Abstract
Background
Kirkia acuminata is a medicinal plant highly renowned in traditional African medicine for its ability to treat various diseases.
Objectives
This study aims to evaluate the pharmacological and phytochemical properties of Kirkia acuminata.
Methods
The Gas Chromatography-Mass Spectrometry (GC-MS) technique was employed to investigate the phytochemical composition, while the antibacterial efficacy of the stembark extract of the plant species was assessed against Vibrio cholerae using the agar well diffusion method. The Brine Shrimp Lethality Assay (BSLA) was utilized to evaluate the cytotoxic characteristics of the plant species.
Results
The GC-MS analysis identified 27 different phytochemical constituents, with the major compounds being tetrapentacontane (23.04%), tetracontane (13.19%), 1-docosanol (12.89%), 2,4-bis(1,1-dimethylethyl)-phenol (9.49%), octacosanol (7.35%), 1-nonadecene (6.97%), and 1-heptadecene (5.52%). These compounds have previously been described as having antimicrobial activity. The stembark extract exhibited notable antibacterial activity, with a minimum inhibitory concentration (MIC) of ≤ 1 mg/mL. Furthermore, the extract showed no signs of toxicity, as indicated by the Lethal Concentration 50 (LC50) values of 1583.34 μg/mL.
Conclusion
These findings offer scientific data that supports the use of the plant species in traditional medicine. However, more research is needed to evaluate any toxicological effects of long-term or repeated plant species concoctions use in traditional medicine. Furthermore, further research is necessary to isolate phytochemical constituents and better understand their mechanisms of action.
Keywords
Introduction
Medicinal plants have historically been employed in folkloric medicine for their bioactive secondary metabolites, which exhibit diverse pharmacological properties. 1 A significant fraction of the African population depends on phytomedicines, particularly in regions with little access to contemporary healthcare.2,3 Among these plants, Kirkia acuminata Oliv. (White Syringa) is commonly used in Southern and Central Africa for the treatment of a variety of diseases, including cholera.2,4 In Zimbabwe, the bark decoction or powdered bark of K. acuminata is used to treat cholera, a life-threatening disease, and other related diarrhoel diseases.2,5,6
Despite its widespread traditional use,6,7 substantial phytochemical and pharmacological research on this plant species is still lacking. Previous research on K. acuminata has relied mostly on qualitative phytochemical screening, which revealed the presence of fatty acids, phenolics, and other bioactive chemicals, 8 many of which have antimicrobial properties against human pathogens. 9 Existing research indicates that K. acuminata extracts have antibacterial activities against pathogens such as Vibrio cholerae, Staphylococcus aureus, and Mycobacterium smegmatis.10,11 However, modern analytical techniques such as Gas Chromatography-Mass Spectrometry (GC-MS), which are required for identifying volatile phytoconstituents, have not been fully applied to this species. Considering that numerous phytoconstituents present in medicinal plants like K. acuminata and similar species may exhibit antibacterial and cytotoxic properties, further research is necessary to validate their traditional uses.2,12
The safety of medicinal plants should not be assumed solely based on their long history of use, as certain plants may exhibit cytotoxic properties. 13 Despite its widespread usage in traditional medicine, K. acuminata preparations have not been extensively investigated for toxicity. 14 While preliminary research on other related Kirkia species suggest low toxicity,15,16 K. acuminata has not been thoroughly toxicologically evaluated. Therefore, due to its extensive historical application, a comprehensive assessment of its cytotoxicity is necessary.
This study aims to bridge these gaps by using GC-MS to analyse the phytochemical constituents of K. acuminata stembark extracts and assess their antibacterial and cytotoxic properties. The findings will shed light on its therapeutic potential and safety, supporting ethnomedicinal applications and directing future drug research efforts.
Materials and Methods
Plant Material
The stem bark of K. acuminata was collected from the parent plants from the main campus of Great Zimbabwe University, Zimbabwe (20.1037° S, 30.8608° E). A qualified botanist, Christopher Chapano from the National Herbarium of Zimbabwe, confirmed the authenticity of the plant species, and sample specimens were deposited in the aforementioned herbarium under the voucher number MZMJ6.
Stem-Bark Extraction
Extraction was carried out following the protocol of Mabadahanye et al, 10 with some few modifications. Bark samples were allowed to air dry in the shade at room temperature and then ground into a powder using a traditional mortar and pestle, to mimic the traditional method of grinding medicinal plants. Large particles were removed using a 2 mm sieve, and the remaining powder was stored at room temperature in airtight containers in the dark. The maceration method was employed at room temperature. Approximately 100 g of powdered samples were extracted over 48 h in 500 mL of water for the antibacterial and cytotoxic assay to mimic traditional remedies preparation. Chloroform was used for the preparation of extracts for GC-MS analysis. The extracts were filtered using Whatman No. 1 filter paper (Whatman International Ltd, England) and concentrated with a rotary evaporator (RE 100-Pro, Biobase, China) under reduced pressure at 50 ± 2 °C. The crude extract was then allowed to air dry at room temperature to remove traces of the solvent.
Gas Chromatography-Mass Spectrometry Analysis of the Extract
The GC-MS analysis was conducted according to the protocol outlined by Kumar et al, 17 with slight adjustments. The analysis was performed using a Shimadzu GCMS-QP-2010 Ultra instrument (Shimadzu Corporation, Japan) equipped with a ZB-5MS column (30 m × 0.25 mm ID × 0.25 µm df) packed with 5% phenyl siloxane. The carrier gas was Helium (99.999%) at a flow rate of 1.0 mL/min and an injection volume of 2 µl was employed (Split ratio of 10:1). The injector temperature was 250 °C; ion-source temperature was 280 °C. The oven temperature was programmed from 110 °C (isothermal for 2 min) with an increase of 10 °C/min to 200 °C, then 5 °C/min to 280 °C, ending with a 9 min isothermal at 280 °C. The Mass spectra were taken at 70 eV. The relative percentage (%) amount of each component was determined by comparing its average peak area to the overall areas. Mass spectra were processed using the software. The NIST database was utilized to identify the specific phytochemical constituents.
Antimicrobial Activity Assessment of the Plant Extract
The antimicrobial activity was assessed on V. cholerae isolates using the agar well diffusion method. Vibrio cholerae is designated as a Biosafety Level 2 pathogen; therefore, all experiments were conducted in a certified Biosafety Level 2 (BSL-2) laboratory, following institutional safety protocols including the use of personal protective equipment and biosafety cabinets. 18 A standardized bacterial suspension equivalent to 0.5 McFarland standard (≈1.5 × 108 CFU/mL) was prepared, and 100 µL was evenly spread on Mueller–Hinton agar (MHA) plates. Wells of 7 mm diameter were aseptically punched into the agar and filled with 100 µL of the extract. An initial screening was performed at 60 mg/mL, followed by assays using serial concentrations of 1, 5, 10, and 20 mg/mL. The negative control was sterile water, whereas the positive control was Ciprofloxacin (30 µg/mL). Plates were incubated at 37 ± 2 °C for a duration of 24 h, and the diameters of the inhibition zones were measured in millimeters utilizing a Vernier caliper. All assays were performed in triplicate.
The minimum inhibitory concentration (MIC) was determined from the same concentration range and defined as the lowest extract concentration producing a measurable zone of inhibition. Based on Ríos and Recio, 19 antibacterial activity was classified as potent (MIC ≤ 1 mg/mL), moderate (1.1-6 mg/mL), or weak (>6 mg/mL).
Standard deviation (SD) ± mean was used to express the data. The evaluation of statistical significance was conducted through one-way analysis of variance (ANOVA), subsequently applying Tukey's Honest Significant Difference (HSD) test with a threshold of p < 0.05. A dose–response relationship was evaluated through regression analysis, with the model's fit assessed using the coefficient of determination (R²).
Cytotoxic Test
The current study employed the Brine Shrimp Lethality Assay (BSLA) to evaluate the cytotoxic activity of the test extract, following the protocols of Karim et al 20 and Waghulde et al. 21 Ten milliliters of artificial seawater were mixed with 0.1 gram of plant extract to achieve a final stock concentration of 10 000 μg/mL. Through serial dilutions, test extracts with different concentrations (1000 μg/mL, 100 μg/mL, 10 μg/mL, and 1 μg/mL) were prepared.
For each dilution, 0.5 mL of test extract was added to 4.5 mL of seawater containing 10 nauplii in separate vials. For every concentration of the test extract, three replications were employed. Distilled water was used as a near control, and Vincristine sulphate as a positive control. After 24 h the number of dead nauplii was recorded, and the percentage mortality for each concentration was calculated. Nauplii were considered dead if they did not exhibit any movement during observation under a magnifying lens. The LC50 (lethal concentration required to kill 50% of the test organisms) was determined using probit regression analysis. Clarkson's toxicity criterion was used to classify extracts in the following order: extracts with LC50 above 1000 μg/mL are non-toxic, LC50 of 500–1000 µg/mL are weakly toxic, extracts with LC50 of 100–500 μg/mL are moderately toxic, while extracts with LC50 of 0–100 µg/mL are highly toxic. 22
Results
Gas Chromatography-Mass Spectrometry Analysis of the Extract
The gas chromatography-mass spectrometry (GC-MS) analysis of the stembark chloroform extract of K. acuminata identified a total of 27 different phytochemical constituents. The identification was based on retention time, peak area, molecular formula, molecular weight, and molecular structure (Table 1; Supplementary Figure S1). The major compounds being tetrapentacontane (23.04%), tetracontane (13.19%), 1-docosanol (12.89%), 2,4-bis(1,1-dimethylethyl)-phenol (9.49%), octacosanol (7.35%), 1-nonadecene (6.97%), 1-heptadecene (5.52%), 1-dodecanol (3.22%), heneicosane (2.29%), (E)-4-Benzylidene-3-phenyl-3,4-dihydronaphtho[2,3-h]quinazoline-2(1H),7,12-trione (1.87%), and n-triacontanol (1.72%).
GC-MS Analysis for K. acuminata Stembark Extract.

Antimicrobial Activity of Extract
The aqueous extract of K. acuminata stembark demonstrated a dose-dependent antimicrobial effect against V. cholerae, with inhibition zones measuring from 10.3 ± 0.6 mm at 1 mg/mL to 19.0 ± 1.0 mm at 20 mg/mL (Table 2). The estimated MIC was found to be ≤1 mg/mL (Figure 1). Significant differences were detected among the concentrations (p < 0.05), with the exception of the comparison between 10 mg/mL and 20 mg/mL, suggesting a plateau in activity. The positive control, ciprofloxacin, produced a zone of inhibition measuring 25.0 ± 1.0 mm. A robust logarithmic dose-response relationship was identified (R² = 0.92) (Figure 2).

Clear Zone of Inhibition at Estimated MIC of (≤1 mg/mL).

Dose-response Relationship of K. acuminata Extract.
Antibacterial Activity of K. acuminata Stembark Aqueous Extract Against V. cholerae.

Different letters indicate significant differences (p < 0.05, Tukey's HSD test).
Cytotoxicity of Kirkia acuminata Extract in Brine Shrimp Assay
The lethality concentration (LC50) of the test extract was determined to be 1583.34 μg/mL (Table 3; Figure 3), indicating low cytotoxicity.

Determination of LC50n of Kirkia acuminata Stembark Extract.
Mortality of Brine Shrimp Nauplii after 24 h Exposure to K. acuminata Extract.
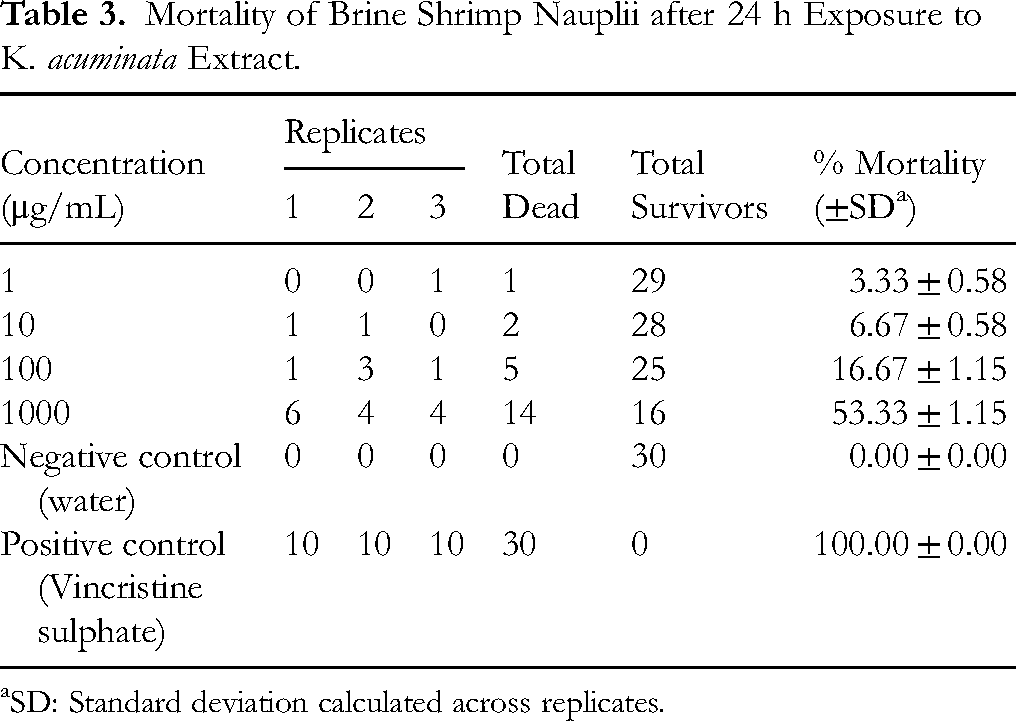
SD: Standard deviation calculated across replicates.
Discussion
Kirkia acuminata is used to treat several diseases, particularly in East, Central, and Southern Africa, where it is exclusively indigenous. 2 The scientific analysis of such therapeutic plants, encompassing their phytochemistry, antibacterial characteristics, and cytotoxic impacts, is essential in confirming their efficacy in traditional medicine.23,24 In this study, gas chromatography-mass spectrometry (GC-MS) analysis of the K. acuminata stem bark chloroform extract revealed 27 different phytochemical constituents, primarily hydrocarbons, alcohols, esters, and sterol-related compounds, in contrast to the previously reported predominance of fatty acids and phenolics.8,25,26 This variation may be attributed, in part, to factors such as the choice of extraction solvent, which can influence the chemical profile obtained. This necessitates employing biphasic extraction (eg, chloroform + methanol/water) to capture both polar and non-polar fractions in future comparative analyses. Furthermore, GC-MS alone may have limited the findings, as it only detects volatile compounds. 27 For comprehensive profiling, combining it with Liquid Chromatography-Mass Spectrometry (LC-MS) would detect both polar and non-volatile compounds. 28
Nonetheless, several studies have reported the biological activities of most major phytochemical constituents found in our study.29-32 This endorses the usage of K. acuminata in traditional treatments for several diseases, including cholera. 2 For example, the most abundant compound, tetrapentacontane (23.04%), was previously discovered in the hydro-alcoholic extract of Aristolochia tagala Cham. a medicinal plant employed in the treatment of stomach aches. 30 This extract demonstrated significant antibacterial activity against Proteus valgaris, Pseudomonas aeruginosa, Escherichia coli and Enterobacter aerogenes, with MIC ranging from 10.93 to 43.7 μg/mL. The same compound was also found (13.97%) in Matthiola tricuspidata ethanol extract with documented antibacterial effects. 32
Several other identified compounds in our extract have documented antimicrobial properties.33-35 For example, heneicosane (57.06%) and 1-heptadecene (10.09%), isolated from Aporosa cardiosperma leaf extract exhibited significant antibacterial properties against drug-resistant bacteria (eg, Staphylococcus aureus, Bacillus subtilis, Mycobacterium tuberculosis, Klebsiella pneumoniae, Aeromonas hydrophila, and Pseudomonas aeruginosa). 33 Similarly, essential oil from Tragopogon graminifolius contained heneicosane (6.6%) and showed notable antibacterial activity against Shigella dysenteriae and Proteus vulgaris. 36 Furthermore, octacosanol isolated from Cibotium barometz (L.) displayed remarkable antioxidant, antibacterial, and antifungal activities. 37 Similarly, behenic alcohol showed antimicrobial activity against human pathogenic bacteria with mean inhibition zones ranging from 9.5 ± 0.91 mm to 11.3 ± 1.6 mm. 32
The aqueous extract of K. acuminata stembark demonstrated pronounced, dose-dependent bactericidal efficacy against V. cholerae, with inhibition zones ranging from 10.3 ± 0.6 mm (1 mg/mL) to 19.0 ± 1.0 mm (20 mg/mL). ANOVA with Tukey's HSD revealed significant differences at most concentrations (p < 0.05); however, no additional increase was noted between 10 and 20 mg/mL, suggesting activity saturation. More importantly, the extract demonstrated a robust logarithmic dose-response (R² = 0.92), indicating its reliable efficacy. Notably, the MIC was determined to be ≤1 mg/mL, indicating a commendable level of potency, albeit approximately 16-fold less effective than ciprofloxacin (MIC = 0.06 μg/mL). The findings underscore K. acuminata's potential as an effective natural antimicrobial agent against V. cholerae. More importantly, the findings also somehow validate the traditional preparation of herbal remedies, which typically employ aqueous extraction. However, due to the limitations of GC-MS in analysing complex aqueous matrices, a non-polar solvent, chloroform, was employed for phytochemical profiling to selectively identify low-polarity compounds, which could lead to a variation in the level of antibacterial efficacy. While this approach does not fully mirror the traditional extraction method; instead, it serves to complement the biological assay by offering an insight into the broader phytochemical profile of the plant.
Another limitation of our study was testing against only one bacterial species. Given the traditional use of K. acuminata in treating a wide range of infections,6,7,38 a broader assessment involving both Gram-positive and Gram-negative bacteria would have provided a more comprehensive evaluation of its antimicrobial potential. Nonetheless, several studies have reported notable antibacterial activity of K. acuminata extracts against various pathogens.10,39,40 These findings reinforce the plant's potential as a broad-spectrum antibacterial agent. For example, extracts of K. acuminata obtained using acetone, dichloromethane, hexane, and methanol demonstrated inhibitory effects against Enterococcus faecalis, Escherichia coli, Pseudomonas aeruginosa, and Staphylococcus aureus. 38 Similarly, Chigayo et al 11 found that aqueous extracts of Kirkia spp. had notable antibacterial activity against V. cholerae, with an average MIC of 3.445 mg/mL. A recent study revealed that crude extracts from K. acuminata stem ascomycete isolates inhibited E. faecalis, E. coli, P. aeruginosa, S. aureus, and Candida albicans with MICs of 0.31 to 2.5 mg/mL, while C. albicans had 1.25 to 2.5 mg/mL. 39
The BSLA demonstrated a significant dose-dependent increase in mortality (linear regression: R² = 0.994, p < 0.05), with the LC50 of K. acuminata extract determined to be 1583.34 μg/mL via probit analysis. This value, substantially higher than the threshold for cytotoxicity (LC50 > 1000 μg/mL), classifies the extract as non-toxic or weakly toxic under experimental conditions. 22 The low variability among replicates (SD ≤ ± 1.15% mortality) underscores the reproducibility of the results. Accordingly, the use of K. acuminata extract in traditional medicine is considered safe. These findings are significant as toxicological evaluations of K. acuminata are scarce in the literature. 14 Only one prior study by Chigayo 15 established that certain Kirkia species are non-cytotoxic, while Suleiman et al 16 reported low toxicity values (ranging from 0.80 to 1.25 mg/mL) for other Kirkia species, including K. wilmsii. Although the BSLA findings from this study support the traditional use of K. acuminata in disease therapies, the assay has limitations. It provides preliminary toxicity data and does not predict mammalian toxicity. To obtain more relevant safety data, cytotoxicity testing on cell lines (eg, human keratinocytes or hepatocytes), other in vitro toxicological assessments and in vivo studies (such as animal models), should be conducted. Additionally, the assay cannot resolve dose-response ambiguities, particularly since traditional remedies often require prolonged use. Thus, the potential toxicological effects of chronic or sub-chronic exposure to K. acuminata extracts remain to be determined.
Conclusion
The GC-MS analysis identified 27 distinct phytochemical constituents in the K. acuminata extract. The major compounds included tetrapentacontane (23.04%), tetracontane (13.19%), 1-docosanol (12.89%), 2,4-bis(1,1-dimethylethyl)-phenol (9.49%), octacosanol (7.35%), 1-nonadecene (6.97%), and 1-heptadecene (5.52%). Our study demonstrated significant antibacterial efficacy of the extract against V. cholerae, with a MIC of ≤ 1 mg/mL. This activity can be attributed to the identified phytochemical constituents, many of which have been previously reported to have antimicrobial effects. Furthermore, the BSLA findings showed that the extracts exhibited no cytotoxic effects. These findings corroborate the traditional use of K. acuminata in herbal medicine, using maceration extraction method to reflect indigenous preparation techniques. Nonetheless, this investigation has some limitations. GC-MS analysis of the extract could not detect highly polar compounds (eg, sugars, glycosides, amino acids, and polypeptides) due to their non-volatility and poor solubility in non-polar chloroform. To address these limitations, future research should combine GC-MS (volatiles/lipids) and LC-MS (polar/non-volatile metabolites) for comprehensive profiling.
Additionally, the antibacterial and cytotoxicity tests performed were in vitro assays, and the conclusions are preliminary, necessitating further validation through more sophisticated experimental models, such as in vivo studies or mechanistic investigations. While the findings offer initial insight into the bioactive potential of K. acuminata, further investigation is required to isolate its active phytochemicals, evaluate their individual and synergistic effects, and clarify their mechanisms of action.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251357459 - Supplemental material for Chemical Profiling of Kirkia acuminata Oliv. Stembark Extracts by GC-MS Analysis and Evaluation of its Antibacterial and Cytotoxic Activity
Supplemental material, sj-docx-1-npx-10.1177_1934578X251357459 for Chemical Profiling of Kirkia acuminata Oliv. Stembark Extracts by GC-MS Analysis and Evaluation of its Antibacterial and Cytotoxic Activity by Jerikias Marumure and Monde A Nyila in Natural Product Communications
Footnotes
Acknowledgements
This work was conducted as part of a broader PhD research project entitled, “Identification of Bioactive Compounds and Evaluation of Antibacterial Activity of Selected Medicinal Plants against Vibrio cholerae.” The project is being supervised by Dr. Monde. A. Nyila. Jerikias Marumure thanks the Department of Life and Consumer Sciences, College of Agriculture and Environmental Sciences, University of South Africa, for hosting the research.
Ethical Considerations
Ethical approval was granted for the PhD research project of which the current study is part of it.
Authors’ Contributions
We declare that the authors listed in this article are the ones who took this work.
Jerikias Marumure - conceptualizing, designing, and writing the draft manuscript.
Monde. A. Nyila - conceptualizing, designing, supervising, and reviewing the draft manuscript.
All the authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: I am the member of the Editorial Review Board.
Data Availability Statement
All relevant research data have been included in the manuscript.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal participants.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
