Abstract
Spontaneous tumors in dogs share several environmental, epidemiologic, biologic, clinical and molecular features with a wide variety of human cancers, making this companion animal an attractive model. Nuclear factor kappa B (NF-kB) transcription factor overactivation is common in several human cancers, and there is evidence that similar signaling aberrations also occur in canine cancers including lymphoma, leukemia, hemangiosarcoma, mammary cancer, melanoma, glioma, and prostate cancer. This review provides an overview of NF-kB signaling biology, both in health and in cancer development. It also summarizes available evidence of aberrant NF-kB signaling in canine cancer, and reviews antineoplastic compounds that have been shown to inhibit NF-kB activity used in various types of canine cancers. Available data suggest that dogs may be an excellent model for human cancers that have overactivation of NF-kB.
Cancer affects approximately 4 million dogs per year and is the most common cause of death in this species. 24 Dogs have recently gained attention as a viable animal model of human cancer for several reasons. Relative to rodent models, dogs are genetically diverse and share many environmental exposures with humans. Unlike laboratory-induced cancer in rodents, dogs develop spontaneous neoplasms over a long period of time, which more closely mirrors cancer development in humans; and for many tumor types, canine cancers are remarkably similar to their human counterparts in microscopic appearance and biologic behavior.23,24,72,87
Investigation of molecular pathways that are dysregulated in canine cancers can help researchers decide whether a canine cancer type is a suitable model for the study of human cancer. Nuclear factor kappa-light-chain-enhancer of B cells (NF-kB) is a transcription factor that is constitutively active in most human hematopoietic and solid tumor cell lines, and has also been demonstrated in diverse human tumor types. Examples include human multiple myeloma, acute myeloid leukemia, acute lymphoblastic leukemia, chronic myelogenous leukemia, and prostate and breast cancer.1,18,82,40
The aim of this article is to provide an overview of what is currently known about NF-kB signaling in human health and cancer, and to summarize information regarding NF-kB and NF-kB overactivation in canine cancer. In addition, a review of the utility of NF-kB inhibition in the treatment of canine cancers is provided.
NF-kB Signaling in Health
NF-kB is a family of structurally related, inducible transcription factors that was first described in 1986 and is highly conserved in animals.48,74,79 Inactive NF-kB is sequestered in the cell cytoplasm until it is activated, when it translocates to the nucleus to bind DNA and initiate transcription of over 400 target genes that are important in normal immunity, growth, apoptosis, and inflammation.18,39,74 Target genes include inflammatory cytokines, adhesion molecules, enzymes related to inflammation (such as cyclooxygenase-2), telomerase, antiapoptotic proteins, and cell cycle-regulatory genes. 74
NF-kB may become activated via a canonical or an alternative signaling pathway (Fig. 1). The canonical pathway is activated by diverse factors, including tumor necrosis factor (TNF), interleukin-1 (IL-1), and lipopolysaccharide (LPS), and this pathway is critical for innate immunity, inflammation, and inhibition of apoptosis. 79 In the canonical pathway, NF-kB is a heterodimer made up of p50 and RelA subunits. NF-kB is sequestered in an inactive form in the cell cytoplasm with one of many inhibitory molecules containing ankyrin repeats that function as protein-protein interaction domains that interact with the Rel homology domains of NF-kB members; the most common inhibitory molecule is IkBa.18,39,74,79,94 Upon activation of a specific IkB kinase (IKK), phosphorylation of two conserved serine residues on the N-terminal domains of IkB proteins leads to polyubiquitination and proteasomal degradation of the inhibitory molecule, which unmasks a nuclear localization signal, allowing the NF-kB heterodimer to enter the cell nucleus and initiate transcription of downstream target genes.18,39,74
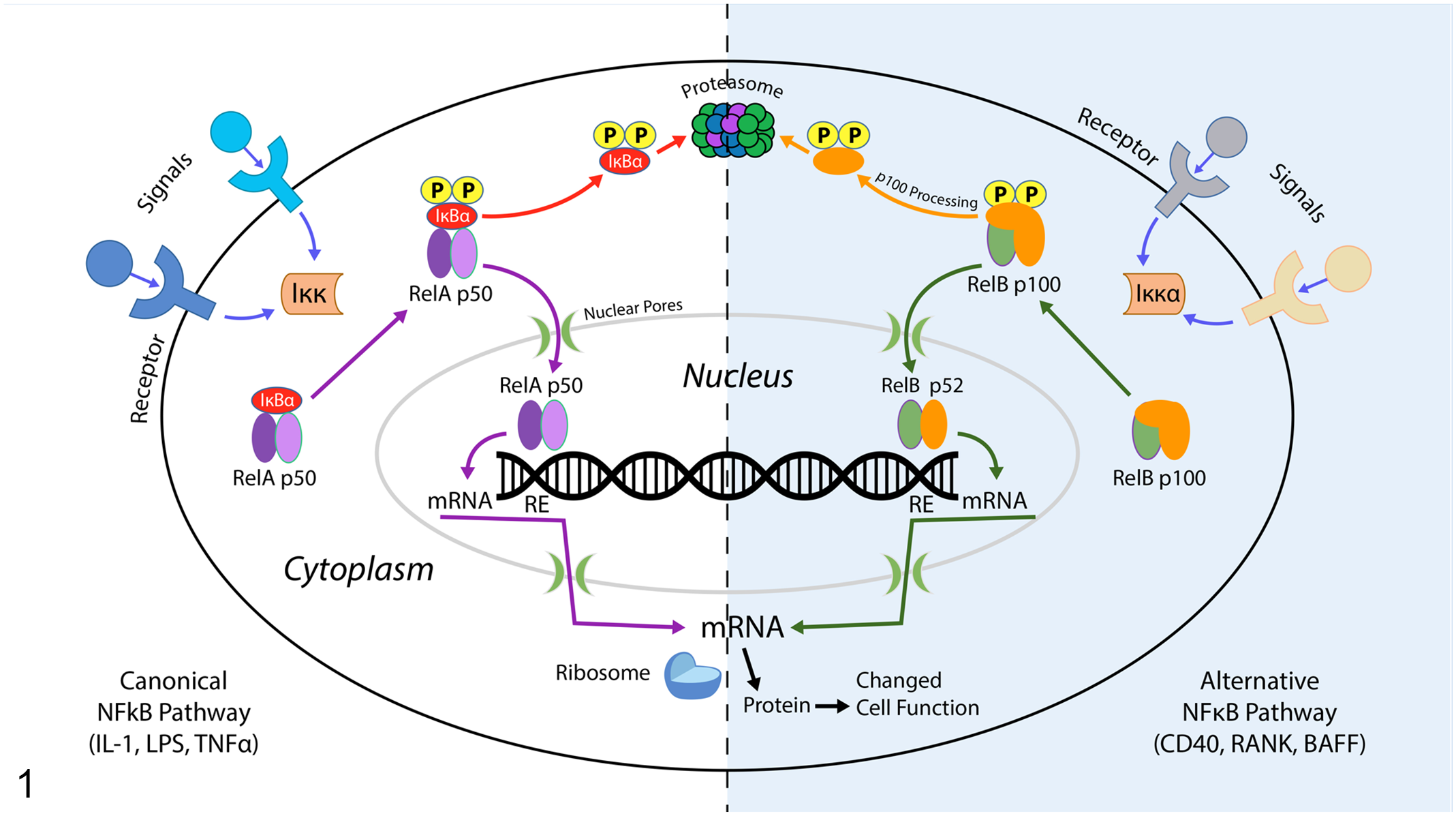
NF-kB signaling cascade. The left side of the diagram (white background) shows the canonical signaling pathway, while the alternative pathway is on the right side of the diagram (blue background). Some agonists for both pathways are listed in the figure. The canonical pathway is critical for innate immunity, inflammation, and inhibition of apoptosis, and in this pathway, NF-kB is a heterodimer made up of p50 and RelA subunits (shown in purple). This heterodimer is sequestered in an inactive form in the cell cytoplasm by an inhibitory molecule; IkBa is most common and is shown in red. Upon activation of a specific IkB kinase (IKK), phosphorylation of 2 conserved serine residues on the N-terminal domains of inhibitory IkB proteins leads to polyubiquitination and proteasomal degradation of the inhibitory molecule, which unmasks a nuclear localization signal, allowing the NF-kB heterodimer to enter the cell nucleus and initiate transcription of downstream target genes. The alternative pathway is important for appropriate B-cell maturation, formation of lymphoid organs and production of high-affinity antibodies. In this pathway, NF-kB-inducing kinase phosphorylates inhibitory IKKa, which then phosphorylates preexisting p100/NF-kB2:RelB heterodimers (shown in green and orange). This triggers the processing of inhibitory p100/NF-kB2 to p52. Active p52:RelB complexes can then translocate and activate downstream target genes. RE = response element, a binding site for active NF-kB.
The alternative pathway, by contrast, is important for B-cell maturation, formation of secondary lymphoid organs, and production of high-affinity antibodies, and is activated by NF-kB-inducing kinase (NIK).74,79 NIK phosphorylates IKKa, which then phosphorylates preexisting p100/NF-kB2:RelB heterodimers, triggering the processing of inhibitory p100/NF-kB2 to p52. Active p52:RelB complexes can then translocate and activate downstream target genes.18,39,74,80
In health, NF-kB overactivation is prevented via tightly regulated negative feedback loops that limit the duration of NF-kB nuclear localization. IkBa has a nuclear export sequence that can bind to NF-kB and remove it from the nucleus. In addition, there are several deubiquitinating enzymes (CYLD, A20, and Cezanne) that are induced by proinflammatory signaling and block IKK activation by removing polyubiquitin chains from IkBa. This results in stabilization of newly synthesized IkBa and prevents additional NF-kB activation. 74
NF-kB has many relevant roles, but most of the inducible activity of NF-kB appears to be important in mounting effective immune responses and functioning as an antiapoptotic prosurvival factor, which may help the body’s immune cells avoid death during infection. 79
NF-kB Dysregulation and Cancer Development
While acute inflammation is self-limiting and readily resolved, marked or chronic inflammation can lead to a permissive state in which precancerous cells evade normal immunosurveillance and are more vulnerable to DNA damage and epigenetic change.20,22,91
Constitutively active NF-kB is implicated in diseases that are associated with inappropriate inflammatory responses (such as asthma and autoimmune diseases) or unnatural survival (cancer). 74 The mechanisms that lead to NF-kB overactivation in tumor cells can be due to upstream activation of NF-kB signaling, direct alteration of NF-kB signaling pathway members (for example, mutation of IkBa, enhanced proteasomal activity), genetic alterations leading to the decoupling of NF-kB factors from their regulators, and autocrine and paracrine production of inflammatory cytokines.1,18,39 Some members of the NF-kB family are directly oncogenic, including c-Rel, which consistently transforms cells in culture and is amplified in lymphomas 43 and v-Rel protein, which leads to rapidly fatal lymphoma/leukemia in birds. 29 Other known oncogenes that signal through NF-kB and lead to its overactivation include Ras, myc, Pim-2 (a transcriptionally regulated oncogenic kinase), and various viral proteins, including Kaposi’s sarcoma-associated herpesvirus, hepatitis B virus protein HBx, Epstein-Barr Virus latent membrane protein, and human T-lymphocytic leukemia virus 1.1,61
There are several studies that demonstrate a link between inflammation and the perpetuation or development of cancer. Inflammation in a nontumorous part of the liver was a risk factor for recurrence of hepatocellular carcinoma following surgery in one study. 81 In another study, BalB/c mice received mammary carcinoma cells via tail vein injection; some of these mice experienced surgical trauma or LPS injection, while controls received anesthesia only. Those that were exposed to surgical trauma or LPS had increased evidence of lung metastasis, as well as increased tumor cell proliferation and decreased apoptotic activity in tumor cells. 67 Finally, modern mathematical evaluation of early B-cell differentiation includes several biological perturbations with NF-kB signaling, and predicts that (1) constant but intermediate NF-kB signaling during B-cell differentiation promotes the emergence of a CXCR7+ B-cell precursor that has a phenotype compatible with leukemia-initiating cells, and (2) constitutively overactive NF-kB signaling can potentially lead to a blockage in lineage commitment. 20
NF-kB-Induced Cellular Alterations in Cancer
There are several distinctive and complementary capabilities that allow tumors to grow and spread in the body, known as the “hallmarks” of cancer. 31 NF-kB is able to induce several of these cellular alterations, including evasion of apoptosis, acquisition of limitless replicative potential, induction of angiogenesis, and induction of invasion and metastasis.
Evasion of Apoptosis
Cellular apoptosis is a form of regulated cell death that is activated following periods of significant physiologic stress or after the cell experiences irreparable damage. Despite exposure to many typically lethal cellular stresses, cancer cells are not only able to survive, but can gain new mutations and continue to evolve, ultimately becoming more aggressive. Following initiation, the apoptotic cascade involves activation of caspase enzymes, ultimately dismantling the cell.
Constitutive activation of NF-kB is seen in many different cancer types, and its activation results in prosurvival and antiapoptotic signaling in most cells, inducing expression of at least 26 survival genes such as c-IAP1, caspase-8/FADD, FAS-associated death domain-like-IL-1B-converting enzyme (FLICE), TRAF, c-FLIP, and members of the BCL2 family, such as Bcl-xl.18,39,41 Most agents that experimentally induce apoptosis also activate NF-kB, consistent with NF-kB acting as part of the cell’s defensive machinery that can mediate chemoresistance and radioresistance.92,93 In addition to mediating resistance to anticancer therapies, NF-kB has an important role in the emergence of neoplastic cells by preventing the death of cells that have undergone significant DNA damage; there is evidence for transcriptional antagonism between active NF-kB signaling and TP53, a checkpoint control colloquially known as the “guardian of the genome.”18,97
NF-kB can also play a key role in regulating oxidative stress-related apoptosis in cells. For example, when it is acutely expressed, NF-kB can induce nitric oxide synthesis, which has been described as a proapoptotic function;13,42 however, when there is chronic production of nitric oxide via constitutively active NF-kB signaling, apoptosis is inhibited.10,15 NF-kB also regulates activation of heme oxygenase-1, which enhances free heme catabolism and protects the cell from damaging effects to lipid bilayer cell membranes.15,37 Upregulation of heme oxygenase-1 has been correlated with resistance to TNF-mediated apoptosis as well as resistance to chemotherapy-induced apoptosis.15,32,70
Acquisition of Limitless Replicative Potential
Overactive NF-kB can induce transcription of the gene for cyclin D1, which is involved in the G1/S transition of the cell cycle and therefore promotes cellular proliferation. 39 Several inflammatory cytokines that are downstream of NF-kB activation can serve as growth factors for tumor cells, including IL-2, granulocyte macrophage colony stimulating factor, IL-1B (AML), TNF (Hodgkin’s lymphoma), and IL-6 (multiple myeloma).1,39,67 In addition, telomerase reverse transcriptase protects the telomeres of neoplastic cells from shortening, which ultimately prevents normal cellular senescence or cell death. Because NF-kB overactivation can lead to telomerase reverse transcriptase-dependent transcription and induction of telomerase activity, this is another means by which cells can acquire limitless replicative potential.7,28,50
Induction of Angiogenesis
Cytokines that are enhanced by NF-kB activity can also lead to constitutive expression of angiogenic chemokines and growth factors (such as IL-8 and vascular endothelial growth factor), which affect tumor vessel growth and, ultimately, lead to tumor dissemination.34,39
Induction of Invasion and Metastasis
Metastasis is mediated in part via the expression of various adhesion molecules, including ICAM-1, VCAM-1, and ELAM-1, which are in turn mediated by NF-kB.18,89 Tumor invasion is also regulated, in part, by NF-kB-regulated gene products, including matrix metalloproteinases, urokinase plasminogen activator, and IL-8. 39 Inducible nitric oxide synthase has also been linked with metastatic activity of cancerous cells. 85
From these available data, it is tempting to conclude that NF-kB activation is often connected with tumorigenesis, but there are some scenarios in which the opposite is true. Interestingly, NF-kB blockade has been experimentally associated with the spontaneous development of squamous cell carcinomas and other cutaneous neoplasms.14,90 The role of constitutively active NF-kB signaling in leukemogenesis was investigated by studying mice that express constitutively active IKK2. These mice demonstrated a reduced pool of hematopoietic stem cells, and the functions of the remaining hematopoietic stem cells were compromised. Mice demonstrated a hyper-proliferative phenotype of hematopoietic stem cells with loss of quiescence. 67 Interestingly, increased turnover and impaired function of hematopoietic stem cells are also seen in mice that have a deficiency of RelA. 78 While it is unclear as to why both loss and gain of NF-kB function would lead to a similar phenotype, the authors speculated that “fine-tuning” of NF-kB signaling is necessary for hematopoietic stem cell biology. 67 Thus, while several NF-kB inhibitory drugs are being developed for cancer treatment, caution is warranted, as the activation or inactivation of NF-kB may lead to tumorigenesis, depending on the specific circumstances. 1
NF-kB Inhibition in Cancer Therapy
Because NF-kB activation requires the activation of multiple steps in signaling, there are numerous ways in which signaling can be interrupted at various points in the process. Examples of anticancer compounds that have been explored in human medicine include those that interfere with IKK activity (antiinflammatory drugs and natural compounds such as curcumin), those that bind NF-kB and prevent its nuclear translocation, and proteasome inhibitors that prevent NF-kB activation by blocking degradation of inhibitory molecules like IkB.18,26,27,76,83,101 Our group has been investigating the therapeutic potential of parthenolide, a sesquiterpene lactone originally purified from the shoots of feverfew (Tanacetum parthenium) to treat various canine cancers. This is a drug that appears effective for several types of human cancer, and mechanistically, exerts its anticancer effects in part via NF-kB inhibition. 26 We have found that many different canine cancer cell lines are sensitive to parthenolide and exhibit NF-kB inhibition following treatment (personal observations, unpublished data). Canine cancer types that have documented NF-kB overactivation are discussed below.
NF-kB Inhibition as a Treatment for Canine Cancers
NF-kB inhibition is a promising anticancer therapeutic strategy, given its importance in multiple cancer types and crosstalk with other pathways of clinical relevance. Ideally, an anticancer therapy will selectively target malignant cancer cells, while limiting systemic side effects and toxicity. Some compounds that target NF-kB in humans include antiinflammatory drugs, proteasome inhibitors, and IKK inhibitors. Long-term consumption of antiinflammatory drugs such as aspirin and COX2 inhibitors has been associated with reduced cancer risk.46,82 Bortezomib is a 26S proteasome inhibitor that has been shown to inhibit IkB degradation and NF-kB nuclear translocation, 11 and its use has been approved for treatment of the plasma cell neoplasm multiple myeloma. Thalidomide and other putative IKK inhibitors have also been used in the treatment of multiple myeloma, but problematically, there are side effects that may be due in part to broad NF-kB suppression, including nephrotoxicity, neuropathy, and relapse of aggressive cancers.57,105
NF-kB signaling is essential in maintaining a normal immune response and long-term use of an NF-kB inhibitor could theoretically result in immunodeficiency.52,106 In addition, NF-kB inhibitors could inhibit T cells that recognize and kill tumor cells, potentially worsening disease. Although the broad role of NF-kB and cell type-specific action complicate its potential as an anticancer target, 82 there are several examples of NF-kB inhibitors that have shown promise in canine medicine.
Leukemias and Lymphomas
Gene expression profiling of human diffuse large B-cell lymphomas (DLBCL) distinguishes two primary neoplastic subtypes: those that arise from cells within germinal centers (GC-DLBCL) and those that are immediately postgerminal center and differentiating toward plasma cells (ABC-DLBCL).3,5 Although these two types of cancer are histologically identical, the chromosomal aberrations and pathways that drive their differentiation are different, and most notably, NF-kB activation is characteristic of ABC-DLBCL but not GC-DLBCL.16,103 By comparison, gene expression profiling of canine DLBCL reveals a gene expression pattern typically most similar to ABC-DLBCL, in part because of constitutively active NF-kB signaling and activation of downstream antiapoptotic genes in most cases.4,5,69,80
Constitutive activation of both the classical and alternative NF-kB signaling pathways has been documented in dogs with DLBCL.25,62,73 Inactivating mutations in TRAF3 were first identified in canine DLBCL, then in some human DLBCL samples. 11 TRAF3 encodes a negative regulator of the noncanonical NF-kB pathway, leading to upregulation of NF-kB. In addition, mutations that lead to constitutively active canonical NF-kB signaling (in TNFAIP3 and CD79b) were observed in low numbers of canine DLBCL cases.5,11 These studies provide convincing evidence that NF-kB overactivation is important in the pathogenesis of canine DLBCL.
The importance of NF-kB signaling in canine lymphoma and leukemia has also been documented during the evaluation of multiple investigational anticancer compounds, as discussed below.
Intracellular transport between nuclear and cytoplasmic compartments is regulated by importins and exportins, and of the 7 known exportins, most tumor suppressor proteins and IkB are transported out of the cell nucleus by exportin 1 (XPO1/CRM1). Stabilization of IkB within the cell nucleus may neutralize constitutively active NF-kB activity in canine lymphoma. KPT-335 is a selective inhibitor of nuclear export (SINE) that has shown biological activity in vitro and in vivo in dogs with previously progressive non-Hodgkin’s lymphoma.51,71,75
A study evaluating parthenolide in inhibition of NF-kB activity in human leukemic stem cells also evaluated cells from dogs with acute leukemia. Researchers found that most acute leukemia cells from dogs (7/8 samples) exhibited constitutive activation of NF-kB and that in vitro exposure to parthenolide led to decreased NF-kB activity and decreased cell survival. 30 Three dogs with acute leukemia also received parthenolide therapy, and peripheral blood from these animals demonstrated a rapid and consistent reduction in immature CD34+ cells following treatment, consistent with differentiation and/or death of primitive leukemia cells. 30 The study showed active NF-kB signaling in part by demonstrating nuclear p65 immunofluorescence that was inhibited by parthenolide therapy. We recapitulated this experiment to show dramatic inhibition of p65 nuclear translocation in DH82, a canine histiocytic sarcoma cell line, treated with parthenolide (Fig. 2 and Fig. 3; previously unpublished data).
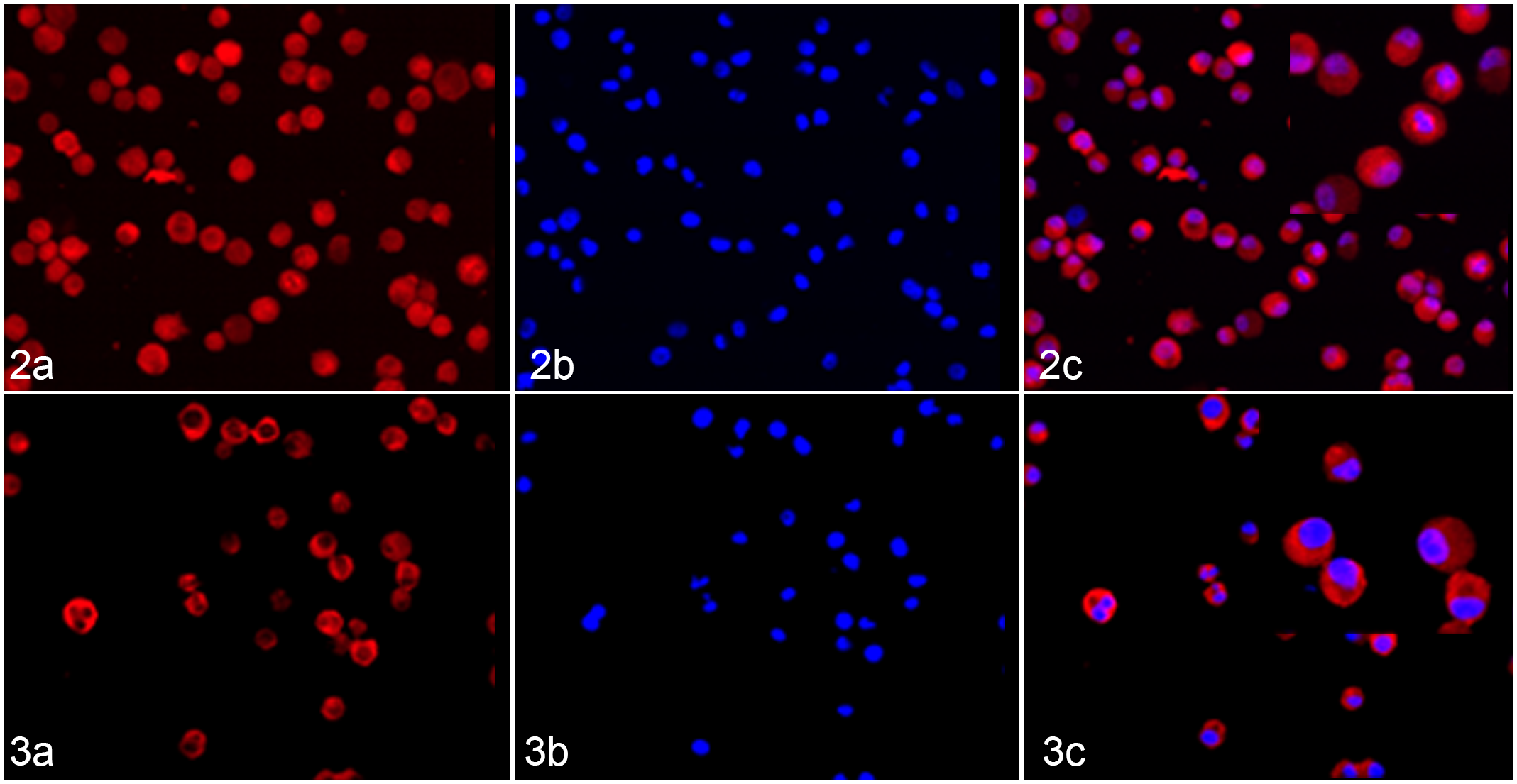
NF-kB signaling in canine histiocytic sarcoma (DH82) cells. From left to right: (a) NF-kB Cy3, (b) DAPI nuclear stain, and (c) merge.
Bortezomib is a proteasome inhibitor that has been utilized for treatment of human multiple myeloma and other hematological malignancies. One study demonstrated constitutive activation of NF-kB in some canine lymphoma and leukemia cell lines, and showed that treatment with bortezomib prevented nuclear translocation of NF-kB and inhibited the growth of several cell lines. 44
Methotrexate (MTX) is an antimetabolite used as an anticancer therapeutic in various types of canine cancer, including lymphoma. MTX-induced cell death was investigated in canine lymphoma and leukemia cell lines. The data suggested that apoptotic cell death was mediated via inhibition of NF-kB, leading to decreased expression of Bcl2, and enabling activation of the intrinsic apoptotic cascade. 66
Finally, inhibition of NF-kB by the synthetic compound IMD-0354 in canine lymphoma cells increased glucocorticoid receptor expression, sensitizing cells to dexamethasone. 55 This work implied that combination therapy with glucocorticoids and NF-kB inhibitors may be beneficial for some dogs with lymphoma.
Hemangiosarcoma/Angiosarcoma
Angiosarcomas (ASs) and hemangiosarcomas (HSAs) are tumors of endothelial origin. The exact cause of these neoplasms in humans, dogs, and rodents is unknown; they may arise from the transformation of endothelial cells that are resident within tissues, or alternatively from circulating stem cells that are recruited from bone marrow or sites of extramedullary hematopoiesis.12,45,102 Because these tumors are exceptionally rare in humans, research efforts to identify driver mutations, identify actionable drug targets, and conduct human clinical trials have stopped short of the need to address this deadly disease. Studies have found mutations in P53 and PTEN in both human AS and canine HSA, implying shared pathogenesis for these cancer types.17,63,84,95
Whole-exome sequencing from formalin-fixed, paraffin-embedded tissues, pathway analysis, comparative genomics, and candidate gene analysis were performed to identify candidate driver mutations in canine visceral HSA. In addition to recurrent mutations in the PI3K pathway, PTEN, and TP53, there was a missense mutation in PLCG1 (S273F) in one canine tumor. 96 S273 is in the kinase domain of PLCG1 and is highly conserved, and the corresponding human mutation results in increased enzyme activation and downstream signaling, including NF-kB signaling.35,53 Although this mutation has only recently been identified in human visceral AS, available data suggest that it may contribute to the pathogenesis of HSA and that some affected canine tumors may share molecular similarity to their human counterparts. 96
Yunnan Baiyao is a Chinese herbal medication that has been used in humans for its hemostatic properties and is used anecdotally in dogs with visceral HSA. A study demonstrated that Yunnan Baiyao induces both time- and dose-dependent apoptotic cell deaths in canine HSA cell lines in vitro. Intriguingly, wild yam root, a component of this medication, contains a plant phytosterol estrogen known as diosgenin, which induces antiproliferative and proapoptotic effects, in part via downregulation of NF-kB.2,56,98
Mammary Cancer
Growing evidence in the literature suggests a role for overactive NF-kB signaling in canine mammary cancer. In neoplastic mammary tissues from dogs, several researchers have found molecular evidence consistent with NF-kB overactivation. In a canine model of breast ductal carcinoma in situ, evaluation of the transcriptome of mammary lesions showed that NF-kB was differentially overexpressed in canine ductal carcinoma in situ and invasive carcinoma as compared with samples from normal tissues and from atypical ductal hyperplasia. 60 Others reported relative miR-497 downregulation in canine mammary cancer tissue and discovered that restoration of miR-497 inhibited active NF-kB signaling in vitro. 106 Protein phosphatase 2A (PP2A) is an evolutionarily conserved serine/threonine protein phosphatase that functions as a tumor suppressor that can be inhibited by SET; increased SET protein levels in canine mammary cancer and osteosarcoma suppresses PP2A activity, which in turn enchances mTOR, B-catenin, and NF-kB signaling in these cancers.38,86
Overexpression of the secreted frizzled protein 2 (sFRP2) in canine mammary tumor cells has been documented as key to resistance to apoptosis. 47 Specifically, using electrophoretic mobility shift assays, the UV-induced level of active NF-kB in mammary gland cells expressing sFRP2 was significantly increased relative to NF-kB activity in control cells, suggestive of NF-kB-mediated apoptotic resistance. 47
There is some evidence suggesting that NF-kB overactivation is correlated with more aggressive tumor behavior. A study evaluating immunohistochemical labeling of NF-kB p65 in canine mammary tumors found that higher nuclear expression of NF-kB was correlated with larger tumor size, lymph node metastasis, and higher mitotic index, while there was no correlation with distant metastasis or cyclin D1 expression. 59 Another study identified an association between increased NF-kB activation and decreased surivival in dogs with mammary cancer. 54
Malignant Melanoma
In a study comparing the transcriptome of canine oral malignant melanoma in comparison with human disease, there was >80% homology in upregulation of significant oncogenes, with activation of the JAK-STAT pathway being the most frequently upregulated pathway in both species. In addition, several target genes of NF-kB were upregulated, consistent with NF-kB activation, and binding motifs for NF-kB were observed in the highest number of upregulated differentially expressed genes in canine melanoma. 68
Glioma
During transcriptome evaluation of canine oligodendroglioma tissues, IRX5 was among the top 10 upregulated genes relative to normal brain tissue. 58 IRX5 is important for cancer cell survival. While IRX5 has not been characterized in glioma, its overexpression has been found to contribute to NF-kB overactivation and interactions with osteopontin in human tongue squamous cell carcinoma. 33
Osteosarcoma
Osteosarcoma is a malignant mesenchymal tumor affecting bones and soft tissues of dogs and humans; in dogs, it is the most common primary bone tumor and shares clinical and biological similarities to the human cancer.99,100 As previously stated, increased SET protein levels in canine osteosarcoma positively regulate NF-kB signaling secondary to decreased PP2A activity.38,86
Genome-wide RNAi was used to screen canine osteosarcoma cells to identify genes correlated with aggressive features (growth and survival in anchorage-independent conditions). The study found that TMIFGD3 (transmembrane and immunoglobulin domain containing 3) expression was associated with decreased cell sphere formation and reduced NF-kB activity. 36
In addition, there is some work demonstrating the effect of NF-kB overactivation within the tumor microenvironment in canine osteosarcoma. Interstitial acidosis in canine osteosarcoma is a microenvironmental condition that mimics wound healing and is suggested to contribute to tumor cell proliferation via the release of mitogenic and chemotactic factors from reactive mesenchymal cells. In this work, acidosis-mediated activation of NF-kB in mesenchymal stem cells enhanced clonogenicity and invasion of cocultured osteosarcoma cells.6,9
Prostate Cancer
The only nonhuman animal known to spontaneously develop prostate cancer is the dog, which makes dogs a good animal model for this human disease.49,77 AR-42 is a histone deacetylase inhibitor that inhibits the proliferation of several different cancer types. AR-42 upregulated PTEN and downregulated PI3K and NF-kB expression in a canine prostate cancer cell line. As downregulation of PTEN and upregulation of PI3K and NF-kB are important in anoikis resistance, this work suggests that modulation of gene expression by AR-42 leads to anoikis and apoptosis in prostate cancer cells. 19
Studies of Multiple Cancer Cell Types
CIGB-552 is an anticancer peptide that induces expression of copper metabolism domain-containing protein 1 (COMMD), leading to ubiquitination of NF-kB RelA and cell death in cancer cell lines.21,88 In a pilot clinical trial of CIBG-552 with eight patients, dogs with lymphoma (n = 3), malignant melanoma (n = 1), and mast cell tumor (n = 1) had stable disease or a response to therapy. 88
Conclusions
As documented in humans, there are many types of canine cancer that exhibit dysregulated NF-kB signaling, and since dogs and humans often share common cancer biology, dogs are well-positioned to provide a natural animal model of human cancers with NF-kB overactivation. Ultimately, continued research efforts will provide a direct benefit in advancing therapeutics for both dogs and humans.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors wish to thank the Morris Animal Foundation (fellowship training grant D20CA-402) and National Institutes of Health (grant number 1TL1TR002533-01) for funding, and SK Richman for assistance with figure illustration and formatting.
