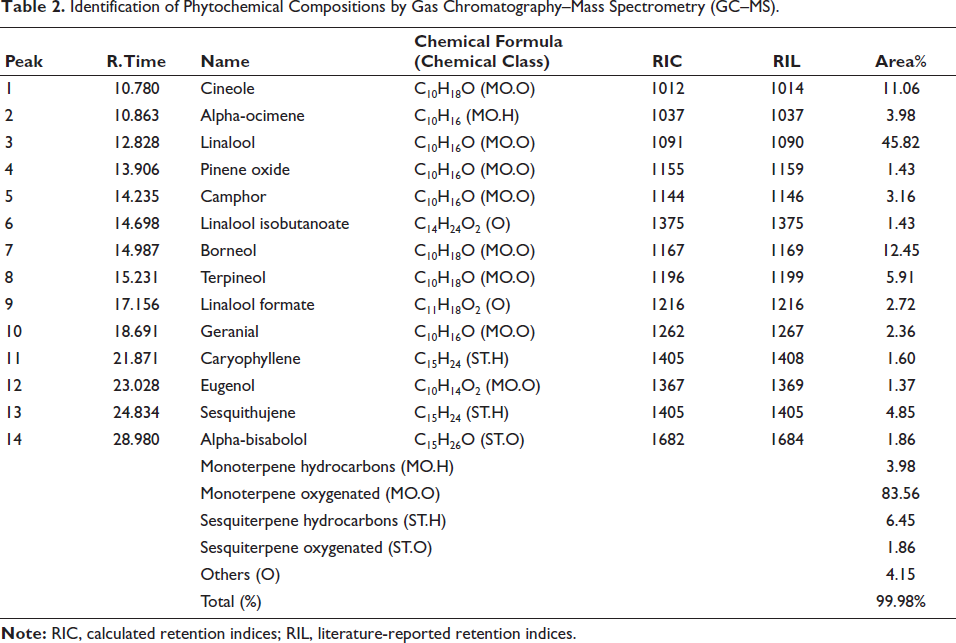
GC–MS chromatographic analysis of J. turbinata essential oils revealed 14 terpenic compounds with a total identification of 99.98% (Table 2 and Figure 1). The compounds are dominated by linalool (45%), followed by borneol (12%) and cineole (11%).
Identification of Phytochemical Compositions by Gas Chromatography–Mass Spectrometry (GC–MS).
Chromatographic Profile of Compounds Identified by Gas Chromatography–Mass Spectrometry (GC–MS).
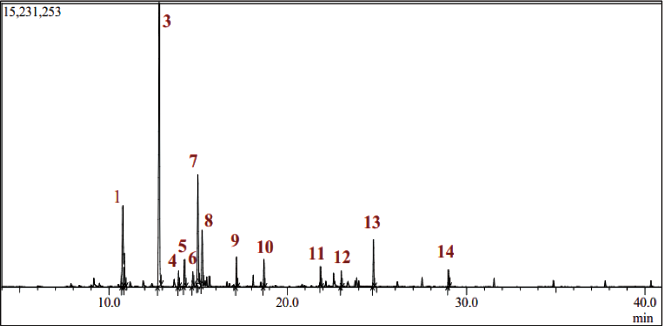
The essential oil research identified various chemical components, each categorized by structure. Monoterpene oxygenated compounds (MO.O) make up 83.56% of the makeup. Monoterpene hydrocarbon compounds (MO.H) make up 3.98% of the content. Sesquiterpene hydrocarbons (ST.H) provide 6.45%, and oxygenated sesquiterpenes (ST.O) provide 1.86% (Table 1). Another (O) group includes chemicals that do not fit into the preceding categories, making up 4.15% of the essential oils (Table 1). This chemical class distribution shows the complexity and variety of essential oils (Table 1). Recent investigations have shown qualitative and quantitative changes in the chemical composition of EOJL leaves in various locales, supporting phytochemical study findings. Chemical categorization showed that 85.1% of all plant oils were monoterpenes. Oxygenated monoterpenes range from 3.3% to 17.5%, whereas monoterpene hydrocarbons from 74.0% to 89.8%. Sesquiterpene hydrocarbons provide 0.5–5.0%, and oxygenated sesquiterpenes provide 1.1–9.2%.
Recent phytochemical characterization of essential oils extracted from this plant’s leaves in the same region of its distribution found qualitative and quantitative variations in essential oil composition, including dominant compounds and chemical classes (El Moussaoui et al., 2021a).
In addition, the leaves of 97 different J. turbinate plants were used to extract essential oils. Gas chromatographic analysis revealed that the majority of the essential oil, ranging from 45.5% to 71.8%, was composed of monoterpene hydrocarbons, with α-pinene accounting for 38.2–55.8% of this total. A number of other monoterpenes and sesquiterpenes, including myrcene, δ-phellandrene, α-terpinyl acetate, (E)-caryophyllene, and germacrene D, were also found in the samples at quite high concentrations (Rajčević et al., 2018). El Moussaoui et al. (2021b) discovered that Withania frutescens’ phytochemical composition varied qualitatively and quantitatively by season (dry and cold) and climatic variables (pH, soil, altitude, and others). Thus, intraspecific variation in phenolic content is attributed to plant climate, harvest or ripening phase, and phytochemical characterization analysis and extraction technique (Ennajar et al., 2009; Moussaoui et al., 2021).
Antioxidant Activity
Antiradical power is assessed by the DPPH method. The results in Figure 2A show that the higher the concentration of EOJL, the higher the rate of inhibition (dose–inhibition). A concentration of 25 µg/mL has an inhibition rate of 53%, 100 µg/mL has an inhibition rate of 69%, and the highest concentration (1,000 µg/mL) has an inhibition rate of 94%. These results are less significant when compared with the positive controls, including BHT and quercetin, where the 100 µg/mL concentration showed inhibition in the order of 95% and 79%, respectively (Figure 2A).
(A) Antioxidant Activity of Essential Oils of Juniperus turbinata Guss. (EOJL) by the 2,2-Diphenylpicrylhydrazyl (DPPH) Method, (B) Ferric Reducing Antioxidant Power (FRAP) Method, (C) and Total Antioxidant Capacity Using the Phosphomolybdate Method.
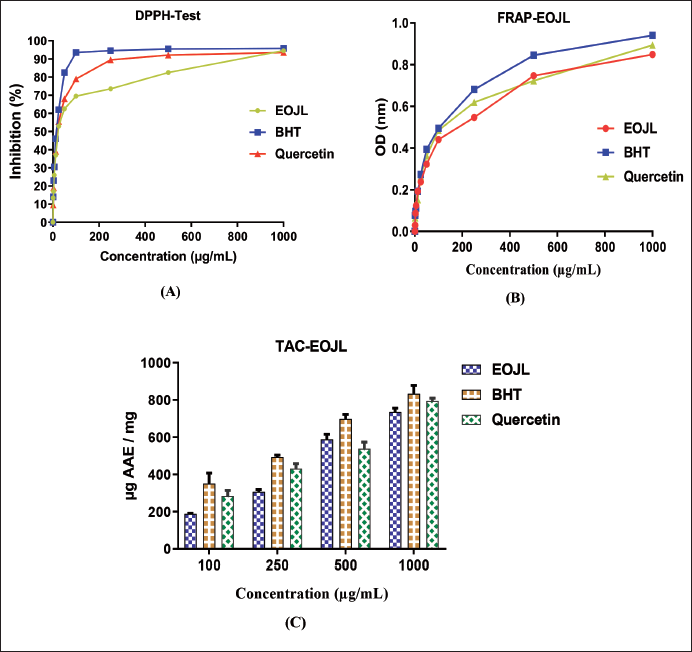
Evaluation of the antioxidant power of the essential oils was determined by IC50 (50% inhibition concentration of DPPH radicals), where the lower the IC50, the greater the antioxidant activity. The antiradical power of EOJL (IC50) was in the order of 24.18 ± 0.85 µg/mL, this activity remaining less effective when compared with BHT (IC50 = 21.47 ± 0.51 µg/mL) and quercetin (IC50 = 23.56 ± 0.72 µg/mL) (Table 3).
The Antioxidant Power of Essential Oils of Juniperus turbinata Guss. (EOJL) and the Positive Control (Butylated Hydroxytoluene (BHT) and Quercetin) by Three Methods (2,2-Diphenylpicrylhydrazyl (DPPH), Ferric Reducing Antioxidant Power (FRAP), and Total Antioxidant Capacity (TAC)).
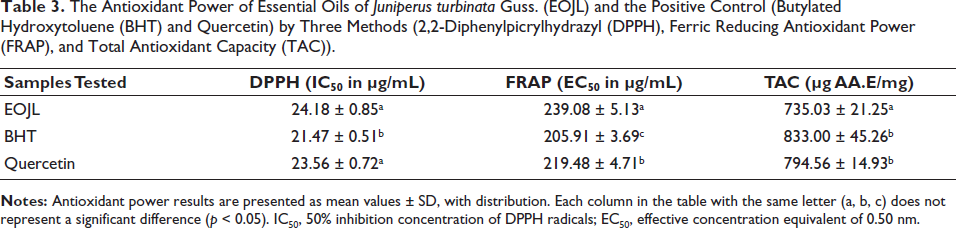
The second evaluation of the antiradical power of EOJL by the FRAP method revealed antioxidant kinetics such that as the concentration of EOJL increases, the OD increases (Figure 2B). A 25 µg/mL concentration of EOJL, BHT, and quercetin had an OD of 0.238, 0.273, and 0.263, respectively. However, a high concentration of 250 µg/mL of EOJL, BHT, and quercetin has an OD of 0.547, 0.681, and 0.619 µg/mL, respectively. The equivalent effective concentration of 0.50 nm (EC50) was in the order of 239.08 ± 5.13 µg/mL (EOJL), 205.91 ± 3.69 µg/mL (BHT), and 219.48 ± 4.71 µg/mL (quercetin) (Table 3).
The total free radical scavenging activity of EOJL was determined by the phosphomolybdate method. The results shown in Figure 2C show that a low concentration of 100 µg/mL EOJL has a free radical scavenging activity of 187.68 ± 3.28 µg AA.E/mg. However, 500 µg/mL of EOJL has an antioxidant activity of 588.75 ± 27.35 µg AA.E/mL. On the other hand, the highest concentration of EOJL (1,000 µg/mL) showed a total antioxidant activity of 735.03 ± 21.25 µg AA.E/mg (Table 3).
Recent studies have revealed that essential oils from Juniperus seeds have an antioxidant activity of around 26 µg/mL. They also found that Juniperus bark essential oils have an anti-free radical activity of 21.25 µg/mL, 23 µg/mL, and 5.36 µg/mL (Ennajar et al., 2009; Lafraxo, El Barnossi et al., 2022; Lafraxo, El Moussaoui et al., 2022). The richness of the essential oils in Juniperus leaves may confirm this plant’s anti-free radical power.
Recent studies have shown that essential oils extracted from J. turbinata seeds were considered to have antioxidant power by the FRAP method; the EC50 was in the order of 216.34 µg/mL. On the other hand, they found an antiradical power in essential oils of Juniperus thurifera with an EC50 was in the order of 190 µg/mL (leaves) and 481 µg/mL (barks), which confirms that the essential oils of these mountain plants have antioxidant activity and can trap free radicals by chelation (Chelouati et al., 2023; Lafraxo, El Moussaoui et al., 2022).
This explains why the higher the concentration, the greater the anti-radical activity. Recent studies on Juniperus plants have shown that a concentration of 1,000 µg/mL of the essential oils of these plants revealed an anti-free radical activity in the order of 720 µg AA.E/mg for the oils extracted from the seeds, 930 µg AA.E/mg for the leaves, and 271 µg AA.E/mg for the barks (Lafraxo, El Barnossi et al., 2022; Lafraxo, El Moussaoui et al., 2022).
Essential oils from J. turbinata, derived from leaves, exhibit antioxidant potential due to terpenic chemicals found in the plant. Intraspecific variation in antioxidant power may result from differences in phytochemical composition within the species. Recent studies explore seasonal, ecological, and environmental influences on phytochemical composition (El Moussaoui et al., 2021b).
The antioxidant efficacy of plant essential oils is associated with both major and minor compounds working synergistically. Terpene richness, particularly in monoterpenes (hydrocarbonated, carbonated, or oxygenated), contributes to antiradical activity. Elevated concentrations of oxygenated monoterpenes in essential oils often correlate with increased antiradical effects (Woerdenbag et al., 1993).
Hydroxyl functions in the essential oils of these plants contribute to in vitro antioxidant efficacy. Terpenes and phenolic compounds extracted from essential oils are recognized as antioxidants due to their potent antioxidant properties (Chun et al., 2005; Fayed, 2009; Zhuang et al., 2009). Essential oils rich in oxygenated monoterpenes (MO.O) demonstrate heightened antioxidant activity, as evidenced by the EOJL antioxidant power in DPPH, FRAP, and TAC tests. In vitro or in vivo assessment of antioxidant potency is vital in preclinical studies, given the association of diseases like aging, rheumatoid arthritis, diabetes, atherosclerosis, and cancer with oxidative stress (Hybertson et al., 2011; Liang et al., 2011).
Hemolytic Activity
The hemolysis test was evaluated because, even if a plant possesses powerful antioxidant power, its use in traditional medicine and pharmacological preparations will be impossible in the presence of its hemolytic effect, which indicates cytotoxicity. The results show that the hemolytic effect percentages are directly proportional to the increase in EOJL concentrations (Figure 3). After 60 minutes and all concentrations tested, hemolysis percentages ranged from 1.45 ± 0.63% to 8.29 ± 0.26%. The rate of hemolysis increased with the concentration of EOJL, with a maximum hemolytic activity of approximately 8.29 ± 0.26%. These essential oils can be slightly hemolytic at high concentrations and after 1 hour’s contact with human erythrocytes (Figure 3).
In Vitro Hemolytic Activity of Essential Oils of Juniperus turbinata Guss. (EOJL).
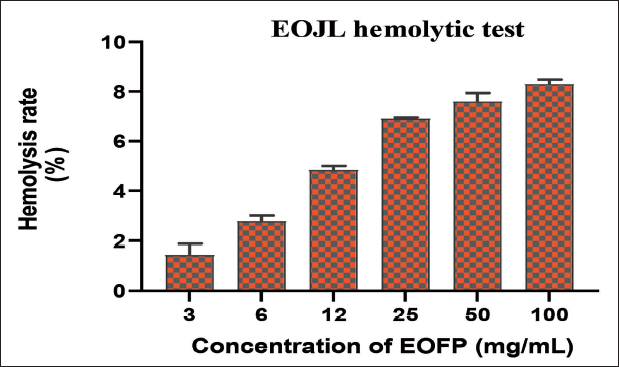
These results show that J. turbinata essential oils have a very low toxic effect on isolated erythrocytes, with a hemolysis rate of no more than 8% at a concentration of 100 mg/mL and therefore no risk of cytotoxicity.
Certain plants used for their therapeutic effect can, at high doses, present a threat to human health. The interest in the plant requires an approach to its toxicity, which led us to evaluate the hemolytic effect of this plant’s EOJL on red blood cells (El Moussaoui et al., 2020). Thus, the daily administration of therapeutic compounds can lead to their dissociation and aggregation due to interactions with blood components, as some free compounds then interact with cell membranes, extracellular matrix proteins, and blood components, resulting in undesirable side effects to human health (Fischer, 2003; Kircheis et al., 2001).
These results found in vitro confirmed that this mountain plant is not toxic, which gave the reason for the use of parts of the plant (leaves, roots) by the Indigenous population to treat certain diseases (Chelouati et al., 2023; Lafraxo, El Barnossi et al., 2022). However, this study needs further evaluation to justify the toxicity of this species, starting with acute toxicity (14 days) and subacute toxicity at repeated doses for 28 days, and evaluating the symptoms of toxicity (animal weight, behavior) in the animals (rats or mice) followed by a study of the biochemical parameters of the serum (alanine aminotransferase (ALAT) and aspartate aminotransferase (ASAT)), the relative weight of the organs (spleen, liver, kidneys), and the histological study of these organs (Chebaibi et al., 2019; El Moussaoui et al., 2020).
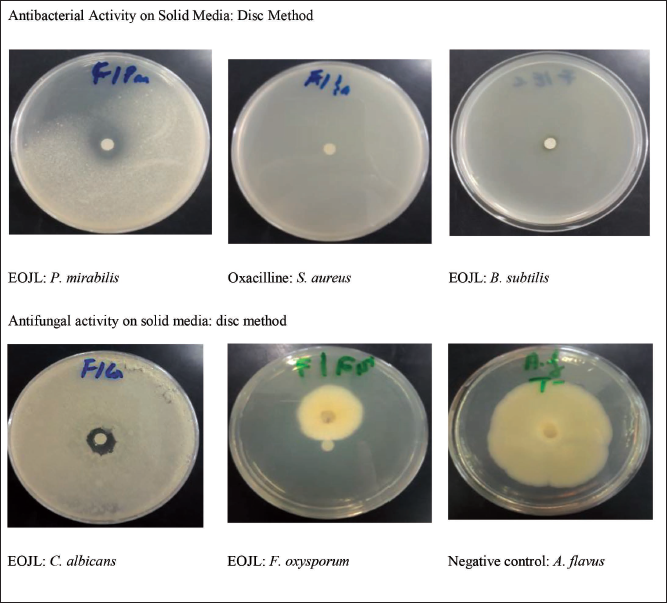
Antibacterial Power of EOJL
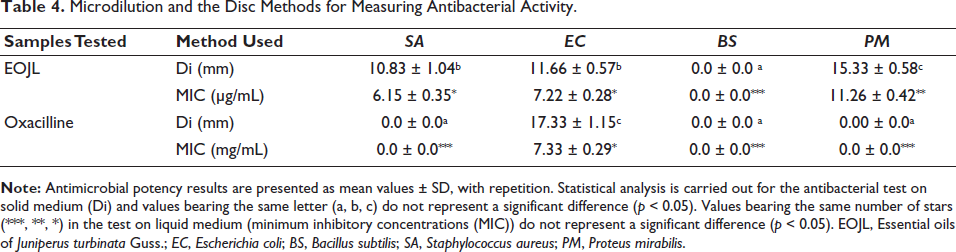
EOJL’s antibacterial activity on solid media was measured by its inhibition diameter in millimeter (Figure 4). Table 4 shows EOJL and positive control (oxacillin) inhibition diameters of bacterial growth. P. mirabilis was inhibited by 15.33 ± 0.58 mm by EOJL, followed by E. coli and S. aureus with inhibition diameters of 11.66 ± 0.57 mm and 10.83 ± 1.04 mm, respectively. Oxacillin-resistant strains, except for E. coli, were tested with an inhibitory diameter of 17.33 ± 1.15 mm (Table 4). The microdilution test revealed antibacterial activity in our essential oils, with S. aureus having the lowest minimum inhibitory concentrations (MIC) (6.15 ± 0.35 µg/mL) and P. mirabilis having the highest MIC (11.26) (Table 4). Since B. subtilis is resistant to EOJL, it was employed as the positive control in this investigation.
Antimicrobial Activity of Essential Oils of Juniperus turbinata Guss. (EOJL), Negative Control, and Positive Control on Solid Media.
Microdilution and the Disc Methods for Measuring Antibacterial Activity.
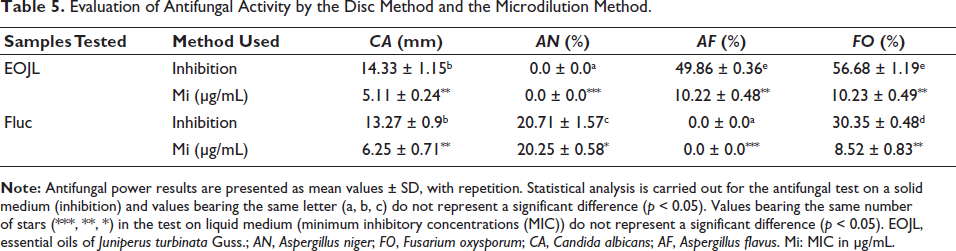
The second antimicrobial activity of these oils studied was their antifungal activity (Table 5 and Figure 4). Antifungal activity on solid media showed that the largest zone of inhibition was 14.33 mm (C. albicans), and a percentage inhibition of 56.68% (F. oxysporum). On the other hand, the microdilution method revealed that the lowest growth inhibition concentration was recorded for C. albicans (5.11 ± 0.24 µg/mL) followed by F. oxysporum (10.23 ± 0.49 µg/mL). The positive control, fluconazole, showed total resistance of the A. flavus fungal strain, while the other strains showed partial resistance by weakly inhibiting fungal mycelium growth compared to EOJL (Table 5).
Evaluation of Antifungal Activity by the Disc Method and the Microdilution Method.
The essential oils extracted from Juniperus seeds showed that terpene compounds from J. turbinata inhibit P. mirabilis, S. aureus, and E. coli at 20 mm, 11 mm, and 18 mm, respectively. However, Lafraxo, El Moussaoui et al. (2022a), Lafraxo et al. (2023), and Lafraxo, El Barnossi et al. (2022) found that Juniperus plant essential oils from seeds, leaves, and bark inhibit E. coli, S. aureus, and P. mirabilis by 11–14 mm. Juniperus plants kill gram-positive and gram-negative bacteria. Juniperus essential oils inhibited P. aeruginosa (18.27 mm), E. coli (13.23 mm), and S. aureus (31.12 mm), according to Rahhal et al. (2019). However, Bahri et al. (2012) showed Juniperus inhibited E. coli and S. aureus by 27 and 18 mm, respectively.
Shi et al. (2019) found that essential oils inhibited five phytopathogens: Phomopsis sp., F. proliferatum, C. gloeosporioides, Candida spp., and A. kikuchiana (Nóbrega et al., 2020). Jemli et al. (2018) found significant antifungal activity against F. oxysporum, Aspergillus alternata, Fusarium solani, Vibrio dahliae, and Rhizopus solani, with inhibition percentages ranging from 24% to 92.1%. These microorganisms are protected against hydrophobic terpenes by their hydrophilic outer membrane, which is rich in lipopolysaccharides and proteins. Certain gram-negative bacteria are resistant to Juniperus essential oils due to this natural alteration (El Abdali et al., 2022; Mansouri et al., 2010). Studies reveal that essential oils rich in the hydroxyl group of terpene compounds (linalool, carvacrol, and others) are more efficient antibacterial agents because they improve permeability and change the cell’s outer membrane. Blocking ATPases allows them to inhibit enzymes (Agati et al., 2012; Hybertson et al., 2011; Liang et al., 2011; Rains & Jain, 2011).
Linalool, a monoterpene alcohol with the molecular formula C10H18O, is present in essential oils derived from more than 200 plants around the globe. Some of these species include thyme (Thymus vulgaris) and juniper (Juniperus communis) (Aprotosoaie et al., 2014; Pereira et al., 2018). Along with antioxidant, anti inflammatory, and anticancer characteristics, this monoterpene alcohol is antibacterial against S. aureus, P. aeruginosa, and E. coli. Hence, it finds extensive application in the pharmaceutical, cosmetic, and food additive industries (Calo et al., 2015). According to Aprotosoaie et al. (2014), the global use of linalool is around 1,000 metric tons per annum. Natural antimicrobials derived from linalool show great promise in this regard.
There has been a lot of research on essential oils as a food preservative due to their safety and naturalness (Calo et al., 2015). Evidence suggests that linalool, a component of several essential oils, may possess antibacterial properties. It is possible that essential oils derived from plants have a stronger antibacterial impact when combined with linalool (Herman et al., 2016).
Another factor that needs to be considered in order to understand how linalool kills microbes is intracellular macromolecules. Bacteria rely on healthy cell membranes and cytoplasm for their growth, which contain macromolecules like proteins and nucleic acids. Proteins carry out important structural functions, and nucleic acids are involved in DNA translation, transcription, and replication (Guo et al., 2021). The integrity of the S. aureus cell membrane was observed to be disrupted by treatment with HE (linalool 1.201%), according to Hu et al. (2019), leading to the leaking of intracellular macromolecules such as proteins and nucleic acids. Increased intracellular permeability of macromolecules was demonstrated by HEs (linalool). However, Diao et al. (2014) found that linalool can enhance intracellular leakage of macromolecules by acting on cell membranes and affecting membrane integrity, which, in turn, releases nucleic acids and proteins across the fault membrane.
Molecular Docking
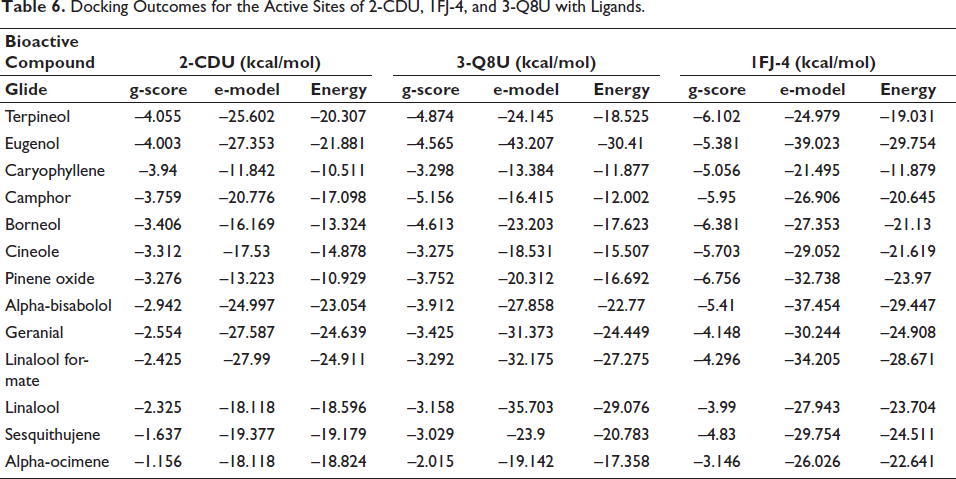
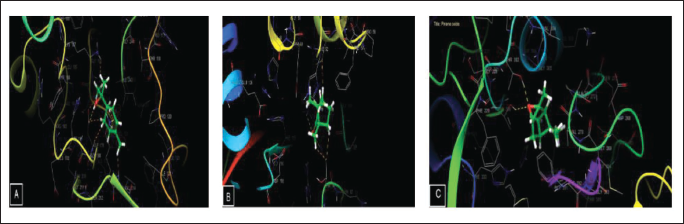
In terms of antioxidant activity, terpineol demonstrated the highest efficacy against nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, achieving a glide score of –4.055 kcal mol–1, while eugenol followed closely with a glide score of –4.003 kcal mol–1. Shifting to antimicrobial activity, camphor exhibited remarkable efficacy against S. aureus nucleoside diphosphate kinase, obtaining a glide score of –5.156 kcal mol–1, whereas terpineol showed slightly lower activity with a glide score of –4.874 kcal mol–1. When considering E. coli beta-ketoacyl-[acyl carrier protein] synthase, pinene oxide and borneol emerged as the most active compounds, with glide scores of –6.756 and –6.381 kcal mol–1, respectively (Table 6, Figures 5 and 6).
Docking Outcomes for the Active Sites of 2-CDU, 1FJ-4, and 3-Q8U with Ligands.
Viewer for the 2D Interactions of Ligands at Active Sites. (A) Terpineol Interactions in the Nicotinamide Adenine Dinucleotide Phosphate (NADPH) Oxidase Active Sites, (B) Camphor Interactions in the Nucleoside Diphosphate Kinase Active Sites of S. aureus, (C) Pinene Oxide Interactions in the Beta-ketoacylsynthase Active Site of E.coli.
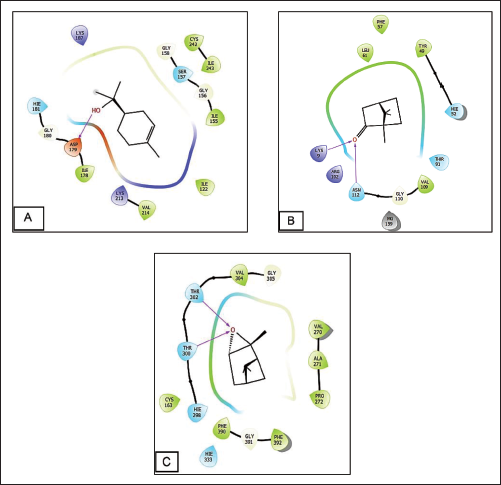
Figures 5 and 6 depict the quantity and types of bonds formed between ligands and active sites. Specifically, within the active site of NADPH oxidase, terpineol interacted with ASP 179 residues, forming one hydrogen bond. Similarly, camphor interacted with residues ASN112 and LYS9 in the active site of S. aureus nucleoside diphosphate-kinase, resulting in the formation of two hydrogen bonds. Pinene oxide established two hydrogen bonds with THR 302 and THR 300 residues in the active site of E. coli beta-ketoacyl-[acyl carrier protein] synthase.
Viewer for the 2D Interactions of Ligands at Active Sites. (A) Terpineol Interactions in the Nicotinamide Adenine Dinucleotide Phosphate (NADPH) Oxidase Active Sites, (B) Camphor Interactions in the Nucleoside Diphosphate Kinase Active Sites of S.aureus, (C) Pinene Oxide Interactions in the Beta-ketoacyl Synthase Active Site of E. coli.
Conclusion
The leaves of this alpine plant have a high concentration of terpenoid chemicals, and an in vitro evaluation of the antioxidant and antibacterial activities of the essential oils of Juniperus suggests that these oils might be used as a food additive for the purpose of preserving food. In addition, they have the potential to act as alternative agents in the treatment of a variety of infectious disorders that are caused by the bacteria that have been studied and have evolved resistance to antibiotics. In light of the relevance of this plant, it is very necessary to carry out a toxicity study, as well as a subacute toxicity evaluation in order to provide evidence for its therapeutic use. In addition, other biological and pharmacological properties, such as the capacity to kill insects, have anti-inflammatory effects, and heal wounds, should be investigated.
Footnotes
Abbreviations
EOJL: Essential oils of Juniperus turbinata Guss; DPPH: 2,2-diphenylpicrylhydrazyl; FRAP: Ferric reducing antioxidant power; TAC: Total antioxidant capacity; BHT: Butylated hydroxytoluene; IC50: 50% inhibition concentration of DPPH radicals; EC50: Effective concentration equivalent of 0.50 nm; TTC: 2,3,5-Triphenyl tetrazolium chloride; FeCl3: Iron III chloride; GC–MS: Gas chromatography with mass spectrometry; MIC: Minimum inhibitory concentrations; CFU: Colony-forming unit; PBS: Phosphate-buffered saline; NADPH: Nicotinamide adenine dinucleotide phosphate; PDB: Protein Data Bank; OPLS: Optimized potential for liquid simulations; SP: Standard precision.
Data Availability Statement
Derived data supporting the findings of this study are available from the corresponding author on request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is financially supported by the Researchers Supporting Project number RSP 2024/236, King Saud University, Riyadh, Saudi Arabia.