Abstract
Background
Over time, the prevalence of allergic rhinitis (AR) has surged due to various risk factors, notably attributed to global urbanization, rendering heightened levels of pollutants, including traffic-related emissions and particulate matter. About 25% of the global children population and 40% of the adult population were reported with AR. Even though AR is recognized as a systemic inflammatory disease, it often results in diverse other comorbidities, such as dermatitis, sinusitis, conjunctivitis, and otitis, requiring extensive and expensive treatment.
Objectives
We assessed the effectiveness of iridoid glucoside catalpol against AR in a mouse model. Catalpol is recommended in traditional Chinese medicine to treat diverse acute and chronic diseases.
Materials and Methods
AR was induced in mice with an ovalbumin-sensitized AR model and treated with 10 and 20 mg of catalpol. Nasal severity scoring was performed to confirm the AR induction in mice. Allergic mediators immunoglobulin E (IgE) and histamine were quantified in the serum to assess the anti-allergic response of catalpol. In nasal lavage fluid (NALF), the inflammatory mediators IgE Ab, prostaglandin D2, leukotriene C4, eosinophil cationic protein (ECP), and pro-inflammatory cytokines were measured to analyze the anti-inflammatory potency of catalpol. The binding capacity of nuclear factor-kappa B (NF-κB) to DNA was evaluated to assess the catalpol inhibitory potency against NF-κB-mediated inflammation in AR mice. To confirm the ameliorative potency of catalpol in AR mice, a histopathological analysis of nasal mucosa was performed.
Results
Catalpol treatment significantly decreased the nasal symptoms and reduced the allergic mediators in the serum of experimental animals. It effectively inhibited the synthesis of inflammatory mediators, ECP, and pro-inflammatory cytokines in the NALF, and also suppressed the NF-κB DNA-binding activity in AR mice. The decrease in ciliary loss, goblet cells, eosinophil infiltration, and vascular congestion observed with our nasal mucosa histopathological analysis confirmed the ameliorative potency of catalpol.
Conclusion
Our findings have proven catalpol inhibits NF-κB-mediated inflammatory response in AR mice. With further analysis, a potent natural compound, catalpol, can be formulated as a drug to treat AR.
Keywords
Introduction
Allergic rhinitis (AR), a prevalent inflammatory condition affecting the nasal mucosa, has a severe impact on a significant portion of the global population (Urrutia-Pereira et al., 2023). According to global statistics, AR affects approximately 40% of the global population and 10–30% of adults (Savouré et al., 2022). The prevalence of AR varies across different regions, with higher rates observed in urban areas and developed countries. In recent years, the incidence of AR has rapidly increased, particularly in urbanized areas undergoing swift industrialization and environmental changes (Song et al., 2023). Factors such as air pollution, pollen exposure, indoor allergens, and changing lifestyles contribute to the rising burden of AR globally (Gu et al., 2024; Zhaobin et al., 2024). This ailment not only affects the well-being of those it afflicts but also places a substantial financial strain on healthcare systems due to healthcare utilization, medication costs, and loss of productivity. Efforts to raise awareness, improve diagnosis, and implement effective management strategies are crucial for addressing the growing public health challenge posed by AR worldwide.
Nuclear factor-kappa B (NF-κB)-mediated inflammatory response plays a pivotal role in orchestrating the immune reaction to allergens in AR pathology (Lawrence, 2009; Sun & Liu, 2011). Upon exposure to allergens, epithelial cells lining the nasal mucosa recognize these foreign substances via pattern recognition receptors (PRRs) like toll-like receptors (TLRs). TLRs further trigger signaling pathways that converge NF-κB signaling, resulting in pro-inflammatory gene transcription. These genes encode various chemokines, cytokines, adhesion molecules, and inflammatory enzymes (Kang et al., 2023). The release of these inflammatory mediators leads to eosinophil recruitment and activation of immune cells such as T lymphocytes, amplifying the inflammatory response in the nasal mucosa (Hayden & Ghosh, 2011). Consequently, this cascade of events results in the characteristic symptoms of AR, including nasal congestion, rhinorrhea, sneezing, and nasal itching. Targeting NF-κB signaling pathways offers a hopeful avenue for therapeutic intervention for managing AR by mitigating the inflammatory response and alleviating symptoms associated with the condition (Wee et al., 2017; Yu et al., 2020a).
Catalpol, a natural iridoid compound found in the roots of plants such as Rehmannia glutinosa, has garnered increasing attention in pharmaceutical research due to its diverse pharmacological properties and potential therapeutic applications (Bhattamisra et al., 2019; Zhang et al., 2019). Multiple investigations on catalpol have highlighted its anti-inflammatory, antioxidant, neuroprotective, antidiabetic (Bai et al., 2019), and cardiovascular protective effects (Zhang et al., 2023), among others. In particular, its ability to modulate various signaling pathways, such as NF-κB and nuclear factor erythroid 2-related factor 2 (Nrf2) (Fu et al., 2023; Zhang et al., 2024), suggests a promising potential for managing inflammatory disorders, neurodegenerative disorders, diabetes, and cardiovascular conditions. Despite these promising findings, additional investigation is warranted to fully interpret catalpol’s pharmacological mechanisms and evaluate its safety and efficacy for clinical use. Hence, we assessed the antiallergic potency of natural iridoid glucoside catalpol in an ovasensitized AR mice model
Materials and Methods
Animals
Healthy BALB/c mice aged about 6–8 weeks were obtained from the institutional animal facility, acclimatized for a week, and maintained under the laboratory conditions prescribed by global laboratory settings for rodents. A sterile environment with standard laboratory conditions was maintained in the animals. A 12-hour light-dark cycle was maintained and the mice were fed a standard rodent pellet diet. The diet and water were provided ad libitum, and only ethical committee-approved procedures were executed on the mice (No. 2023/45).
AR Rodent Mole
AR was established in BALB/c mice with aluminum hydroxide treatment followed by ovalbumin (OVA) sensitization. Acclimatized mice were treated intraperitoneally with four doses of 75 µg of OVA and 2 mg aluminum hydroxide dissolved in 200 µL of phosphate-buffered saline (PBS) at intervals of 7 days from 0 to 21st day of the experiment. The mice were then sensitized with 25 mg/mL of OVA through nasal injections on the experimental days 23, 25, and 27 (Bui et al., 2017).
Experimental Groupings (n = 6)
Group I (Control): Received normal saline; Group II (OVA-induced): Mice were sensitized with OVA along with aluminum hydroxide via intraperitoneal injection on days 0, 7, 14, and 21. They were then administered OVA solution via nasal cavity injection on days 23, 25, and 27; Group III (OVA + catalpol 10 mg/kg): OVA-sensitized mice treated with 10 mg/kg of catalpol from days 22 to 28; Group IV (OVA + catalpol 20 mg/kg): OVA-sensitized mice treated with 20 mg/kg of catalpol from days 22 to 28; Group V (OVA + dexamethasone 2.5 mg/kg): OVA-sensitized mice treated with 2.5 mg/kg of dexamethasone from days 22 to 28.
The animals were euthanized 24 hours after the last dose treatment, and samples of nasal and lung tissues, as well as nasal lavage fluid (NALF), were collected for subsequent analysis (Sagit et al., 2017).
Nasal Symptom Severity Evaluation
After the final dose of OVA sensitization, the mice were placed in the observation chamber for 10 minutes and observed for nasal severity symptoms such as rubbing the nostrils and sneezing. Individually, each mouse from all the experimental groups was placed in an observation chamber, and the nasal symptoms were recorded. The observation chamber was sterilized before the initiation of the experiment. The experiment was performed by observers unaware of animal grouping (Suzuki et al., 2017).
NALF Collection
Anesthetized mice were securely positioned by extending the neck to facilitate access to the nasal cavity. Sterile saline solution was gently administered into one nostril, allowing it to flow into the nasal cavity before being aspirated back out. The procedure was repeated with the other nostril, and the collected fluid was preserved at –20°C for further analysis (Fan et al., 2019).
Quantification of Allergic Serum Biochemical Markers
The allergic biochemical markers immunoglobulin E (IgE) antibody and the chemical histamine released in response to allergens were quantified with the enzyme-linked immunosorbent assay (ELISA) kit, Abcam. The reagents were brought to room temperature, and the standard solutions were prepared before the initiation of the experiment. The ELISA plate was set up with standards, samples, and controls in designated wells. After an incubation period to allow antibody binding, the plate was washed to remove unbound substances. Detection antibodies were added, followed by another incubation. Substrate solution was then added to induce a colorimetric reaction, and the reaction was stopped. The absorbance of each well was measured, and IgE and histamine concentrations in the samples were calculated by comparing absorbance values to a standard curve.
Quantification of Inflammatory Mediators in NALF
IgE antibody and the allergic lipid mediators, prostaglandin D2 (PGD2) and leukotriene C4 (LTC4) were measured in the NALF of the experimental mice. IgE antibody was detected with the ELISA kit, Abcam. PGD2 and LTC4 were measured using the ELISA kits procured from Cayman Chemicals. The final absorbance of standards and samples was measured in the microplate reader. A standard curve was drawn with the standard optical density (OD) values, and the concentrations of PGD2 and LTC4 were calculated with the standard curve.
Measurement of NALF Pro-inflammatory Cytokines
Eosinophil cationic protein (ECP), interleukins IL-4, IL-5, IL-6, IL-33, and tumor necrosis factor (TNF)-alpha were determined in the NALF of the experimental mice. The pro-inflammatory cytokine levels were measured with an ELISA kit, MyBioSource. The absorbance of both standards and samples was measured using a microplate reader. A standard curve was generated using the OD values of the standards, and the concentrations of ECP and pro-inflammatory cytokines in the samples were determined by extrapolating from this curve.
Evaluation of Eosinophil Count in the Nasal Tissues
The euthanized mice heads were fixed with buffered formalin and subjected to decalcification with 0.1 M ethylenediaminetetraacetic acid (EDTA) buffer. The decalcified tissue was embedded in paraffin wax and sectioned into 4-µm slices using a microtome. The tissue sections were mounted onto slides and stained with hematoxylin and eosin (H&E). The eosinophils’ characteristic morphology, including bilobed nuclei and eosinophilic granules in the cytoplasm, were counted under a light microscope. Multiple fields of view were examined, and eosinophils were counted to obtain an average count per unit area of tissue.
NF-κB DNA-binding Activity
The binding of NF-κB DNA was quantified with the kit procured at Abcam, following the protocol provided in the kit. Initially, nuclear extracts (10 µL) were combined with Complete Transcription Factor Binding Assay Buffer (CTFB) (90 µL) and incubated for 12 hours at 4°C. Subsequently, the plate underwent five washes with 200 µL of 1X wash buffer. Then, 100 µL of NF-κB (p65) antibody was added to each well and incubated for 1 hour in a shaking incubator at room temperature, followed by additional washing steps. Following this, a secondary antibody (100 µL) at a dilution of 1:100 was applied and incubated for 1 hour with shaking, followed by further washing. Finally, the plate was exposed to a developing solution (100 µL) at room temperature (RT) for 30 minutes in the dark with shaking. The color transition to blue prompted the addition of a stop solution, resulting in a yellow color change. OD was measured at 450 nm.
Nasal Tissue Histopathological Analysis
The euthanized mice heads were immediately fixed in 10% neutral buffered formaldehyde for a period of 7 days and then transferred to 0.1 M EDTA buffer for 14 days to decalcify the tissue. The decalcified tissue was embedded in paraffin and subjected to sectioning of 4-µm thickness. The tissue sections were then stained with periodic acid-Schiff and Giemsa stain to assess ciliary loss, eosinophil infiltration, goblet cell count, and vascular congestion. The slides were scanned with slide digital scanners Pannoramic DESK and analyzed with Pannoramic software.
Statistical Analysis
The data were evaluated utilizing GraphPad Prism software v5.0, USA, and presented as mean ± SEMs across multiple experiments. Student’s t-test and analysis of variance (ANOVA) with Dunnett’s test for comparison were done. p < 0.05 at the 95% confidence level was considered statistically significant. *Control versus OVA-sensitized AR-induced untreated group, #OVA-sensitized AR-induced untreated group versus 10 and 20 mg catalpol-treated and dexamethasone-treated group.
Results
Protective Effect of Iridoid Glucoside Catalpol Against Nasal Symptoms in OVA-triggered AR Mice Model
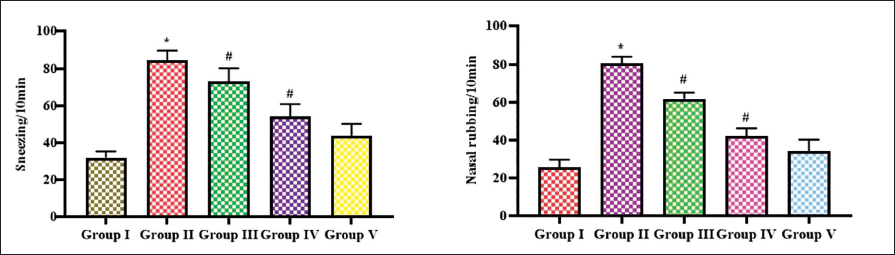
The primary manifestations of allergic induction are sneezing and nasal rubbing; hence, we analyzed and counted the nasal symptoms in the experimental mice, and the results were tabulated in Figure 1. The OVA-sensitized untreated mice showed significantly increased sneezing and nasal rubbing count than the other group mice. The control mice performed 34 sneezing counts and 22 counts of nasal rubbing for a period of 10 minutes, whereas the OVA-sensitized untreated mice showed 85 and 76 counts of sneezing and nasal rubbing, respectively. The catalpol-treated mice showed decreased nasal symptoms such as 67 and 59 counts of sneezing and nasal rubbing, respectively, in 10 mg catalpol-treated mice, and it is further reduced to 49 and 37 counts of sneezing and nasal rubbing, respectively, in 20 mg catalpol-treated mice. 2.5 mg dexamethasone-treated mice showed significantly decreased nasal symptoms compared to catalpol-treated mice.
Impact of Iridoid Glucoside Catalpol Against Allergic Mediators in OVA-induced AR Mice
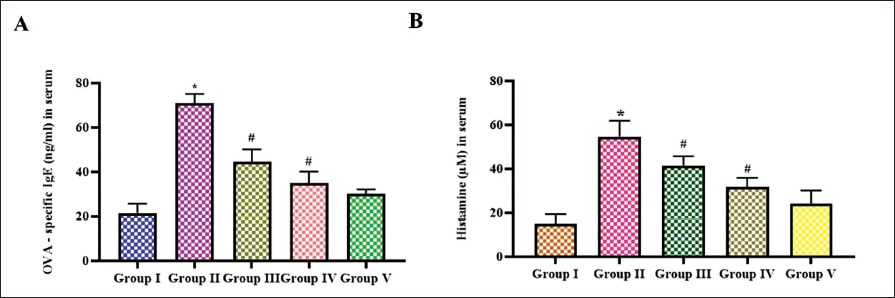
Figure 2 illustrates catalpol potency against allergic mediators, such as IgE antibody (A) and histamine (B), in the experimental mice. IgE antibodies significantly increased to 64 ± 0.05 ng/mL in the OVA-triggered AR-induced untreated mice compared to other groups, such as control mice with 15 ± 0.02 ng/mL, 10 and 20 mg catalpol-treated mice with 41 ± 0.04 and 38 ± 0.07 ng/mL, respectively, and in 2.5 mg dexamethasone-treated mice, the IgE levels were 31 ± 0.07 ng/mL. OVA-sensitized untreated group exhibited histamine levels than the control mice. 54 ± 0.04 µM of histamine is quantified in the serum of OVA-sensitized untreated group whereas it is decreased to 37 ± 0.06 and 29 ± 0.09 µM, respectively, in 10 and 20 mg catalpol-treated mice. The histamine levels were 9 ± 0.009 µM in the control and 18 ± 0.04 µM in the standard drug dexamethasone-treated mice.
Anti-allergic Effect of Iridoid Glucoside Catalpol in OVA-induced AR Mice
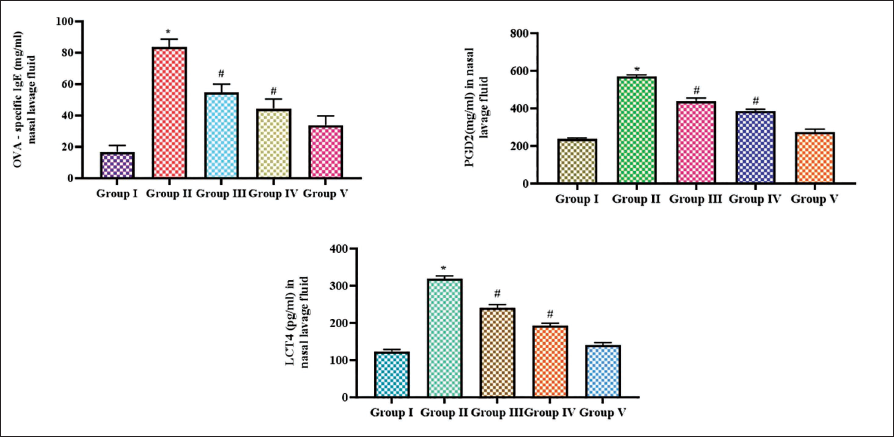
Figure 3 depicts the levels of IgE antibody, and the allergic lipid mediators PGD2 and LTC4 were measured in the NALF of the experimental mice. In NALF also, the levels of IgE antibody were significantly increased to 78 ± 0.02 ng/mL in the OVA-triggered AR-untreated mice compared to other groups, such as control mice which showed 5 ± 0.006 ng/mL, 10 and 20 mg catalpol-treated mice showed 61 ± 0.07 and 39 ± 0.05 ng/mL, respectively, and in 2.5 mg dexamethasone-treated mice, the IgE levels were 29 ± 0.04 ng/mL. Both PGD2 and LTC4 were significantly increased in the OVA-sensitized untreated group to 580 ± 0.4 pg/mL and 256 ± 0.1 pg/mL, respectively, compared to the control mice, which showed 217 ± 1.8 pg/mL of PGD2 and 98 ± 0.08 pg/mL of LTC4. Catalpol dose-dependent decreases the PGD2 and LTC4 levels. 10 mg catalpol-treated mice showed 310 ± 0.3 pg/mL of PGD2 and 182 ± 0.2 pg/mL of LTC4, which was further decreased to 280 ± 0.3 pg/mL of PGD2 and 155 ± 0.09 pg/mL of LTC4 in 20 mg catalpol-treated mice. Dexamethasone-treated mice showed 275 ± 0.2 pg/mL of PGD2 and 138 ± 0.07 pg/mL of LTC4.
Inhibitory Effect of Iridoid Glucoside Catalpol Against ECP and Cytokines in OVA-induced AR Mice
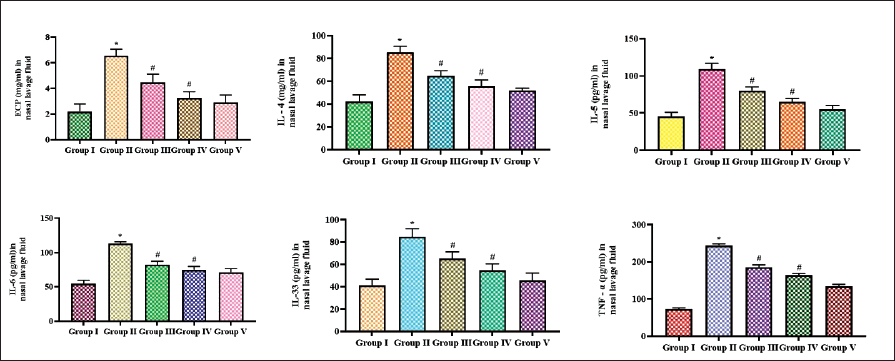
Eosinophilic cationic protein and cytokines were measured in the NALF of the experimental mice, and the results are represented in Figure 4. Catalpol treatment significantly decreased the ECP in a dose-dependent manner compared to the OVA-sensitized untreated group mice. The pro-inflammatory interleukins IL-4, IL-5, IL-6, and IL-33 were increased in the OVA-sensitized untreated group compared to the other group. Marked reduction in the pro-inflammatory interleukin levels was decreased in the catalpol- and dexamethasone-treated mice. IL-6 was comparatively increased in the OVA-triggered AR untreated mice group compared to other interleukins. TNF-alpha was significantly declined in the catalpol and dexamethasone treatment than the OVA-sensitized untreated group.
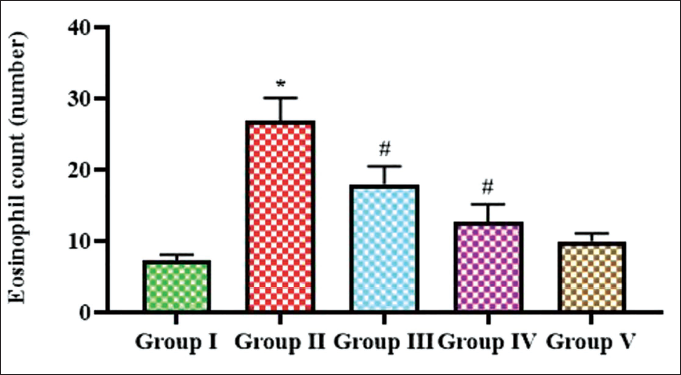
Antagonistic Effect of Iridoid Glucoside Catalpol Against Eosinophilic Infiltration in OVA-induced AR Mice
Eosinophil count in nasal mucosa serves as a valuable marker for assessing allergic inflammation and the severity of AR; hence, we quantified the eosinophil count, and the results are depicted in Figure 5. Increase in eosinophil count was observed in the OVA-sensitized untreated group, which showed about 26 eosinophils, whereas it reduced to 17 and 8 in 10 and 20 mg catalpol-treated mice. Control mice exhibited four counts of eosinophils and the standard drug exhibited seven eosinophil counts.
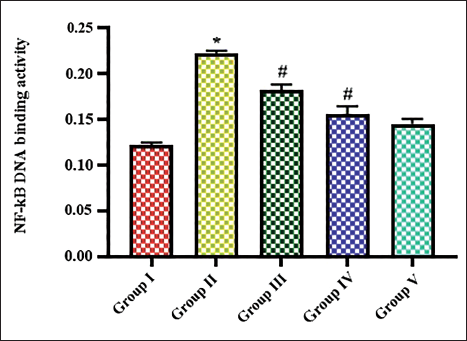
Suppressing Effect of Iridoid Glucoside Catalpol Against NF-κB DNA-binding Activity in OVA-triggered AR-induced Mice Model
To assess the role of NF-κB in mediating allergic response in OVA-sensitized AR-induced rats, NF-κB-binding activity was assessed and the results are presented in Figure 6. The control mice showed NF-κB DNA-binding activity of 0.14 increased to 0.28 in OVA-sensitized untreated group. 10 and 20 mg catalpol-treated mice showed reduced levels of 0.23 and 0.18 NF-κB DNA-binding activity, respectively, to OVA-sensitized untreated group. The dexamethasone-treated group showed 0.17 NF-κB DNA-binding activity.
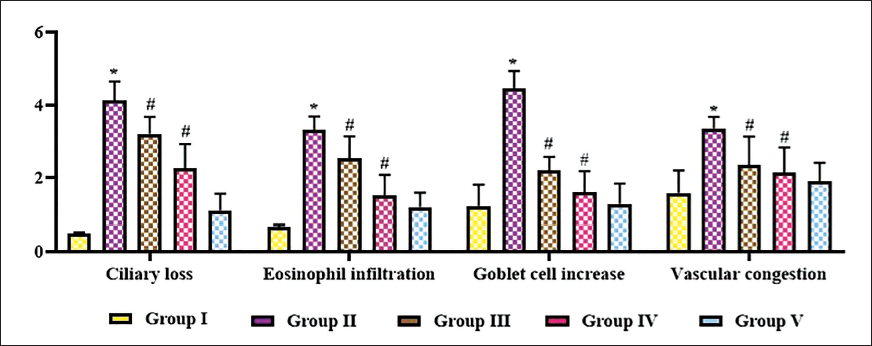
Ameliorative Effect of Iridoid Glucoside Catalpol in OVA-triggered AR Mice Model
The ameliorative effect of catalpol in OVA-sensitized AR-induced mice was analyzed with histopathological analysis of nasal mucosa. Figure 7 illustrates the ciliary loss, eosinophil infiltration, goblet cell count, and vascular congestion in the experimental animals. The cilia were significantly decreased, and increased eosinophil infiltration was observed in the epithelium of OVA-sensitized untreated group. An increased number of goblet cells and vascular congestion was also observed in the OVA-sensitized untreated group. Catalpol treatment decreased the ciliary loss and goblet cells in the epithelium, and prevented eosinophil infiltration and vascular congestion in the nasal mucosa.
Ameliorative Effect of Iridoid Glucoside Catalpol in Ovalbumin (OVA)-induced Allergic Rhinitis (AR) Mice. Histopathological Analysis of Nasal Mucosa was Performed in the Experimental Animals to Evaluate the Ciliary Loss, Eosinophil Infiltration, Goblet Cell Count, and Vascular Congestion in the Nasal Mucosa.
Discussion
AR is a prevalent disorder that affects 400 million population globally and affects all age groups, and the prevalence of AR is rapidly escalating the past few decades (Savouré et al., 2022). AR often coexists with other severe morbidities such as asthma, sinusitis, and conjunctivitis. It severely affects the quality of life of individuals, which has a greater impact on the global economy (Nur et al., 2022). The first line of therapy prescribed to AR patients is intranasal corticosteroids. These drugs are proven to be effective in patients with varied grades of severity (May & Dolen, 2017). The other drugs prescribed are antihistamines and decongestants, which help to alleviate nasal stuffiness (Simons & Simons, 2011). Despite their efficacy, some individuals may experience adverse effects, such as drowsiness, dry mouth, headache, or local irritation. Balancing the benefits with potential side effects is crucial in tailoring the treatment approach to each patient’s needs (Abdullah et al., 2022; Small et al., 2018).
Traditional Chinese medicine (TCM) offers a holistic approach to treating AR, focusing on rebalancing the body’s energy flow rather than merely alleviating symptoms. Glycyrrhizin, tanshinone IIA, saikosaponin A, and Baicalin are some of the phytocompounds prescribed in TCM to clear nasal congestion, reduce inflammation, and strengthen the body’s immune response (Qin et al., 2024; Yu et al., 2020b). Catalpol, an iridoid glycoside that crosses the blood–brain barrier (BBB), is rapidly absorbed, possesses high absolute bioavailability, and has a relatively prolonged half-life (Fu et al., 2023). Extensive research across various disease conditions has unveiled its diverse biological effects, including anticancer (Wang & Zhan-Sheng, 2018), neuroprotective, anti-inflammatory, antioxidant, hepatoprotective, and antidiabetic properties (Bhattamisra et al., 2019; Xu et al., 2020, 2015), as well as cardiovascular protection (He et al., 2021).
Exposure to allergens triggers the B-cell synthesis in the blood, which, in turn, produces allergen-specific IgE antibodies. It also activates the nasal mucosal mast cells, thereby triggering histamine secretion (Bousquet et al., 2020). On binding to high-affinity, IgE Fc receptors located on mast cells or to the allergens sensitize the IgE of targeted cells (Gould & Soutton, 2008). This interaction triggers histamine, kininogenases, leukotrienes (LTs), and cytokines, causing contraction of smooth muscles and excessive glandular secretion (Watts et al., 2019). These processes collectively result in symptoms such as sneezing, rhinorrhea, and nasal itching congestion. (Li et al., 2023; Rondón et al., 2009). In our study, catalpol treatment reduced both serum and NALF IgE levels. This may be the reason for reduced histamine in the sensitized AR mice model. IgE antibodies and histamine inhibition were confirmed with the decrease in nasal severity symptoms exhibited by the catalpol-treated AR mice model.
Activated mast cells release lipid mediators PGD2 and cysteinyl leukotrienes (cysLTs), which are involved in the smooth contraction of the respiratory tract, vasodilation, and glandular secretion (Peinhaupt et al., 2017). Animal models exposed to exogenous PGD2 agonists had aggravated the eosinophilia in blood, lungs, nostrils, conjunctiva, and skin (Shiraishi et al., 2014). Inhibiting PGD2 activity with drugs ameliorated rhinitis, asthma, conjunctivitis, and atopic dermatitis (Spik et al., 2005; Stebbins et al., 2012; Xue et al., 2015). PGD2 is a potent eosinophil attractant, and it is also involved in eosinophil lipid body-driven LTC4 synthesis (Hirai et al., 2001). Allergic inflammation triggers intracellular signaling in eosinophils through mediators such as platelet-activating factor, IL-5, and CC chemokines, which directly interact with their eosinophil surface receptors. This cascade ultimately leads to the lipid bodies generation and primes the eosinophils for LTC4 generation (Mesquita-Santos et al., 2006; Lee et al., 2020). Catalpol treatment in OVA-triggered AR-induced mice decreased the levels of eosinophils infiltration in nasal mucosa. This may be due to the inhibitory action of PGD2, which, in turn, decreased the synthesis of LTC4 and infiltration of eosinophils in the nasal mucosa.
Amplified expression of IgE, along with inflammatory factors such as TNF-α, IL-4, and IL-6, plays a significant role in the pathogenesis of AR. The level of expression closely aligns with the seriousness of the ailment (Shin et al., 2019). In our study, OVA-sensitized untreated AR mice showed a significant increase in IL-4, IL-6, and TNF-α, which may be due to the increase in IgE (Lee et al., 2016). Catalpol effectively inhibited the IgE antibody synthesis, thereby preventing the generation of pro-inflammatory cytokines IL-4, IL-6, and TNF-α. It has previously been reported that IL-5 and IL-13 trigger allergens-induced eosinophilic infiltration (Chung, 2015). Catalpol exposure reduced IL-5 and ECP, thereby attenuating the eosinophilic infiltration, which is crucial to the event of allergic response.
IL-33, also called alarmin, is the cytokine released by the first-line cells of the immune system in response to external stimuli (Cayrol et al., 2018; Martin & Martin, 2016). The whole-genome analysis reported that IL-33 gene loci play a crucial role in numerous inflammatory diseases such as AR and asthma. IL-33 stimulates the NF-κB p65 subunit, initiating an inflammatory response in endothelial cells (Choi et al., 2012; Ding et al., 2018). Targeting IL-33 with an antagonist may effectively the NF-κB-mediated inflammation in allergic conditions. Catalpol treatment significantly attenuated the IL-33 levels in OVA-sensitized AR mice, proving its antiallergic potency.
NF-κB plays a pivotal part in the pathology of the airway by orchestrating cytokines, chemokines, and cell adhesion molecules (CAMs) (Schuliga, 2015). Prolonged NF-κB activation was reported in the airway epithelium of asthma patients (Hayden et al., 2006). These inflammatory mediators impact both the type and quantity of inflammatory cells that penetrate the airway tissue in chronic obstructive airway diseases (Kato & Schleimer, 2007). Attenuating NF-κB activation in mouse models evidenced a decrease in Th2 cytokines, IgE antibodies, and interleukins IL-4, IL-5, and IL-13, which are key players in asthma. These cytokines stimulate hyperplasia of goblet cells and cause airway constriction and eosinophilia (Ather et al., 2011). Catalpol, a potent anti-inflammatory phytochemical, effectively suppressed the NF-κB DNA-binding activity, thereby preventing NF-κB-mediated allergic response in OVA-sensitized AR mice. Our histopathological analysis of nasal mucosa exhibited decreased goblet cells, ciliary loss, eosinophil infiltration, and vascular congestion in catalpol-treated AR mice. This confirms that catalpol has effectively inhibited NF-κB-mediated inflammation, thereby preventing bronchostriction and mucous production in OVA-sensitized AR mice.
Conclusion
AR is a prevalent inflammatory disease that impacts a significant portion of the global population. Rapid urbanization has made AR an unpreventable disease. It has a severe impact on the quality of the individual, which directly affects the global economy. It often leads to various other comorbidities such as asthma and COPD, making it more complicated to be treated. Hence, treating AR at the initial stage with cost-effective phytochemicals, which renders null side effects, is needed today. The present investigation assessed the potency of the iridoid glucoside compound catalpol potency in ameliorating AR in mice. Catalpol effectively attenuated the NF-κB-mediated inflammatory response in the OVA-sensitized AR mice model. It significantly decreased the nasal symptoms, ciliary loss, vascular congestion, eosinophil infiltration, and goblet cell number in the nasal tissue of AR mice. Our finding had proven catalpol effectively inhibits NF-κB-mediated inflammatory response and protects OVA-sensitized AR mice from AR. However, additional studies are still recommended in the future to fully comprehend the therapeutic roles of catalpol against rhinitis.
Footnotes
Abbreviations
AR: Allergic rhinitis; TLRs: Toll-like receptors; NALF: Nasal lavage fluid; IgE: Immunoglobulin E; PGD2: Prostaglandin D2; LTC4: Leukotriene C4; ECP: Eosinophil cationic protein; OD: Optical density.
Acknowledgments
This work was supported by the Department of Otolaryngology, Head and Neck Surgery, Xi’an International Medical Center Hospital, Xi’an, China.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The ethical committee approved procedures were executed on the mice (No. 2023/45).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
