Abstract
Previous studies revealed that foals inoculated with virulent Rhodococcus equi had significantly higher pulmonary levels of interleukin-1β, interleukin-12 p40, interferon-γ, and tumor necrosis factor-α mRNA compared to foals inoculated with an avirulent plasmid-cured derivative. The purpose of this study was to determine if the increases in cytokine expression were associated with increased pulmonary activation of nuclear factor-kappa;B (NF-kappa;B). Electrophoretic mobility shift assays were performed on pulmonary nuclear protein extracted from foals treated with phosphate-buffered saline, or inoculated with either virulent or avirulent R. equi. NF-kappa;B activation was increased in the nuclear extracts from foals inoculated with virulent R. equi at 14 days after inoculation when increased cytokine expression was also observed. Southwestern histochemistry revealed activated NF-kappa;B in multinucleated giant cells that often contained bacteria. These results indicate that the cytokine response to R. equi is at least partially mediated by NF-kappa;B activation.
Rhodococcus equi, a facultative intracellular pathogen of macrophages, is one of the most important causes of respiratory disease in foals between the ages of 1 and 5 months. R. equi is widespread in the environment, with isolates from pneumonic foals typically containing an 85- to 90-kilobase plasmid encoding a family of seven closely related virulence-associated proteins. 8,16,17 Plasmid-cured derivatives of virulent R. equi strains lose the ability to replicate and survive in macrophages and fail to induce pneumonia in foals. 8,9 The pulmonary cytokine responses associated with development of the typical granulomatous pneumonia induced in foals by R. equi have been characterized using wild-type and avirulent, plasmid-cured derivatives of R. equi. 9,10 Infection with virulent versus avirulent R. equi induces increased interleukin-1β (IL-1β), tumor necrosis factor-α (TNF-α), and IL-12 p40 mRNA expression in lung tissue 3 and 14 days after inoculation. Increased interferon-γ (IFN-γ) mRNA expression was only detected at 14 days after inoculation. 10 All of these inflammatory mediators are at least partially regulated by the transcription factor nuclear factor-κB (NF-κB). 2
NF-κB is a critical regulator of cytokine-mediated inflammation. It is sequestered in the cytoplasm of most cells and is activated by a series of events involving signaled phosphorylation, ubiquitination, and proteolysis of its inhibitor, IκB. 2,4 Degradation of IκB exposes a nuclear localization signal on NF-κB, which allows it to move into the nucleus, bind DNA, and induce gene transcription. 2 NF-κB regulates the transcription of many genes encoding inflammatory mediators, and induction of many NF-κB-regulated mediators is an important component of the both the innate and acquired immune response. Activation of NF-κB can be directly stimulated by a wide range of bacterial, protozoal, and viral pathogens, including Bordetella pertussis, 1 Borrelia burgdorferi, 5 enteroinvasive bacteria, 6 Trypanosoma cruzi, 11 Toxoplasma gondii, 3 rhinovirus, 15 and respiratory syncycial virus. 7 Although NF-κB activation may be critical to resistance or clearance of infection, NF-κB activation also contributes to tissue damage, and inappropriate activation of NF-κB has been implicated in the pathogenesis of several respiratory diseases. 2 The purpose of this study was to determine if the increases in pulmonary expression of inflammatory cytokines were associated with increased pulmonary activation of NF-κB.
Eighteen healthy, 3-week-old mixed-breed pony foals were used in this study, which was performed in conjunction with other studies on the role of the large plasmid in the virulence of, 8 and cytokine induction by, R. equi. 10 Foals were randomly assigned to treatment groups, and those inoculated with R. equi received 2.5 × 109 colony-forming units suspended in phosphate-buffered saline (PBS) intrabronchially. Six foals were inoculated with the virulent plasmid-containing strain (103+), six were inoculated with the avirulent, plasmid-cured derivative (103−), and six foals received only PBS. Half of the foals from each group were euthanatized 3 days after inoculation and the other half were sacrificed at 14 days after inoculation. All organs were examined grossly and representative tissue samples were cultured, whereas others were fixed in formalin and examined histologically. Samples of lung tissue were frozen in liquid nitrogen and stored at −80 C until processed. Pulmonary cytokine mRNA expression was previously determined on the same samples used in the electrophoretic mobility shift assays (EMSAs).
NF-κB activation was assessed by comparing the level of DNA binding in pulmonary nuclear extracts from the foals by means of EMSAs. Nuclear protein was extracted from lung tissue and EMSAs were performed as previously described.
14
The fractionated nuclear extract (20 µg total protein) was incubated (20 minutes, 25 C) with double-stranded oligonucleotides, encoding the target sequence NF-κB, (AGTTGAGGGACTTTCCCAGGC), end labeled with [gamma-32P] adenosine triphosphate and T4 polynucleotide kinase (0.1–0.5 ng; 20,000–100,000 counts per minute). Protein–DNA binding complexes were resolved on a 6% polyacrylamide gel with 0.1 × TAE (Tris–acetic acid–ethylenediaminetetraacetic acid [EDTA]) running buffer. Specificity was determined by addition of excess unlabeled double-stranded oligonucleotides, or by use of a probe with a mutated NF-κB binding site (AGTTGAG
Southwestern in situ histochemistry (SWISH) was performed to identify cells with activated NF-κB in formalin-fixed, paraffin-embedded tissue sections. The protocol was modified from that described previously. 12 In brief, 4-µm sections of lung tissue were deparaffined, rehydrated, incubated in 2 mM levamisole for 15 minutes at room temperature, post fixed in 0.2% paraformaldehyde for 30 minutes at room temperature, and pretreated with pepsin A (433 U/mg, Sigma, St. Louis, MO) in 1 N HCl for 30 minutes at 37 C. After two washes in HEPES-bovine serum albumin (BSA) buffer, the sections were incubated in 0.4 mg/ml DNAse I in HEPES-BSA for 30 minutes at 30 C, and washed twice in HEPES-EDTA. Prehybridization in probe buffer for 30 minutes was followed by incubation with a digoxigenin-labeled double-stranded DNA binding site for NF-κB at 37 C overnight in a humidified box. Detection was performed by incubating the sections with anti-digoxigenin antibody conjugated with alkaline phosphatase (1:250 in blocking solution; Roche molecular Biochemicals, Indianapolis, IN) overnight at 4 C, followed by incubation in detecting solution in the dark at 37 C for 30 minutes as previously described. 12 Sections were then dehydrated, counterstained with eosin, and mounted in Permount (Fisher Scientific, Pittsburgh, PA). Specificity of the binding was determined by comparing the staining to that present in serial sections incubated with a digoxigenin-labeled double-stranded DNA probe containing a mutated NF-κB binding site. Comparisons were also made between these sections and lung sections stained with hematoxylin and eosin, and Gram's stain (Brown and Brenn). 13 Cell types were identified based upon histologic appearance.
The complete clinicopathologic description and cytokine mRNA expression of the foals in this study has been reported elsewhere. 8,10 As indicated by the increased densities of the shifted bands, foals infected with virulent (103+) R. equi had increased pulmonary activation of NF-κB at 14 days after inoculation compared to foals inoculated with PBS or avirulent (103−) R. equi (Fig. 1, 2). At 3 days after inoculation, NF-κB band densities were slightly, but not significantly increased in the nuclear protein extracts from foals inoculated with virulent (103+) R. equi (Fig. 1). SWISH for NF-κB, with the same DNA probe utilized in the EMSA, was performed on lung sections taken from foals 14 days after inoculation (Fig. 3). Positive nuclear staining for activated NF-κB was most pronounced at the edges of pyogranulomatous lesions. In these areas, roughly 40% of epithelial cells, 30% of macrophages, and 60% of giant cells (Fig. 3b) were stained. Many of the multinucleated giant cells also contained Gram's stain–positive bacteria compatible with R. equi (Fig. 3d).
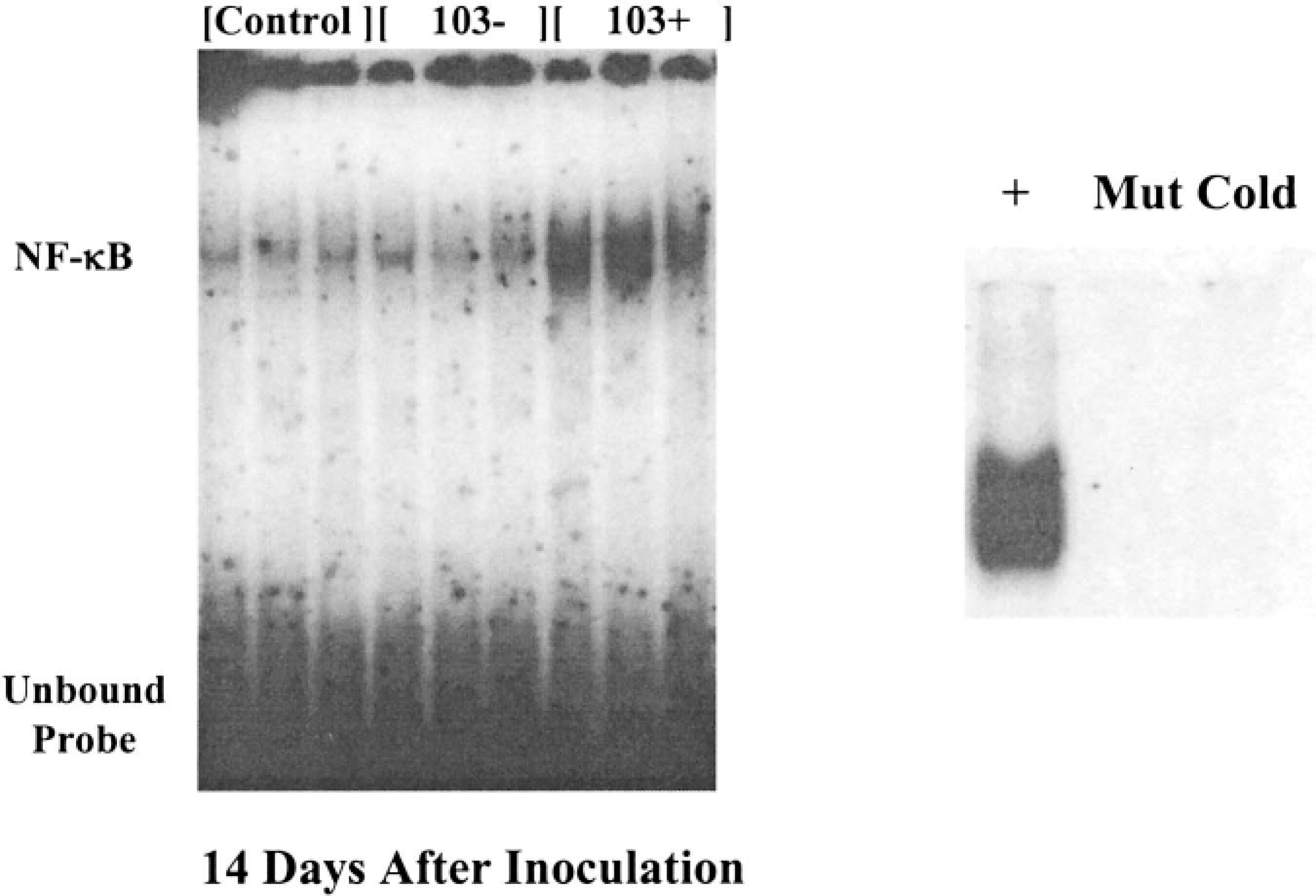
Representative electrophoretic mobility shift assays (EMSAs) for nuclear factor-κB (NF-κB) performed on pulmonary nuclear protein extracted from foals. Each lane was loaded with 20 µg of pulmonary nuclear protein that had been extracted from individual foals and incubated with radiolabeled double-stranded oligonucleotides encoding the NF-κB binding sequence. Increased activation of NF-κB, as indicated by the increased DNA binding, was present in the samples from foals 14 days after inoculation with virulent (103+) Rhodococcus equi compared to samples from foals inoculated with saline (control) or avirulent (103−) R. equi. Positive and negative controls for the NF-κB EMSAs are shown in the small panel. The positive control (+) sample was pulmonary nuclear protein extracted from a rat inoculated with Sendai virus, which is known to directly activate NF-κB. Dense shifted bands, similar to those observed in the foals inoculated with the virulent (103+) form of R. equi, were present. DNA binding was not observed in samples that were either incubated with radiolabeled DNA coding for a mutated NF-κB binding site (Mut), or incubated with unlabeled DNA coding for the NF-κB binding site before incubation with the labeled DNA (cold).
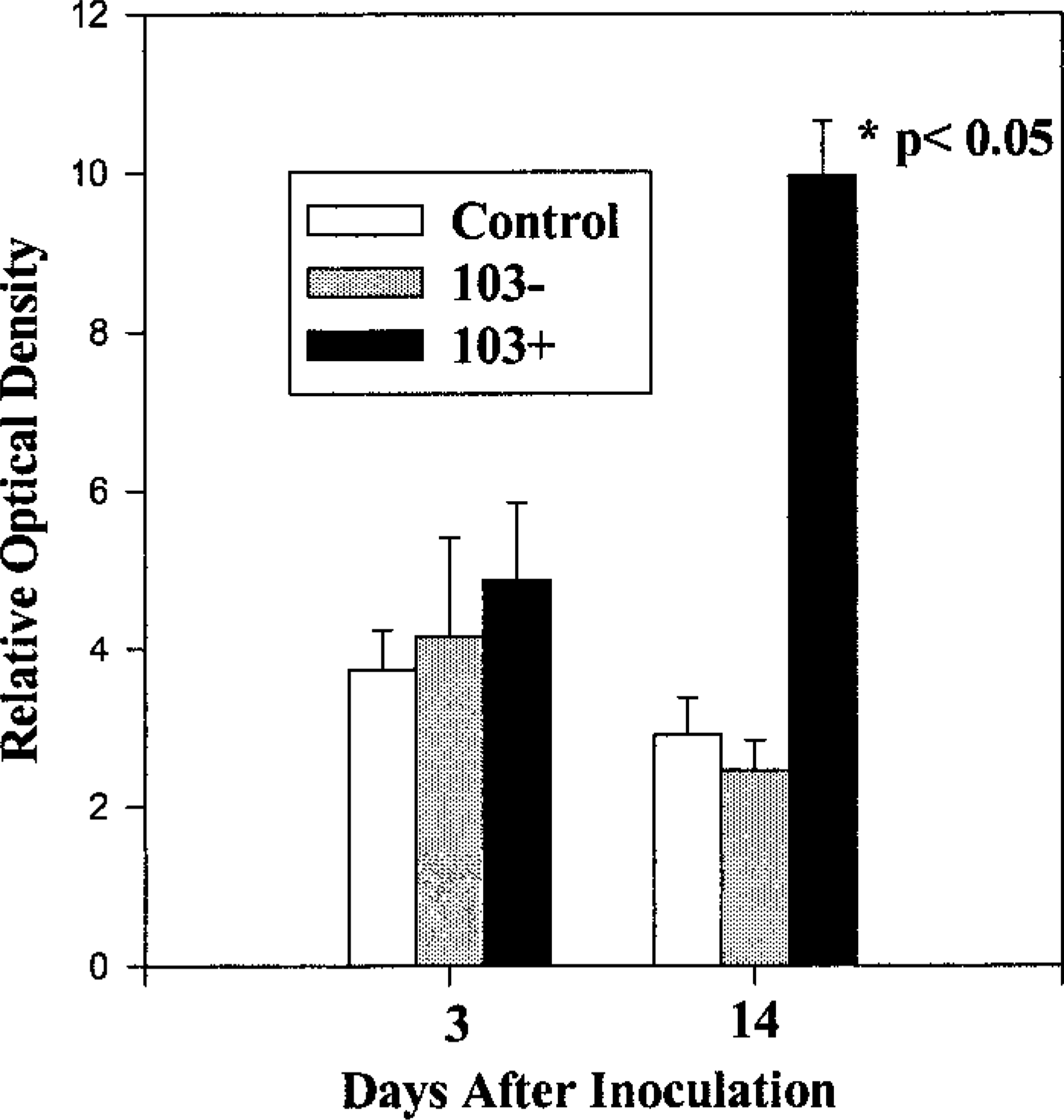
The relative optical densities of the shifted bands were compared between samples taken from all three groups at both 3 and 14 days after inoculation. Compared to those of all the other groups, the band densities in the samples taken from foals 14 days after they were inoculated with the virulent (103+) form of Rhodococcus equi were significantly increased. Nuclear protein from three animals per group was analyzed. Multiple gels were run to confirm the results and band density comparisons were only made on samples run on the same gel.
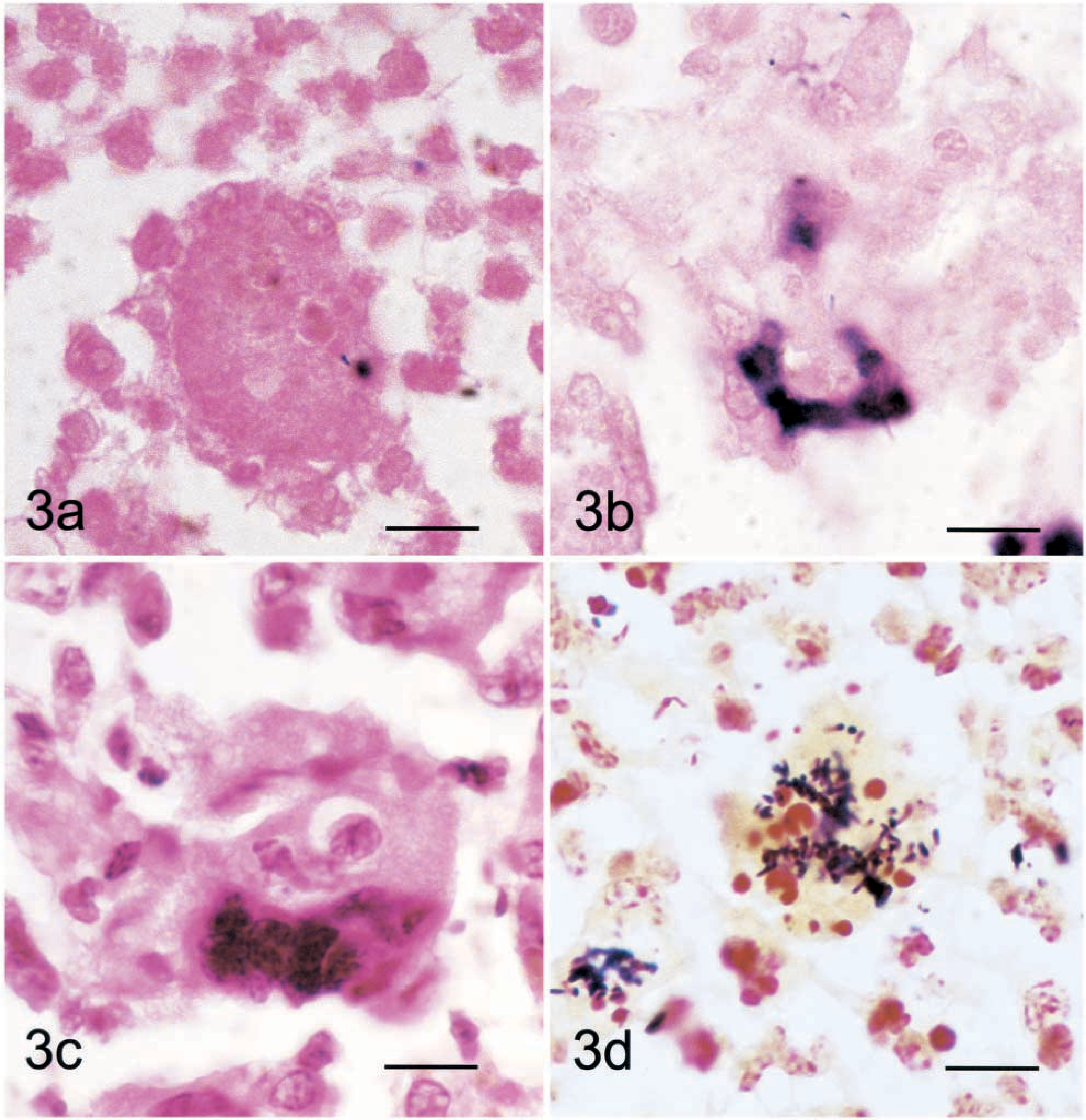
Lung; foal. Lung tissue sections from a foal inoculated with the virulent (103+) form of Rhodococcus equi 14 days before sampling. Nuclear staining for nuclear factor-κB (NF-κB) was detected in multinucleated giant cells by southwestern in situ histochemistry. Fig. 3a Sections were incubated with digoxigenin-labeled, double-stranded DNA probes coding for a mutated (AGTTGAG
One advantage of SWISH over immunohistochemistry is that SWISH detects all forms of NF-κB binding to the -κB target DNA sequence as opposed to only those containing the NF-κB subunit targeted by an antibody. A second advantage is that SWISH detects NF-κB that is able to bind DNA, an indication that the NF-κB is activated, rather than simply detecting the presence of the subunit proteins. However, some of the active NF-κB protein most likely was denatured in tissue processing, and, therefore, the technique probably underestimated the number of cells with activated NF-κB.
Previous studies demonstrated increased pulmonary expression of the proinflammatory cytokines IL-1β, TNF-α, IL-12 p40, and IFN-γ in foals with R. equi-induced pneumonia. 9 NF-κB is known to up-regulate gene expression of all of these cytokines in a variety of species. 2,4 Increased pulmonary activation of NF-κB was associated with increased cytokine expression at 14 days after inoculation. Although a slight trend toward increased activation was observed, NF-κB activation was not significantly increased in foals inoculated with either the avirulent or the virulent forms of R. equi at 3 days after inoculation, even though increases in cytokine mRNA were observed at this time point. 10 Day 3 is early in the infection and increased NF-κB activation may not have been detected if the activation was occurring in a localized area of the lung and the assays of whole lung tissue were not sensitive enough to detect this increase. Alternatively, differentially regulated acute and chronic activation of NF-κB has been described in models of endotoxemia, 18 and the 3-day time point may be within a transition phase between the acute activation of NF-κB, stimulated by the initial bacterial infection, and the chronic phase of NF-κB activation induced by the persistence of infection.
The observation that NF-κB was activated in multinucleated giant cells, many of which commonly contained Gram's stain–positive bacteria, indicates that R. equi could be directly activating NF-κB. This conclusion is consistent with the results of a previous study that documented that R. equi could directly up-regulate cytokine expression in murine macrophages. 9 NF-κB activation is also enhanced by many inflammatory mediators, including TNF-α and IL-1β, 2 that are present in the chronic inflammatory lesions typical of R. equi pneumonia.
Activation of NF-κB has been demonstrated to be critical to the innate and adaptive immune response to a variety of infectious agents; however, its role can be very complex. For example, both cell-specific activation of NF-κB and the presence of specific NF-κB subunits have been shown to be critical to resistance to parasitic infections. 3,11 Given the evidence that abnormal NF-κB signaling results in an impaired ability to respond to a variety of infectious agents, foals susceptible to the development of severe R. equi pneumonia possibly could have defects in NF-κB signaling. Further studies are needed to address this possibility.
The results of this study indicate that one potential mechanism by which virulent R. equi modulates pulmonary expression of inflammatory cytokines is through increased activation of NF-κB, and that some of this increase may be a direct effect of bacterial infection.
Footnotes
Acknowledgements
This study was supported in part by a grant from the State of Florida's Pari-mutuel Wagering Trust Fund.
