Abstract
The aim of this study was to investigate the anti-inflammatory properties of
The inflammation process is tightly regulated by both initiation and maintenance signals and considered to be a major risk factor in the pathogenesis of chronic diseases where the macrophages are important immune cells, which regulate inflammation-producing expression of inflammatory proteins and pro-inflammatory chemokines, cytokines, and nitric oxide (NO).
1,2
Macrophages are highly sensitive to initiators of inflammation, such as lipopolysaccharide (LPS), which respond by the release of mediators, not only interleukins (ILs) and cytokines, but also inducible NO synthase (iNOS) and reactive oxygen species (ROS), which induce inflammatory gene expression where each one is associated somehow with the pathophysiology of the inflammation.
3
-5
Because macrophages produce a wide range of biologically active molecules that participate in both beneficial and detrimental outcomes in inflammation, modulation of macrophage activation is a good strategy to prevent inflammatory diseases. Red fruit (
Materials and Methods
Chemicals and Reagents
RFO (APOTEK) was supplied from Smile International Co., Ltd (Seoul, Korea); Dulbecco’s modified Eagle medium (DMEM), fetal bovine serum (FBS), and penicillin–streptomycin from Corning (Oneonta, NY, USA); 2ʹ7ʹ-dichlorofluorescein diacetate (DCFH-DA) and anti-iNOS antibody from BD (San Jose, CA, USA); peroxidase-conjugated secondary antibodies and TriZol were purchased from Life Technologies (Grand island, NY, USA); and phospho-JNK (c-Jun N-terminal kinase), phospho-ERK (extracellular signal-regulated kinase), phospho-p38, phospho-IκB, and nuclear factor κB (NF-κB) antibodies from Cell Signaling Technology Inc. (Beverly, MA, USA). The enzyme immunoassay (EIA) kit used for prostaglandin E2 (PGE2) was obtained from R&D Systems (Minneapolis, MN, USA); the ECL detection reagents from GE Healthcare (Buckinghamshire, UK); LPS (
Cell Culture
RAW 264.7, a murine macrophage cell line, was purchased from American Type Culture Collection and maintained in DMEM supplemented with 1 mg/mL glucose, 10% FBS, and 100 mg/mL penicillin-streptomycin at 37 °C in 5% CO2.
Cell Viability Assay
The cytotoxic effect of RFO against the RAW264.7 cell line was evaluated by MTT assay. Briefly, cells were seeded at a density of 5 × 103 cells/well in a 96-well plate for 24 hours. Then, the cells were treated with various concentrations of fractions with or without 1 µg/mL LPS. After 24 hours, 2 mg/mL MTT was added to each well, and incubated for 3 hours. The formazan formed was dissolved in dimethylsulfoxide and the absorbance at 550 nm was measured using a microplate reader (Molecular Devices, Sunnyvale, CA, USA). Cell viability was calculated as the percentage of viable cells in drug-treated group vs the untreated control. Each experiment was repeated 3 times.
Nitrite Assay
Cells were treated with various concentrations of RFO for 30 minutes, and then incubated with 1 µg/mL LPS for 24 hours. Because NO production is reflected in the accumulation of nitrite in the cell culture medium, 50 µL of supernatant was removed and mixed with the same volume of Griess reagent (Promega, Madison, WI, USA). After incubation for 10 minutes, the absorbance of the mixture at 550 nm was measured using a spectrophotometer (TECAN, Austria). The nitrite levels were estimated as the percentage of absorbance of the sample relative to the respective control.
Cyclooxygenase-2 Assay
Cells were treated with various concentrations of RFO for 30 minutes, and then incubated with 1 µg/mL LPS for 24 hours. After incubation, the supernatants were removed and cyclooxygenase-2 (COX-2) measured. The COX-2 concentrations were evaluated using a specific EIA kit (Cayman, Ann Arbor, MI, USA), according to the manufacturer’s instructions.
PGE2 Assay
Cells were treated with various concentrations of RFO for 30 minutes, and then incubated with 1 µg/mL LPS for 24 hours. After incubation, the supernatants were removed and PGE2 measured. The PGE2 concentrations were evaluated using a specific EIAkit (Cayman, Ann Arbor, MI, USA), according to the manufacturer’s instructions.
iNOS Gene Measurement by Real-Time Polymerase Chain Reaction
The cells from the supernatants, after being removed, were subjected to RNA isolation, which was performed using TRIzol reagent according to the manufacturer’s instructions. cDNA was synthesized using hyperscript RT master mix (GeneAll, Daejeon, Korea). The primers were iNOS forward: 5ʹ-ATGTCCGAAGCAAACATCAC-3ʹ, reverse: 5ʹ-TAATGTCCAGGAAGTAGGTG-3ʹ, and GAPDH forward: 5ʹ-TGTGATGGTGGGAATGGGTCAG-3ʹ, reverse: 5ʹ-TTTGATGTCACGCACGATTTCC-3ʹ. The polymerase chain reaction (PCR) was amplified using ABI 7500 and Taqman gene expression master mix (Applied Biosystems, Waltham, MA, USA). Quantitative analysis was performed to compare the ΔΔCt after normalization by GAPDH as an internal control. After analysis, the PCR products were electrophoresed on 3% agarose gel and images were taken by cybergreen detection using a Kodak imagestation FX (Kodak, Rochester, NY, USA)
Analysis of ROS by Flow Cytometry
Cells were treated with various concentrations of RFO for 30 minutes, and then incubated with 1 µg/mL LPS for 24 hours. This was followed by the addition of 10 µg/mL DCFH-DA. The suspensions were washed with PBS after incubation for 20 minutes. The suspensions were then assayed with a flow cytometer (C6 Accuri, BD, Bedford, MA, USA) according to Rhee et al. 4
Western Blot Analysis
Cells were treated as described previously, then total lysates were prepared with lysis buffer (50 mM Tris [pH 7.4], 300 mM NaCl, 5 mM EDTA (pH 8.0), 0.5% Triton X-100, 1 mM aprotinin, 1 mM leupeptin, 1 mM pepstatin, 10 mM iodoacetamide, and 2 mM phenylmethylsulfonyl fluoride. Each nucleus and cytosol extract was isolated using an NE-PER nuclear and cytoplasmic extraction reagent kit (Pierce, Rockford, IL, USA). Briefly, cells were washed with PBS, and were prepared with ice-cold extraction buffers sequentially. After centrifugation at 16 000×
High-Performance Liquid Chromatography
To determine the content of β-cryptoxanthin in RFO, high-performance liquid chromatography (HPLC) analysis was used according to previous studies. 17 HPLC analysis was performed using an Agilent 1100 model with a pump (G1311C), auto sampler (G1329B), YMC C30 carotenoid column (250 × 4.6 mm, 9 µm), diode array detector (Agilent, Santa Clara, CA, USA), mobile-phase methanol: MTBE: 0.1% H3PO4: 16:80:4, flow rate 0.9 mL/min, injection volume 120 µL, running time 35 minutes, temperature 25 °C, and detection wavelength 474 nm.
Statistical Analysis
All results are presented as mean ± SD, and represent 3 or more independent experiments. Data were compared using 1-way analysis of variance using Prism (GraphPad, La Jolla, CA, USA) with
Results
RFO Did Not Affect Cell Viability
Figure 1(A) showed the effect of RFO on viability of RAW 264.7 with and without LPS. Cell viability was not affected by 10-1000 μg/mL of RFO with or without LPS.
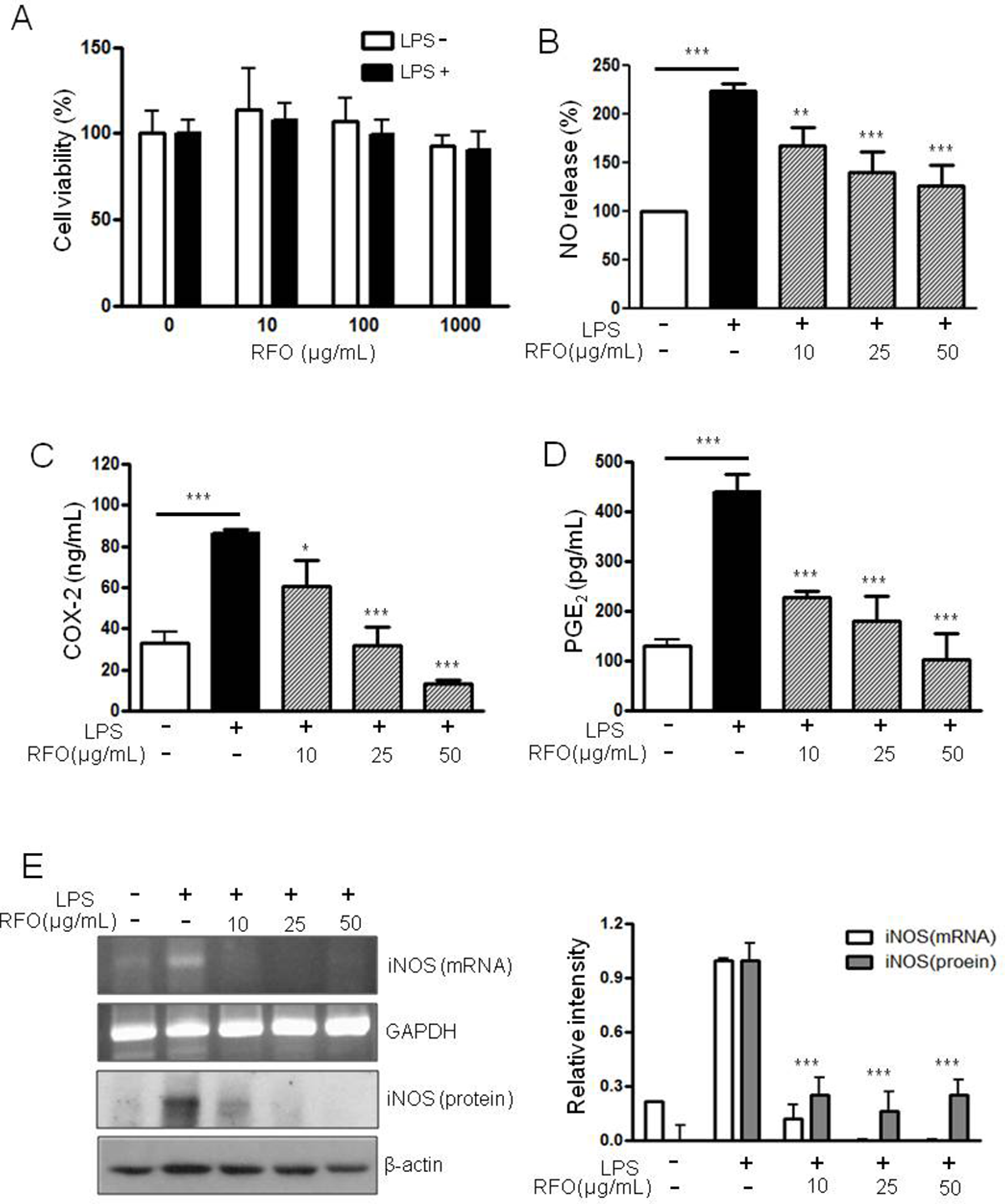
Effect of RFO on cell viability, NO release, PGE2 production, protein level of iNOS, and mRNA level of iNOS in LPS-induced RAW 264.7 cells. The cytotoxic effect of all fractions from RFO against RAW 264.7 cell lines was evaluated by MTT assay. Cell viability was calculated as a percentage of viable cells in the drugs-treated group vs the untreated control. The data are represented as the mean ± SD (
RFO Reduced NO in LPS-Induced Macrophages
To assess the effects of RFO on NO production in LPS-induced RAW 264.7 macrophages, cells were treated with various concentrations of RFO for 30 minutes, then incubated with 1 µg/mL LPS for 24 hours. NO release was elevated by 224% ± 19.2% (
RFO Decreased COX-2 Production in LPS-Induced Macrophages
COX-2 production was significantly increased from 33.2 ± 5.2 to 86.2 ± 1.9 ng/mL (
RFO Also Decreased PGE2 Production in LPS-Induced Macrophages
PGE2 production was significantly increased to 440.6 ± 35.4 pg/mL (
RFO Suppressed Both mRNA and Protein Levels of iNOS in LPS-Induced Macrophages
To determine the inhibitory effects of RFO on pro-inflammatory mediator NO, COX-2, and PGE2 production, the biosynthesis of transcriptional levels of iNOS was performed with semi-quantitative reverse-transcription PCR and western blot analysis on LPS-induced RAW 264.7 macrophages. Consistent with the findings shown in Figure 1(B), RFO had a significant concentration-dependent inhibitory effect on the inflammation through pro-inflammatory mediator NO in LPS-induced RAW 264.7 macrophages. Figure 1(E) indicates that both the mRNA level (ΔCt value) and protein level of iNOS were significantly decreased by treatment with RFO (
RFO Attenuated ROS in LPS-Induced Macrophages
Excess ROS is known to damage intracellular proteins, lipids, and nucleic acids and induce inflammation.
18
Thus, we investigated ROS production in response to LPS using flow cytometry. DCFH-DA binds to ROS-produced cells. Figure 2 shows that the DCFH-DA-positive cells were increased following LPS treatment from 40.7% ± 2.1% to 70.9% ± 3.1%. However, ROS production was also significantly inhibited by RFO in a dose-dependent manner; 47.1% ± 2.45% at 10 µg/mL (

Effect of RFO on ROS in LPS-induced RAW 264.7 cells The cells were incubated with 10-50 μg/mL of various fractions in either the presence or absence of LPS (1 µg/mL) for 24 h. The cells were preincubated for 1 h in the presence or absence of LPS before the addition of RFO fractions, followed by the addition of 10 µg/mL 2′7′-dichlorofluorescein diacetate. The results are expressed as the mean ± SD (
RFO Suppressed Nuclear Translocation of the NF-κB P65 Subunit via MAPK
Since p65 is a major component of NF-κB activated by LPS in macrophages, we evaluated the levels of p65 in nuclear extracts by western blotting analysis. Phosphorylation of I-κB resulted in degradation and release of NF-κB, which then translocated to the nucleus. Therefore, we examined whether RFO could prevent phosphorylation of I-κB induced by LPS treatment. Figure 3(A) shows that I-κB phosphorylation was decreased by treatment with LPS alone at the cytosol level, but that such phosphorylation was significantly increased in the presence of RFO, similar to results for the nuclear translocation of p65. Taken together, these data suggest that the inhibitory effect of RFO on the LPS-induced translocation of p65 might be involved in the suppression of I-κB phosphorylation. To investigate further whether the inhibition of pro-inflammatory mediators by RFO is modulated through the mitogen-activated protein kinase (MAPK) pathway, we evaluated the effects of RFO on the LPS-induced phosphorylation of p38, ERK, and JNK (Figure 3(B)). RFO suppressed LPS-induced phosphorylation of p38, ERK, and JNK. These results suggest that RFO blocks MAPK pathways to suppress the inflammatory response in LPS-induced RAW 264.7 macrophages.
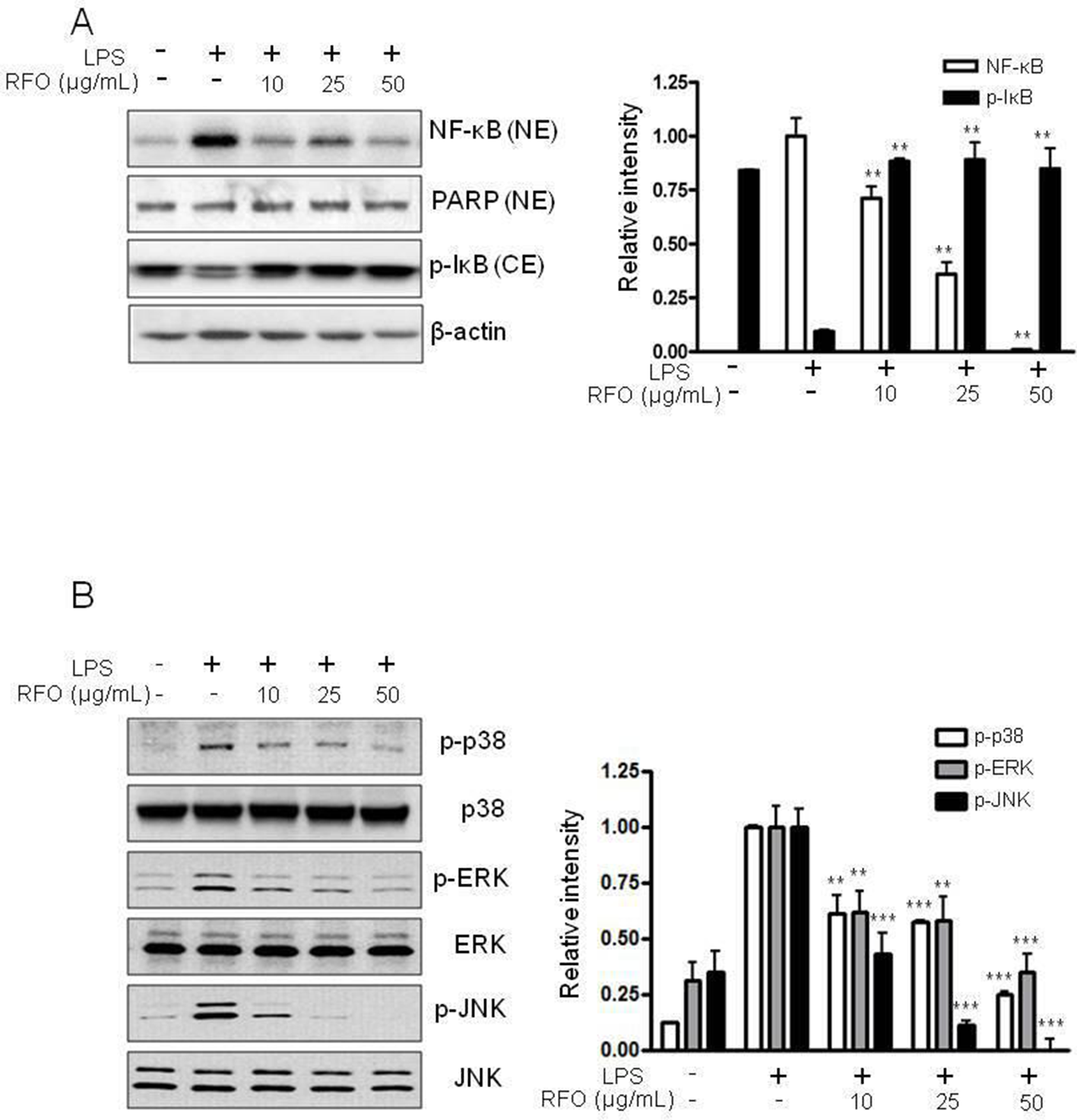
Effect of RFO on the expression of NF-κB, phospho-IκB, and MAPK levels in LPS-induced RAW 264.7 cells. The cells were incubated with 10-50 μg/mL of RFO in either the presence or absence of LPS (1 µg/mL) for 24 h. Each nucleus and cytosol extract was isolated using an NE-PER nuclear and cytoplasmic extraction reagent kit, and the expression of NF-κB and phosphorylation of I-κB were detected by western blot analysis. (A) The relative protein expression was densitometrically quantified using a BioRad GS-670 densitometer and normalized to PARP and β-actin. The expression of MAPK in whole lysates was assessed by the same method. (B) The relative protein expression was densitometrically quantified and normalized to total MAPK proteins. The results from replications are expressed as the mean ± SD (
HPLC Analysis of RFO
Table 1 shows the HPLC analysis of RFO. One gram of RFO had 14.05 ± 0.8 mg of β-cryptoxanthin and 7.4 ± 0.7 mg of β-carotene.
Component of Red Fruit Oil.

Discussion
Inflammation is an immune response that protects our body against host response to infection and injury. 19,20 All inflammatory responses act through mononuclear cells, macrophages, and lymphocytes. Macrophages play on important innate immune effectors and increase pro-inflammatory factors including nitric oxide (NO), PGE2, and cytokines.
The excessive amounts of NO and PGE2 produced by activation of iNOS and COX-2 in response to LPS play an important role in inflammation. 21,22 The overproduction of iNOS-derived NO is involved in the pathology of various inflammatory disorders and tissue damage conditions. A change in the NO level through the inhibition of iNOS enzyme activity or iNOS induction provides a means of assessing the effect of these agents on the inflammatory process. iNOS is implicated in the synthesis of prostaglandin H2 starting with arachidonic acid, which is a precursor of PGE2, in activated macrophages with LPS. 23 PGE2 plays a critical role in the pathogenesis of inflammatory diseases and requires 2 enzymes acting sequentially, COX-2 and PGE synthase. 20,24 Thus, modulation of iNOS and NO expressions could be one of the strategies to reduce inflammatory diseases. The production of inflammatory cytokines is a crucial part of regulating inflammation and tumor progression. The key signaling pathway mediating the inflammatory response, the NF-κB transcription factor, has been well established in various inflammatory diseases and cancers. 25,26 It is also well known that NF-κB is a significant role factor regulating the expression of inflammation-associated enzymes and cytokine genes, such as iNOS, COX-2, tumor necrosis factor α, and IL-1β, which contain NF-κB binding motifs within their respective promoters. 1,27 Therefore, this signaling pathway is a good target for anticancer and anti-inflammatory drug development. Many of the upstream kinases and downstream substrates are the same for each of the major cascades. Our results revealed that the anti-inflammatory activities of RFO are mediated through the phosphorylation of I-κB and nuclear translocation of the NF-κB p65 subunit. Besides, these results also indicate that the inhibitory effects of RFO on MAPK and NF-κB signaling are related to a decrease in ROS. It is well known that oxidative stress stimulates ROS production in the RAW 264.7 cell line. 11,28 Our data showed that the pretreatment with RFO significantly decreased ROS production in LPS-induced RAW264.7 cells using DCFH-DA staining, which demonstrated that RFO had the ability to reduce the oxidative stress. We also suggested that RFO regulated MAPK and NF-κB signaling of inflammation operating through oxidative stress. These results demonstrated that RFO could act either as a scavenging agent or act on the redox state of the cell. In previous study, we already demonstrated that RFO regulated cellular senescence through ROS modulation in H2O2-induced endothelial cells. 5
Carotenoids such as β-cryptoxanthin and β-carotene are antioxidants, which are not produced in the human body and must be ingested from outside. Many studies indicated that healthy people have a high level of β-cryptoxanthin in blood. 29 -32 β-Cryptoxanthin is the only provitamin A component of carotenoid-based xanthophylls. 14,33 Carotenoids are lipid-soluble components that must be ingested with fat to be absorbed completely in the body. Carotenoids affect the inflammation levels in blood as strong antioxidants, and help purify the blood. Park et al 34 showed that daily oral administration of β-cryptoxanthin prevented the progression of osteoarthritis and inhibited proinflammatory cytokines in mice. Therefore, we examined the effects of RFO on the production of several inflammatory mediators and on the expression levels of iNOS in LPS-induced RAW 264.7 macrophage cells. Our results demonstrated that RFO inhibited the expression of iNOS, as well as the production of NO and PGE2, and the mechanisms underlying the suppression of the inflammatory response of the NF-κB and ROS. According to the USDA database, the β-carotene content of RFO is 335 times higher than that of blackberry, 119 times that of broccoli, 13.9 times that of pumpkin, and 5.2 times that of carrot. 35 -37 In addition, the β-cryptoxanthin content of RFO was 76 times higher than that of orange and 15 times that of papaya. 31,38 These findings suggest that RFO might be a beneficial therapeutic agent in the treatment of a variety of inflammatory diseases.
Footnotes
Acknowledgment
We would like to express our sincere appreciation to Tae-Hong Park, Smile International Co. Ltd. for kindly providing Red fruit oil.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2020R1I1A1A01052306).
